Abstract
Convection-enhanced delivery (CED) of compounds into brain tumors reportedly circumvents the blood brain barrier. CED intends to increase drug delivery to malignant cells, reaching high local therapeutic concentration and decreasing or eliminating systemic side effects. Clinical experience and published data on catheter placement (CP) surgery are scarce. We propose practical and technical guidelines for planning CED based on our experience. We retrospectively analyzed the medical charts and relevant neuroimages of 25 patients following the insertion of 64 CED catheters. The patients were enrolled in at least one of four clinical trials using CED for treating recurrent glioblastoma multiforme in our institution between 2003–2006. Intra- and postoperative complications related to CP surgery and the difficulties and pitfalls of planning were evaluated. There were 29 CP surgeries. Forty-four peritumoral brain tissue catheters were inserted in 16 CP surgeries following tumor resection in 16 patients, and 20 catheters were placed into the tumor in 13 procedures in 10 patients. The lesions were in or near eloquent brain tissue areas in 13 of all CP surgeries. Complications included increased edema (31%), infection (6.9%), bleeding (6.9%) and seizures (13.8%). Significant neurological deterioration occurred in 4 patients (13.8%). Difficulties in adhering to CP surgery guidelines included lesion site (superficial, mesial temporal lobe, proximity to CSF spaces), proximity to eloquent cortical areas, tissue density that interfered with the trajectory, and technical limitations of stereotactic instruments. CED procedures for high-grade gliomas may be associated with surgical morbidity. Adherence to guidelines might be difficult because of lesion site and complicated by brain and tumor tissue characteristics. This should be considered while planning clinical trials that use convection-based technology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
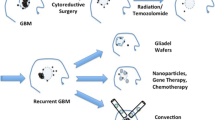
High-grade gliomas (HGGs) are the most common malignant primary intracranial tumor in adults. Despite multimodality treatments, including surgery, radiation and chemotherapy, the prognosis remains poor, with a median survival of approximately 1 year following diagnosis [1]. Factors limiting the success of treatment include the invasive nature of the lesion, intrinsic cellular resistance to most treatment compounds, and the presence of the blood brain barrier (BBB) which limits the passage of drugs into infiltrated brain tissue and subsequently requiring higher drug levels to achieve therapeutic concentration. Local drug delivery methods have been developed in an attempt to increase local drug concentration without intensifying systemic side effects. This includes intracavity direct injection or administration of drugs [2], placement of controlled-release impregnated polymers, such as Gliadel wafers [3], chronic low flow microinfusion [4] or convection-enhanced delivery (CED) [5, 6]. CED results in high-flow micro-infusion and the delivery of macromolecules through the extracellular space. It can be distributed over a large brain volume and at relatively homogeneous concentrations [5, 7–10]. CED of various drugs and molecules are currently under investigation for the treatment of malignant brain tumors. Most clinical trials using a CED strategy deliver tumor-specific ligands linked to a bacterial toxin into the tumor or its surrounding infiltrated tissue [11–16]. Other approaches include the delivery of chemotherapeutic agents (e.g., paclitaxel) [17, 18], gene therapy (e.g., HSV-1-tk) [19] and local radiotherapy with a cytotoxic dose of 131I [20]. The treatment potentials of the CED approach have been demonstrated in many preclinical and clinical studies in addition to having been the subject of many publications. Large randomize clinical trials, however, have failed to establish a survival benefit derived from using this approach with different compounds, partly because of technical issues related to suboptimal catheter placement (CP) and poor drug distribution [21].
The clinical experience of CP is limited to a few specialized centers, and our literature search failed to produce any data on complications and technical difficulties associated with it. We performed a relatively large number of CP surgeries and now describe the intra- and postoperative complications as well as the difficulties and pitfalls related to this procedure in order to enhance surgical planning.
Patients and methods
This is a retrospective analysis of prospectively collected clinical and neuroimaging data. This review has been approved by our local institutional ethics board (0608-10 TLV). The study group consisted of 25 patients who underwent a total of 29 CP surgeries that were performed at the Tel Aviv Medical Center Department of Neurosurgery between March 2003 and December 2006. They were all diagnosed as having recurrent glioblastoma multiforme (GBM) and were enrolled in one of four clinical trials using implantation of intracranial catheters for CED. All patients received anticonvulsive medications, high does steroids and prophylactic antibiotic perioperatively and during the treatment period.
The three surgical protocols included implantation of catheters into the tumor prior to resection (using 1 catheter), implantation into the peritumoral brain tissue (using between 2–4 catheters) after tumor resection, and implantation of catheters into the tumor without resection (using up to 2 catheters). All catheters used in the studies were commercially available ventricular catheters that were inserted using supplied stiletto (Medtronic Cardiac/Peritoneal Catheters, Medtronic, CA, USA and Vygon, Norristown, PA, USA). CP planning was based on a current magnetic resonance imaging (MRI) study (gadolinium-enhanced T1-weighted sequence, 3D SPGR, axial scan) and BrainLab, iPlan® stereotaxy software (BrainLab, Germany). The CP guidelines that were followed [22] specified the distance and depth of catheter tip from pial surfaces/deep sulci (>25 mm), cysts, cavities or the ventricular ependymal layer (>5 mm) and distance between catheters (>20 mm). CP surgeries were performed as frameless stereotactic procedures, and two systems were used for catheter insertion: the Navigus Trajectory Guide system (Image-Guided Neurologics, Melbourne, Florida) before December 2004, and a modified BrainLab stereotactic fixation arm afterwards. CP was done free-handed with the aid of a neuro-navigation system in the first four surgeries. All patients underwent one non-contrast head computerized tomographic (CT) scan immediately after the procedure for the evaluation of the catheter position and surgery-related complications, and another CT 4–7 days later after catheter removal. The CP surgeries and the study patients’ characteristics (including methods of insertion and number of catheters placed) are summarized in Table 1.
Results
The 25 patients who underwent 29 CP surgeries included 18 males and 7 females, whose mean age was 54.2 ± 11.8 years (range 28 to 70 years). The mean preoperative Karnofsky performance score (KPS) for the group was 88 ± 12.1 (range 70–100). A total of 64 catheters were inserted in 29 frameless stereotactic procedures. The lesions were in or near eloquent brain areas in 13 of the 29 operations (45%).
One of the patients underwent three separate procedures for two different clinical trials and another patient underwent two separate procedures for the same clinical trial. Forty-four peritumoral brain tissue catheters were inserted in 16 procedures following tumor resection in 16 patients. Twenty catheters were placed into the tumor bed in 13 frameless stereotactic procedures in 10 patients. Surgical resection of the tumor followed the CED treatment period occurred in 6 of the patients.
One catheter was placed per procedure in five (17.2%) surgeries, and one of these patients underwent two separate surgeries for the placement of one catheter each time. Two catheters were placed per procedure in 13 (44.8%) surgeries. Two of these 13 patients were treated more than once: one of them was treated twice, each time with two catheters (numbers 28 and 29 in Tables 1, 2). The other patient was treated three times for two different clinical trails, each time with two catheters (numbers 9, 19 and 20 in Tables 1, 2). In 11 CP surgeries (37.9%) the insertion of three catheters per procedure was performed.
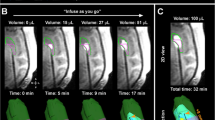
Twenty-three catheters (35.9%) were inserted using the Navigus Trajectory Guide system in nine procedures (31%) for nine patients. Using the modified BrainLab fixation arm for frameless stereotactic navigation, 36 (56.3%) catheters were inserted in 16 (55.2%) separate CP surgeries for 13 patients. Five catheters (7.8%) were inserted free-hand with the aid of a navigation system in four (13.8%) operations. One patient was operated using both of the systems at different times for different clinical trials, and three patients underwent more than one CP procedure. A single pass of the ventricular catheter was sufficient for placement in all cases, but postoperative CT scan adjustments were subsequently needed following six CP surgeries (20.7%) and involved eight catheters (12.5%). The adjustment involved modification of the catheter depth by pulling the catheter out in all cases by 5–15 mm, depending on the required correction. Three of these eight catheters had been placed using the Navigus Trajectory Guide system (13% of the total number of catheters placed with that system) and the other five catheters had been placed using the modified BrainLab fixation arm (13.9% of the total number of catheters placed with that device). All catheters were used for the delivery of local treatment. Of note, 5 out of 6 procedures requiring adjustment of catheter depth were associated with complications.
The complications and adverse effects for CP surgery included edema (31%), infections (6.9%), seizures (13.8%), hemorrhage (6.9%) and neurological deterioration (13.8%). Mild, asymptomatic increase of brain edema occurred following nine (31%) procedures and after the initiation of CED treatment. Seven of these nine patients had three catheters and the other two patients had two catheters.
Postoperative meningitis developed after two (6.9%) operations. Rhinorrhea developed following tumor resection surgery and bacterial meningitis was diagnosed one week later in one patient (number 11, Tables 1, 2). The other patient (number 18, Tables 1, 2) developed a CSF leak after catheter removal, and this was followed by severe bacterial meningitis. There was a new onset of seizures in two patients and a worsening of preexisting seizures in two other patients following four (13.8%) procedures.
No hemorrhage was observed following CP surgery. Post-catheter removal CT scans demonstrated hemorrhages in two cases (6.9%). One was an asymptomatic minimal hemorrhage that was noted on the CT scan of a patient who had three CED catheters. The other patient had significant hemorrhage in all three tracts, leading to severe neurological deterioration and an additional systemic complication [pulmonary embolism (PE)], which was eventually fatal. Although the coagulation profile was within the normal range the clinical presentation of hemorrhage and PE may suggest an occult coagulopathy.
Significant neurological deterioration, defined as a decrease in the KPS of more than 20 or permanent neurological deficit, occurred following four CP surgeries (13.8%), including one patient who died within 24 days postoperatively. Mild and reversible neurological deterioration (e.g., transient hemiparesis and dysarthria) occurred following eight surgeries (27.6%). We observed a neurological deterioration—significant as well as mild and reversible in eight of the thirteen CP procedures (61.5%) where the lesion was located in or near eloquent brain areas. Whereas, only four CP procedures (25%) were associated with neurological deterioration when the lesion was located away from eloquent brain areas. Table 2 summarizes all the surgeries and related complications.
Technical difficulties were also encountered while planning and performing CP surgery. Following the resection of superficial frontal and parietal lesions or mesial temporal lobe lesions, it became difficult to adhere to the CP guidelines due to the vicinity of the lesion or its resection cavity to CSF spaces. In addition, the proximity of lesions to eloquent cortical areas would have led to suboptimal placement of the catheters if the CP guidelines were to be followed. We also found that insertion of intra-tumoral catheters into tumors with firm consistency interfered with the planned trajectory and led to bending or deviation of the catheter from its planned trajectory. Trajectory planning through a craniotomy with underlying artificial dura posed another obstacle for catheter insertion due to the inability to penetrate that dura with a monopolar probe. In addition, steep insertion angles led to deflection and bending of catheters on the cortical pial surface. As for technical limitations related to stereotactic instruments, we found that real-time modification of catheter insertion was limited (trajectory angles smaller than 25°) while using the Navigus Trajectory Guide system, and that this should be taken into consideration during the planning process.
Discussion
This study includes the largest reported experience with CED CP surgery for HGGs from a single site that focuses on practical and technical issues that should be considered in planning future clinical trials involving CED. CED has been studied in preclinical and clinical settings for more than a decade. Phase I/II and III studies using CED have recently been conducted [2, 6, 11–17, 20, 23], but there are no publications describing CP surgical procedures and associated complications. The current study focused on these procedure-related complications and the results indicated that the morbidity associated with CP for the purpose of CED in HGG patients is acceptable and consists mostly of reversible neurological sequelae.
Not surprisingly, we found a strong associated risk of neurological deterioration following catheter insertion in lesions near or in the eloquent cortical areas. The observed edema and seizures were not directly related to the surgical procedure since these events occurred after the infusion treatment had been started. Edema occurred much more frequently following the insertion of three catheters compared to the insertion of two catheters (63.6 vs. 15.4%, respectively). None of the patients treated with one catheter per procedure developed brain edema.
A limitation of this study is our inability to definitively identify which of the two sequential procedures (tumor resection surgery and CP surgery) gave rise to the described complication. However, the two procedures were separated in time by 48–72 h and in all cases the patient baseline characteristics used for comparison were recorded immediately prior to the CP surgery.
Optimal planning was restricted by regional factors (eloquent areas, superficial tumor, vicinity to resection cavity, CSF space or small volume residual brain tissue). Factors that interfered with placement included steep insertion angle (in particular when the Navigus Trajectory Guide system was used), the use of artificial dura and high tissue density tumors. In addition, adherence to CP recommendations[22] was not always technically possible. We recommend that the limitations we found and describe herein should be taken into consideration during the planning process and the insertion of presently available catheters. Currently there are a number of modified catheter prototypes in development to overcome the existing limitations of CED therapeutic modality (personal communication). More comprehensive data collected from a larger patient population will provide a better risk–benefit assessment and will help in guiding treatment recommendations and patient selection.
References
Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, Lang FF, McCutcheon IE, Hassenbusch SJ, Holland E, Hess K, Michael C, Miller D, Sawaya R (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95:190–198
Chamberlain MC (2006) Treatment options for glioblastoma. Neurosurg Focus 20:E2
Brem H, Mahaley MS Jr, Vick NA, Black KL, Schold SC Jr, Burger PC, Friedman AH, Ciric IS, Eller TW, Cozzens JW et al (1991) Interstitial chemotherapy with drug polymer implants for the treatment of recurrent gliomas. J Neurosurg 74:441–446
Bouvier G, Penn RD, Kroin JS, Beique R, Guerard MJ (1987) Direct delivery of medication into a brain tumor through multiple chronically implanted catheters. Neurosurgery 20:286–291
Bobo RH, Laske DW, Akbasak A, Morrison PF, Dedrick RL, Oldfield EH (1994) Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci USA 91:2076–2080
Ferguson S, Lesniak MS (2007) Convection enhanced drug delivery of novel therapeutic agents to malignant brain tumors. Curr Drug Deliv 4:169–180
Laske DW, Morrison PF, Lieberman DM, Corthesy ME, Reynolds JC, Stewart-Henney PA, Koong SS, Cummins A, Paik CH, Oldfield EH (1997) Chronic interstitial infusion of protein to primate brain: determination of drug distribution and clearance with single-photon emission computerized tomography imaging. J Neurosurg 87:586–594
Chen MY, Lonser RR, Morrison PF, Governale LS, Oldfield EH (1999) Variables affecting convection-enhanced delivery to the striatum: a systematic examination of rate of infusion, cannula size, infusate concentration, and tissue-cannula sealing time. J Neurosurg 90:315–320
Lieberman DM, Laske DW, Morrison PF, Bankiewicz KS, Oldfield EH (1995) Convection-enhanced distribution of large molecules in gray matter during interstitial drug infusion. J Neurosurg 82:1021–1029
Morrison PF, Laske DW, Bobo H, Oldfield EH, Dedrick RL (1994) High-flow microinfusion: tissue penetration and pharmacodynamics. Am J Physiol 266:R292–R305
Debinski W (2002) Local treatment of brain tumors with targeted chimera cytotoxic proteins. Cancer Invest 20:801–809
Kawakami M, Kawakami K, Puri RK (2003) Interleukin-4-Pseudomonas exotoxin chimeric fusion protein for malignant glioma therapy. J Neurooncol 65:15–25
Kioi M, Kawakami K, Puri RK (2004) Analysis of antitumor activity of an interleukin-13 (IL-13) receptor-targeted cytotoxin composed of IL-13 antagonist and Pseudomonas exotoxin. Clin Cancer Res 10:6231–6238
Kunwar S, Prados MD, Chang SM, Berger MS, Lang FF, Piepmeier JM, Sampson JH, Ram Z, Gutin PH, Gibbons RD, Aldape KD, Croteau DJ, Sherman JW, Puri RK (2007) Direct intracerebral delivery of cintredekin besudotox (IL13-PE38QQR) in recurrent malignant glioma: a report by the Cintredekin Besudotox Intraparenchymal Study Group. J Clin Oncol 25:837–844
Laske DW, Youle RJ, Oldfield EH (1997) Tumor regression with regional distribution of the targeted toxin TF-CRM107 in patients with malignant brain tumors. Nat Med 3:1362–1368
Weaver M, Laske DW (2003) Transferrin receptor ligand-targeted toxin conjugate (Tf-CRM107) for therapy of malignant gliomas. J Neurooncol 65:3–13
Lidar Z, Mardor Y, Jonas T, Pfeffer R, Faibel M, Nass D, Hadani M, Ram Z (2004) Convection-enhanced delivery of paclitaxel for the treatment of recurrent malignant glioma: a phase I/II clinical study. J Neurosurg 100:472–479
Mardor Y, Roth Y, Lidar Z, Jonas T, Pfeffer R, Maier SE, Faibel M, Nass D, Hadani M, Orenstein A, Cohen JS, Ram Z (2001) Monitoring response to convection-enhanced taxol delivery in brain tumor patients using diffusion-weighted magnetic resonance imaging. Cancer Res 61:4971–4973
Verwijnen SM, Sillevis Smith PA, Hoeben RC, Rabelink MJ, Wiebe L, Curiel DT, Hemminki A, Krenning EP, de Jong M (2004) Molecular imaging and treatment of malignant gliomas following adenoviral transfer of the herpes simplex virus-thymidine kinase gene and the somatostatin receptor subtype 2 gene. Cancer Biother Radiopharm 19:111–120
Patel SJ, Shapiro WR, Laske DW, Jensen RL, Asher AL, Wessels BW, Carpenter SP, Shan JS (2005) Safety and feasibility of convection-enhanced delivery of Cotara for the treatment of malignant glioma: initial experience in 51 patients. Neurosurgery 56:1243–1252; discussion 1252–1243
Sampson JH, Archer G, Pedain C, Wembacher-Schroder E, Westphal M, Kunwar S, Vogelbaum MA, Coan A, Herndon JE, Raghavan R, Brady ML, Reardon DA, Friedman AH, Friedman HS, Rodriguez-Ponce MI, Chang SM, Mittermeyer S, Croteau D, Puri RK (2010) Poor drug distribution as a possible explanation for the results of the PRECISE trial. J Neurosurg 113:301–309
Sampson JH, Brady ML, Petry NA, Croteau D, Friedman AH, Friedman HS, Wong T, Bigner DD, Pastan I, Puri RK, Pedain C (2007) Intracerebral infusate distribution by convection-enhanced delivery in humans with malignant gliomas: descriptive effects of target anatomy and catheter positioning. Neurosurgery 60:ONS89-98; discussion ONS98-89
Degen JW, Walbridge S, Vortmeyer AO, Oldfield EH, Lonser RR (2003) Safety and efficacy of convection-enhanced delivery of gemcitabine or carboplatin in a malignant glioma model in rats. J Neurosurg 99:893–898
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shahar, T., Ram, Z. & Kanner, A.A. Convection-enhanced delivery catheter placements for high-grade gliomas: complications and pitfalls. J Neurooncol 107, 373–378 (2012). https://doi.org/10.1007/s11060-011-0751-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0751-x