Abstract
Plants play an important role in the fate of nanoparticles in the environment through their uptake, bioaccumulation, and transfer to trophic chains. However, the impacts of nanoparticles on plants as essential components of all ecosystems are not well documented. In the present study, the toxic effects of l-Cysteine-capped CdS nanoparticles on Spirodela polyrrhiza as an aquatic higher plant species were studied. l-Cysteine-capped CdS nanoparticles were synthesized using hydrothermal method and their characteristics were determined by XRD, SEM, HR-TEM, and FT-IR techniques. The diameter of majority of synthesized nanoparticles was about 15–20 nm. Subsequently, the uptake of l-Cysteine-capped CdS nanoparticles by the plant species was confirmed using epifluorescence microscopy. The activity of peroxidase and superoxide dismutase as antioxidant enzymes was assayed and the relative frond number was calculated in the presence of different concentrations of l-Cysteine-capped CdS nanoparticles. The obtained results revealed the toxic effects of the synthesized nanoparticles on S. polyrrhiza, leading to growth reduction and significant changes in antioxidant enzymes’ activity.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nanoparticles can be defined as atomic or molecular aggregates with at least one dimension between 1 and 100 nm (Ball 2002; Khataee and Kasiri 2011; Roco 2003). These nanoscale particles own particular physical and chemical characteristics than their bulk materials (Gubbins et al. 2011; Niemeyer 2001; Syed et al. 2013). Such properties include very large specific surface area and high surface energy (Christian et al. 2008). Nanomaterials have been the subject of high interest owing to their potential applications in industrial, biomedical, and electronic applications (Philip 2010).
Cadmium sulfide (CdS) is a type of semiconductor with the narrow band gap of 2.5 eV, and its valence electron can be easily evoked to conduction band under visible light radiance. CdS semiconductor nanoparticles are of high interest owing to their unique photophysicochemical characteristics (Jiang et al. 2009). Two main applications of CdS nanoparticles are known for optoelectronic and biological purposes. Light-emitting diodes, solar cells, photodiodes, nonlinear optics, and heterogeneous photocatalysis are examples of optoelectronic applications of CdS nanoparticles (Prabhu and Khadar 2005). Furthermore, fluorescent labeling of biomolecules such as cellular proteins is one of the biological applications of CdS nanoparticles (Jamieson et al. 2007). In addition, l-Cysteine-capped CdS nanoparticles are utilized as fluorescent probe for the detection of some ions such as mercury (II) ion (Cai et al. 2006) and silver ion (Chen and Zhu 2005) in aqueous solutions. The large scale production and wide application of nanoparticles may eventually lead to increasing their end up in the environment (Khataee et al. 2011; Nowack and Bucheli 2007). Even at low concentrations that are environmentally correlated, CdS nanoparticles are still able to induce significant toxic effects in the locomotion and biochemical activities of organisms. The unique properties of nanoparticles, including size, large specific surface area, reactivity, and shape enable them to enter organisms and transport through tissues, cells, and even into cell organelles in ways that larger particles may not do (Kovochich et al. 2007). However, published ecotoxicity studies of CdS nanoparticles are still few and mostly focus on the therapeutically used nanoparticles and their effects on human health (El-Ansary and Al-Daihan 2009; Pujalte et al. 2011). One of the few studies on ecotoxicity of CdS nanoparticles demonstrates the effects on photosynthetic microorganisms (Brayner et al. 2011).
Plants are an essential component of all ecosystems and play a crucial role in the fate of nanoparticles in the environment via uptake and bioaccumulation. Therefore, plants are considered as organisms that can be directly affected by nanoparticles (Monica and Cremonini 2009). Study of phytotoxic effects of nanoparticles was accomplished on crop plants in many cases to assess their risks on human. However, few studies have been reported on aquatic higher plants such as S. polyrrhiza (Lemnaceae). S. polyrrhiza is a perennial plant usually growing in dense colonies, forming a mat on the water surface. It is an ideal system for biofuels, bioremediation, and carbon cycling owing to its aspects of fast growing, direct contact with media, and smallest genome size. Small size, simple structure, and ease of culture of S. polyrrhiza allow its use as a model plant species for toxicological research (Xing et al. 2010). Investigating the toxicity of nanoparticles on this plant as an indicator may reveal the probable toxic effect of nanoparticles on other organisms.
In the present study, l-Cysteine-capped CdS nanoparticles were synthesized and their potential uptake and adverse effects on S. polyrrhiza were investigated. Although toxic effects of CdS nanoparticles had been previously reported on animals (Hossain and Mukherjee 2013; Li et al. 2011; Ma-Hock et al. 2012), few studies were conducted on plants (Mishra et al. 2013). l-Cysteine is a water-soluble amino acid which is also a stable and nontoxic compound. In addition to its structural role in proteins, l-Cysteine is the metabolic precursor of essential biomolecules including vitamins, cofactors, antioxidants, and several defense compounds in plants (Alvarez et al. 2012). The surface modification of CdS nanoparticles by l-Cysteine reduces their precipitation in plant culture media (Kortan et al. 1990). The mercapto group of l-Cysteine can be bound to the sulfur atom of CdS nanoparticles, thus acting as a stabilizer for nanoparticles. Moreover, the polar carboxylic acid group of l-Cysteine helps nanoparticles to keep their colloidal state for a long time (Koneswaran and Narayanaswamy 2009).
According to the previous studies, nanomaterials can produce reactive oxygen species (ROS) that lead to the oxidative stress (Klaine et al. 2008; Santos et al. 2013; Tedesco et al. 2010). Therefore, changes of antioxidant enzymes’ activity such as peroxidase (POD, EC 1.11.1.7) and superoxide dismutase (SOD, EC 1.15.1.1) have been extensively utilized as the biomarker of oxidative stress (Morelli et al. 2012; Pinto et al. 2003). Hence, another aspect of the present study is to assay the activity of peroxidase and superoxide dismutase after treatment of plants with l-Cysteine-capped CdS nanoparticles.
The principal aims of the present study were, therefore, to determine the possible effect of exposure to l-Cysteine-capped CdS nanoparticles of different concentrations on the growth of Spirodela polyrrhiza and investigate the possibility of l-Cysteine-capped CdS nanoparticles uptake by the treated plants.
Materials and methods
Plant growth
The plant materials were collected from Anzali Lagoon in Northern Iran (Fig. 1). The nutrient solution utilized for S. polyrrhiza growth contained 0.099 mM CaCl2, 0.0002 mM CoCl2.6H2O, 0.0005 mM CuCl2, 0.021 mM FeSO4·7H2O, 0.092 mM H3BO3, 0.155 mM K2HPO4, 0.097 mM K2SO4, 0.367 mM KH2PO4, 1.997 mM KNO3, 0.198 mM MgSO4·7H2O, 0.012 mM MnCl2·2H2O, 0.019 mM Na2-EDTA·2H2O, and 0.00003 mM (NH4)6Mo7O24. The pH of medium was 6.5 ± 0.5 (Dosnon-Olette et al. 2010).
Synthesis of l-Cysteine-capped CdS nanoparticles
The hydrothermal synthesis method was used for the synthesis of l-Cysteine-capped CdS nanoparticles (Fig. 2). To explain briefly, 14 mL of distilled water was added into 250 mL round bottom flask; afterward, 0.175 mmol of Cd(CH3COO)2·2H2O and 0.289 mmol of Na2S were added into flask. Subsequently, 0.395 mmol of l-Cysteine was solubilized in 20 mL of distilled water and added to the solution and then the pH value was adjusted to 10 using 1 M NaOH. The solution was purged with nitrogen bubbling for 30 min and transferred into a 100-mL Teflon-lined autoclave. The autoclave was preserved at 150 °C for 6 h and then cooled down to room temperature.
Nanoparticles characterization
Structural characterization of l-Cysteine-capped CdS nanoparticles was determined utilizing XRD, SEM, HR-TEM, and FT-IR techniques. X-ray powder diffraction (XRD) spectrum of l-Cysteine-capped CdS nanoparticles was recorded at room temperature by utilizing Siemens X-ray diffractometer D5000 (Germany), with Cu Kα radiation (λ = 1.54065 A°). The scanning electron microscope (SEM) (Hitachi S-4200, Japan) was used for the morphologic study of the nanoparticles. The High-resolution transmission electron microscopy (HR-TEM) images and the selected area electron diffraction (SAED) patterns of synthesized nanoparticles were taken by a Cs-corrected high-resolution TEM (JEM-2200FS, JEOL, Japan) operating at 200 kV. Fourier-transform infra-red (FT-IR) spectra were recorded using a Bruker IR-spectrometer (Tensor 27, Bruker, Germany).
Treatment conditions for growth rate determination and antioxidant enzymes activity assay
In order to investigate the effect of l-Cysteine-capped CdS nanoparticles on relative frond number (RFN), a series of nanoparticle concentrations (1, 1.5, 2, and 2.5 mg/L) and control samples (without nanoparticle) containing equal proportions of nutrient solution were prepared. During 8 days, the number of healthy fronds was counted by the stereomicroscope (Olympus, Japan), every 2 days. Relative frond number was calculated through Eq. (1) (Khataee et al. 2013; Mitsou et al. 2006).
where N0 and N1 are the number of fronds on day 0 and day n, respectively. Each experiment was conducted in four replicates and mean values of data were reported. Since standard deviations did not exceed ± 1.6 %, the error bars are not shown in Fig. 9.
For antioxidant enzymes activity assay, a series of nanoparticle concentrations (2 and 4 mg/L) and control samples containing equal proportions of nutrient solution were prepared. The plants were exposed to concentrations of 2 and 4 mg/L of nanoparticles and the activities of enzymes were assayed on days 1, 2, and 4, after treatment. Based on the study of the effect of nanoparticles on growth of S. polyrrhiza, we decided to perform enzymes activity assay at these two concentrations. Actually, the plants died at concentrations up to 4 mg/L at the period of 4 days. Moreover, enzymes activities were not significantly different between concentrations that the plant could tolerate them.
Epifluorescence microscopy
The localization of the l-Cysteine-capped CdS nanoparticles in the treated plants was studied by means of epifluorescence microscopy techniques. Collection of the treated and control plants was performed 4 days after the beginning of the treatment, and 10 well-developed roots were randomly selected. Plant fresh materials were stained with the aqueous solution of Auramine O (0.1 % in 100 mL water). The samples were viewed through an Olympus BX51 (Olympus optical Co., Ltd. Tokyo, Japan) research fluorescence microscope equipped with the catadioptric objectives UMPlanFL-BDP and the BX-RFA (Olympus optical Co., Ltd. Tokyo, Japan) fluorescence illuminator. The best fluorescence excitation was observed when U-MWB3 (480–510 nm) and U-MWG3 (510–550 nm) mirror cube units were used. To capture all desired structures with the extended depth of the focal field, an ordered series of images from successive focal plates (with 5 μm increment per focal step) were taken by means of an Evolution MP cooled CCD (Media Cybernetics, USA) as described by Movafeghi et al. (2010). Depth of field was improved using the stack z-projection to produce the final superimposed images.
Enzyme activity analysis
Antioxidant enzymes activity assay was performed in four replicates and 0.25 gram of plant material was used for each of them. The extraction buffer contained 0.01 M potassium phosphate (K2HPO4/KH2PO4) buffer (pH 7) and 0.2 % polyvinylpyrrolidone. The homogenates were centrifuged at 4,500 rpm for 20 min and the supernatant was utilized to determine enzymatic activities and protein content. SOD and POD activities were computed per milligram of protein. The protein content in the plant extract was measured according to Bradford procedure (Bradford 1976) and bovine serum albumin was utilized as the standard.
SOD activity was determined via assessing the ability of enzyme in inhibiting nitrobluetetrazolium (NBT) photoreduction (Winterbourn et al. 1976). There were 67 mM potassium phosphate buffer (pH 7.8), 0.12 mM riboflavin, 1.5 mM NBT, and 50 µL enzyme extract in the reaction mixture. Absorbance of the commixture was assessed at the wavelength of 560 nm. One unit of SOD was hypothesized as the amount of enzyme needed for the inhibition of NBT photoreduction by 50 %.
POD activity was assayed according to the procedure of Chance and Maehly (1955). There were 1 M citrate–phosphate–borate buffer (pH = 7), 3.3 mM H2O2, 15 mM guaiacol, and 25 µL enzyme extract in the reaction mixture. The increase in absorbance was recorded at the wavelength of 470 nm for 3 min. One unit of POD activity was hypothesized as the amount of enzyme needed to produce 1 µmol/L tetraguaiacol min−1 (ε = 26.6 mM−1 cm−1).
Statistical analysis
Statistical analysis was performed using one-way analysis of variance (ANOVA) with Tukey–Kramer multiple comparisons test and utilizing GraphPad InStat 3 software package. Experiments were done in four replicates (n = 4) and degrees of freedom between groups and within groups were 2 and 9, respectively. P values less than 0.05 were considered as significant.
Results and discussion
Structural characterization of l-Cysteine-capped CdS nanoparticles
The XRD pattern of l-Cysteine-capped CdS nanoparticles is shown in Fig. 3. The peaks in the XRD spectrum of l-Cysteine-capped CdS nanoparticles corresponded to (100), (002), (101), (102), (110), (103), and (112) planes of hexagonal CdS phase (Wang et al. 2000). According to Debye–Scherrer formula, the average crystalline size of synthesized l-Cysteine-capped CdS nanoparticles was 3 nm. Figure 4a and b shows SEM images of l-Cysteine-capped CdS nanoparticles. Accordingly, l-Cysteine-capped CdS nanoparticles were spherical in shape and the diameter of majority of them was about 15–20 nm (Fig. 5). The HR-TEM image of l-Cysteine-capped CdS nanoparticles (Fig. 6 a) confirmed the results obtained by SEM. The SAED pattern (Fig. 6 b) indicated the crystallinity and small size of l-Cysteine-capped CdS nanoparticles in the range of quantum dots. The FT-IR spectra of free l-Cysteine and l-Cysteine-capped CdS nanoparticles, as shown in Fig. 7, confirmed the existence of l-Cysteine on the surface of CdS nanoparticles. The peaks at 1,550–1,600 cm−1 (s- COO−), 1,400 cm−1 (m- COO−), and 3,500–3,000 cm−1 (m- OH, COOH) represented the COO− group. The FT-IR absorption band at 2,900–3,420 cm−1 (m- N–H) indicated –NH2 group. The peak of -SH group was at the 2,550 cm−1 that was present only in the FT-IR spectrum of l-Cysteine. The formation of covalent bonds between -SH groups of l-Cysteine and the surface of CdS was the reason for the absence of -SH group vibration on CdS nanoparticles.
Microscopic evidence for the uptake of l-Cysteine-capped CdS nanoparticles
The high uptake of l-Cysteine-capped CdS nanoparticles by the roots of treated S. polyrrhiza plants was confirmed by epifluorescence microscopy (Fig. 8). Figure 8b indicates the remarkable presence of l-Cysteine-capped CdS nanoparticles inside the root tissues. Interestingly, aggregates of nanoparticle appeared as optically dense signals under fluorescence microscope. The nanoparticle aggregates were detected as particles with different sizes in intracellular spaces. Nanoparticles may enter roots of plants via osmotic pressure, capillary forces, pores in cell walls and intercellular plasmodesmata, or via the highly regulated symplastic route (Nowack and Bucheli 2007). The properties of nanoparticles, such as size, shape, composition, and surface charge, may affect the uptake and translocation inside plant (Jia et al. 2005; Zhu et al. 2009).
The effect of l-Cysteine-capped CdS nanoparticles on the growth of S. polyrrhiza
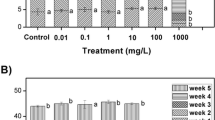
Relative frond number was used to study the effect of l-Cysteine-capped CdS nanoparticles on the growth of S. polyrrhiza. Eight days after exposing the plants to l-Cysteine-capped CdS nanoparticles, RFN was reduced by increasing the nanoparticles concentration in comparison to control (Fig. 9). Reduction in RFN by increasing the concentration of l-Cysteine-capped CdS nanoparticles confirmed the inhibitory effect of the nanoparticles on S. polyrrhiza growth. On the basis of the results obtained, l-Cysteine-capped CdS nanoparticles were toxic for S. polyrrhiza at the concentrations up to 1 mg/L. This finding is in agreement with that of Jiang et al. (2012), thereby demonstrating the phytotoxicity of silver nanoparticles on S. polyrrhiza.
Cadmium (Cd) as a nonessential toxic heavy metal can have considerable effects on higher plants. Although not essential for plant growth, Cd2+ ions are readily taken up by roots and translocated into the leaves in many plants, depressing growth by affecting photosynthesis, chlorophyll fluorescence, and nutrient uptake by plants (Mishra and Tripathi 2008). Using 2.0 mg/L of Cd, the aquatic plant Pistia stratiotes indicated the highest accumulation of it as 0.39 g/kg (Mishra and Tripathi 2008). Based on the studies on the toxicity of Cd on S. polyrrhiza, this plant appears to be a phytoremediator species in aquatic environments moderately contaminated with Cd owning to its capacity for metal accumulation coupled with its defense potential (Mishra and Tripathi 2008; Rai et al. 1995; Seth et al. 2007). However, in our study, the results indicate the significant negative effect of l-Cysteine-capped CdS nanoparticles on growth of S. polyrrhiza. The toxicity of l-Cysteine-capped cadmium sulfide nanoparticles on S. polyrrhiza is possibly due to the presence of either nanoparticles or their aggregates in plant tissues.
The effect of l-Cysteine-capped CdS nanoparticles on the activity of antioxidant enzymes
The effect of l-Cysteine-capped CdS nanoparticles on SOD and POD activities in S. polyrrhiza plant after 1, 2, and 4 days treatment is presented in Figs. 10 and 11, respectively. The SOD activity was significantly decreased (p < 0.05), whereas the POD activity was notably increased (p < 0.05) in the plants treated with nanoparticles compared to the control sample. However, the activities of SOD and POD are not significantly different between two different nanoparticle concentrations. Probably, the oxidative stress by these concentrations of NPs is not sufficient to change the activity of SOD and POD.
SOD activity in control Spirodela polyrrhiza plants and the plants exposed to 2 and 4 mg/L l-Cysteine-capped CdS NPs. The SOD activity was significantly decreased in the plants treated with NPs compared to the control sample. a and b indicate significant difference at p < 0.05. Bars show average of four replicate measurements per treatment; vertical lines show the standard deviation (SD)
POD activity in control Spirodela polyrrhiza plants and the plants exposed to 2 and 4 mg/L l-Cysteine-capped CdS NPs. The POD activity was notably increased in the plants treated with NPs compared to the control sample. Statistics as for Fig. 10
It is known that SOD acts as a key antioxidant enzyme in higher plants and provides protection for cellular components against ROS (Alscher et al. 2002). SOD converts superoxide radicals to H2O2 and this product is detoxified by other antioxidant enzymes including Catalase (CAT) and POD (Khataee et al. 2012). Reduction in SOD activity has been used as a sign for free radical scavenging ability, thereby indicating that the antioxidant defense system is suppressed by ROS (Van der Oost et al. 2003). In a study on the physiological effects of anatase TiO2 nanoparticles on one species of duckweed, the decrease in SOD activity in concentrations up to 200 mg/L was observed (Song et al. 2012). Also, Wen-wen et al. (2010) reported the reduced activity of SOD in HeLa cells exposed to Fe3O4 nanoparticles. Therefore, the decrease in SOD activity in the present study could be a consequence of the high production of superoxide radicals which led to the repression of antioxidant defense system.
Peroxidases convert hydrogen peroxide (H2O2) to H2O and O2 using a variety of organic and inorganic compounds (Havaux 2003; Vafaei et al. 2013). H2O2 produced by SOD activity can be converted into water and oxygen by CAT and POD. Therefore, peroxidase activity increment can be due to the conversion of some generated H2O2 from superoxide dismutase activity and H2O2 created directly in the presence of nanoparticles to water and oxygen.
Conclusions
The present work provided insights on the uptake and toxicity of l-Cysteine-capped CdS nanoparticles in an aquatic higher plant. Epifluorescence microscopic images confirmed the high uptake of l-Cysteine-capped CdS nanoparticles by S. polyrrhiza. The toxicity of l-Cysteine-capped CdS nanoparticles on S. polyrrhiza was established by changes in antioxidant enzymes’ (POD and SOD) activity and the decrease in plant growth. S. polyrrhiza was susceptible to concentrations up to 1 mg/L of synthesized nanoparticles. It can be concluded that such studies may be very useful for the assessment of potential environmental risks posed by the newly synthesized nanomaterials.
References
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341
Alvarez C, Angeles Bermudez M, Romero LC, Gotor C, Garcia I (2012) Cysteine homeostasis plays an essential role in plant immunity. New Phytol 193:165–177
Ball P (2002) Natural strategies for the molecular engineer. Nanotechnology 13:15–28
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brayner R, Dahoumane SA, Nguyen JN-L, Yepremian C, Djediat C, Coute A, Fievet F (2011) Ecotoxicological studies of CdS nanoparticles on photosynthetic microorganisms. J Nanosci Nanotechnol 11:1852–1858
Cai Z-X, Yang H, Zhang Y, Yan X-P (2006) Preparation, characterization and evaluation of water-soluble l-cysteine-capped-CdS nanoparticles as fluorescence probe for detection of Hg(II) in aqueous solution. Anal Chim Acta 559:234–239
Chance B, Maehly A (1955) Assay of catalases and peroxidases. Methods Enzymol 2:764–775
Chen J-L, Zhu C-Q (2005) Functionalized cadmium sulfide quantum dots as fluorescence probe for silver ion determination. Anal Chim Acta 546:147–153
Christian P, Von der Kammer F, Baalousha M, Hofmann T (2008) Nanoparticles: structure, properties, preparation and behaviour in environmental media. Ecotoxicology 17:326–343
Dosnon-Olette R, Couderchet M, El Arfaoui A, Sayen S, Eullaffroy P (2010) Influence of initial pesticide concentrations and plant population density on dimethomorph toxicity and removal by two duckweed species. Sci Total Environ 408:2254–2259
El-Ansary A, Al-Daihan S (2009) On the toxicity of therapeutically used nanoparticles: an overview. J Toxicol. doi:10.1155/2009/754810
Gubbins EJ, Batty LC, Lead JR (2011) Phytotoxicity of silver nanoparticles to Lemna minor L. Environ Pollut 159:1551–1559
Havaux M (2003) Spontaneous and thermoinduced photon emission: new methods to detect and quantify oxidative stress in plants. Trends Plant Sci 8:409–413
Hossain ST, Mukherjee SK (2013) Toxicity of cadmium sulfide (CdS) nanoparticles against Escherichia coli and HeLa cells. J Hazard Mater 260:1073–1082
Jamieson T, Bakhshi R, Petrova D, Pocock R, Imani M, Seifalian AM (2007) Biological applications of quantum dots. Biomaterials 28:4717–4732
Jia G, Wang H, Yan L, Wang X, Pei R, Yan T, Zhao Y, Guo X (2005) Cytotoxicity of carbon nanomaterials: single-wall nanotube, multi-wall nanotube, and fullerene. Environ Sci Technol 39:1378–1383
Jiang R, Zhu H, Li X, Xiao L (2009) Visible light photocatalytic decolourization of CI Acid Red 66 by chitosan capped CdS composite nanoparticles. Chem Eng J 152:537–542
Jiang H-S, Li M, Chang F-Y, Li W, Yin L-Y (2012) Physiological analysis of silver nanoparticles and AgNO3 toxicity to Spirodela polyrhiza. Environ Toxicol Chem 31:1880–1886
Khataee AR, Kasiri MB (2011) Artificial neural network modeling of water and wastewater treatment processes. NOVA Science Publisher, Inc, USA
Khataee AR, Aber S, Zarei M, Sheydaei M (2011) Environmental applications of activated carbon and carbon nanotubes. NOVA Science Publisher, Inc, USA
Khataee A, Movafeghi A, Torbati S, Salehi Lisar S, Zarei M (2012) Phytoremediation potential of duckweed (Lemna minor L.) in degradation of CI Acid Blue 92: artificial neural network modeling. Ecotoxicol Environ Saf 80:291–298
Khataee A, Fathinia M, Joo S (2013) Simultaneous monitoring of photocatalysis of three pharmaceuticals by immobilized TiO2 nanoparticles: chemometric assessment, intermediates identification and ecotoxicological evaluation. Spectrochim Acta Part A 112:33–45
Klaine SJ et al (2008) Nanomaterials in the environment: behavior, fate, bioavailability, and effects. Environ Toxicol Chem 27:1825–1851
Koneswaran M, Narayanaswamy R (2009) l-Cysteine-capped ZnS quantum dots based fluorescence sensor for Cu 2+ ion. Sens Actuator B Chem 139:104–109
Kortan A, Hull R, Opila R, Bawendi M, Steigerwald M, Carroll P, Brus LE (1990) Nucleation and growth of cadmium selendie on zinc sulfide quantum crystallite seeds, and vice versa, in inverse micelle media. J Am Chem Soc 112:1327–1332
Kovochich M, Xia T, Xu J, Yeh JI, Nel AE (2007) Principles and procedures to assess nanomaterial toxicity environmental nanotechnology: applications and impacts of nanomaterials. McGraw Hill, New York, pp 205–229
Li H, Li M, Shih WY, Lelkes PI, Shih W-H (2011) Cytotoxicity tests of water soluble ZnS and CdS quantum dots. J Nanosci Nanotechnol 11:3543–3551
Ma-Hock L, Brill S, Wohlleben W, Farias P, Chaves C, Tenorio D, Fontes A, Santos B, Landsiedel R, Strauss V (2012) Short term inhalation toxicity of a liquid aerosol of CdS/Cd (OH)2 core shell quantum dots in male Wistar rats. Toxicol Lett 208:115–124
Mishra VK, Tripathi B (2008) Concurrent removal and accumulation of heavy metals by the three aquatic macrophytes. Bioresour Technol 99:7091–7097
Mishra P, Shukla VK, Yadav RS, Pandey AC (2013) Toxicity concerns of semiconducting nanostructures on aquatic plant Hydrilla verticillata. J Stress Physiol Biochem 9:287–298
Mitsou K, Koulianou A, Lambropoulou D, Pappas P, Albanis T, Lekka M (2006) Growth rate effects, responses of antioxidant enzymes and metabolic fate of the herbicide Propanil in the aquatic plant Lemna minor. Chemosphere 62:275–284
Monica RC, Cremonini R (2009) Nanoparticles and higher plants. Caryologia 62:161–165
Morelli E, Cioni P, Posarelli M, Gabellieri E (2012) Chemical stability of CdSe quantum dots in seawater and their effects on a marine microalga. Aquat Toxicol 122:153–162
Movafeghi A, Dadpour MR, Naghiloo S, Farabi S, Omidi Y (2010) Floral development in Astragalus caspicus Bieb. (Leguminosae: Papilionoideae: Galegeae). Flora 205:251–258
Niemeyer CM (2001) Nanoparticles, proteins, and nucleic acids: biotechnology meets materials science. Angew Chem Int Ed 40:4128–4158
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150:5–22
Philip D (2010) Rapid green synthesis of spherical gold nanoparticles using Mangifera indica leaf. Spectrochim Acta A 77:807–810
Pinto E, Sigaud-kutner T, Leitao MA, Okamoto OK, Morse D, Colepicolo P (2003) Heavy metal-induced oxidative stress in algai. J Phycol 39:1008–1018
Prabhu RR, Khadar MA (2005) Characterization of chemically synthesized CdS nanoparticles. Pramana 65:801–807
Pujalte I, Passagne I, Brouillaud B, Treguer M, Durand E, Ohayon-Courtes C, L’Azou B (2011) Cytotoxicity and oxidative stress induced by different metallic nanoparticles on human kidney cells. Part Fibre Toxicol 8:1–16
Rai U, Sinha S, Tripathi R, Chandra P (1995) Wastewater treatability potential of some aquatic macrophytes: removal of heavy metals. Ecol Eng 5:5–12
Roco MC (2003) Broader societal issues of nanotechnology. J Nanopart Res 5:181–189
Santos SMA, Dinis AM, Rodrigues DMF, Peixoto F, Videira RA, Jurado AS (2013) Studies on the toxicity of an aqueous suspension of C60 nanoparticles using a bacterium (gen. Bacillus) and an aquatic plant (Lemna gibba) as in vitro model systems. Aquat Toxicol 142–143:347–354
Seth CS, Chaturvedi PK, Misra V (2007) Toxic effect of arsenate and cadmium alone and in combination on giant duckweed (Spirodela polyrrhiza L.) in response to its accumulation. Environ Toxicol 22:539–549
Song G, Gao Y, Wu H, Hou W, Zhang C, Ma H (2012) Physiological effect of anatase TiO2 nanoparticles on Lemna minor. Environ Toxicol Chem 31:2147–2152
Syed A, Saraswati S, Kundu GC, Ahmad A (2013) Biological synthesis of silver nanoparticles using the fungus Humicola sp. and evaluation of their cytoxicity using normal and cancer cell lines. Spectrochim Acta A 114:144–147
Tedesco S, Doyle H, Blasco J, Redmond G, Sheehan D (2010) Oxidative stress and toxicity of gold nanoparticles in Mytilus edulis. Aquat Toxicol 100:178–186
Vafaei F, Movafeghi A, Khataee A, Zarei M, Salehi Lisar S (2013) Potential of Hydrocotyle vulgaris for phytoremediation of a textile dye: Inducing antioxidant response in roots and leaves. Ecotoxicol Environ Saf 93:128–134
Van der Oost R, Beyer J, Vermeulen NP (2003) Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environ Toxicol Pharmacol 13:57–149
Wang S, Yang S, Yang C, Li Z, Wang J, Ge W (2000) Poly (N-vinylcarbazole)(PVK) photoconductivity enhancement induced by doping with CdS nanocrystals through chemical hybridization. J Phys Chem B 104:11853–11858
Wen-wen C, Si-jia H, Chen-xi W, Qiang Z, Chuan-lu H, Juan D, Shu-mao D (2010) Cytotoxicity effects of nano-Fe3O4 on HeLa cells. In: Bioinformatics and Biomedical Engineering (iCBBE), 2010 4th International Conference on,. IEEE, pp 1–4
Winterbourn CC, McGrath BM, Carrell RW (1976) Reactions involving superoxide and normal and unstable haemoglobins. Biochem J 155:493–502
Xing W, Huang W, Liu G (2010) Effect of excess iron and copper on physiology of aquatic plant Spirodela polyrrhiza (L.) Schleid. Environ Toxicol 25:103–112
Zhu RR, Wang SL, Chao J, Shi DL, Zhang R, Sun XY, Yao SD (2009) Bio-effects of Nano-TiO2 on DNA and cellular ultrastructure with different polymorph and size. Mater Sci Eng C 29:691–696
Acknowledgments
The authors thank the University of Tabriz, Iran, for all support provided.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Khataee, A., Movafeghi, A., Nazari, F. et al. The toxic effects of l-Cysteine-capped cadmium sulfide nanoparticles on the aquatic plant Spirodela polyrrhiza . J Nanopart Res 16, 2774 (2014). https://doi.org/10.1007/s11051-014-2774-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-014-2774-7