Abstract
Different photochemical approaches have been investigated to prepare alumina supported Pd nanoparticles to be used as heterogeneous catalysts. The employed techniques were: (i) direct photodeposition, (ii) sensitized photodeposition, and (iii) photodeposition in the presence of a protecting agent (polyvinylpirrolidone). The sensitized photodeposition with acetone resulted to be the most proper technique, allowing to obtain, in very short irradiation time, Pd particles homogeneously distributed over the support surface, with a very narrow and symmetrical monomodal distribution centered at 4–5 nm, found to be appropriate for the deep oxidation of methanol.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Metal nanoparticles (NPs) have become a subject of intense interest in various fields of chemistry and physics during the past decades. Such interest stems from their unique physical and chemical properties (Doyle et al. 2003; White et al. 2009), which are different from their bulk counterparts and hence lead to novel applications in a variety of areas, such as sensors, electronics, optics, magnetics, and catalysts.
In particular in the case of applications of nanoparticles in the field of catalysis, the synthesis of metal particles with a controlled distribution is a critical factor chiefly for those catalytic reactions which are dependent on particle dimensions, the so-called structure sensitive reactions (Gates, 1992). Recently, the authors reported the preparation of supported Pt catalysts with a narrow particle size distribution through a photochemical approach, namely the liquid phase photodeposition (LPPD) technique, which allows the direct deposition of active metallic species on the support from the liquid phase at room temperature by means of quite simple and economic equipment (Crisafulli et al. 2006). LPPD involves the chromofore of a metal complex which is able to absorb the light producing a photoexcited state which may decompose, by a photoredox reaction, to give an unprotected solid metallic phase cluster able to spread over the support surface (Peled 1997; Giuffrida et al. 2005). Recently the authors also reported the preparation of supported Ag catalysts through a modified photochemical approach consisting in the photoassisted preparation of stable unprotected Ag colloids in water, followed by the deposition of Ag clusters on the support (Scirè et al. 2009).
Among all transition metals, Pd is probably one of the most versatile in catalyzing reactions, many of which are not easily achieved with other transition metals. It is one of the most efficient metals for the formation of C–C bond (Astruc 2007) in organic reaction such as Suzuchi, Heck, and Still coupling (Franzèn 2000; Li et al. 2000; Kim et al. 2002; Son et al. 2004) and for hydrogenation of polyunsaturated hydrocarbons (Ouchaib et al. 1989; Redjala et al. 2006). Palladium is known as an efficient electro-catalyst for ethanol oxidation for fuel cell applications (Gupta and Datta 2005; Liu et al. 2007) and also displays a remarkable performance in H2 storage and sensing (Favier et al. 2001; Langhammer et al. 2007). It has shown that the catalytic activity of noble metal nanostructures highly depends on the morphology of the nanoparticles (Narayanan and El-Sayed 2004). Traditionally, supported metal catalysts are typically prepared by impregnation of high-area supports with an aqueous solution of metal salt, followed by heating and reduction in hydrogen. The resultant metal particles on the support are usually heterogeneously dispersed with broad size distribution.
Herein, the authors report a photochemical study of the reduction of the Pd(acac)2 complex in alcohol in order to prepare Pd nanoparticles supported on alumina so that the authors continue to seek alternative routes to prepare supported catalysts from the liquid phase photodeposition (Crisafulli et al. 2006; Scirè et al. 2009). The main advantages of this photochemical approach are the short reaction time, the high reproducibility, the use of a clean reagent as the light as reducing agent, the possibility to work at room temperature with simple and low cost equipment, and finally it is possible to follow spectrophotometrically both the complex photoreduction during the irradiation and the catalyst washing process in order to obtain a pure catalyst. In the attempt to obtain NPs with different size, the authors performed the reaction in different conditions of irradiation namely direct and sensitized photoreduction. In the direct photoreduction, the incident light is directly absorbed from the molecules of the Pd complex, whereas in the sensitized method a molecule different from the palladium complex (in this case acetone) is involved in the light absorption process. In this latter case, the excited species of this different molecule causes the reduction of the Pd complex through a specific mechanism of electron or energy transfer. Moreover, the photoreduction in the presence of a protective agent, namely polyvinylpirrolidone (PVP), was also carried out, maintaining constant the complex concentration.
The catalytic activity of so prepared Pd catalysts was evaluated in the deep oxidation of methanol, which was chosen as representative of volatile organic compounds (VOC).
Experimental
The bis (2,4-pentandionato) Pd(II) complex, Pd(acac)2, and PVP (Aldrich) were reagent grade. Methanol, ethanol, isopropanol, and acetone (Carlo Erba) were spectrophotometric grade.
For the preparation of alumina supported Pd catalysts by LPPD, Pd(acac)2 1.5 × 10−4 M containing 0.32 g of Al2O3 (Alfa Aesar, grain size: 106–180 μm, BET surface area: 216 m2 g−1) was suspended in the specific solvent and irradiated in nitrogen atmosphere at 25 °C in a quartz vessel (200 mL) using a Rayonet Photochemical Reactor equipped with various numbers of lamps, provided by Italquartz, with monochromatic emission at either 254 or 300 nm. The light intensity, measured by a ferric oxalate actinometer (Calvert and Pitts 1966), was in the range 2.0 × 10−6–1.0 × 10−5 Nhν min−1 (5–27 mW cm−2). The kinetics of Pd(acac)2 photodegradation, with or without the protecting agent, was followed spectrophotometrically in a quartz cuvette (3 mL) with a Jasco V-560 spectrophotometer using a light intensity of 2.0 × 10−6 Nhν min−1. Preliminary experiments were performed to determine the suitable concentration of the protective agent.
Different photodeposition approaches have been used to obtain the Pd catalysts, namely (i) direct (sample coded Pd-D), where the photodeposition was carried out in the presence of the complex and the support; (ii) sensitized (sample coded Pd-S), where the photodeposition was carried out in the presence of the complex, the support and acetone as sensitizer; and (iii) Pd-protected (sample coded Pd-PVP), where the photodeposition was carried out in the presence of the complex, the support, and polyvinylpirrolidone as protecting agent. Moreover, for comparison a Pd sample was also prepared suspending the support only after that the complete palladium photoreduction occurred (sample coded Pd-R). The vessel suspension was subjected, during the irradiation, to fast and continuous stirring to avoid the sedimentation of the γ-Al2O3. The Pt(acac)2 photoreduction was followed spectrophotometrically on a clear solution obtained through centrifugation of a little amount of suspension, monitoring the disappearance of the adsorption peak at 324 nm (at this wavelength, the released ligand Hacac does not absorb). After completion of the photochemical deposition of the metal onto γ-Al2O3, the suspended material was recovered by centrifugation and washed several times with the solvent and then dried in an oven at 90 °C. All Pd samples were prepared in order to have a nominal 1 wt% Pd content. Real Pd loadings, determined by atomic adsorption spectrometry, are reported in Table 1.
Dynamic light scattering (DLS) was carried out using a LB-550 HORIBA Size Analyzer equipped with a Laser diode at 650 nm.
High resolution transmission electron microscopy (TEM) was carried out on a 200 keV JEOL JEM 2010 analytic electron microscope (LaB6 electron gun), equipped with a Gatan 794 Multi-Scan CCD camera for digital imaging. Samples for TEM were prepared by ultrasonic treatment in isopropanol for 2 min. A drop of the suspension of the sample was placed onto a carbon-coated copper micro-grid followed by evaporating off the solvent under ambient conditions. In order to obtain a good statistical particle size distribution several different areas of the grid were observed and more than 150 Pd particles measured for each sample. The average size diameter estimated by TEM was calculated using the following formula: d = Σ(n i d i )/n, where n i is the number of Pd particles of diameter d i and n is the total number of Pd particles.
Pd dispersion was measured by H2 titration (Prelazzi et al. 1999) in a pulse reactor system according to the following procedure: samples were treated in H2 at 150 °C for 1 h, maintained at this temperature in flowing Ar for 30 min, cooled at room temperature always in Ar, saturated with pulses of O2/Ar (20/80 v/v). Pulses of H2/Ar (5/95 v/v) were then introduced at room temperature through an injection valve in order to titrate the oxygen chemisorbed on Pd.
Catalytic activity tests were performed in a continuous-flow fixed-bed microreactor in the gas phase at atmospheric pressure, using 0.05 g of catalyst (80–140 mesh) diluted with an inert glass powder. The reactant mixture was fed to the reactor by flowing a part of the He stream through a saturator containing the VOC and then mixing with O2 and He before reaching the catalyst. The reactant mixture was 0.7 vol.% methanol and 10 vol.% O2, balance in helium. A space velocity (GHSV) of \( 7. 6 \times 10^{ - 3} \,{\text{mol}}\,{\text{h}}^{ - 1} \,{\text{g}}^{ - 1}_{\text{cat}} \) was always used. The effluent gases were analyzed on-line by a gas chromatograph (Thermo), equipped with a packed column with 10% FFAP on Chromosorb W and FID detector, and by a quadrupole mass spectrometer (VG quadrupoles). For all experiments, CO2 was the main carbon-containing product, only very small amounts of CO were found at low conversions. The carbon balance was always higher than 95%. Before activity tests samples were treated in H2 at 150 °C for 1 h and then calcined in air at 200 °C for 1 h. Preliminary runs carried out at different flow-rates showed the absence of external diffusional limitations. The absence of internal diffusion limitations was verified by running experiments with catalyst powders at different grain size.
Code and characteristics of Pd samples are summarized in Table 1.
Results
Kinetic of the Pd complex photoreduction
Formation of metallic Pd by direct photoreduction of alcohol Pd(acac)2 solutions
In the direct photolysis of alcohol Pd(acac)2 solutions, the incident light of 254 nm is absorbed from the Pd(acac)2 complex. Irradiation of deoxygenated isopropanol, ethanol, or methanol solutions of Pd(acac)2 at 254 nm caused similar spectral changes, consisting of the decrease of the absorption band at 329 nm, the appearance of a new band at 273 nm, typical of Hacac (λmax = 273 nm), due to a π − π* (HOMO-LUMO) transition (Nakanishi et al. 1977), and of a moderate structureless absorbance over 350 nm, attributed to the light scattering of Pd nanoparticles. It must be noted that Pd nanoparticles do not exhibit the UV–Vis plasmone band (Shon et al. 2002) due to the damping effect of d–d transitions. In Fig. 1, the spectral changes of the isopropanol solution are reported as a representative example. As the photoreaction proceeded, the solution color turned from pale yellow to brown. As shown in Fig. 1, the 329-nm band, due to the adsorption of PdII(acac)2, completely disappears after about 50 min irradiation, pointing to the complete photoreduction of the complex to Pdo. It must be underlined the Pd(acac)2 complex is unreactive upon irradiation at wavelength >260 nm, due to the presence of the low-energy metal-localized d,d singlet state (Lewis et al. 1993).
The spectral patterns were consistent with a mechanism (Scheme 1) which involves the photoreduction of PdII to Pdo and the release of the free Hacac ligand, exhibiting a maximum peak at 273 nm (Lewis et al. 1993). According to the literature (Giuffrida et al. 2004), the mechanism shows that, in the direct reaction, which competes with the reverse one, the Pd–O homolytic cleavage of PdII(acac)2 occurs, with formation of PdI(acac) and acetylacetonyl radical that abstracts one hydrogen atom from a molecule of the solvent. In the second step, the PdI intermediate undergoes fast homolytic dissociation to give Pdo and another acetylacetonyl radical which in turn gains a hydrogen atom at the expense of the solvent. The quenching of radicals causes the oxidation of the isopropanol to acetone. The same mechanism occurs with ethanol and methanol leading to formaldehyde and acetaldehyde, respectively, pointing that alcohols work both as solvent and reductant, being oxidized into aldehydes or ketones.
In order to choose the most proper alcohol leading to the smallest Pd particles, the authors evaluated the size of the Pd particles in solution through the DLS analysis, even if the authors are aware that the particle size obtained by this technique can be overestimated, in so as DLS measures the hydrodynamic diameter in solution, including the solvent, shell moving with the particle.
The DLS analysis of the Pd particles obtained with the three different alcohols showed that the size of hydrodynamic diameters is in the order: isopropanol < ethanol < methanol (Fig. 2). This finding can be attributed to the irradiation time required for each solvent, which in turn depends on the photoreduction mechanism. It have been found, in fact, that in the same conditions of precursor concentration and light intensity, the kinetic of the photoreduction in isopropanol is faster that in ethanol and methanol. This agrees with the fact that the abstraction of a hydrogen atom, which is the rate determining step, occurs more easily from a secondary carbon compared to a primary or a methanol carbon (Morrison and Boyd 1992). Really it has been reported in the literature that the metal cluster dimension depends on the ratio of the nucleation rate to the growing rate of metal particles, an higher ratio leading to smaller particles (Park et al. 2007; Kasture et al. 2008). However, it is not possible to rule out that the above reported order of Pd nanoparticles is also due to the molecular complexity of the solvent which should protects the metal clusters from their further aggregation, larger molecular size of the solvent resulting in a more effective protection. In fact, it has been reported that nanoparticles can be also stabilized by the bare solvent (Franke et al. 1996; Vidoni et al. 1999).
For the above reasons, isopropanol was chosen as solvent for the direct photoreduction of Pd(acac)2 in order to prepare supported Pd catalysts.
Formation of metallic Pd by sensitized photoreduction
As reported in the previous section, by direct irradiation at wavelengths higher than 260 nm the alcohol Pd(acac)2 solutions resulted to be photochemically inert (Lewis et al. 1995). However, the photoreduction of Pd(acac)2 to metallic Pd could be carried out at 300 nm in the presence of acetone which acts as a sensitizer absorbing the incident light thus leading to the complex reduction. The acetone is a very interesting triplet sensitizer due to its volatility which makes it suitable for several applications as, for instance, photo-CVD (Condorelli et al. 2003).
Concerning to the mechanism, it can be proposed (Scheme 2) that the key reaction step is the electron transfer (e.t.) from the palladium complex to the excited triplet state of acetone (produced by light absorption at 300 nm) with consequent formation of an excited complex (3CH3COCH3 in the Scheme 2) which can give a back electron transfer (b.e.t.) to the reagents or lead to the products (Marciniak and Buono-core 1990). The competition between these two processes determines the quantum efficiency of the sensitized photoreduction. Then, as found for aromatic ketones with triplet energy values close to that of acetone (ET = 75 kcal) (Chow et al. 1986), the sensitized photoreduction involves a charge transfer within an excited state complex, rather than a triplet energy transfer, with formation of palladium (I) species, which, in turn, can be reduced to metallic palladium through ketyl radicals or fast decomposition. Scheme 2 also points out that the presence of a hydrogen donor compound, such as isopropanol, is essential to allow the complex photoreduction. This was confirmed by the lack of reaction when only acetone was used as solvent. Therefore, a acetone–isopropanol mixture (80:20 v/v) which allows to reach the maximum photoreduction rate was used. A comparison between direct and sensitized photoreduction evidenced that the rate of photoreduction, in the sensitized process by acetone, is much higher than in the direct one. For example, the time necessary to reduce completely PdII to metallic Pdo in 1.5 × 10−4 M Pd(acac)2 solution is 1 h in the direct photoreduction and only 5 min in the acetone sensitized process, using the same light intensity (1.0 × 10−5 Nhν min−1).
Formation of metallic Pd by photoreduction of isopropanol Pd(acac)2 solutions in presence of the protective agent (PVP)
Ligand or polymers, especially solvent-soluble polymers, either natural or synthetic, with some affinity for metals are often used as stabilizers of metal nanoparticles, preventing them from aggregating through the adsorption of these molecules at the particle surfaces thus providing a protective layer (Hirai and Toshima 1986; Hunter 1987; Napper 1983).
The protective effect of polyvinylpyrrolidone (PVP) results in the stabilization of the nanoparticles by the polymer chain on the palladium particle surface via the coordination of the PVP carbonyl group to the palladium atoms. Preliminary experiments were carried out with different complex/PVP ratios in order to choose the optimal ratio. A PVP concentration of 1.5 × 10−3 M of the monomeric unit (ratio complex/PVP 1:10) was then used (Taranishi and Miyake 1998). The spectral changes observed by irradiation of isopropanol solution of PVP-Pd(acac)2 in the ratio 10:1 (Fig. 3) were similar to that found in the absence of PVP (Fig. 1) in so as the absorption of PVP in the examined UV spectral region is negligible. It must be noted that on the final sample no residual PVP was detected (FT–IR analysis) indicating that the washing procedure was effective in the removal of the polymer from the surface.
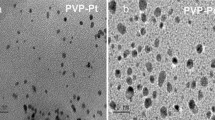
TEM analysis of supported Pd catalysts
Transmission electron microscopy was carried out in order to characterize metallic palladium nanoparticles of prepared alumina supported catalysts and the results are reported in Figs. 4, 5, 6, 7. Analysis of the spacing of the lattice fringes of 0.224 and 0.12 nm for the (111) lattice planes of palladium and alumina nanoparticles, respectively provided unambiguous discrimination of the two phases.
Figure 4 reports TEM photos, with different magnification, of the sample prepared by direct irradiation (Pd-D). The images show almost spherical Pd particles mixed with bigger and squared alumina grains. The size distribution reported in Fig. 4d shows that the diameter of the Pd particles ranges from 3 to 15 nm, with a broad and bimodal distribution with a main maximum at 6 nm and a second less intense one at 13 nm. The average size diameter estimated by TEM was around 7.6 nm. It must be noted that the Pd nanoparticles appear quite homogeneously distributed over the support surface, forming however in some cases cluster aggregates of a few units.
TEM microphotographs of the sample prepared by carrying out the photoreduction in the absence of the support (sample Pd-R) are reported in Fig. 5. It can be immediately noted that the Pd-R sample shows a significantly different morphology compared to the Pd-D sample, exhibiting extensive clustering of Pd particles, resulting in massive aggregates (around 30–50 nm) of not well distinguishable units. Due to this feature it was not possible to carry out the particle size distribution of this sample.
TEM images of the sample obtained through acetone sensitized photoreduction are reported in Fig. 6. It is possible to observe that Pd clusters appear small, well distinct from each other with rare particles aggregates, and homogeneously distributed over the entire support surface. The size distribution reported in Fig. 6d shows that the diameter of the Pd particles ranges from 2 to 9 nm, with a very narrow monomodal distribution of palladium nanoparticles, centered at 4–5 nm with an average value of 5.2 nm.
TEM images of the sample obtained through direct photoreduction in the presence of PVP as protective agent (sample coded as Pd-PVP) are reported in Fig. 7. In this case the morphology of Pd clusters appears similar to that found in the Pd-S sample. However, the distribution of Pd nanoparticles (Fig. 7d) is much broader than the Pd-S sample with particles ranging from 2 to 11 nm, with an average diameter of 5.3 nm.
Catalytic activity
The conversion of methanol as a function of the reaction temperature over investigated Pd catalysts is reported in Fig. 8. It must be noted that on Pd/γ-Al2O3 catalysts, analogously to that reported in the case of Pt/γ-Al2O3 (Crisafulli et al. 2006) the oxidation reaction proceeds directly to CO2 with no significant formation of intermediate oxidation products (formaldehyde or formic acid). It must be also underlined that on the pure alumina support no reaction was observed up to 200 °C. The figure shows that the oxidation activity of investigated samples was in the order: Pd-S > Pd-PVP > Pd-D > > Pd-R. In particular, on the most active sample (Pd-S sample), the light off curve was significantly shifted (around 55 °C) to lower temperature with respect to the less active one (Pd-R sample). In order to allow an easier comparison among various samples, the extrapolated temperatures at which the 50% conversion was reached (T50) are reported in Table 1. Data of Table 1 and Fig. 8 clearly point out that the different photochemical approach used for the preparation of Pd samples has a strong influence on their catalytic properties.
Discussion
As evidenced by TEM analysis, the different photochemical approach significantly affects the size and morphology of the obtained Pd particles over the alumina support surface. When the catalyst is prepared by carrying out the photoreduction in the absence of the support (sample Pd-R), added only successively, massive aggregates of Pd particles (30–50 nm diameter) are obtained (Fig. 5). This suggests that metallic Pd, just formed in alcohol solution by photoreduction, easily tend to aggregate. If photoreduction is instead carried out in the presence of the alumina support (Pd-D sample) the aggregation of Pd particles is somehow inhibited, resulting in smaller particles, with an average diameter of 7.6 nm. It must be underlined that on this sample some bigger particles (10–15 nm) are also present, as shown by the bimodal distribution with two maxima centered at 6 and 13 nm, respectively (Fig. 4). The above behavior is different from that observed in the case of Pt particles prepared by the same photochemical approach. In this latter case, in fact, small and stable Pt metal particles (with diameters in the range 1.6–2 nm) were obtained both in the presence and in the absence of a support (Crisafulli et al. 2006), suggesting that Pt has a lower tendency to grow compared to Pd.
When a capping agent, as PVP, is also present during the photodeposition process, the obtained Pd sample (Pd-PVP) shows even smaller particles with an average diameter of 5.3 nm (Fig. 7). It is noteworthy that in this sample the size distribution is monomodal with a relevant presence of very small particles (23% in the 2–3 nm range), pointing out that PVP has a strong protective effect against the growth of Pd particles over the support surface.
The case of photoreduction carried out in the presence of a sensitizer such as acetone is highly interesting. In this case, in fact, TEM data show that Pd particles are small and homogeneously distributed over the entire support surface, with a very narrow and symmetrical monomodal distribution centered at 4 nm with an average diameter of 5.2 nm (Fig. 6). This behavior can be explained considering that sensitized photoreduction was found to occur with a much higher rate compared to the direct process. It has been, in fact, reported that the metal cluster dimension depends on the ratio of the nucleation rate to the growing rate of metal particles, an higher ratio leading to smaller particles (Park et al. 2007; Kasture et al. 2008). In the presence of the support, these Pd metal particles spread over the support surface thus avoiding their aggregation.
The different features above observed can be taken into account to rationalize the catalytic activity of investigated samples toward the methanol combustion (Fig. 8), which was found to be in the order: Pd-S > Pd-PVP > Pd-D > > Pd-R. In order to interpret this trend, it is important to draw the attention to the fact that over supported platinum and palladium catalysts the deep oxidation of light alkanes, aromatic hydrocarbons, and oxygenated compounds is regarded as a structure sensitive reaction, involving the activation of the oxygen on the noble metal. However, conflicting results were reported in the literature on the extent of this effect as well as on which particle size of the active metal is the optimal one (Minicò et al. 2000; Baldi et al. 1998; Cordi et al. 1997; Garetto and Apesteguia 2000; Garetto and Apesteguia 2001; Radic et al. 2004).
These data suggest that catalytic activity of Pd samples can be related to the size of the obtained Pd particles. In particular, the deep oxidation activity of investigated catalysts appears to decrease as the average Pd particle size increases (Fig. 8), as also confirmed by turnover frequencies (TOF), namely the specific activity per surface Pd atom, which are reported in Table 1. This points out that methanol combustion is a structure sensitive reaction, at least under the experimental condition and in the examined range of particles. However, it is noteworthy that Pd-S and Pd-PVP samples, despite they exhibit almost the same average Pd size (about 5 nm), have a quite different catalytic behavior, with the Pd-S sample more active than the Pd-PVP. In order to explain the above behavior, the different size distribution of the Pd particles can be taken into account. In fact the Pd-S sample presents a highly symmetric monomodal distribution with a very high percentage of particles in the range 4–5 nm (57%) and a low number of particles lower than 4 nm (8%), whereas the Pd-PVP sample exhibits a larger distribution shifted toward smaller particles, which are more abundant (23% lower than 4 nm). Therefore, it can be suggested that the particle size distribution has a strong effect toward the activation of the oxygen on Pd atoms, which is the rate determining step of the oxidation reaction. On the basis of the data, it can be proposed that the presence of Pd particles with highly uniform diameter in the range 4–5 nm can provide an efficacious oxygen activation. The occurrence of an optimal Pd metal size for oxygen activation has been very recently demonstrated over Pd/TiO2 catalysts (Kaden et al. 2010). A similar behavior has been also reported in the VOC oxidation over Pt/Al2O3 catalysts (Crisafulli et al. 2006).
Conclusions
On the basis of the results reported in this article, the following conclusions can be drawn:
-
Different photochemical techniques can be used to prepare alumina supported Pd nanoparticles, namely (i) direct photoreduction, (ii) sensitized photoreduction, and (iii) photoreduction in the presence of PVP as protecting agent.
-
Among the above approaches, the sensitized photoreduction with acetone allowed to obtain, in very short irradiation time, Pd particles homogeneously distributed over the support surface, with a very narrow and symmetrical monomodal distribution centered at 4–5 nm, found to be appropriate for the deep oxidation of VOC.
References
Astruc D (2007) Palladium nanoparticles as efficient green homogeneous and heterogeneous carbon–carbon coupling precatalysts: a unifying view. Inorg Chem 46:1884–1894
Baldi M, Finocchio E, Milella F, Busca G (1998) Catalytic combustion of C3 hydrocarbons and oxygenates over Mn3O4. Appl Catal B 16:43–51
Calvert J, Pitts JN (eds) (1966) Experimental methods in photochemistry. Wiley, New York, p 783
Chow YL, Buono-Core GE, Lee CWB, Scaiano JC (1986) Sensitized photoreduction of bis(acetylacetonato)nickel(II) by triplet state aromatic ketones. J Am Chem Soc 108:7620–7627
Condorelli GG, Anastasi G, Giuffrida S, Fragalà IL (2003) In: Allendorf M, Maury F, Teyssandier F (eds) Proceedings of the CVD XVI/EUROCVD 14, vol 08. The Electrochemical Society, Pennington, pp 112–115
Cordi EM, O’Neill PJ, Falconer JL (1997) Transient oxidation of volatile organic compounds on a CuO/Al2O3 catalyst. Appl Catal B 14:23–36
Crisafulli C, Scirè S, Giuffrida S, Ventimiglia G, Lo Nigro R (2006) An investigation on the use of liquid phase photodeposition for the preparation of supported Pt catalysts. Appl Catal A 306:51–57
Doyle A, Shaikhutdinov SK, Jackson SD, Freund HJ (2003) Hydrogenation on metal surfaces: why are nanoparticles more active than single crystals? Angew Chem Int Ed 42:5240–5243
Favier F, Walter EC, Zach MP, Benter T, Penner RM (2001) Hydrogen sensors and switches from electrodeposited palladium mesowire arrays. Science 293:2227–2231
Franke R, Rothe J, Pollmann J, Hormes J, Bonnemann H, Brijoux W, Hinderburg T (1996) A study of the electronic and geometric structure of colloidal Tio 0.5THF. J Am Chem Soc 118:12090–12097
Franzèn R (2000) The Suzuki, the Heck, and the Stille reaction—three versatile methods for the introduction of new C–C bonds on solid support. Can J Chem 78:957–962
Garetto TF, Apesteguia CR (2000) Oxidative catalytic removal of hydrocarbons over Pt/Al2O3 catalysts. Catal Today 62:189–199
Garetto TF, Apesteguia CR (2001) Structure sensitivity and in situ activation of benzene combustion on Pt/Al2O3 catalysts. Appl Catal B 32:83–94
Gates BC (ed) (1992) Catalytic chemistry. Wiley, New York
Giuffrida S, Condorelli GG, Costanzo LL, Fragala IL, Ventimiglia G, Vecchio G (2004) Photochemical mechanism of the formation of nanometer-sized copper by UV irradiation of ethanol bis(2,4-pentandionato)copper(II) solutions. Chem Mater 16:1260–1266
Giuffrida S, Costanzo LL, Condorelli GG, Ventimiglia G, Fragala IL (2005) Photochemistry of bis(1,1,1,5,5,5-hexafluoro-2,4-pentanedionate)strontium tetraglyme solutions for eventual liquid phase photochemical deposition. Inorg Chim Acta 358:1873–1881
Gupta SS, Datta J (2005) Electrode kinetics of ethanol oxidation on novel CuNi alloy supported catalysts synthesized from PTFE suspension. J Power Sources 145:124–132
Hirai H, Toshima N (1986) Polymer-attached catalysts. In: Iwasawa Y (ed) Tailored metal catalysts. Reidel D, Dordrecht, pp 87–140
Hunter RJ (ed) (1987) Foundations of colloid science, vol 1. Oxford University Press, New York, p 316
Kaden WE, Kunkel WA, Kane MD, Roberts FS, Anderson SL (2010) Size-dependent oxygen activation efficiency over Pdn/TiO2(110) for the CO oxidation reaction. doi: 10.1021/ja103347v
Kasture MB, Patel P, Prabhune AA, Ramana CV, Kulkarni AA, Prasad BLV (2008) Synthesis of silver nanoparticles by sophorolipids: effect of temperature and sophorolipid structure on the size of particles. J Chem Sci 120:515–520
Kim SW, Lee WY, Hyeon T (2002) Fabrication of hollow palladium spheres and their successful application to the recyclable heterogeneous catalyst for suzuki coupling reactions. J Am Chem Soc 124:7642–7643
Langhammer C, Zoric I, Kasemo B (2007) Hydrogen storage in Pd nanodisks characterized with a novel nanoplasmonic sensing scheme. Nano Lett 7:3122–3127
Lewis FD, Salvi GD, Kanis DR, Ratner MA (1993) Electronic structure and spectroscopy of nickel(II), palladium(II), and platinum(II) acetylacetonate complexes. Inorg Chem 32:1251–1258
Lewis FD, Miller AM, Salvi GD (1995) Spectroscopy and photochemistry of nickel(II), palladium(II), and platinum(II) β-diketonates. Inorg Chem 34:3173–3181
Li Y, Hong XM, Collard DM, El-Sayed MA (2000) Suzuki cross-coupling reactions catalyzed by palladium nanoparticles in aqueous solution. Org Lett 2:2385–2388
Liu J, Ye J, Xu C, Jiang SP, Tong Y (2007) Kinetics of ethanol electrooxidation at Pd electrodeposited on Ti. Electrochem Commun 9:2334–2339
Marciniak B, Buono-core GE (1990) Quenching of triplet state benzophenone by metal(II)1,3-diketones in solution. Spectrosc Lett 23:149–160
Minicò S, Scirè S, Crisafulli C, Maggiore R, Galvagno S (2000) Catalytic combustion of volatile organic compounds on gold/iron oxide catalysts. Appl Catal B 28:245–251
Morrison RT, Boyd RN (eds) (1992) Organic chemistry, 6th edn. Prenctise Hall International, London, pp 110–111
Nakanishi H, Morita H, Nagakura S (1977) Electronic structures and spectra of the keto and enol forms of acetylacetone. Bull Chem Soc Jpn 50:2255–2261
Napper DH (ed) (1983) Polymeric stabilization of colloidal dispersions. Academic Press, London
Narayanan R, El-Sayed MA (2004) Shape-dependent catalytic activity of platinum nanoparticles in colloidal solution. Nano Lett 4:1343–1348
Ouchaib T, Massardier J, Renouprez A (1989) Competitive hydrogenation of butadiene and butene on palladium and platinum catalysts. J Catal 119:517–523
Park J, Joo J, Kwon GS, Jang Y, Hyeon T (2007) Synthesis of monodisperse spherical nanocrystals. Angew Chem Int Ed 46:4630–4660
Peled A (1997) State of the art in liquid phase photodeposition processes and applications (LPPD). Lasers Eng 6:41–79
Prelazzi G, Cerboni M, Leofanti G (1999) Comparison of H2 adsorption, O2 adsorption, H2 titration, and O2 titration on supported palladium catalysts. J Catal 181:73–79
Radic N, Grbic B, Terlecki-Baricevic A (2004) Kinetics of deep oxidation of n-hexane and toluene over Pt/Al2O3 catalysts: platinum crystallite size effect. Appl Catal B 50:153–159
Redjala T, Remita H, Apostulescu G, Mostafavi M, Thomazeau C, Uzio D (2006) Bimetallic Au–Pd and Ag–Pd clusters synthesised by or electron beam radiolysis and study of the reactivity/structure relationships in the selective hydrogenation of buta-1,3-diene. Gas Oil Sci Technol 61:789–797
Scirè S, Crisafulli C, Giuffrida S, Mazza C, Riccobene PM, Pistone A, Ventimiglia G, Buongiorno C, Spinella C (2009) Supported silver catalysts prepared by deposition in aqueous solution of Ag nanoparticles obtained through a photochemical approach. Appl Catal A 367:138–145
Shon YS, Dawson GB, Porter M, Murray RW (2002) Monolayer-protected bimetal cluster synthesis by core metal galvanic exchange reaction. Langmuir 18:3880–3885
Son SU, Jang Y, Park J, Na HB, Park HM, Yun HJ, Lee J, Hyeon T (2004) Designed synthesis of atom-economical pd/ni bimetallic nanoparticle-based catalysts for sonogashira coupling reactions. J Am Chem Soc 126:5026–5027
Taranishi T, Miyake M (1998) Size control of palladium nanoparticles and their crystal structures. Chem Mater 10:594–600
Vidoni O, Pholppot K, Amiens C, Chaudret B, Balmes O, Malm JO, Bovin JO, Senocq F, Casanove MJ (1999) Novel, spongelike ruthenium particles of controllable size stabilized only by organic solvents. Angew Chem Int Ed 38:3736–3738
White RJ, Luque R, Budarin V, Clark JH, Macquarrie DJ (2009) Supported metal nanoparticles on porous materials. Methods and applications. Chem Soc Rev 38:481–494
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Scirè, S., Giuffrida, S., Crisafulli, C. et al. Direct and sensitized liquid phase photodeposition for the preparation of alumina supported Pd nanoparticles for applications to heterogeneous catalysis. J Nanopart Res 13, 3217–3228 (2011). https://doi.org/10.1007/s11051-011-0222-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-011-0222-5