Abstract
Wild and cultured mushrooms have been extensively used for food and medicinal purposes all around the world. However, there is limited information on chemical composition, health enhancing effects and contributions on diet of some mushrooms (e.g., Agaricus arvensis) widely distributed in many countries including United Kingdom, Australia, Turkey etc. Therefore, the present study was aimed to analyse the bioactive composition and ameliorative effects of A. arvensis via evaluating in vitro and in vivo antioxidant properties in CCl4 induced rat model. The extract exhibited higher antioxidant capacities in vitro than that of the positive control (Reishi-Shiitake-Maitake standardized extract). Administration of the extract had significant regulative effects in the levels of AST, ALT, LDH, Urea and TRIG levels according to CCl4 group. Additionally, lipid peroxidation and GSH in the brain, kidney and liver tissues was regulated by extract treated groups compared to the CCI4 group. The supplementation of the extract at the dose of 100 mg/kg regulated the levels of GST, GR, CAT and GPx enzyme activities in brain and liver, but not in kidney tissue. There was approximately three fold increase in CAT enzyme activity in kidney tissue of extract treated groups compared to Control and CCl4 groups. The extract contained a rich composition of bioactive compounds including phenolics (protocatechuic acid and p-hydroxybenzoic acid), volatile compounds (benzaldehyde, palmitic acid and linoleic acid) and mineral compounds (K, Si, Mg and Na). Data obtained within this study suggests that A. arvensis might be used for food industries in order to obtain nutritional products.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Many environmental pollutants, alcohol, drugs and toxic materials engender oxidative stress such as hepatotoxicity, neurotoxicity, nephrotoxicity and lipid peroxidation. Carbon tetrachloride (CCl4) is an extremely toxic chemical compound which has been commonly used to induce hepatic injury in experimental animal models [1]. CCl4 induced oxidative stress might cause lipid peroxidation, protein and DNA damage in various rat tissues such as liver, kidneys and lungs [2]. A large amount of mushrooms have been investigated to eliminate the hepatic damage stimulated by toxic chemical substances [3, 4].
Oxidative stress generates free radicals such as peroxyl radicals, superoxide radicals, hydroxyl radicals etc. These free radicals suppress in vivo and in vitro antioxidant defence systems in living systems. Organisms avoid from harmful effects of free radicals by their in vivo protective enzymes such as catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione reductase (GR), reduced glutathione (GSH) and glutathione S-transferase (GST) [2,3,4,5].
Bioactive compounds present in mushrooms have been reported to increase the activity of antioxidant enzymes and suppress the formation of possible free radical sources [5, 6]. Several macrofungus samples exhibited pronounced antioxidant activity which has been associated to the presence of biologically active compounds including phenolics, glycosides, flavonoids, tocopherols, polysaccharides, carotenoids, ergothioneine, vitamins, ascorbic acid and minerals [7,8,9,10,11]. It was propounded that protective effects of antioxidants compounds present in macrofungus samples functioned mainly by two mechanisms: (I) chain breaking and free radical scavengers, (II) deactivation of metals, inhibition or breakdown of lipid hydroperoxides, regeneration of primary antioxidants and singlet oxygen quenching [11, 12].
Agaricus arvensis Schaeff. (Agaricaceae) known as horse mushroom has a widespread distribution in Europe (including in the United Kingdom and Turkey), Asia, Australia, New Zealand, and northern America. It is consumed globally in summer and autumn seasons and is collected from wild, particularly from scattered and rotten meadow areas. The hat zone is 7–20 cm in diameter and young white or pale yellowish as color. It has a thick structure, white or whitish colour without changing when damaged. A. arvensis has been traditionally utilized in Turkey for nutritional purposes. It is generally consumed freshly in salads or cooked with spices and vegetables in summer as food.
There is no or limited data regarding in vitro and in vivo antioxidant capacities, hepatoprotective and nephroprotective properties and chemical composition of A. arvensis in the scientific literature. Therefore, we aimed to determine health attributing properties of A. arvensis extract by evaluating antioxidant capacity in vitro and in vivo, toxicity level, protective properties on liver, kidney and brain, and general antioxidant defence system. Additionally, chemical composition of the extract was analysed by evaluating phenolic, volatile and mineral compounds.
Methods
Mushroom material
Fruiting bodies of Agaricus arvensis were collected from Akalin village, Yüksekova district, Hakkari City, in the Eastern Anatolia Region of Turkey, (GPS coordinates 37°34′022″N; 44°14′582″E) on June, 2016. The identity of samples was confirmed by Prof. Dr. Kenan Demirel at the Department of Biology, Science Faculty, Van Yuzuncu Yil University, Turkey and a voucher specimen was deposited at the university’s fungarium (Fungarium code: YYU-VAN, FUNGARIUM-7487). The fruiting bodies of fresh mushroom samples were cleaned properly and dried at room temperature in the dark until dry. Subsequently, the samples were ground and stored at − 20 °C until analysed.
Reagents
Unless otherwise stated, all chemicals were purchased from Sigma-Aldrich, Inc. (St Louis, MO, USA) and were of analytical or HPLC grade. Folin–Ciocalteu reagent was purchased from Merck (Darmstadt, Germany). Standardized Reishi-Shiitake-Maitake extract was purchased from Solgar (Istanbul, Turkey). Kits for antioxidant enzymes analysis were supplied by Randox Laboratories L.T.D. (Antrim, UK).
Preparation of lyophilized extract
The lyophilized extract was prepared according to method by Dalar and Konczak [13]. Briefly, the ground mushroom material was mixed with a 10-fold volume of aqueous ethanol (80% ethanol, 20% H2O, v/v), shaken for 2 h at room temperature (25 °C) and centrifuged for 20 min (15,320×g) at 4 °C (Sorvall RC-5B; DuPont, Wilmington, DE, USA; rotor Beckman JA14 (137 mm) serial No. 02U8152, USA) with the supernatant collected. The extraction procedure was repeated one more time. The supernatants from the consecutive extractions were combined. Solvent was evaporated under reduced pressure at 40 °C. The derived fraction was dissolved in purified water and freeze-dried under a vacuum at—51 °C to obtain a fine lyophilized powder used for analysis.
Antioxidant capacity in vitro assay
Folin–Ciocalteu reducing capacity (Total phenolic content), total reducing capacity (FRAP assay) and oxygen radical scavenging capacity of the extract were determined as described previously by Dalar and Konczak [13]. Standardized Reishi-Shiitake-Maitake extract was used as positive control. The analyses were conducted in triplicate.
Analysis of phenolic compounds
Identification and quantification of phenolic compounds by high liquid chromatography–diode array–mass spectrometry (LC–DAD–MS/MS) analysis were conducted as described previously with minor modifications [13].
Analysis of volatile and fatty acid compounds
Volatile compounds and fatty acids present in extracts were analysed by gas chromatography mass spectrometry (GC/MS) using a head space solid phase micro extraction and identified by the fragment ions and relative retention indices of their peaks with those of the MS library standards as described previously [14]. The headspace volume, heating temperature and time of the extraction were optimized according to Verzera et al. [15].
Mineral compounds analysis
Mineral composition of the extract was evaluated by using AAS, ICP-MS and ICP-OS. The analysis solution was prepared by dissolving the extract in HNO3. Subsequently, the solution was subjected to microwave assisted extraction procedure. The identity of mineral compounds was confirmed by comparison of authentic standards.
Animals
Twenty-four Wistar albino male rats 2–6 months of aged were provided by the Experimental Animal Research Centre, Van Yuzuncu Yil University (Van, Turkey). The animals were housed in four groups, each group containing six rats. The animals were housed at 25 ± 1 °C in a daily light/dark (~ 12/12) cycle. All animals were fed a diet based on wheat and soybean meal and water ad libitum in stainless cages, and received humane care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Science and published by the National Institutes of Health. The ethic regulations were followed in accordance with national and institutional guidelines for the protection of animal welfare during experiments. This study was approved by The Ethic Committee of the Van Yuzuncu Yil University (Protocol Number: 27552122-604.01.02).
Acute toxicity test
Acute toxicity was determined according to the OECD guidelines, test 423: acute oral toxicity method; 2002. A range of the extract (20, 50, 100, 250, 500, 1000 and 2000 mg/kg) administrated to the three rats orally via gastric gavage for 0.5, 2, 4, 8, 24, 48 and 72 h respectively. The animals were fed on food and water ad libitum. There is no symptoms of toxicity and mortality were observed over a period of 72 h.
Experimental design
The rats were randomly distributed into four groups; Control, CCl4, CCl4 + A. arvensis 100 mg/kg extract and CCl4 + A. arvensis 500 mg/kg extract. Food and drinking water were supplied for all animals as ad libitum during experiments.
Control group (n = 5): the rats received drinking water and fed with standard pellet diet as ad libitum.
CCl4 group (n = 5): the rats received 0.5 ml CCl4/kg rat weight intraperitonally twice per week. Dose of CCl4 was selected on the basis of a 0.5 ml CCl4/kg rat weight diluted in olive oil (1:1 dilution) [16].
CCI4 + A. arvensis 100 mg/kg extract group (n = 6) CCI4 + A. arvensis 100 mg/kg extract group (n = 6): the rats received 0.5 ml CCl4/kg rat weight intraperitonally injection twice per week and A. arvensis extract (100 mg/kg, per day) was applied to rats via orally during 28 days.
CCI4 + A. arvensis 500 mg/kg extract Group (n = 6): CCI4 + A. arvensis 500 mg/kg extract group (n = 6): the rats received 0.5 ml CCl4/kg rat weight intraperitonally injection twice per week and A. arvensis extract (500 mg/kg, per day) was applied to rats via orally during 28 days.
Preparation of serum and tissue supernatants
At the end of the experiments, the rats were anesthetized by injection of ketamine (5 mg/100 g body weight) intraperitonally. The blood samples were taken using a cardiac puncture and were put immediately into silicon disposable glass biochemical tubes. Subsequently, the samples were centrifuged at 4000×g for 15 min at 4 °C in order to obtaining serum samples for the measurement of biochemical parameters. The liver, brain and kidney tissues were dissected and put into Petri dishes. Subsequently, the samples were taken and kept at − 78 °C until analysis. The tissues were homogenized for 5 min in 50 mM ice-cold potassium dihydrogen phosphate (KH2PO4) solution (1:5 w/v) using stainless steel probe homogenizer (SONOPULS HD 2200, Bandelin, Berlin, Germany), and subsequently centrifuged at 7000×g for 15 min. All processes were carried out at 4 °C. Supernatants were used to determine antioxidant defence systems (ADS) constituents and Malondialdehyde (MDA) contents as described previously [17, 18].
Antioxidant defence system enzyme analysis
The liver, brain and kidney in tissue supernatants reduced glutathione (GSH) levels, malondialdehyde (MDA) content, glutathione reductase (GR) activity, glutathione-S transferase (GST) activity, superoxide dismutase (SOD) activity, glutathione peroxidase (GPx) activity, catalase (CAT) activity were determined as described previously [18].
Measurement of biochemical parameters
Serum biochemical parameters [aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), urea, uric acid (UA), creatinine (CREA), total protein (TP), sodium (Na), potassium (K), chlorine (CI), cholesterol (CHOL), triglyceride (TRIG) and HDL-cholesterol (HDL-C)] were measured by an auto analyser (ARCHITECT 16200, Abbott Park, IL 60064, USA) using the Abbott biochemistry kits (USA).
Analysis of data
All data were expressed as mean ± standard deviation (SD). The statistical analyses were made using the Minitab 13 for Windows packet program. One-way analysis of variance (ANOVA) statistical test was used to determine the differences between means of the experimental groups accepting the significance level at (p ≤ 0.05).
Results
Extraction yield, total phenolic content and antioxidant capacity in vitro
A high extraction yield of (23.5%) was obtained in consequence of the extraction procedure. The extract exhibited high antioxidant capacities. As shown in (Table 1), the extract exhibited 2.6–2.8 fold higher antioxidant capacities (ORAC and FRAP) and had 2.8 fold higher amount of total phenolics (FCR) than that of the positive control (Reishi-Shiitake-Maitake standardized extract), which indicated high antioxidant potential of the extract.
Chemical composition
Chemical composition of the extract was presented in (Table 2) as evaluated by HPLC–MS/MS (phenolics), GC–MS (volatiles and fatty acids) and AAS, ICP–MS VE ICP–OS (minerals). Phenolic composition of the extract mainly comprised of phenolic acids which had the maximum absorbance at 280 nm. No any compound was detected at 520 nm in the extract. The dominated phenolic compound identified based on m/z transition data and spectral and absorbance characteristics of HPLC peaks of the extract was detected as protocatechuic acid, followed by p-hydroxybenzoic acid as shown in (Table 2) and (Fig. 1). Volatile composition of the extract was mainly formed of benzaldehyde, palmitic acid and linoleic acid which contributed 80% of volatile composition as shown in (Table 2) and (Fig. 2). Major volatile compounds of the extract were identified as linoleic acid (54.03% contribution), followed by benzaldehyde (14.37% contribution) and palmitic acid (11.57% contribution) based on mass data (Table 2; Fig. 2). With regards to mineral composition of the extract, K, Si, Mg and Na were found as the dominant compounds of the extract, followed by Ca and B (Table 2). The heavy metals such as Cd, Co, As and Pb were found as trace levels in the extract (Table 2).
Acute toxicity test
Optimum tolerance dose was detected in animals against the application of Agaricus arvensis lyophilized extract. Non-lethal dose was obtained until 2000 mg/kg lyophilized extract. No any noticeable signs of toxicity and mortality were observed after daily administration of the extract orally at the end of the 72 h. Two different doses (100 and 500 mg/kg) were selected for further studies.
Effect of the extract on body weight
Changes in the initial and final body weight of the control and experimental groups were presented as supplementary material. Weight gain was significantly (p ≤ 0.05) observed in final weight of all of the rat groups compared to initial weight.
Effect of the A. arvensis extract on biochemical parameters
As shown in (Table 3), the levels of AST, ALT and LDH significantly increased in CCl4 group compared to control group. However, administration of the extract caused significant decreases in the levels of AST, ALT and LDH levels according to CCl4 group. However, the effect of the extract didn’t regulate the level of ALT compared to control group. Urea level was substantially lower in CCl4 + A. arvensis 500 mg/kg group than all other groups. No significant correlation was found between UA, CREA, TP, Na, K, Cl and CHOL levels and extraction doses applied. TRIG level significantly increased in control group than other all groups. HDL level in the CCl4 + A. arvensis 500 mg/kg group was significantly lower according to control group.
Effect of the A. arvensis extract on lipid peroxidation and antioxidant defence systems
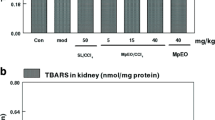
Effects of A. arvensis extract and CCl4 on MDA content, GSH level, enzyme activities of GST, GR, CAT, GPx and SOD in brain, kidney and liver tissues are presented in (Table 4). MDA content of CCl4 group significantly increased in brain, kidney and liver tissues according to control group. On the other hand, lipid peroxidation in the brain, kidney and liver tissues was regulated by extract treated groups compared to the CCI4 group (Table 4). GSH level of brain in CCl4 + A. arvensis 100 mg/kg group markedly decreased compared to CCI4 group. Additionally, GSH levels in liver markedly decreased in extract treated and CCI4 groups compared to control group. Relatively decreases in GST, GR, CAT and GPx enzyme activities in tissues of CCl4 group were observed according to control group. Generally, the supplementation of the extract at the dose of 100 mg/kg regulated the levels of GST, GR, CAT and GPx enzyme activities in brain and liver, but not in kidney tissue. There was approximately three fold increase in CAT enzyme activity in kidney tissue of extract treated groups compared to Control and CCl4 groups (Table 4).
Discussion
Oxidative stress is a complicated consecution occurring as endogenous and exogenous sources, playing an important role in aging processes, increasing the risk of chronic disease and causing fatal consequences. It is known that oxidative stress plays an important role in various diseases such as cardiovascular disease, aging, mutations, cancer and neurodegenerative diseases [19]. Natural sources obtained from plant, fungi and lichens are rich in physiologically beneficial bioactive substances such as phenolics, glycosides, tocopherols, polysaccharides, carotenoids, alkaloids and ascorbic acid which possesses pronounced antioxidant capacities in vitro and in vivo [9, 11]. Therefore, investigation of bioactive rich edible mushroom samples is crucially important in order to find new and safe antioxidant agents which able to prevent and/or minimize the negative effects of oxidative stress.
Within this study, in vitro antioxidant capacity of the extract was evaluated by using three complementary antioxidant testing methods representing two basic antioxidant mechanisms. ORAC assay represented hydrogen atom transfer (HAT) mechanism and FCR and FRAP tests represented single electron transfer (SET) mechanism. ORAC values can be used as reference antioxidant effectiveness since ORAC assay is able to measure the radical chain breaking ability of antioxidants by monitoring the inhibition of peroxyl radical-induced oxidation. In order to distinguish dominant mechanisms for different antioxidants, ORAC, FRAP and FCR methods were suggested to reveal the complementary antioxidant potential of food sources [20]. As shown in (Table 1), the extract had more antioxidant activities than positive control (Reishi-Shitake-Maitake standardized extract) which was investigated by Islam et al. [21] and reported for their high antioxidant capacities. Pronounced ORAC, FRAP and FCR capacities of the extract might be due to the presence of rich chemical composition of the extract. Protocatechuic acid was found as the major phenolic compound of the A. arvensis extract in this study. Li et al. [22] investigated the antioxidant potential of protocatechuic acid using several antioxidant tests and detected more antioxidant activity of protocatechuic acid than trolox and associated its activity to both chelating metal transition ions as well as scavenging free radicals via donating hydrogen atom (H⋅) or electron (e) properties.
CCl4 was used to create oxidative stress in different experimental animal models. CCl4 activated by phase-II detoxifying enzymes to generate oxidative radicals in liver such as trichloromethyl (CCl3) and peroxytrichloromethyl (CCl3O2). Peroxyl radicals induce lipid peroxidation causing damage on cell membrane in many tissues mainly liver, brain, kidney, lung and erythrocyte [23, 24]. In this study, in vivo antioxidant studies were conducted against CCl4 induced oxidative stress. Our findings showed positive effect of CCl4 on increasing the final body weights (Supplementary material) which was in accordance with previous studies [25, 26]. On the other hand, Connor et al. [27] suggested different doses (1.58 and 3.16 g/kg) of CCl4 could cause an increase for the first 5 days following by a decrease in live body weight. Hence it might be speculated that dose concentration and application period of CCl4 might have different effects on live body weight.
As shown in (Table 2), within this study protocatechuic acid as major hydrophilic compound; linoleic acid as major lipophilic compound and K and Si as major mineral compounds were detected in A. arvensis extract. The levels of heavy metals were determined in the extract was substantially at low levels, which indicate the reliability of this mushroom in the context of nutrition. It was reported that economically important wild edible mushrooms contained significant amounts of valuable fatty acids [28]. Similar to our findings Barros et al. [29] detected linoleic acid as one of the major fatty acids in various mushroom samples including A. arvensis, L.deliciosus, L. giganteus, S. imbricatus, and T. portentosum. In another study conducted on pharma-nutritional properties of wild mushrooms, protocatechuic acid and p-hydroxybenzoic acid were detected as major phenolic compounds and linoleic and palmitic acids as major fatty acids of mushroom extracts [30]. Protocatechuic acid was reported for its potential hepatoprotective and nephroprotective activities in vivo [31] and p-hydroxybenzoic acid and its derivatives were detected as effective antioxidant compound for their abilities to scavenge free radicals and trigger endogenous antioxidant activity [11], which are in accordance with our findings.
As shown in (Table 3), although there was an increase in the levels of AST, ALT and LDH in the CCl4 group compared to control group, supplementation of the extract regulated the levels of AST, ALT and LDH levels compared to CCl4 group. Increases in serum AST, ALT, LDH, CRE and Urea levels generally indicate liver and kidney injuries. Özkol et al. [32] and Dogan and Celik [18] reported a positive correlation between serum liver enzymes and reactive oxygen species (ROS) which were associated with damage to the liver cells, leading to enzyme leakage into the plasma and cellular necrosis. Similar to our findings, some researchers reported increase in the levels of those enzymes in CCl4 groups [16, 27]. Jayakumar et al. [5] evaluated Pleurotus ostreatus extract against CCl4-induced hepatotoxicity and found regulative effects of the extract on serum AST, ALT and ALP levels. Furthermore, Jeong et al. [33] and Aniya et al. [34] demonstrated that the administration of Artemisia campestris extract decreased the toxicity induced by CCl4 by healing of abovementioned enzymes. Treatment of A. arvensis extract might have improved biochemical parameters due to the presence of an antioxidative active compound—protocatechuic acid. We therefore suggest that pretreatment with the A. arvensis extract markedly reversed hepatotoxicity and nephrotoxicity caused by the CCl4. The positive effect of extract on triglycerides (TRIG) and high density lipoprotein cholesterol (HDLC) was shown in Table 3. Goyal and Grewal [35] investigated the effects of Agaricus bisporus on serum total lipid and triglyceride levels and found a negative correlation between applied dose and levels of TRIG and HDLC. Additionally, ameliorative effects of various mushroom samples on TC, TRIG, LDL, HDL and VLDL levels were reported previously [36, 37].
As presented in (Table 4), the negative effect of CCl4 which caused a significant increase in lipid peroxidation was regulated by extract applied in brain, kidney and liver tissues. Lipid peroxidation especially occurs on cell membrane as a result of unsaturated fatty acids oxidation. Malondyaldehyde (MDA) forms as a final product of lipid peroxidation in cells. MDA is one of the significant biomarker which have been used to observe lipid peroxidation and oxidative stress. Moreover, MDA might have important mutagenic, atherogenic, and cancerogenic actions, as it is capable of forming different, biologically relevant carbohydrates, nucleic acids and protein adducts [18]. It was reported that CCI4 significantly increased MDA content in rat erythrocyte, liver, brain, kidney and spleen tissues compared to the control group [16, 24]. Similarly, Coballase-Urrutia et al. [26] reported significant increase in MDA levels and decrease in antioxidant enzyme activities in the liver and brain regions (cerebellum, cortex and cerebral hemispheres) of CCl4 group. Treatment of rats with A. arvensis extract reversed the increase of lipid peroxidation compared to the control group. This could be explained by the presence of antioxidant compounds present in the extract including protocatechuic and linoleic acids. Our findings are in agreement with those of Lin et al. [38] and Jayakumar et al. [5] who reported the inhibitory potential of various mushroom samples on lipid peroxidation induced by CCl4.
As shown in (Table 4), A. arvensis extract regulated the levels of GST, GR, CAT and GPx enzyme activities in tissues except kidney. Furthermore, the extract increased the CAT activity in kidney tissue threefold higher than those of Control and CCl4 groups. Administration of CCl4 causes generation of peroxyl radical and inactivation of antioxidant enzymes such as GPx, SOD, CAT and GST. Inactivation of these enzymes in organisms is associated with the accumulation of highly reactive molecules such as superoxide anion (O2·−), hydroxyl radical (OH·), hydroxyl ion (OH−), nitric oxide (NO·) and hydrogen peroxide (H2O2) which contribute to the deleterious effects such as loss of coherence and function of cell membranes [5, 39,40,41]. When all the data are evaluated, this study suggests that A. arvensis has a protective capacity and antioxidant activity against CCl4 toxicity in rtas, probably by promoting antioxidative defense systems.
In conclusions, A. arvensis extract which was rich in phenolics (protocatechuic acid and p-hydroxybenzoic acid), fatty acids (linoleic and palmitic acid) and various mineral compounds exhibited pronounced antioxidant capacities in vitro and in vivo. Data obtained within this study suggest that A. arvensis extract might help gain protection against unwanted and harmful effects of oxidative stress such as hepatotoxicity, nephrotoxicity and lipid peroxidation induced by admistration of carbon tetrachloride and we suggest further clinical and industrial studies on A. arvensis extract in order to obtain a nutritional health enhancing food product both for food and pharmaceutical industries.
Abbreviations
- Ag:
-
Silver
- ALT:
-
Alanine aminotransferase
- As:
-
Arsenic
- AST:
-
Aspartate aminotransferase
- B:
-
Boron
- Ca:
-
Calcium
- CAT:
-
Catalase
- CCl4 :
-
Carbon tetrachloride
- Cd:
-
Cadmium
- CHOL:
-
Cholesterol
- Cl:
-
Chlorine
- Co:
-
Cobalt
- Cr:
-
Chromium
- CREA:
-
Creatinine
- Cu:
-
Copper
- FCR:
-
Folin–Ciocalteu reagent
- Fe:
-
Iron
- FRAP:
-
Ferric reducing antioxidant power
- GPx:
-
Glutathione peroxidase
- GR:
-
Glutathione reductase
- GSH:
-
Reduced glutathione
- GST:
-
Glutathione S-transferase
- HDL-C:
-
High-density lipoprotein cholesterol
- K:
-
Potassium
- LDH:
-
Lactate dehydrogenase
- MDA:
-
Malondialdehyde
- Mg:
-
Magnesium
- Mn:
-
Manganese
- Mo:
-
Molybdenum
- Na:
-
Sodium
- ORAC:
-
Oxygen radical absorbance capacity
- Pb:
-
Lead
- Se:
-
Selenium
- Si:
-
Silicon
- SOD:
-
Superoxide dismutase
- Na:
-
Sodium
- TG:
-
Triglycerides
- TP:
-
Total protein
- UA:
-
Uric acid
- Zn:
-
Zinc
References
Li S, Tan HY, Wang N et al (2015) The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 16:26087–26124
Feng Y, Wang N, Ye X et al (2011) Hepatoprotective effect and its possible mechanism of Coptidis rhizoma aqueous extract on carbon tetrachloride-induced chronic liver hepatotoxicity in rats. J Ethnopharmacol 138:683–690
Zhang J, Meng G, Zhang C et al (2015) The antioxidative effects of acidic-, alkalic-, and enzymatic-extractable mycelium zinc polysaccharides by Pleurotus djamor on liver and kidney of streptozocin-induced diabetic mice. BMC Complement Altern Med 15:440
Okolo KO, Siminialayi IM, Orisakwe OE (2016) Protective effects of Pleurotus tuberregium on carbon- tetrachloride induced testicular injury in Sprague Dawley rats. Front Pharmacol 7:480
Jayakumar T, Ramesh E, Geraldine P (2006) Antioxidant activity of the oyster mushroom, Pleurotus ostreatus, on CCl4-induced liver injury in rats. Food Chem Toxicol 44:1989–1996
Kalaras MD, Richie JP, Calcagnotto A et al (2017) Mushrooms: a rich source of the antioxidants ergothioneine and glutathione. Food Chem 233:429–433
Cheung LM, Cheung PCK, Ooi VEC (2003) Antioxidant activity and total phenolics of edible mushroom extracts. Food Chem 81:249–255
Klaus A, Kozarski M, Niksic M et al (2011) Antioxidative activities and chemical characterization of polysaccharides extracted from the basidiomycete Schizophyllum commune. LWT-Food Sci Technol 44:2005–2011
Chen SY, Ho KJ, Hsieh YJ et al (2012) Contents of lovastatin, γ-aminobutyric acid and ergothioneine in mushroom fruiting bodies and mycelia. LWT-Food Sci Technol 47:274–278
Naguib YM, Azmy RM, Samaka RM et al (2014) Pleurotus ostreatus opposes mitochondrial dysfunction and oxidative stress in acetaminophen-induced hepato-renal injury. BMC Complement Altern Med 14:494
Kozarski M, Klaus A, Vunduk J et al (2015) Nutraceutical properties of the methanolic extract of edible mushroom Cantharellus cibarius (Fries): primary mechanisms. Food Funct 6:1875–1886
Brewer MS (2011) Natural antioxidants: sources, compounds, mechanism of action, and potential applications. Compr Rev Food Sci Food Saf 10:221–247
Dalar A, Konczak I (2013) Phenolic contents, antioxidant capacities and inhibitory activities against key metabolic syndrome relevant enzymes of herbal teas from Eastern Anatolia. Ind Crops Prod 44:383–390
Uzun Y, Dalar A, Konczak I (2017) Sempervivum davisii: phytochemical composition, antioxidant and lipase-inhibitory activities. Pharm Biol 55:532–540
Verzera A, Zino M, Condurso C et al (2004) Solid-phase microextraction and gas chromatography/mass spectrometry for the rapid characterization of semi-hard cheeses. Anal Bioanal Chem 380:930–936
Suzek H, Celik I, Dogan A et al (2016) Protective effect and antioxidant role of sweetgum (Liquidambar orientalis) oil against carbon tetrachloride-induced hepatotoxicity and oxidative stress in rats. Pharm Biol 54:451–457
Dogan A, Celik I, Kaya MS (2015) Antidiabetic properties of lyophilized extract of acorn (Quercus brantii Lindl.) on experimentally STZ-induced diabetic rats. J Ethnopharmacol 176:243–251
Dogan A, Celik I (2012) Hepatoprotective and antioxidant activities of grapeseeds against ethanol-induced oxidative stress in rats. Br J Nutr 107:45–51
Sachindra NM, Airanthi MKWA, Hosokawa M et al (2010) Radical scavenging and singlet oxygen quenching activity of extracts from Indian seaweeds. J Food Sci Technol 47:94–99
Shahidi F, Zhong Y (2015) Measurement of antioxidant activity. J Funct Foods 18:757–781
Islam T, Xiaoming Y, Xu B (2016) Phenolic profiles, antioxidant capacities and metal chelating ability of edible mushrooms commonly consumed in China. LWT Food Sci Technol 72:423–431
Li X, Wang X, Chen D et al (2011) Antioxidant activity and mechanism of protocatechuic acid in vitro. Funct Food Health Dis 1:232–244
Srinivasan M, Rukkumani A, Sudheer R et al (2005) Ferulic acid, a natural protector against carbon tetrachloride induced toxicity. Fundam Clin Pharmacol 19:491–496
Suzek H, Celik I, Dogan A (2017) Nephroprotective hepatoprotective potential and antioxidant role of carob pods (Cerotonia siliqua L.) against carbon tetrachloride-induced toxicity in rats. Indian J Pharm Educ 51:312–320
Mahli A, Koch A, Czech B et al (2015) Hepatoprotective effect of oral application of a silymarin extract in carbon tetrachloride-induced hepatotoxicity in rats. Clin Phytosci 1:5
Coballase-Urrutia E, Cárdenas-Rodríguez E, González-García MC et al (2017) Biochemical and molecular modulation of CCl4-induced peripheral and central damage by Tilia americana var. mexicana extracts. Saudi Pharm J 25:319–331
Connor SC, Wu W, Sweatman BC et al (2004) Effects of feeding and body weight loss on the 1H-NMR-based urine metabolic profiles of male Wistar Han Rats: implications for biomarker discovery. Biomarkers 9:156–179
Günç Ergönül P, Akata I, Kalyoncu F et al (2013) Fatty acid compositions of six wild edible mushroom species. Sci World J. https://doi.org/10.1155/2013/163964
Barros L, Baptista P, Correia DM et al (2007) Fatty acid and sugar compositions, and nutritional value of five wild edible mushrooms from Northeast Portugal. Food Chem 105:140–145
Leal AR, Barros L, Barreira J et al (2013) Portuguese wild mushrooms at the “pharma–nutrition” interface: nutritional characterization and antioxidant properties. Food Res Int 50:1–9
Kakkar S, Bais S (2014) A review on protocatechuic acid and its pharmacological potential. ISRN Pharmacol. https://doi.org/10.1155/2014/952943
Özkol H, Musa D, Tülüce Y et al (2011) Ameliorative influence of Urtica dioica L against cisplatin-induced toxicity in mice bearing Ehrlich ascites carcinoma. Drug Chem Toxicol 35:251–257
Jeong TC, Kim HJ, Park J et al (1996) Protective effect of red ginseng saponins against carbon tetrachloride induced hepatoxicity in Sprague dawley rats. Planta Med 63:136–140
Aniya Y, Shimabukuro M, Shimoji et al (2000) Antioxidant and hepatoprotective actions of the medicinal herb Artemisia campestris from the Okinawa Islands. Biol Pharm Bull 23:309–312
Goyal R, Grewal R (2017) Changes in lipid profile of rats fed with Agaricus bisporus (white button) mushroom. Nutr Food Sci 47:288–296
Bobek P, Hromadova M, Ozdin L (1995) Oyster mushroom (Pleurotus ostreatus) reduces the activity of 3-hydroxy-3-methyl-glutaryl coA reductase in rat liver microsomes. Experientia 51:589–591
Alam N, Amin R, Khan A et al (2009) Comparative effects of oyster mushrooms on lipid profile, liver and kidney function in hypercholesterolemic rats. Mycobiology 37:37–42
Lin S, Zhu P, Liao H (1998) Blocking effect of 4 edible mushroom beverages on the carbon tetrachloride induced rat liver lipid peroxidation. Zhongguo Gonggong Weisheng Xuebao 17:15
Krishnakantha TP, Lokesh BR (1993) Scavenging of super oxide anions by spice principles. Indian J Exp Biol 30:133–134
Sheela CG, Angusti K (1995) Antiperoxide effects of S-allyl cystein sulphoxide isolated from Allium sativum Linn and gugulipid in chlosterol diet fed rats. Indian J Exp Biol 33:337–341
Ozkol H, Bulut G, Balahoroglu R et al (2017) Protective effects of selenium, N-acetylcysteine and vitamin E against acute ethanol intoxication in rats. Biol Trace Elem Res 175(1):177–185
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has a conflict of commercial interest, financial interest, and/or other relationship with manufacturers of pharmaceuticals, laboratory supplies and/or medical devices or with commercial providers of medical services. The authors are grateful to the University of Van Yuzuncu Yil Grant Commission for providing financial assistance of the research (Project Number: THD-2016-5377).
Research involving human and animal participants
Animals used in this study (24 Wistar albino male rats 2–6 months of aged, weighting 200–350 g) were provided by the Experimental Animal Research Centre, Van Yuzuncu Yil University (Van, Turkey). All animals were fed and received human care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Academy of Science and published by the National Institutes of Health. The ethic regulations were followed in accordance with national and institutional guidelines for the protection of animal welfare during experiments. This study was approved by The Ethic Committee of the Van Yuzuncu Yil University (Protocol Number: 27552122-604.01.02). No human participants involved in the present research.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dogan, A., Dalar, A., Sadullahoglu, C. et al. Investigation of the protective effects of horse mushroom (Agaricus arvensis Schaeff.) against carbon tetrachloride-induced oxidative stress in rats. Mol Biol Rep 45, 787–797 (2018). https://doi.org/10.1007/s11033-018-4218-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-018-4218-4