Abstract
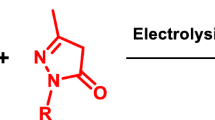
Electrochemically induced catalytic multicomponent transformation of isatins, 3-methyl-2-pyrazolin-5-ones and malononitrile in ethanol in an undivided cell in the presence of sodium bromide as an electrolyte results in the formation of spirooxindoles with fused functionalized pyrano[2,3-c]pyrazole system in 78–99} yields. The developed efficient electrocatalytic approach to medicinally relevant spirocyclic [indole-3,4′-pyrano[2,3-c]pyrazoles] is beneficial from the viewpoint of diversity-oriented large-scale processes and represents a novel example of facile environmentally benign synthetic concept for electrocatalytic multicomponent reaction strategy.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Thompson LA (2000) Recent applications of polymer-supported reagents and scavengers in combinatorial, parallel, or multistep synthesis. Curr Opin Chem Biol 4: 324–337. doi:10.1016/S1367-5931(00)00096-X
Nefzi A, Ostresh JM, Houghten RA (1997) The current status of heterocyclic combinatorial libraries. Chem Rev 97: 449–472. doi:10.1021/cr960010b
Weber L (2002) Multi-component reactions and evolutionary chemistry. Drug Discov Today 7: 143–147
Dömling A (2002) Recent advances in isocyanide-based multicomponent chemistry. Curr Opin Chem Biol 6: 306–313. doi:10.1016/S1367-5931(02)00328-9
Mironov MA (2006) Design of multi-component reactions: from libraries of compounds to libraries of reactions. QSAR Comb Sci 25: 423–431. doi:10.1002/qsar.200540190
Ramón DJ, Yus M (2005) Asymmetric multicomponent reactions (AMCRs): the new frontier. Angew Chem Int Ed 44: 1602–1634
Orru RVA, de Greef M (2003) Recent advances in solution-phase multicomponent methodology for the synthesis of heterocyclic compounds. Synthesis 10: 1471–1499. doi:10.1055/s-2003-40507
Williams RM, Cox RJ (2003) Paraherquamides, brevianamides, and asperparalines: laboratory synthesis and biosynthesis. An interim report. Acc Chem Res 36: 127–139. doi:10.1021/ar020229e
Cui CB, Kakeya H, Osada H (1996) Novel mammalian cell cycle inhibitors, spirotryprostatins A and B, produced by Aspergillus fumigatus, which inhibit mammalian cell cycle at G2/M phase. Tetrahedron 52: 12651–12666. doi:10.1016/0040-4020(96)00737-5
Leclercq J, de Pauw-Gillet MC, Bassleer R, Angenot L (1986) Screening of cytotoxic activities of Strychnos alkaloids (methods and results). J Ethnopharmacol 15: 305–316. doi:10.1016/0378-8741(86)90169-8
Alper PB, Meyers C, Lerchner A, Siegel DR, Carreira EM (1999) Facile, novel methodology for the synthesis of spiro[pyrrolidin-3,3-oxindoles]: catalyzed ring expansion reactions of cyclopropanes by aldimines. Angew Chem Int Ed 38: 3186–3189. doi:10.1002/(SICI)1521-3773(19991102)38:21<3186::AID-AN IE3186>3.0.CO;2-E
Foloppe N, Fisher LM, Howes R, Potter A, Robertson AGS, Surgenor AE (2006) Identification of chemically diverse Chk1 inhibitors by receptor-based virtual screening. Bioorg Med Chem 14: 4792–4802. doi:10.1016/j.bmc.2006.03.021
Mishriky N, Girgis AS, Asaad FM, Ibrahim YA, Sobieh UI, Fawzy NG (2001) Simple synthesis of condensed pyran containing compounds and their antimicrobial properties. Boll Chim Farm 140: 129–139
Ebtisam AAH, Galil FMA, Sherif SM, Elnagdi MH (1986) Nitriles in heterocyclic synthesis. A novel synthesis of spiropyran-ylindolidene derivatives. J Heterocycl Chem 4: 1375–1378
Dworczak R (1991) Synthesen mit nitrilen, 88 Mitt.: spiro[indol- und spiro[inden-pyrano[2,3-c]pyrazole] aus cyanmethylenderivaten und pyrazolonen. Monatsh Chem 122: 731–737. doi:10.1007/BF00811473
Higashiyama K, Otomasu H (1980) Spiro heterocyclic compounds. III. Synthesis of spiro[oxindole-4′-(4′H-pyran] compounds. Chem Pharm Bull (Tokyo) 3: 648–651
El-Latif FFA, Gohar AEMN, Fahmy AM, Badr MZA (1986) Novel synthesis of furo[2,3-b]indole derivatives. Bull Chem Soc Jpn 59: 1235–1238. doi:10.1246/bcsj.59.1235
Shanthi G, Subbulakshmi G, Perumal PT (2007) A new InCl3- catalyzed, facile and efficient method for the synthesis of spirooxindoles under conventional and solvent-free microwave conditions. Tetrahedron 63: 2057–2063. doi:10.1016/j.tet.2006.12.042
Lund H (2000) Organic electrochemistry, 4 edn. Marcell Dekker Inc, New York
Elinson MN, Feducovich SK, Lizunova TL, Nikishin GI (2000) Electrochemical transformation of malononitrile and carbonyl compounds into functionally substituted cyclopropanes: electrocatalytic variant of the Wideqvist reaction. Tetrahedron 56: 3063–3069. doi:10.1016/S0040-4020(00)00195-2
Elinson MN, Dorofeev AS, Miloserdov FM, Ilovaisky AI, Feducovich SK, Belyakov PA, Nikishin GI (2008) Catalysis of salicylaldehydes and two different C–H acids with electricity: first example of an efficient multicomponent approach to the design of functionalized medicinally privileged 2-amino-4H-chromene scaffold. Adv Synth Catal 350: 591–601. doi:10.1002/adsc.200700493
Elinson MN, Ilovaisky AI, Dorofeev AS, Merkulova VM, Stepanov NO, Miloserdov FM, Ogibin YN, Nikishin GI (2007) Electrocatalytic multicomponent transformation of cyclic 1,3-diketones, isatins, and malononitrile: facile and convenient way to functionalized spirocyclic (5,6,7,8-tetrahydro-4H-chromene)-4,3′-oxindole system. Tetrahedron 63: 10543–10548. doi:10.1016/j.tet.2007.07.080
Elinson MN, Dorofeev AS, Feducovich SK, Nasybullin RF, Gorbunov SV, Nikishin GI (2006) Electrocatalytic chain transformation of salicylaldehydes and malononitrile into substituted 4H-chromenes. Electrochem Commun 8: 1567–1571. doi:10.1016/j.elecom.2006.07.009
Dandia A, Arya K, Sati M, Sharma R (2003) Facile microwave-assisted solid phase synthesis of spiro[3H-indole-3,4′-pyrazolo[3,4-b]pyridines]. Heterocycl Commun 9: 415–420
Patai S, Israeli Y (1960) The kinetics and mechanisms of carbonyl–methylene condensations. Part VII. The reaction of malononitrile with aromatic aldehydes in ethanol. J Chem Soc 2025–2030. doi:10.1039/jr9600002025
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elinson, M.N., Dorofeev, A.S., Miloserdov, F.M. et al. Electrocatalytic multicomponent assembling of isatins, 3-methyl-2-pyrazolin-5-ones and malononitrile: facile and convenient way to functionalized spirocyclic [indole-3,4′-pyrano[2,3-c]pyrazole] system. Mol Divers 13, 47–52 (2009). https://doi.org/10.1007/s11030-008-9100-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-008-9100-1