Abstract
Parkinson's disease (PD) is a complex neurodegenerative disease in which the understanding of the underlying molecular mechanisms can be constructive in the diagnosis and treatment. Matrix metalloproteinase (MMPs) elevation and damage to the blood–brain barrier (BBB) are critical mechanisms involved in the PD separation. Studies have revealed that changes in miR-149-5p and CoQ10 are associated with BBB damage, and CoQ10 can affect the levels of some miRs. Hence, in the present study, we aimed to evaluate CoQ10 and miR-149-5p mimic on miR-149-5p, MMPs and TH expression, and behavioral functions of the PD models. PD was induced by injection of 6-OHDA into the rats' Medial Forbrain Bundle (MFB). The behavioral tests, including the Rotation test, Rotarod test, and Open field test, have been directed two weeks after PD induction. Next, the MiR-149-5p mimic (miR-mimic) and CoQ10 have been administered to rats. The same behavioral tests have been evaluated two weeks after administration to investigate the effect of miR-149-5p mimic and CoQ10. The rats were followed extra four weeks, and the behavioral tests have performed again. Finally, the expression of MMPs and miR-149-5p genes was measured using RT-qPCR, and tyrosine hydroxylase (TH) was assessed through immunohistochemistry analysis. According to the obtained results, the level of miR-149-5p has decreased, followed by PD induction in rats. RT-qPCR analysis has represented upregulation and downregulation of miR-149-5p and MMP-2,9, respectively, after miR-mimic and CoQ10 treatment. The treated rats have also represented improved motor function and increased TH + cells in the striatum according to the behavioral tests and immunohistochemistry assay. Taking together miR-149 and CoQ10 has shown to have an impressive potential to prevent damage to dopaminergic neurons caused by 6-OHDA injection through reducing MMP-2,9, increased TH expression, and improved motor function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since Parkinson's disease (PD) was described in 1817 by James Parkinson, many studies have been conducted regarding its mechanisms and potential therapies. PD is inducing their destructive effect through severe progressive dopaminergic neuron destruction in the nigrostriatal pathway that eventually leads to neurons loss in the substantia nigra dense part (Mendes Filho et al. 2018). The PD disease is characterized by motor complications such as tremor, stiffness, bradykinesia, gait and balance disorders (Obeso et al. 2017). Despite dopaminergic therapies such as L-Dopa and other pharmacological and surgical procedures, the disease is still progressive (Fox et al. 2018; Jankovic 2019). It seems crucial to find more effective treatment strategies to recover the neuronal damage followed by PD development.
The blood–brain barrier (BBB), as a highly critical element of the central nervous systems (CNS), plays a tremendous role in neuronal activity and information processing through controlling molecules influx. BBB deformation has shown to be a primary biomarker for brain injury, leading to the secondary cascade of injuries such as apoptosis. According to previous studies, PD development caused BBB damages, leading to more extensive PD complications (Chen et al. 2008; Cabezase et al. 2014; Sweeney et al. 2018; Nation et al. 2019; Lin et al. 2020). Endothelial cell tight junctions (TJs) are considered an essential factor in the BBB integrity. TJs have several complex proteins such as claudin, occluding, and zonula occlumency that are susceptible to matrix metalloproteinase (MMPs). It has been well established that MMPs' high expression following neurodegenerative disorders, including PD, leads to TJs destruction that enhances the BBB permeability (Chen et al. 2008; Winkler et al. 2021). In this regard, MMPs inhibition could be considered as a possible therapeutic strategy to alleviate PD complications.
Several small molecules in cells, including microRNAs (miRs), can be applied for specific purposes. miRs are short, endogenous, and highly conserved non-coding RNAs containing approximately 22 nucleotides. These molecules are well known as gene post-transcriptional regulation agents by binding to the mRNAs 3′-untranslated region (3′-UTR) specifically, inhibiting translation or induction of mRNA degradation of the (Qadir et al. 2020). As the most common pathway of miRs generation, the canonical path is accomplished in two separate steps. In which they have synthesized in the nucleus and processed in the cytoplasm (O’Brien et al. 2018). However, non-canonical pathways of miRs synthesize have been recently recognized. Scientists have been recruiting this approach to the chemical production of miRs-like oligonucleotides known as miR mimics that mimic the miRs functions in cells (O’Brien et al. 2018; Titze-de-Almeida and Titze-de-Almeida 2018). According to genetic studies, several miRs levels have been altered following PD development, which plays a significant role in its pathogenesis. Such alterations are ascribed to the miR-155-5p and miR-449a increasing and miR-146a-5p and miR-221-3p decreasing during PD (Caggiu et al. 2018; Oh et al. 2018; Tolosa et al. 2018). Among them, miR-149-5p has been critically decreased in brain disorders such as stroke (Wan et al. 2018) and Alzheimer's (Chen et al. 2020). Based on the previous studies, miR-149-5p mimic molecules have reduced brain disease complications by reducing MMP-2,9 mRNA by targeting the 3′-UTR region (Lu et al. 2017; Feng et al. 2019; Ghasemloo et al. 2021).
Coenzyme Q10 (CoQ10; chemically 2,3-dimethoxy-5-methyl-6-decaprenyl benzoquinone) is an essential endogenous lipophilic molecule found in the mitochondrial respiratory chain of the eukaryotic cell membranes (Belhaj et al. 2012). Q10 deficiency has been linked to neurodegenerative disorders (Mancuso et al. 2010), heart disease (Mugoni et al. 2013), and cancer (Contin et al. 2015). CoQ10 improves ATP production through electrons capture from complexes I and II. Complex I has shown to be decreased in the substantia nigra of patients with PD, leading to the dopaminergic neurons apoptosis. CoQ10 showed to restore the dopaminergic neurons function and protect dopaminergic axons against excitotoxin-induced degradation. The antioxidant effect of Q10 leads to increasing the protein's activity responsible for free radical scavenging Seet et al. 2014). Besides, it has been shown that Q10 reduces brain disease complications by increasing the expression of miRs (Schmelzer et al. 2008; Olivieri et al. 2013; Wang et al. 2014).
Previous studies have shown that the miR-149-5p molecules play an essential role in decreasing several brain disorders' complications. Hence, in the present study, the effect of miR-149-5p alteration following PD induction has been evaluated for the first time by measuring motor-balance function, TH level, and MMP-2,9 expression in the striatum of rats. The effects of CoQ10 administration on miR-149-5p expression and PD complications have been analyzed to determine whether Q10 can prevent the development of PD complications by upregulating miR-149-5p and decreasing MMPs.
Materials and methods
Experimental design
Two weeks after stereotactic injection of 6-OHDA into the right MBF and induction of PD model in rats, a rotation test was performed to confirm the PD induction. Ultimately, rotarod and open field tests were performed, and then coenzyme Q10 and miR-149-5p mimic were administered to the treatment groups, Q10 solvent to the vehicle group, scramble miR to the NC group, and distilled water to the PD group. All behavioral tests were conducted two and four weeks after the administrations. After deep anesthesia of the animals, perfusion was performed, and the brains were removed for immunohistochemistry and gene expression evaluation (Fig. 1).
Study population
Male Wistar rats weighing 250–300 g were purchased from the Karaj Pasteur Institute and kept under the standard condition with the temperature of 22 ± 2 °C, the light cycle of 12 h of light and 12 h of darkness (light from 8 am to 8 pm), and unlimited access to water and special food (pellets). The animals were treated according to the International Organization for Medical Science Working with Laboratory Animals guidelines. This study has been approved by the ethics committee of Zanjan University of Medical Sciences (Ethical No: IR.ZUMS.REC.1400.005).
Study groups
Animals were randomly divided into six groups, each consisting of six Wistar rats. The Study groups were labeled as sham (Healthy rats that undergo surgical stress only), PD model (Parkinson's rats that without any treatment), the treatment groups receiving CoQ10 and miR-149-5p mimic (miR-mimic), respectively, vehicle group (Parkinson's rats receiving Q10 solvent) and NC (Negative Control) group (receiving Scramble miR (S-miR)).
PD models
The animals were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg) and fixed in a stereotactic device (Stoelting, USA) to induce the PD. The surface of the skull was dissected using a razor blade. The Medial Forbrain Bundle (MFB) coordinates of rats were defined as AP: − 4 mm from Bregma, ML: 1.8 mm from Midline, and DV: 8.8 mm from the skull based on the Paxinos and Watson Atlas. A small hole was made in the skull bone using a dental drill, and 6-OHDA (6-hydroxydopamine) was injected through into the target area with a Hamilton syringe (8 µg/2 µl normal saline containing 0.01% ascorbic acid, pH = 5), leading to the destruction of the nigrostriatal pathway (Fig. 1a).
miR-149-5p mimic and CoQ10 treatment of PD rats
To intracerebral injection miR-149-5p mimic and CoQ10, the animals were anesthesia first, and the lateral ventricular coordinates were determined according to Paxinos & Watson atlas (1 mm posteriorly relative to Bergma, 2.5 mm from the sagittal line, and 3.5 mm deep from the surface of the skull) (Wan et al. 2018). The cannulation was performed at the target point using a stereotactic device. The cannula was secured with dental cement, and a cap was placed on it to prevent contamination. After regaining consciousness, the animals were transferred to the cages and were observed for two weeks to reduce pain and discomfort. A nozzle 0.5 mm larger than the prepared cannula connected to a 5 μl Hamilton syringe have been used to inject the miR-149-5p mimic and CoQ10. 5 μl of rno-miR-149-5p mimic (MIMAT0035726, MC12788, Thermo Fisher, USA) and mirVana™ miRNA mimic negative control #1 with a concentration of 0.5 nmol were injected into the right lateral ventricle by Lipofectamine™ RNAiMax (Thermo Fisher Scientific) in the miR treatment group according to manufacturer's instructions (Ruili et al. 2020). The injection was performed a week at the same time, followed by the behavioral studies. In The Q10 treatment group, Q10 was administered orally by gavage by the 200 mg/kg/day dose for two weeks (Prajapati et al. 2017).
Rotation test
This test is based on dopamine receptors' sensitivity in the substantia nigra to dopamine and dopamine agonists agents such as apomorphine. Hence, the animals begin to rotate towards the opposite direction of the affected area after apomorphine injection. The number of rotations is directly related to the extent of the lesion. The animals' rotational behavior was measured by apomorphine injection (0.5 mg/kg, i.p) both before and after treatments consecutively. The rats were fixed in a rotameter (Borj sanat azma RT-5300, Tehran, Iran) for 5 min to adapt. 1 mg/kg of apomorphine hydrochloride dissolved in saline was injected intraperitoneally into rats, and the number of rotations towards the opposite direction of the lesion (left) was measured using a rotameter. Data are expressed in the form of a complete rotation of the body every minute. (Outcome of rotations: rotations towards the direction of lesion—rotations towards the opposite direction of the lesion) (Mostafavi et al. 2019). The animals were evaluated at week zero (two weeks after Parkinson's induction and right before treatment), second (two weeks after treatment), and fourth (four weeks after treatment).
Rotarod test
The rats' motor-balance coordination was evaluated by the Rotarod device (Stoelting USA) at week zero, second and fourth after treatment. Adopting the animals was accomplished through two exercises for two consecutive days (4 rpm on the first day and 15 rpm on the second day). The device automatically starts recording time 0.1 s after the rat is placed on the rotating rod. The device's rotation speed increased from 4 to 40 rpm in 180 s, and the animals' motor coordination was assessed. Each rat's retention time on the rotating rod was recorded after three replications with an interval of 5 min (Mostafavi et al. 2019).
Open field test
The improvement of cognitive activities of the treated animals has been executed through an Open field test. The animals were first adopted to the device. PD-induced rats were placed in the center of the open field device (OPF; insight model open field EP 154C, Borj Sanat azma RT-5300, Tehran, Iran), and their performance was evaluated through the frequency of movements recording for 5 min (Mostafavi et al. 2019).
RT-qPCR
Quantitative Real-Time PCR (RT-qPCR) was used to evaluate the expression of miR-149-5p and MMP-2,9. Briefly, the total RNA was extracted using Trizol. The nanodrop device (Thermo Scientific NanoDrop 2000c) has been employed to measure the quantity and purity of the extracted RNA. According to the manufacturer's instructions, the mRNA/miRNA cDNA Synthesis Kit (Stem Cell Technology Research Center BON209002) has been used to synthesize cDNA. Real Plus 2 × Master Mix green and Real-Time PCR Instrument (Applied BiosystemsTM) were recruited to the miR-149-5p, MMP-2, and MMP-9 genes using specific primers (Table 1).
Immunohistochemistry analysis
Tyrosine hydroxylase (TH) has been determined through Immunohistochemistry analysis. In summary, the rats were deeply anesthetized using Ketamine/xylazine (100/10 mg/kg, i.p.) injection. Then, were transcranial perfused using 250 ml sodium chloride 0.9% and 100 ml formaldehyde 4%, respectively. The brains were removed, fixed in formalin 4%, and embedded in paraffin. After preparing thin brain sections, xylene was used for deparaffinization. The sections were then dehydrated in a serial dilution of ethanol (100%, 96%, and 70% ethanol). Next, the sections were treated with 0.5% Triton X-100 for 10 min, followed by 12 h incubation at 4 °C with TH primary antibodies. The sections were also incubated with the PE-conjugated IgG as the secondary antibody for 1 h at room temperature. Diaminobenzidine (DAB) staining was performed on the slides, the sections were incubated with the DAB solution for 10 s at room temperature and were analyzed by a fluorescence microscope (Olympus, Japan).
Statistical analysis
All statistics were reported as mean ± SD. The One-Way ANOVA and Tukey post hoc test was used to analyze data, and p < 0.05 was considered the significant threshold.
Results
miR-149-5p expression
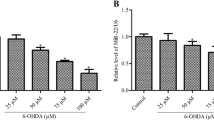
According to the qRT-PCR results, PD induction has significantly decreased the miR level in the striatum region than the sham group (Fig. 2a). However, miR-mimic and CoQ10 treated rats have been led to upregulation of miR-149-5p (Fig. 2a, b). In contrast, the use of Q10 and S-miR solvents did not affect the expression of miR-149-5p (Fig. 2c).
miR-149-5p alterations in the striatum region. PD induction result in the miR-149-5p significant decrease in the PD group (*P < 0.05 compared to the sham group). miR-mimic injection resulted in miR considerable increase in the rat's striatum (**P < 0.05 compared to the PD group) (a). The use of CoQ10 leads to a significant rise in miR-149-5p level in the treated rats' striatum compared to the PD group (#P < 0.05 compared to the PD group) (b). The use of CoQ10 and S-miR solvents did not affect the miR-149-5p level (C) (*P < 0.05 compared to the sham group, **P < 0.05 compared to PD and vehicle groups, #P < 0.05 compared to PD and NC groups). Values include mean ± standard deviation (n = 6)
Rotation tests
According to the obtained results, 6-OHDA injection leads to the force rotation in the opposite lesion direction in all study groups (Fig. 3a). However, a significant reduction in the rotations number was observed following miR-149-5p mimic and CoQ10 administration compared to the PD rats two weeks after treatments. On the other hand, Q10 (vehicle group) and S-miR (NC group) injection did not represent any significant decrease in the rotations number compared to the PD rats (Fig. 3b). The same reduction in the rotations number has been observed in subsequent weeks (Fig. 3c). However, the effect of miR-149-5p mimic and CoQ10 administration have shown a time-dependent manner regarding the rotation number in animals. The treated groups showed a significant decrease in the fourth week compared to the second week and week zero, and a significant difference was revealed between the rotations number in the second week and week zero (Fig. 3d).
Graphs are comparing the rotations number between groups during different weeks. 6-OHDA injection resulted in apomorphine-induced rotations in the opposite direction of the lesion in animals (*P < 0.05 compared to the sham group) (a). Two weeks after miR-mimic and coQ10 treatment, the rotations number significantly decreased in the treatment groups compared to the PD group (b). The same reduction was also observed two weeks later (week 4) (c) (*P < 0.05 compared to the sham group, **P < 0.05 compared to PD and vehicle groups, #P < 0.05 compared to PD and NC groups). The number of rotations in both treatment groups in weeks 2 and 4 showed a significant decrease compared to week zero. Also, a significant difference was observed in the treatment groups between weeks 2 and 4 (*P < 0.05 compared to week two and zero, **P < 0.05 compared to week zero) (d). Values include mean ± standard deviation (n = 6)
Rotarod test
A significant reduction of retention time on the rotating rod has been indicated for rats following PD induction than the sham group (Fig. 4a). Nevertheless, miR-mimic and CoQ10 treatment promoted rats' retention time on the rotating rod compared to the PD rats group, but there was no significant difference in NC and vehicle groups compared to PD rats (Fig. 4b). The balance improvement of the miR-mimic and CoQ10 treated groups compared to the PD group was also observed at week fourth (Fig. 4c). Notwithstanding, the retention time of miR-149-5p mimic and CoQ10 treated groups significantly increased in the fourth week compared to the second week and week zero and in the second week compared to week zero (Fig. 4d).
Comparison of differences in the rats' balance before, after and during different treatment weeks. 6-OHDA injection reduced the rats' retention time on the rotating rod (*P < 0.05 compared to the sham group) (a). Two weeks after miR-mimic and CoQ10 treatment, there was a significant improvement in the animal balance compared to the PD group (b). The same trend was also observed in the performed test two weeks later (week 4) (c) (*P < 0.05 compared to the sham group, **P < 0.05 compared to PD and vehicle groups, #P < 0.05 compared to PD and NC groups). The animals' retention time length on the rotating rod had a significant increase in both treatment groups in weeks 2 and 4 compared to week 0. Also, a significant difference was observed in the treatment groups between weeks 2 and 4 (d) (*P < 0.05 compared to week 2 and 0 **P < 0.05 compared to weeks 0 and 2). Values include mean ± standard deviation (n = 6)
Open field test
PD induction following 6-OHDA injection has reduced the mobility and locomotion frequency of rats (Fig. 5a). CoQ10 and miR-mimic treatments have significantly improved the rats' functional ability and movement frequency two and four weeks after Parkinson's induction, compared to the PD group (Fig. 5b,c). From a temporal perspective, the CoQ10 and miR-mimic treated animals' movement frequency significantly increased in week fourth compared to week second and zero and in week second compared to week zero (Fig. 5d).
analyzing the movement frequencies. PD induction reduced the movement frequencies (*P < 0.05 compared to the sham group) (a). Two weeks after miR-mimic and CoQ10treatment of animals, the rats motor abilities significantly improved compared to the PD group (b), and the same improvement was also observed four weeks after treatment (c) (*P < 0.05 compared to the sham group, **P < 0.05 compared to PD and vehicle groups, #P < 0.05 compared to PD and NC groups). The increase in the fourth and second weeks compared to week zero was significant, and also a significant difference was observed between the movement frequencies of the fourth and third weeks between the treated groups (d) (*P < 0.05 compared to weeks two and zero, **P < 0.05 compared to week zero and 2). Values include mean ± standard deviation (n = 6)
Gene analysis
Gene expression analysis has indicated significant MMP-2,9 upregulation following MFB degradation in PD rats compared to the sham group. CoQ10 and miR-mimic treatment resulted in a considerable decrease of MMP-2,9 in rats than the PD rats' group. No significant difference was observed between vehicle and NC groups compared to the PD group (Fig. 7a, b).
Histochemical analysis
TH+ cells have been investigated through histochemical analysis. PD induction significantly decreased the TH+ cell number in rats' striatum compared to healthy rats' striatum. 6-OHDA injection reduced the number of TH+ cells (Fig. 6a, c). There was a significant increase in TH expression in the CoQ10 and miR-mimic receiving rat's striatum region compared to the PD group (Fig. 6b, c).
Differences in the striatum TH expression (100 µm). 6-OHDA injection and MBF degradation resulted in a significant reduction in the number of striatum TH+ cells (a, c) (*P < 0.05 compared to the sham group). Injection of miR-mimic and CoQ10 resulted in a significant increase in the striatum region TH+ cells number (b, c) (**P < 0.05 compared to the PD and vehicle groups, #P < 0.05 compared to the PD and NC groups). Values include mean ± standard deviation (n = 4)
Discussion
According to the previous studies, miR-149-5p reduced level following brain damages such as stroke and Alzheimer's disease can be elevated by a miR-149-5p mimic (Wan et al. 2018; Chen et al. 2020). It has been reported miR-mimic treatment following cerebral ischemia has increased miR-149-5p expression that eventually promoted the BBB integrity and reduced neuronal death (Wan et al. 2018; Xiong et al. 2020). Here in the present study, the miR-149-5p level has been reduced through PD induction, which plays an essential role in PD development. Even so, miR-mimic and CoQ10 injection leads to an upregulation of miR-149-5p in PD rats (Fig. 1). Various studies have investigated the effect of CoQ10 in different miRs regulation. CoQ10 has been shown to regulate the miR-378 levels by affecting Activator Protein-1 (Wang et al. 2014). Besides, the modulatory effect of Q10 on the miR-146a expression has been indicated through inflammatory pathways (Olivieri et al. 2013). In a comprehensive study, the impact of Q10 consumption has been investigated on more than 100 miRs expression for four years. The results have signified the Q10 modulatory effect on various miRs, both upregulation and downregulation. Among the upregulation miRgs, 2.2, 3, and twofold increasing of miR-29b-3p, miR-19a, and a miR-30e-3p, respectively, were outstanding (Alehagen et al. 2017). Each has been reported to be a vital element in different pathological conditions. Rapid reduction of miR-29b-3p and miR-30e-3p level have been observed in myocardial injury compared to normal conditions (Singh et al. 2007; Fan et al. 2017) and the level of miR-19a have been represented to be downregulated in patients with cerebral ischemia (Jickling et al. 2014). Because Q10 is influenced the pathological conditions by upregulating the reduced miRs level, such as decreased miR-149-5p in brain injuries, it seems that Q10 reducing the PD progression through miR-149-5p upregulate (Fig. 7).
MMP-2,9 change comparison chart. PD modeling leads to increased MMP-2 expression and treatment of rats using miR-mimic and CoQ10 resulted in a significant reduction in the level of this enzyme in striatum compared to the model group, NC and vehicle (a). Significant increase in MMP-9 striatum level of PD rats was observed, which due to CoQ10 and miR-mimic injection, its level decreased significantly compared to untreated groups.) (#P < 0.05 compared to sham group, ##P < 0.05 compared to the model and vehicle groups, *P < 0.05 compared to the model and NC groups). Values include mean ± standard deviation (n = 4)
It has been shown that the promotion of downregulated miRs following PD has a vital impact on mobility and TH expression. In this regard, the decreased miR-124-3p level in animals PD brain has been upregulated through miR-124-3p mimic stimulation, leading to TH high expression and improved motor abilities (Li et al. 2020). miR-29c is another studied molecule that downregulated in PD and has been successfully upregulated using miR-29c mimic, which in turn causes increasing TH expression in the striatum region and inducing a protective effect against dopaminergic neurons damages(Ruili et al. 2020). In line with previous studies, our results indicated that miR-149-5p high expression following miR-mimic and Q10 administration leads to raising TH expression in the striatum and improving rats' motor abilities.
Several studies have represented that miR-149-5p and CoQ10 can affect the pathways involved in apoptosis and thus prevent cell death. For example, miR-149-5p-mimic leads to increased miR-149-5p, which cased dimenshing the expression of P53 and a variety of caspases. In this regard, one of the reduced caspases is attributed to caspase-3, an involved factor in mitochondrial-mediated cell death and thus reduced neuronal death (Teertam et al. 2020). CoQ10 inhibits mitochondrial-induced cell death and increases neuronal survival by maintaining mitochondrial membrane stability, inhibiting cytochrome C release, and suppressing caspase-9 (Prajapati et al. 2017). Furthermore, Zamani et al. (2012) Also reported that CoQ10 administration reduced apoptosis following cerebral ischemia by modulating the BAX-Bcl-2 signaling cascade. Thus, we hypothesize that our interventions may increase TH cells by reducing neuronal death. Park et al. (2020) reported that CoQ10 injection into the striatum and its oral administration significantly reduced the number of asymmetric rotations in animals diagnosed with PD and significantly increased TH expression in the striatum and substantia nigra compared to the PD group. Besides, Q10 has been revealed to prevent PD development through improving motor functions, including reduced stiffness, increased muscle function, and swimming ability in animals with PD (Guapta et al. 2018). Although, 14 days orally administration of Q10 has increased animals' motor capabilities, balance, and body's antioxidant capacity (Prajapati et al. 2017). Inconsistent with the previous studies, at the present study, the treated rats with CoQ10 represented decreased rotations numbers, increased animal balance, increased frequency of movements, and increased TH expression in the striatum that ascribed as the improved motor functions.
MMPs expression, particularly MMP-2,9, have been observed to be upregulated in various brain disorders, including PD that have an important role in the PD complications. Twenty-four hours after PD induction, the MMP-9 level has increased with the rate of 1.8 and 2.2-fold in substantia nigra and striatum, and have been raised 2.5-fold in the SN reached 48 h after PD induction (Annesa et al. 2015). Chen et al. (2008) showed that the MMP-9 gelatinase activity increased in PD animals' striatum, leading to BBB damages and PD complications development through tight junctions degradation. BBB disruption due to the MMP-9 levels increase following PD caused CNS homeostasis disruption and mitophagy inhibition, leading to damaged mitochondria accumulation, cell death promotion, and PD progression. Thus, MMP-9 inhibitor can be used for BBB integrity improvement and eventually functional abilities promotion of animals (Lin et al. 2020). Furthermore, studies showed that MMP-2,9 could be targeted by miR-149-5p through the 3′-UTR region of the mRNA and decreased. (Feng et al. 2019; Nyariki et al. 2019; Lin et al. 2020). On the other hand, Q10 has reduced the MMP-2,9 level in malaria, leading to oxidative stress and neuroinflammation abatement (Nyariki et al. 2019). The present study has shared similar results, in which miR-149-5p uprising following miR-mimic and CoQ10 injection and subsequently MMP-2,9 decreasing, could improve the BBB integrity and reduces the PD complications.
Studies have shown that neurodegenerative agents such as caspase-3 and BAX increased during the early days following the 6-OHDA injection. Although, the Bcl-2 activity decreases in a time-dependent manner, leading to TH+ cells number decrease during 3 to 14 days after the neurotoxin injection (Chi et al. 2019). It was also found that PD induction by neurotoxin injection resulted in about a 58% decrease of the neuronal population compared to the opposite hemisphere (Haddadi et al. 2020).
By examining the rat's behavior in different groups during consecutive weeks after treatment, it may be stated that the protective effects of CoQ10 and miR-149-5p are likely to begin during the early stages (two weeks after treatment). It is due to the significant difference in the number of rotations and motor performance of rats in the second week after treatment that the time before treatment and compared to the model group (Figs. 3, 4, 5). Therefore, it appears that CoQ10 in the early stages of disease progression can reduce the expression of MMPs by inducing its antioxidant effects and thus prevent further damage to the BBB. However, further studies need to reveal other mechanisms involved in the protective development of CoQ10. On the other hand, by upregulation of miR-149-5p by CoQ10 and its mimic, the favorite effects may continue in the following weeks because a significant difference between rats' motor and behavioral actions was revealed in the next weeks.
PD is a common neurodegenerative disease with a complex mechanism. Hence the detailed molecular mechanisms exploration may be useful for emerging new therapeutic or diagnostic strategies (Ruili et al. 2020). Our results indicted PD complications reduction through MMP-2,9 inhibition. Despite the expanding MMP inhibitors' use, their harmful side-effects, including abnormal blockade, should be vigorously considered (Annesa et al. 2015). Nonetheless, using biological pathways to inhibit MMPs could be a potential therapeutic strategy for PD. Here at the present study, we have shown for the first time that miR-149-5p mimic and CoQ10 improved PD complications by reducing the involved factors in disrupting BBB integrity. Besides, miR-149-5p mimic and CoQ10 treatments may lead to dopaminergic neurons protect against PD induction through TH high expression in the striatum and reduced the motor complications caused by PD.
Data availability
The analyzed data of this study will be made available by the corresponding author upon reasonable request.
Abbreviations
- PD:
-
Parkinson's disease
- MMP:
-
Matrix metalloproteinase
- BBB:
-
Blood brain barrier
- miR:
-
MicroRNA
- miR-mimic:
-
MiRNA-149-5p mimic
- CoQ10:
-
Coenzyme Q10
- 6-OHDA:
-
6-Hydroxypopamine
- MFB:
-
Medial Forbrain Bundle
- qRT-PCR:
-
Quantitative real time-polymerase chain reaction
- TH:
-
Tyrosine hydroxylase
- 3′-UTR:
-
3′-Untranslated region
- S-miR:
-
Scramble microRNA
- NC:
-
Negative control
References
Alehagen U, Johansson P, Aaseth J, Alexander J, Wagsater D (2017) Significant changes in circulating microRNA by dietary supplementation of selenium and coenzyme Q10 in healthy elderly males. A subgroup analysis of a prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. PLoS ONE 12(4):e0174880. https://doi.org/10.1371/journal.pone.0174880
Annesa V, Herrero MT, Di-Pentima M, Gomez A, Lombardi A, Ros CM et al (2015) Metalloproteinase-9 contributes to inflammatory glia activation and nigro-striatal pathway degeneration in both mouse and monkey models of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced Parkinsonism. Brain Struct Funct 220:7.3-27. https://doi.org/10.1007/s00429-014-0718-8
Belhaj N, Dupuis F, Arab-Tehrany E, Denis FM, Paris C, Lartaud I et al (2012) Formulation, characterization and pharmacokinetic studies of coenzyme Q10PUFA’s nanoemulsions. Eur J Pharm Sci 47(2):305–312. https://doi.org/10.1016/j.ejps.2012.06.008
Cabezase R, Avila M, Gonzalez J, El-Bacha RS, Baez E, Garcia-Segura LM et al (2014) Astrocytic modulation of blood brain barrier: perspectives on Parkinson’s disease. Front Cell Neurosci. https://doi.org/10.3389/fncel.2014.00211
Caggiu E, Paulus K, Mameli G, Arru G, Sechi GP, Sechi LA (2018) differential expression of miRNA 155 and miRNA 146a in Parkinson’s disease patients. eNeurologicalSci 13:1–4. https://doi.org/10.1016/j.ensci.2018.09.002
Chen X, Lan X, Roche L, Liu R, Geiger JD (2008) Caffeine protects against MPTP-induced blood-brain barrier dysfunction in mouse striatum. J Neurochem 107(4):1147–1157. https://doi.org/10.1111/j.1471-4159.2008.05697.x
Chen F, Chen H, Jia Y, Lu H, Tan Q, Zhou X (2020) miR-149-5p inhibition reduces Alzheimer’s disease β-amyloid generation in 293/APPsw cells by upregulating H4K16ac via KAT8. Exp Therap Med. https://doi.org/10.3892/etm.2020.9216
Chi H, Guan Y, Li F, Chen Z (2019) The effect of human umbilical cord mesenchymal stromal cells in protection of dopaminergic neurons from apoptosis by reducing oxidative stress in the early stage of a 6-OHDA-induced Parkinson’s disease model. Cell Transplant 28(1s):875–995. https://doi.org/10.1177/0963689719891134
Contin M, Flor S, Martinefski M, Lucangioli S, Tripodi V (2015) New analytical strategies applied to the determination of Coenzyme Q10 in biological matrix. Methods Mol Biol 1208:409–420. https://doi.org/10.1007/978-1-4939-1441-8_29
Fan L, Feng U, Chen GC, Qin LQ, Fu CL, Chen LH (2017) Effects of coenzymeQ10 supplementation on inflammatory markers: a systematic review and meta-analysis of randomized controlled trials. Pharmacol Res 119:128–136. https://doi.org/10.1016/j.phrs.2017.01.032
Feng Q, Zhang H, Nie X, Li Y, Chen W-D, Wang Y-D (2019) miRNA-149* suppresses liver cancer progression by down-regulating tumor necrosis factor receptor 1-associated death domain protein expression. Am J Pathol 190(2):469–483. https://doi.org/10.1016/j.ajpath.2019.10.010
Fox SH, Katzenschlager R, Lim S-Y, Barton B, de Bie RMA, Seppi K et al (2018) International Parkinson and movement disorder society evidence-based medicine review: update on treatments for the motor symptoms of Parkinson’s disease. Mov Disord 33(8):1248–1266. https://doi.org/10.1002/mds.27372
Ghasemloo E, Oryan S, Reza M, Hossein B, Mehdi M, Eskandari (2021) The neuroprotective effect of MicroRNA-149-5p and coenzymeQ10 by reducing levels of inflammatory cytokines and metalloproteinases following focal brain ischemia in rats. Brain Res Bull 169205-213 https://doi.org/10.1016/j.brainresbull.2021.01.013
Guapta BK, Kumar S, Kuar H, Ali J, Baboota S (2018) Attenuation of oxidative damage by coenzyme Q10 loaded nanoemulsion through oral route for the management of Parkinson’s disease. Rejuv Res 21(3):232–248. https://doi.org/10.1089/rej.2017.1959
Jankovic J (2019) Pathogenesis-targeted therapeutic strategies in Parkinson’s disease. Mov Disord 34(1):41–44. https://doi.org/10.1002/mds.27534
Jickling GC, Ander BP, Zhan X, Noblett D, Stamova B, Liu D (2014) microRNA expression in peripheral blood cells following acute ischemic stroke and their predicted gene targets. PLoS ONE 9(6):e99283. https://doi.org/10.1371/journal.pone.0099283
Li Y, Gong Z, Jin X, Zhao P, Zhang Y, Wang Z (2020) LncRNA MALAT1 targeting miR-124-3p regulates DAPK1 expression contributes to cell apoptosis in Parkinson’s disease. Cell Biochem 121(12):4838–4848. https://doi.org/10.1002/jcb.29711
Lin C, Wu W, Lu H, Li W, Bao Z, Wang Y et al (2020) MMP-9 inhibitor GM6001 prevents the development of ssTBI-induced Parkinson’s disease via the autophagy pathway. Cell Mol Neurobiol 40(6):66. https://doi.org/10.1007/s10571-020-00933-z
Lu CJ, Guo YZ, Zhang Y, Yang L, Chang Y, Zhang JW, Jing L, Zhang JZ (2017) Coenzyme Q10 ameliorates cerebral ischemia-reperfusion injury in hyperglycemic rats. Pathol Res and Pract 213(9):1191–1199. https://doi.org/10.1016/j.prp.2017.06.005
Mancuso M, Orsucci D, Volpi L, Calsolaro V, Siciliano G (2010) Coenzyme Q10 in neuromuscular and neurodegenerative disorders. Curr Drug Targets 11(1):111–121
Mendes Filho D, Ribeiro P, Oliveira LF, Paula DR, Capuano V, Assunção SF et al (2018) Therapy with mesenchymal stem cells in Parkinson disease. Neurol J 23(4):141–147. https://doi.org/10.1097/NRL.0000000000000188
Mostafavi H, Ghasemifard L, Rostamo A, Alipour A, Nadri S (2019) Trabecular meshwork mesenchymal stem cell transplantation improve motor symptoms of parkinsonian rat model. Biologicals 61:61–67. https://doi.org/10.1016/j.biologicals.2019.06.006
Mugoni V, Postel R, Catanzaro V, De Luca E, Turco E, Digilio G et al (2013) Ubiad1 is an antioxidant enzyme that regulates eNOS activity by CoQ10 synthesis. Cell 152(3):504–518. https://doi.org/10.1016/j.cell.2013.01.013
Nation DA, Sweeney MD, Montagne A, Sagare AP, D’Orazio LM, Pachicano M et al (2019) Blood–brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat Med 25(2):270–276. https://doi.org/10.1038/s41591-018-0297-y
Nyariki JN, Ochola AL, Jillani NE, Nyamweya NO, Amwayi PE, Yole DS (2019) Oral administration of Coenzyme Q10 protects mice against oxidative stress and neuro-inflammation during experimental cerebral malaria. Parasitol 71:106–120. https://doi.org/10.1016/j.parint.2019.04.010
O’Brien J, Hayder H, Zayed Y, Peng C (2018) Overview of MicroRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol. https://doi.org/10.3389/fendo.2018.00402
Obeso JA, Stamelou M, Goetz CG, Poewe W, Lang AE, Weintraub D et al (2017) Past, present, and future of Parkinson’s disease: a special essay on the 200th anniversary of the shaking palsy. Mov Disord 32(9):1264–1310. https://doi.org/10.1002/mds.27115
Oh SE, Park HJ, He L, Skibiel C, Junn E, Mouradian MM (2018) The Parkinson’s disease gene product DJ-1 modulates miR-221 to promote neuronal survival against oxidative stress. Redox Biol 19:62–73. https://doi.org/10.1016/j.redox.2018.07.021
Olivieri F, Lazzarini R, Baini L, Prattichizzo F, Rippo MR, Tiano L et al (2013) Anti-inflammatory effect of ubiquinol-10 on young and senescent endothelial cells via miR-146a modulation. Free Radic Biol Med 63:410–420. https://doi.org/10.1016/j.freeradbiomed.2013.05
Park HW, Park CG, Park M, Lee SH, Park HR, Lim J et al (2020) Intrastriatal administration of coenzyme Q10 enhances neuroprotection in a parkinson’s disease rat model. Sci Rep. https://doi.org/10.1038/s41598-020-66493-w
Prajapati SK, Garabadu D, Krishnamurthy S (2017) Coenzyme Q10 prevents mitochondrial dysfunction and facilitates pharmacological activity of atorvastatin in 6-OHDA induced dopaminergic toxicity in rats. Neurotox Res 31:478–492. https://doi.org/10.1007/s12640-016-9693-6
Qadir MI, Bukhat S, Rasul S, Manzoor H, Manzoor M (2020) RNA therapeutics: identification of novel targets leading to drug discovery. J Cell Biochem 121(2):898–929. https://doi.org/10.1002/jcb.29364
Ruili W, Ying Y, Hui W, Ya H, Chen L (2020) MiR-29c protects against inflammation and apoptosis in Parkinson’s disease model in vivo and in vitroby targeting SP. Clin Exp Pharmacol Physiol 47(3):372–382. https://doi.org/10.1111/1440-1681.13212
Schmelzer C, Lindner I, Rimbach G, Niklowitz P, Menke T, Doring F (2008) Functions of coenzyme Q10 in inflammation and gene expression. BioFactors 32(1–4):179–183. https://doi.org/10.1002/biof.5520320121
Seet RC, Lim EC, Tan JJ, Quek AM, Chow AW, Chong WL et al (2014) Does high-dose coenzyme Q10 improve oxidative damage and clinical outcomes in Parkinson’s disease? Antioxid Redox Signal 21:211–217. https://doi.org/10.1089/ars.2013.5801
Singh U, Devaraj S, Jialal I (2007) CoenzymeQ10 supplementation and heart failure. Nutr Rev 65(96):286–293. https://doi.org/10.1111/j.1753-4887.2007.tb00306.x
Sweeney MD, Sagare AP, Zlokovic BV (2018) Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nature Rev Neurol 14:133–150. https://doi.org/10.1038/nrneurol.2017.188
Teertam SK, Jha S, Babu PP (2020) Up-regulation of Sirt1/miR-149–5p signaling may play a role in resveratrol induced protection against ischemia via p53 in rat brain. Clin Neurosci 72:402–11. https://doi.org/10.1016/j.jocn.2019.11.043
Titze-de-Almeida R, Titze-de-Almeida SS (2018) miR-7 replacement therapy in Parkinson’s disease. Curr Gene Ther 18(3):143–153. https://doi.org/10.2174/1566523218666180430121323
Tolosa E, Botta-Orfila T, Morat X, Calatayud C, Ferrer-Lorente R, Martí MJ et al (2018) MicroRNA alterations in iPSC-derived dopaminergic neurons from Parkinson disease patients. Neurobiol Aging 69:283–291. https://doi.org/10.1016/j.neurobiolaging.2018.05.032
Wan Y, Jin HJ, Zhu YY, Fang Z, Mao L, He Q, Xia YP, Li M et al (2018) MicroRNA-149-5p regulates blood–brain barrierpermeability after transient middle cerebral arteryocclusion in rats by targeting S1PR2 of pericytes. FASEB J 32(6):3133–3148. https://doi.org/10.1096/fj.201701121R
Wang D, Yan X, Xia M, Yang Y, Li D, Li X et al (2014) Coenzyme Q10 promotes macrophage cholesterol efflux by regulation of the activator protein-1/miR-378/ATP-binding cassette transporter G1–signaling pathway. Arterioscler Thromb Vasc Biol 34(9):1861–1870. https://doi.org/10.1161/ATVBAHA.113.302879
Winkler L, Blasig R, Breitkreuz-Korff O, Berndt F, Dithmer S, Helms HC et al (2021) Tight junctions in the blood–brain barrier promote edema formation and infarct size in stroke—ambivalent effects of sealing proteins. Cereb Bllod Flow and Metabol 41(1):132–145. https://doi.org/10.1177/0271678X20904687
Xiong L, Zhou H, Zhao Q, Xue L, Al-Hawwas M, He J, Wu M (2020) Overexpression of miR-124 protects against neurological dysfunction induced by neonatal hypoxic-ischemic brain injury. Cell Mol Neurobiol 40(5):737–750. https://doi.org/10.1007/s10571-019-00769-2
Zamani M, Katebi M, Mehdizadeh M, Mohamadzadeh F, Soleimani M (2012) Coenzyme Q10 protectshippocampal neurons against ischemia/reperfusion injury via modulationof BAX/Bcl-2 expression. Basic Clin Neurosci 3(5):5–10
Acknowledgements
This study was supported by Grant No: A-12-871-10 from Deputy of Research and Technology, Zanjan University of Medical Sciences (Ethical No: IR.ZUMS.REC.1400.005).
Author information
Authors and Affiliations
Contributions
Elham Ghasemloo: Conceptualization, Methodology, Software, Validation, Writing-Reviewing, Resources. Hossein Mostafavi: Conceptualization, Methodology, Data curation, Software, Validation, Writing-Reviewing, Investigation, Project administration, Supervision. Meysam Forouzandeh: Methodology, Writing-Original draft preparation and Editing. Masoumeh Hosseini: Data curation, Software, Validation, Writing-Reviewing. Mehdi Eskandari: Software, Validatio. Seyyed Saeid Mousavi: Software, Validatio.
Corresponding author
Ethics declarations
Conflict of interest
The author declare that they have no conflict of interest.
Ethics approval
The animals were tested according to the guidelines of the International Organization for Medical Science Working with Laboratory Animals. The protocol for working with animals was approved by the ethics committee of Zanjan University of Medical Sciences.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghasemloo, E., Mostafavi, H., Hosseini, M. et al. Neuroprotective effects of coenzyme Q10 in Parkinson's model via a novel Q10/miR-149-5p/MMPs pathway. Metab Brain Dis 36, 2089–2100 (2021). https://doi.org/10.1007/s11011-021-00795-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00795-4