Abstract
Hepatic encephalopathy (HE) is a neuropsychiatric syndrome which develops in patients with severe liver diseases and/or portal-systemic shunting. Minimal HE, the earliest manifestation of HE, has drawn increasing attention in the last decade. Minimal HE is associated with a series of brain functional changes, such as attention, working memory, and so on. Blood oxygen level dependent (BOLD) functional MRI (fMRI), especially resting-state fMRI has been used to explore the brain functional changes of HE, yielding important insights for understanding pathophysiological mechanisms and functional reorganization of HE. This paper briefly reviews the principles of BOLD fMRI, potential applications of resting-state fMRI with advanced post-processing algorithms such as regional homogeneity, amplitude of low frequency fluctuation, functional connectivity and future research perspective in this field.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatic encephalopathy (HE) is a common and serious neuropsychiatric syndrome that develops in patients with severe liver diseases and/or portal-systemic shunting as a serious complication of acute liver failure and chronic liver failure (Córdoba 2011). Clinically overt HE is characterized by a wide spectrum of clinical manifestations from alterations of mild upsets to stupor and coma (Córdoba 2011; McPhail et al. 2012). In the last decade, minimal HE (MHE) has drawn increasing attention, becoming one of focal topics in gastroenterology and hepatology. Early recognition of neurocognitive functional impairment in patients with cirrhosis may allow the avoidance or delay of a disease. One study by Bajaj et al. showed detection of MHE and subsequent treatment with lactulose could substantially reduce societal costs (Bajaj et al. 2012).
Neuroimaging plays an important role in uncovering the neuropathological mechanism of HE (McPhail et al. 2012; Zhang et al. 2013b). For example, [11C](R)-PK11195 positron emission tomography can in vivo map cerebral “peripheral benzodiazepine binding sites” in patients with HE (Cagnin et al. 2006), supporting the role of neuroinflammation in the development of HE. 1H-magnetic resonance spectroscopy studies indicated mild brain edema occurring in the brain of HE patients, indicated by reduced myo-inositol and increased glutamine and glutamate complex level (Zhang et al. 2013b). Recently, blood oxygen level dependent (BOLD) functional MRI (fMRI) was introduced in the studies of HE, which provides some new insights to help clarify the mechanism of neurocognitive dysfunction and brain reorganization of HE. Especially, resting-state fMRI gains more attention in recent years because of its simpler experimental design than task-related fMRI studies. Some advanced algorithms such as regional homogeneity, amplitude of low frequency fluctuation and functional connectivity have been used to analyze the brain functional changes and brain reorganization mechanism in HE patients (Chen et al. 2012a; Qi et al. 2012b; Ni et al. 2012; Lv et al. 2013a). However, these advanced resting-state fMRI investigations of HE are performed at major medical centers. This review will focus on the principles of BOLD fMRI, the applications of above-mentioned advanced post-processing algorithms and future research perspectives in this field.
Clinical features of HE
Clinically overt HE has a wide spectrum of clinical manifestations, ranging from alterations of psychometric performance such as impaired mental state, neuromuscular abnormalities, asterixis to stupor and coma (Córdoba 2011; McPhail et al. 2012). Hyperammonemia and/or neuroinflammation are believed to be involved in the pathogenesis of HE. HE is classified in three types, i.e., (1) type A HE related to acute liver failure; (2) type B HE related to porto-systemic shunts; and (3) type C related to cirrhosis (Ferenci et al. 2002). Clinically, West Haven criteria are used to semiquantitatively grade HE (Córdoba 2011). However, West Haven scale is subjective, other methods such as The Hepatic Encephalopathy Scaling Algorithm (HESA) and the Clinical Hepatic Encephalopathy Staging Scale (CHESS) have been developed to evaluate the severity of HE (Hassanein et al. 2008; Ortiz et al. 2007).
MHE refers to the subtle changes in cognitive function, electrophysiological parameters, cerebral neurochemical/neurotransmitter homeostasis, cerebral blood flow, metabolism, and fluid homeostasis that can be observed in patients with cirrhosis that have no clinical evidence of HE (Amodio et al. 2004). MHE as the earliest manifestation of HE, is estimated to affect up to 60% of patients with cirrhosis and may seriously impair a patient’s daily functioning and quality of life (Riordan and Williams 2010). A large amount of behavioral studies have demonstrated the existence of neuropsychological abnormalities, such as attention, working memory, and fine motor alterations, even in cirrhotic patients without overt HE (Ferenci et al. 2002; Bajaj et al. 2009). No “gold” standard can be used to diagnose MHE, although a battery of psychometric tests are recommended (Ferenci et al. 2002). Thus, it is urgent to develop objective and quantitative methods for early diagnosis of MHE.
The principle of BOLD fMRI
BOLD fMRI is a safe, noninvasive, radiation-free, and repeatable technique and has widespread potential applications in both clinical (such as neurosurgery preoperative planning, neurological or psychiatric diseases) and basic neurosciences to map brain areas of abnormal neuronal activity in processing of different stimuli or performing various cognitive tasks (Zhang et al. 2013b). fMRI can detect BOLD changes in MRI signal that arise when changes in neuronal activity occur following a change in brain state, for example, responding to a stimulus or performing a specific task (Gore 2003). The BOLD fMRI signal change is the result of combined changes of hemoglobin oxygenation and cerebral blood flow during task-induced brain activation for task-related fMRI studies. For resting-state fMRI, it detects spontaneous low frequency fluctuations (<0.1 Hz) in the BOLD signal, which was initially regarded as noise. Subsequently, these spontaneous low-frequency fluctuations were shown to be of neural origin and specialty to gray matter (Raichle 2011). Resting-state fMRI investigates synchronous activations between brain regions that are spatially distinct, occurring even in the absence of a task or stimulus. Detailed introduction of BOLD principle is beyond the scope of this review, some excellent reviews published elsewhere are recommended for readers interested in this topic (Gore 2003; Glover 2011; Logothetis 2008).
BOLD fMRI can be roughly classified to task-related fMRI and resting-state fMRI according to experimental paradigms used. In task-related fMRI design, the subject is placed in the magnet of an MRI machine, where various kinds of stimulus, such as visual or motion stimulus, are continuously administered in a controlled fashion (Zhang et al. 2013b; Gore 2003). Two main experimental paradigms are in common use in task-related fMRI studies: block design and event-related paradigms (Gore 2003). Nearly all task-related fMRI studies on HE used block design fMRI acquisition scheme, focusing on different brain functions such as attention (Zafiris et al. 2004; Zhang et al. 2007a), working memory (Liao et al. 2012), and motion (McPhail et al. 2013). Task-related fMRI is rather difficult even for the patients without overt HE, which limits the generalization of this technique. In contrast to task-related fMRI, resting-state fMRI investigates the brain without overt task performance or stimulation. In resting-state fMRI studies, the subjects are not required to perform specific stimulating task or make response in contrast to task-related fMRI. In resting-state fMRI, the participants are required to close their eyes and keep their heads still and not to think of anything in particular during MRI scans lasting several minutes. Resting-state fMRI could have a potentially broader and faster translation into clinical practice thus it is suitable for investigating neuropathological mechanism of HE and performing longitudinal studies. In recent years, several resting-state fMRI studies on HE have been published (Chen et al. 2012a; Qi et al. 2012b; Ni et al. 2012; Lv et al. 2013a). Resting-state fMRI data acquisition scheme is rather easy, however, data analysis is complex, various data post-processing algorithms which will be introduced later have been developed to resolve different clinical and research issues.
Algorithms for resting-state fMRI studies
Various algorithms has been developed to investigate the brain at the resting state, such as independent component analysis, region of interest analysis, regional homogeneity, amplitude of low frequency fluctuation, whole brain network analysis, and small world network. The two most popular techniques for performing resting-state fMRI studies are independent components analysis and region of interest analysis. Independent components analysis is a data-driven signal processing method based on blind source separation. Independent components analysis analyzes the entire BOLD dataset and decomposes the signals into several independent components that are maximally statistically independent but there are connections between different brain regions (Liu et al. 2008). The independent components analysis separates correspond to each functional network related to mental activities, thus the functional connectivity network can be obtained (McPhail et al. 2013). Compared with seed-based methods, independent components analysis requires few priori assumptions of region of interest but asks the user to manually select the important components and distinguish noise from physiologic signals (Lee et al. 2013). Independent components analysis can only get the spatial distribution of the network but cannot measure the joint strength between the brain regions. Independent components analysis can not simultaneously examine multiple networks (Sheline and Raichle. 2013). Region of interest analysis is the most common method to investigate the functional connectivity of a specific region by selecting this region as a ‘seed’ with a hypothesis-driven algorithm and is able to clearly visualize the anatomy of correlated regions (Liu et al. 2008). The seed-voxel approach consists of extracting the BOLD time course from a ROI and it determines the temporal correlation between this signal (seed) and the time course from all other brain voxels. Seed correlation analysis has been used to define various specific brain networks but it can not simultaneously examine multiple networks. Other advanced algorithms will briefly introduced in the following potential applications of resting-state fMRI.
Potential applications of resting-state fMRI with various algorithms
Various algorithms has been developed to investigate the brain at the resting state, here we introduce recent findings of resting-state fMRI in the studies of HE according to these advanced data post-processing algorithms.
Independent component analysis
Resting state brain network can be extracted by using independent components analysis algorithm. Resting state brain networks normally represent a remarkably robust phenomenon, which are present in rudimentary form early in human life and later reorganize as brain development proceeds through childhood and persist during task performance, sleep, and under sedation (Pizoli et al. 2011). Most frequently reported resting-state network includes the following sub-networks: default mode network, sensorimotor network, executive control network, mesial visual network, lateralized fronto-parietal networks, auditory network, temporo-parietal network (Rosazza and Minati. 2011). These resting-state networks consist of anatomically separated, but functionally connected regions displaying a high level of correlated activity with similar BOLD signal pattern (Rosazza and Minati. 2011). Among the subnetworks, the most studied network is the default mode network, a system of areas involving the precuneus/posterior cingulate, the lateral parietal area and the medial prefrontal cortex (Lee et al. 2013). It has been demonstrated that default mode network is more intensely engaged under resting conditions and relatively de-activated whenever the participant is involved in active tasks. What the default mode network represents is unclear, but it is known to have a high metabolic activity during rest and is suppressed during cognitively demanding tasks, such as visual and auditory attention, language processing, memory, and motoric activity (Zhang et al. 2013b, b). Default mode network is believed to be involved in the core process of human cognition, including the integration of cognitive and emotional processing, monitoring the world around us, and mind-wandering (van den Heuvel and Hulshoff Pol 2010).
Abnormal resting-state brain network of HE patients has been demonstrated by some studies (Zhang et al. 2007b, 2012a; Chen et al. 2013; Qi et al. 2012c). Among the subnetworks of resting-state brain networks, abnormal default mode network in HE patients was first reported. In one study by Zhang et al. (2007b), the default mode network of the cirrhotic patients was investigated with subtraction method using a blocked-design fMRI in which modified Chinese Stroop task was used as the target stimulus. They found the absence of deactivation in the posterior cingulate cortex and precuneus may be a sensitive rather than specific marker in cirrhotic patients. Recently, Zhang et al. (2012a) investigated the default mode network in the patients with HE and its underlying physiological relevance using resting-state fMRI. They found significantly reduced functional connectivity in the right middle frontal gyrus and left posterior cingulate cortex consisting of the default mode network compared to healthy controls and negative correlation between the functional connectivity changes in left angular gyrus and the venous blood ammonia levels. Their findings support the notion of damages in functional organization of the central nervous system in HE patients. Chen et al. (2013) found that the reduced resting-state functional connectivity within default mode network was associated with neurocognitive impairments in MHE compared to healthy controls and after clinical resolution of overt HE and previous overt HE episodes rather than current MHE might be primarily related to brain dysfunction in patients with latent overt HE.
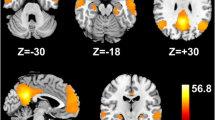
In addition to default mode network, other resting-state subnetworks of cirrhotic patients were investigated. Qi et al. (2012c) applied a resting-state fMRI approach with independent components analysis to assess the differences of resting-state networks between MHE patients and healthy controls. Independent components analysis was used to identify 6 resting state networks [dorsal attention network, default mode network, visual network, auditory network, sensorimotor network, self-referential network] in each subject. They found that MHE patients showed significantly decreased functional connectivity in dorsal attention network, both decreased and increased functional connectivity in default mode network, auditory network and visual network compared to healthy controls. No significant differences were found in self-referential network and sensorimotor network between two groups (Fig. 1). This study indicates MHE patients have selective impairments of resting-state networks, with aberrant functional connectivity in dorsal attention network, default mode network, visual network, auditory network, and spared sensorimotor network and self-referential network.
Group comparison maps of 6 resting state networks between MHE patients, and healthy controls. Compared with the healthy controls, MHE patients showed a generally decreased functional connectivity within dorsal attention network (DAN), both decreased and increased functional connectivity in default mode network (DMN), visual network (VN), and auditory network (AN), as well as unchanged sensorimotor network (SMN) and self-referential network (SRN). Significant thresholds were set at a corrected P<0.05 using AlphaSim program. The sensorimotor network (SMN) and and self-referential network (SRN) had no statistical significance between the two groups
Region of interest analysis
Seed correlation analysis has been used to investigate brain functional connectivity changes in HE patients. Qi et al. (2013b) investigated the thalamic functional connectivity in cirrhotic patients with MHE by using resting-state fMRI with seed-voxel approach. They found MHE patients had disrupted thalamic functional connectivity compared to healthy controls, which mainly located in the bilateral ventral anterior nuclei and ventral posterior medial nuclei. The decreased connectivity between thalamus and many cortices, and basal ganglia indicated reduced integrity of thalamic resting state network in MHE. Qi et al. (2013a) also investigated whether the effective connectivity network associated with basal ganglia is disturbed in HE patients by using resting-state fMRI. They found the patients with low-grade HE had mutually decreased influence between the globus pallidus and the anterior cingulate cortex, cuneus, bi-directionally increased influence between the globus pallidus and the precuneus, and either decreased or increased influence from and to the globus pallidus in many other frontal, temporal, parietal gyri, and cerebellum compared to healthy controls. Blood ammonia levels in HE patients negatively correlated with effective connectivity from the globus pallidus to anterior cingulate cortex, and positively correlated with that from the globus pallidus to precuneus; and the number connection test scores in patients negatively correlated with the effective connectivity from the globus pallidus to anterior cingulate cortex, and from superior frontal gyrus to globus pallidus (Fig. 2). Zhang et al. (2013a) also evaluated the changes of functional connectivity of the anterior cingulate cortex in patients with cirrhosis without overt HE using resting-state fMRI. They found that compared to healthy controls, decreased anterior cingulate cortex functional connectivity predominated with the increasing severity of HE, which was associated with cognitive dysfunction in cirrhotic patients. Zhu et al. (2012) analyzed the functional connectivity between the striatum and the remaining brain regions using seed-based correlation analyses. They found decreased connectivity of the caudate was detected in the anterior/middle cingulate gyrus, and increased connectivity of the caudate was found in the left motor cortex compared to healthy controls. A reduction in functional connectivity was found between the putamen and several regions, including the anterior cingulate cortex, right insular lobe, inferior frontal gyrus, left parahippocampal gyrus, and anterior lobe of the right cerebellum; increased connectivity was detected between the putamen and right middle temporal gyrus. These studies support the role of disrupted basal ganglia-thalamocortical circuit in the development of HE.
Correlations results between the altered effective connectivity of the globus pallidus and blood venous ammonia and neuropsychological performances in low-grade HE patients (P<0.05, uncorrected). Pearson correlation analyses reveals that the blood venous ammonia levels of low-grade HE patients negatively correlate with the decreased effective connectivity from the bilateral globus pallidus to the iso-lateral anterior cingulate cortex (a/b), and positively correlate with the influence from the right globus pallidus to the right precuneus (c). The other regions with aberrant effective connectivity show no correlation with venous blood ammonia levels. In addition, the number connectivity test scores in patients negatively correlated with the effective connectivity from the left globus pallidus to left anterior cingulate cortex (e), and from left superior frontal gyrus to left globus pallidus (f)
Regional homogeneity
Regional homogeneity developed by Zang et al. (2004) measures the functional coherence of a given voxel with its nearest neighbors, which is based on the hypothesis that significant brain activities would more likely occur in clusters than in a single voxel. Regional homogeneity assumes that the hemodynamic characteristics of every voxel within a functional cluster would be similar or synchronous with that of others, and such similarity could be changed or modulated by different conditions (Lv et al. 2013a). Although the exact biological mechanism of regional homogeneity remains uncover, it might reveal the spontaneous brain activity and help improve our understanding of the neuropathological mechanisms underlying many neuropsychological diseases. More recently, Chen et al. (2012c) used regional homogeneity to investigate the resting brain activity in MHE patients, and found abnormal local neuronal activity in MHE patients compared to healthy controls, including decreased and increased regional homogeneity, which was correlated with neurocognitive impairment. Ni et al. (2012) evaluated the evolution pattern of spontaneous brain activities in cirrhotic patients using resting-state fMRI with a regional homogeneity method. They found that diffused abnormal homogeneity of baseline brain activity was nonspecific for MHE, and only the progressively decreased regional homogeneity value in the supplementary motor area and the cuneus, especially for the latter, might be associated with the development of MHE (Fig. 3). Lv et al. (2013a) recently investigated regional activity abnormalities in patients with hepatitis B virus-related cirrhosis without overt HE using resting-state fMRI with regional homogeneity analysis. They found the cirrhosis group showed high regional homogeneity in the prefrontal cortex, and widespread low regional homogeneity value in visual association areas, motor association areas and the bilateral precuneus compared to healthy controls. These studies demonstrate the feasibility of resting-state fMRI with regional homogeneity analysis to detect the progression of cognitive changes and provide information on the pathophysiological mechanisms underlying cognitive alterations in HE patients.
Regional homogeneity differences among non-HE, MHE, and healthy controls (P<0.05, AlphaSim corrected). Compared with the healthy controls, MHE patients show significantly decreased regional homogeneity value in the bilateral frontal lobes including the left anterior cingulate cortex, parietal lobes including the precuneus temporal lobes, occipital lobes including the cuneus and increased regional homogeneity value in the bilateral caudate, and non-HE patients show decreased regional homogeneity value in the bilateral frontal lobes, parietal lobes, temporal lobes and increased regional homogeneity value in the bilateral caudate. Compared with the non-HE patients, the MHE patients show decreased regional homogeneity value in the bilateral parietal lobes including the precuneus, supplementary motor area, frontal lobes and occipital lobes including the cuneus. MHE = minimal hepatic encephalopathy; non-HE = non-hepatic encephalopathy; HC = healthy controls
Amplitude of low frequency fluctuation
Amplitude of low frequency fluctuation developed by Zang et al. (2007) has been widely used in studies of various mental disorders. Although the exact biological mechanism of amplitude of low frequency fluctuation remains unravel to date, these spontaneous low-frequency fluctuation changes in BOLD signal are regarded as being associated with local neuronal activity. Compared with other algorithms such as functional connectivity analysis, amplitude of low frequency fluctuation has the advantage of directly reflecting the amplitude or intensity of spontaneous activity. Resting-state fMRI with amplitude of low frequency fluctuation analysis has been used to detect aberrant brain activity at the baseline state in cirrhotic patients and the progression of HE (Qi et al. 2012b; Lv et al. 2013b; Chen et al. 2012a). For instance, Qi et al. (2012b) explored the spatial patterns of amplitude of low frequency fluctuation in patients with HE of varying severity and to correlate these fluctuations with clinical markers of HE. They found, compared with control subjects, patients with overt and MHE showed decreased amplitude of low frequency fluctuation mainly in regions within the default mode network and increased amplitude of low frequency fluctuation in the cerebellum and middle temporal gyrus. Compared with MHE patients, overt HE patients showed decreased amplitude of low frequency fluctuation in default mode network regions and increased amplitude of low frequency fluctuation in the posterior insular cortex (Fig. 4). They concluded that the levels of decreased amplitude of low frequency fluctuation in the default mode network and increased amplitude of low frequency fluctuation in the posterior insular cortex are dependent on the severity of HE, suggesting continuous impairment of the default mode network and a compensatory role of the insula during the progression of HE.
Amplitude of low frequency fluctuation differences in healthy control, overt HE, and MHE groups (corrected P <0.05). a Comparison of healthy control subjects (Ctrl) and patients with overt HE (OHE) shows significant amplitude of low frequency fluctuation decreases in posterior cingulate cortex, precuneus, medial prefrontal cortex, supplementary motor area and/or superior frontal gyrus, left anterior insular cortex, right postcentral gyrus, left inferior parietal lobule, and right superior parietal lobule (cold color) and increased amplitude of low frequency fluctuation in bilateral posterior insular cortex, bilateral superior temporal gyri, right middle temporal gyrus, and left posterior lobe of the cerebellum (warm color). b Comparison of control subjects (Ctrl) and patients with minimal HE (MHE) shows decreased amplitude of low frequency fluctuation in posterior cingulate cortex, superior frontal gyrus and/or supplementary motor area, bilateral inferior frontal gyri and/or anterior insular cortex, and left IPL and increased amplitude of low frequency fluctuation in right middle temporal gyrus and left posterior lobe of the cerebellum. c Comparison of patients with minimal HE (MHE) and patients with overt HE (OHE) shows significantly decreased amplitude of low frequency fluctuation in posterior cingulate cortex, precuneus, left medial prefrontal cortex, left inferior frontal gyrus, right postcentral gyrus, left inferior parietal lobule, and right superior parietal lobule and increased amplitude of low frequency fluctuation in bilateral posterior insular cortex, bilateral superior temporal gyri, and right middle temporal gyrus. Color scale indicates t values. X and Z represent Montreal Neurologic Institute coordinates of brain regions
Resting-state fMRI with amplitude of low frequency fluctuation analysis has been used to detect change in brain functional connectivity following transjugular intrahepatic portosystemic shunt (TIPS) (Qi et al. 2012d). TIPS is a percutaneously created shunt through the liver parenchyma connecting the right or left portal vein to one of the three main hepatic veins, which is used increasingly in patients with variceal hemorrhage and refractory ascites. HE is one main complication of TIPS procedure, with the prevalence of up to 30% in patients with TIPS insertion (Madoff et al. 2004). The majority of post-TIPS HE happened within the first 3 months (Madoff et al. 2004). Qi et al. (2012d) found patients who underwent the median 8-day follow-up fMRI examinations showed decreased amplitude of low frequency fluctuation in posterior cingulate cortex/precuneus and increased amplitude of low frequency fluctuation in anterior cingulate cortex. In the median 3-month follow up observation, patients displayed persistently decreased amplitude of low frequency fluctuation in posterior cingulate cortex, anterior cingulate cortex and medial prefrontal cortex, while no increased regional amplitude of low frequency fluctuation was observed (Fig. 5). They concluded that TIPS insertion altered cirrhotic patients’ amplitude of low frequency fluctuation patterns in the resting state, which may imply different short-term and moderate-term effects on cirrhotic patients, i.e., both impairment and compensatory mechanism of brain functions in peri-TIPS and continuous impairment of brain function 3 months following TIPS. However, this study is limited to a small patient cohort, and the follow-up examinations are only available for some of the patients after TIPS insertion. Thus, further studies with more patients in each follow up period are warranted to confirm these findings.
Amplitude of low frequency fluctuation differences between patients in pre-TIPS and post-TIPS examinations. In the median 8-day follow-up examinations, post-TIPS patients demonstrated decreased amplitude of low frequency fluctuation in the left middle/posterior cingulate cortex, right superior frontal cortex, left dorsal lateral prefrontal cortex, supplementary motor area and increased amplitude of low frequency fluctuation in the left anterior cingulate cortex and medial prefrontal cortex. In 3 months following TIPS, patients with TIPS insertion showed decreased amplitude of low frequency fluctuation in the left posterior cingulate cortex, left dorsal lateral prefrontal cortex, left anterior cingulate cortex and left medial prefrontal cortex, but no increased regional amplitude of low frequency fluctuation was detected
Whole brain network analysis
The completion of human brain’s advanced activity needs the cooperation of each brain region because each brain region has its own functions, and together they form an organic network to make sense. Thus, it has implication to observe functional connectivity of HE patients from the global perspective. Whole-brain functional connectivity analysis can provide a platform to examine how functional connectivity and information integration relates to human behavior and how this organization may be altered in diseases. Zhang et al. (2012b) investigated patterns of whole-brain functional connectivity in MHE patients by using resting-state fMRI. They found among the 51 connectivity of interest that were significantly different in MHE patients from those in control participants, 44 connectivity were weaker in patients with MHE. All 22 connectivity of interests related to subcortical regions of interest (bilateral putamen, pallidum, and thalamus) were weaker in patients with MHE (Fig. 6). Of 29 cortical connectivity of interests, 22 connectivities were weaker and seven were stronger in patients with MHE. Nearly all connectivity of interests with significant differences correlated with digital symbol test scores (P < .05, false discovery rate corrected). They concluded that MHE patients present widespread cortical and subcortical network connectivity changes that correlated with neuropsychologic impairment. In particular, impairment in the basal ganglia-thalamocortical circuit could play an important role in mediating neurocognitive dysfunction, especially for psychomotor speed and attention deficits in MHE patients.
Axial MR images show functional connectivities in MHE patients between cortical and subcortical regions compared with healthy controls. A, decreased positive functional connectivities between cortical and subcortical regions, B, decreased negative functional connectivities between cortical and subcortical regions. Green nodes = cortical ROIs, red nodes = subcortical ROIs, light blue lines = decreased positive connectivities in patients with minimal HE, dark blue lines = decreased negative connectivities in patients with minimal HE. R = right, L = left, ACG = anterior cingulum gyrus, SMG = supramarginal gyrus, PUT = putamen, PAL = pallidum, THA = thalamus, CUN = cuneus, LING = lingual gyrus, SOG = superior occipital gyrus, MOG = middle occipital gyrus, IOG = inferior occipital gyrus, FFG = fusiform gyrus, INS = insula
Small world network
The small-world network has been demonstrated in human brain by using graph method. Small world networks have high clustering coefficients implying high levels of local connections and a short average path length between any two nodes (Lee et al. 2013). A ‘small-world’ network model can quantify the effectiveness of information transfer within brain networks (He et al. 2009; Hsu et al. 2012), which has been used in various neuropsychiatric diseases such as Alzheimer’s disease, depression, and others. Hsu et al. (2012) investigated abnormalities of neuronal connectivity in patients with cirrhosis using resting-state fMRI with small-world topology analysis. They found that the local and global topological efficiencies of the functional connectivity network were significantly disrupted in HE patients; showing abnormal small-world properties. Alterations in regional characteristics, including nodal efficiency and nodal strength, occurred predominantly in the association, primary, and limbic/paralimbic regions. The degree of network organization disruption depended on the severity of HE. Ammonia levels were also significantly associated with the alterations in local network properties. This study indicated that alterations in the resting-state fMRI network topology of the brain were associated with HE grade; and that focal or diffuse lesions disturbed the functional network to further alter the global topology and efficiency of the whole brain network. These findings provide new insights into the neuropathological mechanism underlying functional changes in the brain of HE patients.
Multimodality studies combining resting-state fMRI and other advanced MRI algorithms
Some investigators also performed multimodality studies combining resting-state fMRI and other MRI techniques, such as diffusion tensor imaging or voxel-based morphometry to provide both structural and functional information (Lin et al. 2012; Qi et al. 2012a; Chen et al. 2012b). For example, Lin et al. (2012) characterized and correlated functional abnormalities in the default mode network to cerebral edema using resting-state fMRI and diffusion tensor imaging in cirrhotic patients and found that cerebral edema can be linked to altered cerebral temporal architecture that modifies both within- and between-network connectivity in HE and reduced functional connectivity correlation coefficient in default mode network is associated with behavior and consciousness deterioration. Qi et al. (2012a) combined diffusion tensor imaging and resting-state fMRI to investigate the changes of structural (path length, tracts count, fractional anisotropy and mean diffusivity derived from diffusion tensor imaging tractography) and functional (temporal correlation coefficient derived from resting-state fMRI) connectivity of the default mode network in MHE patients. They found MHE patients showed both decreased fractional anisotropy and increased mean diffusivity in the tract connecting the posterior cingulate cortex/precuneus to left parahippocampal gyrus, and decreased functional connectivity between the posterior cingulate cortex/precuneus and left parahippocampal gyrus, and medial prefrontal cortex compared to healthy controls (Figs. 7 and 8). Mean diffusivity values of the tract connecting posterior cingulate cortex/precuneus to the left parahippocampal gyrus positively correlated to the ammonia levels, the temporal correlation coefficients between the posterior cingulate cortex/precuneus and the medial prefrontal cortex showed positive correlation to the digital symbol tests scores of patients. They concluded that MHE patients have both disturbed structural and functional connectivity within the default mode network. The decreased functional connectivity was also detected between some regions without abnormal structural connectivity, suggesting that the former may be more sensitive in detecting the early abnormalities of MHE. Chen et al. (2012b) investigated the continued influence of overt HE on neurological function by measuring brain resting-state inherent connectivity, based on an investigation of structural abnormalities identified by voxel-based morphometry in cirrhotic patients with a history of overt HE. They found that patients exhibited a decrease in gray matter volume primarily in the midline regions, bilateral insular cortex and caudates, left parahippocampal gyrus, and right cerebellum posterior lobe, while the volume of the bilateral thalamus showed an increase compared to healthy controls. Of these regions, the posterior cingulate cortex with peak atrophy was selected as the seed for the analysis of functional connectivity. Decreased functional connectivity was found in the medial prefrontal cortex, left inferior parietal lobule, and left middle temporal gyrus in the patients. Thus, both functional and structural impairments were evident after apparent recovery from overt HE, demonstrating that brain dysfunction induced by HE persisted after clinical resolution and provided a basis for further evolution of the disease.
Example of diffusion tensor imaging based fiber tractography on one healthy control and one MHE patient. Only three fiber bundles connecting the posterior cingulated cortex/precuneus and medial prefrontal cortex, bilateral parahippocampal gyri were detected in all the subjects. The color-coding of the obtained fibers is based on the standard RGB (Red, Green, Blue) code applied to the vector at every segment of each fiber. Red indicates the medio-lateral plane. Green indicates the dorsoventral orientation. Blue indicates the rostro-caudal direction
Functional connectivity within the default mode network in healthy control and MHE patient groups. Averaged temporal correlation coefficient r (Blue) across all subjects in the healthy control group (a) and in the MHE patient group (b). Compared with healthy controls, MHE patients show decreased temporal correlation coefficients between the posterior cingulate cortex/precuneus and the medial prefrontal cortex, as well as between the posterior cingulate cortex/precuneus and the left parahippocampal gyrus. Medial prefrontal cortex is color-coded yellow, posterior cingulate cortex/precuneus color-coded red, the left parahippocampal gyrus color-coded pink, the right parahippocampal gyrus color-coded cyan
HE is multi-facet and can involve many features including redistribution of cerebral blood flow, metabolic rate of glucose, and ammonia from various cortical regions (e.g., the frontal and parietal cortices) to subcortical grey matter regions (the basal ganglia and thalamus) and so on, requesting the multimodality studies by the way of noninvasive imaging approaches. The advanced fMRI and diffusion tensor imaging combination provides and expands the applications of the MR imaging study of HE with the capability of assessing functional changes in brain affected by HE, also providing the opportunity to uncover the pathophysiologic mechanisms of HE. Magnetic resonance spectroscopy investigations should provide more evidence as an important factor for the development of HE, illustrating increase of cerebral ammonia metabolism, and other brain metabolism. These studies extend our understanding of the pathophysiology of MHE and can provide new biomarkers for the clinical diagnosis of HE.
Perspectives
Functional performance in HE could be delineated directly by network integrity assessment, in addition to cerebral edema, and other factors. The default mode network derived from coherent spontaneous BOLD fluctuations in resting-state fMRI has been increasingly used to evaluate neuropsychiatric disorders, neurodevelopmental disorders and other diseases (Fox et al. 2005; McKiernan et al. 2006). The default mode network is different between HE patients and normal controls, and resting-state fMRI can be applied to demonstrate variability of structural and functional connectivity and other features among HE patients with differing symptom profiles, including MHE. This may serve as a new biomarker for clinical diagnosis, also at the same time providing supplemental information for monitoring HE patients.
Recently, investigations of neuropsychiatric disorders on brain network changes have been largely advanced by attempts to demonstrate inter-regional interactions comprising the brain’s intricate web of connectivity; i.e., the human connectome (Sporns 2013; Glasser et al. 2013). As one major modality in the Human Connectome Project, resting-state fMRI has many aspects of estimating the “network matrix” that demand considerable attention from researchers in future. Structure–function relationship is the particularly important point in Imaging Connectomics since the detailed characterization of multiple neurobiological mechanisms requires appropriate applications of methodology and related interpretation. For this new field in neuroscience that relatively comprehensive mapping of brain network connectivity is needed. There is a need to develop the optimum techniques for resting-state fMRI to better detect functional connectivity differences between neuropsychiatric disorders, and different stages groups and normal healthy population. In future, multi-parameter MR imaging can simultaneously provide multiple information about structure–function relationships in HE. For example, combination of blood flow provided by arterial spin labeling MR perfusion and functional connectivity by resting-state fMRI can uncover how cerebral blood flow influences functional connectivity of the brain.
There is also a need for studies to determine correlation between network changes and brain edema in HE by multiple imaging modalities, including the causal-relationship between morphology and function in cellular level (Lin et al. 2012). BOLD-based fMRI should also be further investigated; the role of BOLD fMRI in the neural basis of cognitive and functional impairment of patients with HE and of the progression of HE from cirrhosis to MHE to overt HE should be clarified. There was limited correlation between decreased default mode network integrity and ammonia level, then it implies an adaptive change of functional network in each individual subject of HE, with a diverse clinical profile, and rather a complicated process in disease development (Zhang et al. 2012a). On the multi-modality platform, investigators explored more clues for the mechanism relating to the alteration of astrocyte morphology and function, ammonia metabolism, and others. For example, the role of glia in the neurobiology of depression is one of focuses of research (Naismith et al. 2012), and changed level of glial fibrillary acidic protein may be explained as an imbalance in toxic (glucocorticoid/glutamate) and trophic factors, lead to a reduction in glia, with resultant neuronal damage and injury in the early course of depression. Hyper-ammonemia has also been demonstrated in preclinical study (Tanigami et al. 2005), to contribute to increased numbers of swollen astrocytes, increased immuno-reactivity of glutamine synthetase, and some cytoskeletal proteins like the intermediate filament glial fibrillary acidic protein, which is involved in cell structure maintenance, cell communication, and functioning of the blood brain barrier. Altering glutamate re-uptake during ammonia metabolism may also cause the intracellular depletion of myo-inositol, which can trigger macromolecule migration to the extracellular space, resulting in increased extracellular fluid accumulation, as well as the increased mean diffusivity value on diffusion tensor imaging (Qi et al. 2012a; Kale et al. 2006). If extra- and intra-cellular glutamate concentrations persist on changing the surrounding glial processes, possible apoptosis will present like the associated mitochondrial dysfunction with oxidative stress, and subsequent change in the brain network occur.
The potential role of γ-aminobutyric acid- or glutamate-mediated neurotransmission in the pathophysiology of HE should be also investigated, because clinical manifestation of a low-grade cerebral edema associated with an altered neuron-astrocyte crosstalk in HE, which exacerbated by hyperammonemia and oxidative stress, are tightly related with alterations in neurotransmission (Palomero-Gallagher and Zilles. 2013). The slow progress in understanding the pathology of γ-aminobutyric acid-ergic transmission in HE is due to the high complexity of brain circuitries controlled by multiple types of γ-aminobutyric acid-ergic interneurons and the large variety of γ-aminobutyric acid-A receptors, which are differently affected by pathological conditions and not yet fully identified (Sergeeva 2013). Meanwhile, proton magnetic resonance spectroscopy measures static concentration of important brain chemicals (i.g., γ-aminobutyric acid), whereas 13C magnetic resonance spectroscopy allows evaluation of dynamic metabolic fluxes by introducing exogenous 13C-labeled substrates, with the flux between neuronal glutamate and astroglial glutamine determined by measuring the kinetics of 13C label incorporation into glutamate or the glia-specific substrate acetate, etc. (Xiang and Shen. 2011; Boumezbeur et al. 2010). However, 13C magnetic resonance spectroscopy has not applicable to patients so far.
Combined positron emission tomography and MRI may have a promising role to set up a bridge between molecular imaging, resting-state fMRI, metabolic and hemodynamic research of HE (Wehrl et al. 2013), especially in vivo neuroinflammation (with molecular marker such as peripheral-type benzodiazepine receptor) (Cagnin et al. 2006; Butterworth 2013 and neurotransmission (Glutamatergic, cholinergic, adenosine, etc.) mapping (García-Ayllón et al. 2008; Palomero-Gallagher and Zilles. 2013) in HE. These can provide pathophysiological interpretation for resting-state fMRI findings in HE.
The longitudinal resting-state fMRI studies following the treatment of HE patients or those studies which observe natural course of MHE can provide new biomarker for monitoring the disease evolution as a noninvasive method. Moreover, the studies of brain connectivity changes in neuropsychiatric disorders by fMRI (Ivanov et al. 2012) and their modification by drug treatment might ultimately help the development of combination of multiple imaging modalities (such as diffusion tensor imaging, or magnetic resonance spectroscopy) for the monitoring of effective interventions.
Conclusions
In conclusion, resting-state MRI offers the great potential for investigating HE and is helpful to clarify the pathophysiological mechanism of HE and functional reorganization pattern during disease progression. Combination of resting-state fMRI and other advanced analysis algorithms expand the applications of MRI in HE which can assess functional, structural and metabolic changes in brain affected by HE and their potential mechanisms. Investigation of drug-induced changes in functional brain connectivity might provide an opportunity for developing useful markers that might inform the development of personalized medicine.
Abbreviations
- BOLD:
-
Blood oxygen level dependent
- fMRI:
-
Functional MRI
- HE:
-
Hepatic encephalopathy
- MHE:
-
Minimal hepatic encephalopathy
- TIPS:
-
Transjugular intrahepatic portosystemic shunt
References
Amodio P, Montagnese S, Gatta A, Morgan MY (2004) Characteristics of minimal hepatic encephalopathy. Metab Brain Dis 19(3–4):253–267
Bajaj JS, Wade JB, Sanyal AJ (2009) Spectrum of neurocognitive impairment in cirrhosis: implications for the assessment of hepatic encephalopathy. Hepatology 50(6):2014–2021
Bajaj JS, Pinkerton SD, Sanyal AJ, Heuman DM (2012) Diagnosis and treatment of minimal hepatic encephalopathy to prevent motor vehicle accidents: a cost-effectiveness analysis. Hepatology 55(4):1164–1171
Boumezbeur F, Petersen KF, Cline GW, Mason GF, Behar KL, Shulman GI, Rothman DL (2010) The contribution of blood lactate to brain energy metabolism in humans measured by dynamic 13C nuclear magnetic resonance spectroscopy. J Neurosci 30(42):13983–13991
Butterworth RF (2013) The liver-brain axis in liver failure: neuroinflammation and encephalopathy. Nat Rev Gastroenterol Hepatol 10(9):522–528
Cagnin A, Taylor-Robinson SD, Forton DM, Banati RB (2006) In vivo imaging of cerebral “peripheral benzodiazepine binding sites” in patients with hepatic encephalopathy. Gut 55(4):547–553
Chen HJ, Zhu XQ, Jiao Y, Li PC, Wang Y, Teng GJ (2012a) Abnormal baseline brain activity in low-grade hepatic encephalopathy: a resting-state fMRI study. J Neurol Sci 318(1–2):140–145
Chen HJ, Zhu XQ, Shu H, Yang M, Zhang Y, Ding J, Wang Y, Teng GJ (2012b) Structural and functional cerebral impairments in cirrhotic patients with a history of overt hepatic encephalopathy. Eur J Radiol 81(10):2463–2469
Chen HJ, Zhu XQ, Yang M, Liu B, Zhang Y, Wang Y, Teng GJ (2012c) Changes in the regional homogeneity of resting-state brain activity in minimal hepatic encephalopathy. Neurosci Lett 507(1):5–9
Chen HJ, Jiao Y, Zhu XQ, Zhang HY, Liu JC, Wen S, Teng GJ (2013) Brain dysfunction primarily related to previous overt hepatic encephalopathy compared with minimal hepatic encephalopathy: resting-state functional MR imaging demonstration. Radiology 266(1):261–270
Córdoba J (2011) New assessment of hepatic encephalopathy. J Hepatol 54(5):1030–1040
Ferenci P, Lockwood A, Mullen K, Tarter R, Weissenborn K, Blei AT (2002) Hepatic encephalopathy– definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology 35(3):716–721
Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME (2005) The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A 102(27):9673–9678
García-Ayllón MS, Cauli O, Silveyra MX, Rodrigo R, Candela A, Compañ A, Jover R, Pérez-Mateo M, Martínez S, Felipo V, Sáez-Valero J (2008) Brain cholinergic impairment in liver failure. Brain 131(Pt 11):2946–2956
Glasser MF, Sotiropoulos SN, Wilson JA, Coalson TS, Fischl B, Andersson JL, Xu J, Jbabdi S, Webster M, Polimeni JR, Van Essen DC, Jenkinson M, WU-Minn HCP Consortium (2013) The minimal preprocessing pipelines for the Human Connectome Project. Neuroimage 80:105–124
Glover GH (2011) Overview of functional magnetic resonance imaging. Neurosurg Clin N Am 22(2):133–139
Gore JC (2003) Principles and practice of functional MRI of the human brain. J Clin Invest 112(1):4–9
Hassanein TI, Hilsabeck RC, Perry W (2008) Introduction to the Hepatic Encephalopathy Scoring Algorithm (HESA). Dig Dis Sci 53(2):529–538
He Y, Dagher A, Chen Z, Charil A, Zijdenbos A, Worsley K, Evans A (2009) Impaired small-world efficiency in structural cortical networks in multiple sclerosis associated with white matter lesion load. Brain 132(Pt 12):3366–3379
Hsu TW, Wu CW, Cheng YF, Chen HL, Lu CH, Cho KH, Lin WC, Lin CP (2012) Impaired small-world network efficiency and dynamic functional distribution in patients with cirrhosis. PLoS One 7(5):e35266
Ivanov I, Liu X, Shulz K, Fan J, London E, Friston K, Halperin JM, Newcorn JH (2012) Parental substance abuse and function of the motivation and behavioral inhibition systems in drug-naïve youth. Psychiatry Res 201(2):128–135
Kale RA, Gupta RK, Saraswat VA, Hasan KM, Trivedi R, Mishra AM, Ranjan P, Pandey CM, Narayana PA (2006) Demonstration of interstitial cerebral edema with diffusion tensor MR imaging in type C hepatic encephalopathy. Hepatology 43(4):698–706
Lee MH, Smyser CD, Shimony JS (2013) Resting-state fMRI: a review of methods and clinical applications. AJNR Am J Neuroradiol 34(10):1866–1872
Liao LM, Zhou LX, Le HB, Yin JJ, Ma SH (2012) Spatial working memory dysfunction in minimal hepatic encephalopathy: an ethology and BOLD-fMRI study. Brain Res 1445:62–72
Lin WC, Hsu TW, Chen CL, Wu CW, Lu CH, Chen HL, Li SH, Yeh PY, Cheng YF, Lin CP (2012) Connectivity of default-mode network is associated with cerebral edema in hepatic encephalopathy. PLoS One 7(5):e36986
Liu Y, Wang K, Yu C, He Y, Zhou Y, Liang M, Wang L, Jiang T (2008) Regional homogeneity, functional connectivity and imaging markers of Alzheimer’s disease: a review of resting-state fMRI studies. Neuropsychologia 46(6):1648–1656
Logothetis NK (2008) What we can do and what we cannot do with fMRI. Nature 453(7197):869–878
Lv XF, Qiu YW, Tian JZ, Xie CM, Han LJ, Su HH, Liu ZY, Peng JP, Lin CL, Wu MS, Jiang GH, Zhang XL (2013a) Abnormal regional homogeneity of resting-state brain activity in patients with HBV-related cirrhosis without overt hepatic encephalopathy. Liver Int 33(3):375–383
Lv XF, Ye M, Han LJ, Zhang XL, Cai PQ, Jiang GH, Qiu YW, Qiu SJ, Wu YP, Liu K, Liu ZY, Wu PH, Xie CM (2013b) Abnormal baseline brain activity in patients with HBV-related cirrhosis without overt hepatic encephalopathy revealed by resting-state functional MRI. Metab Brain Dis 28(3):485–492
Madoff DC, Wallace MJ, Ahrar K, Saxon RR (2004) TIPS-related hepatic encephalopathy: management options with novel endovascular techniques. Radiographics 24(1):21–36
McKiernan KA, D’Angelo BR, Kaufman JN, Binder JR (2006) Interrupting the “stream of consciousness”: an fMRI investigation. Neuroimage 29(4):1185–1191
McPhail MJ, Patel NR, Taylor-Robinson SD (2012) Brain imaging and hepatic encephalopathy. Clin Liver Dis 16(1):57–72
McPhail MJ, Leech R, Grover VP, Fitzpatrick JA, Dhanjal NS, Crossey MM, Pflugrad H, Saxby BK, Wesnes K, Dresner MA, Waldman AD, Thomas HC, Taylor-Robinson SD (2013) Modulation of neural activation following treatment of hepatic encephalopathy. Neurology 80(11):1041–1047
Naismith SL, Norrie LM, Mowszowski L, Hickie IB (2012) The neurobiology of depression in later-life: clinical, neuropsychological, neuroimaging and pathophysiological features. Prog Neurobiol 98(1):99–143
Ni L, Qi R, Zhang LJ, Zhong J, Zheng G, Zhang Z, Zhong Y, Xu Q, Liao W, Jiao Q, Wu X, Fan X, Lu GM (2012) Altered regional homogeneity in the development of minimal hepatic encephalopathy: a resting-state functional MRI study. PLoS One 7(7):e42016
Ortiz M, Cordoba J, Doval E, Jacas C, Pujadas F, Esteban R, Guardia J (2007) Development of a clinical hepatic encephalopathy staging scale. Aliment Pharmacol Ther 26(6):859–867
Palomero-Gallagher N, Zilles K (2013) Neurotransmitter receptor alterations in hepatic encephalopathy: a review. Arch Biochem Biophys 536(2):109–121
Pizoli CE, Shah MN, Snyder AZ, Shimony JS, Limbrick DD, Raichle ME, Schlaggar BL, Smyth MD (2011) Resting-state activity in development and maintenance of normal brain function. Proc Natl Acad Sci U S A 108(28):11638–11643
Qi R, Xu Q, Zhang LJ, Zhong J, Zheng G, Wu S, Zhang Z, Liao W, Zhong Y, Ni L, Jiao Q, Zhang Z, Liu Y, Lu G (2012a) Structural and functional abnormalities of default mode network in minimal hepatic encephalopathy: a study combining DTI and fMRI. PLoS One 7(7):e41376
Qi R, Zhang L, Wu S, Zhong J, Zhang Z, Zhong Y, Ni L, Zhang Z, Li K, Jiao Q, Wu X, Fan X, Liu Y, Lu G (2012b) Altered resting-state brain activity at functional MR imaging during the progression of hepatic encephalopathy. Radiology 264(1):187–195
Qi R, Zhang LJ, Xu Q, Zhong J, Wu S, Zhang Z, Liao W, Ni L, Zhang Z, Chen H, Zhong Y, Jiao Q, Wu X, Fan X, Liu Y (2012c) Lu G (2012c) Selective impairments of resting-state networks in minimal hepatic encephalopathy. PLoS One 7(5):e37400
Qi R, Zhang LJ, Zhong J, Wu S, Zhang Z, Zhong Y, Ni L, Zheng G, Jiao Q, Wu X, Fan X, Liu Y, Lu G (2012d) Dynamic changes of intrinsic brain activity in cirrhotic patients after transjugular intrahepatic portosystemic shunt: a resting-state FMRI study. PLoS One 7(10):e46681
Qi R, Zhang LJ, Zhong J, Zhang Z, Ni L, Jiao Q, Liao W, Zheng G, Lu G (2013a) Altered effective connectivity network of the basal ganglia in low-grade hepatic encephalopathy: a resting-state fMRI study with Granger causality analysis. PLoS One 8(1):e53677
Qi R, Zhang LJ, Zhong J, Zhang Z, Ni L, Zheng G, Lu GM (2013b) Disrupted thalamic resting-state functional connectivity in patients with minimal hepatic encephalopathy. Eur J Radiol 82(5):850–856
Raichle ME (2011) The restless brain. Brain Connectivity 1(1):3–12
Riordan SM, Williams R (2010) Gut flora and hepatic encephalopathy in patients with cirrhosis. N Engl J Med 362(12):1140–1142
Rosazza C, Minati L (2011) Resting-state brain networks: literature review and clinical applications. Neurol Sci 32(5):773–785
Sergeeva OA (2013) GABAergic transmission in hepatic encephalopathy. Arch Biochem Biophys 536(2):122–130
Sheline YI, Raichle ME (2013) Resting state functional connectivity in preclinical Alzheimer’s disease. Biol Psychiatry 74(5):340–347
Sporns O (2013) The human connectome: origins and challenges. NeuroImage 80:53–61
Tanigami H, Rebel A, Martin LJ, Chen TY, Brusilow SW, Traystmana RJ, Koehler RC (2005) Effect of glutamine synthetase inhibition on astrocyte swelling and altered astroglial protein expression during hyper-ammonemia in rats. Neuroscience 131(2):437–449
van den Heuvel MP, Hulshoff Pol HE (2010) Exploring the brain network: a review on resting-state fMRI functional connectivity. Eur Neuropsychopharmacol 20(8):519–534
Wehrl HF, Hossain M, Lankes K, Liu CC, Bezrukov I, Martirosian P, Schick F, Reischl G, Pichler BJ (2013) Simultaneous PET-MRI reveals brain function in activated and resting state on metabolic, hemodynamic and multiple temporal scales. Nat Med 19(9):1184–1189
Xiang Y, Shen J (2011) Simultaneous detection of cerebral metabolism of different substrates by in vivo 13C isotopomer MRS. J Neurosci Methods 198(1):8–15
Zafiris O, Kircheis G, Rood HA, Boers F, Häussinger D, Zilles K (2004) Neural mechanism underlying impaired visual judgement in the dysmetabolic brain: an fMRI study. Neuroimage 22(2):541–552
Zang Y, Jiang T, Lu Y, He Y, Tian L (2004) Regional homogeneity approach to fMRI data analysis. Neuroimage 22(1):394–400
Zang YF, He Y, Zhu CZ, Cao QJ, SuiMQ LM, Tian LX, Jiang TZ, Wang YF (2007) Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain Dev 29(1):83–91
Zhang LJ, Yang G, Yin J, Liu Y, Qi J (2007a) Neural mechanism of cognitive control impairment in patients with hepatic cirrhosis: a functional magnetic resonance imaging study. Acta Radiol 48(5):577–587
Zhang LJ, Yang G, Yin J, Liu Y, Qi J (2007b) Abnormal default-mode network activation in cirrhotic patients: a functional magnetic resonance imaging study. Acta Radiol 48(7):781–787
Zhang L, Qi R, Wu S, Zhong J, Zhong Y, Zhang Z, Zhang Z, Lu G (2012a) Brain default-mode network abnormalities in hepatic encephalopathy: a resting-state functional MRI study. Hum Brain Mapp 33(6):1384–1392
Zhang LJ, Zheng G, Zhang L, Zhong J, Wu S, Qi R, Li Q, Wang L, Lu G (2012b) Altered brain functional connectivity in patients with cirrhosis and minimal hepatic encephalopathy: a functional MR imaging study. Radiology 265(2):528–536
Zhang LJ, Qi R, Zhong J, Ni L, Zheng G, Xu J, Lu GM (2013a) Disrupted functional connectivity of the anterior cingulate cortex in cirrhotic patients without overt hepatic encephalopathy: a resting state fMRI study. PLoS One 8(1):e53206
Zhang LJ, Zhong J, Lu GM (2013b) Multimodality MR imaging findings of low-grade brain edema in hepatic encephalopathy. AJNR Am J Neuroradiol 34(4):707–715
Zhu XQ, Chen HJ, Wang Y, Cui Y, Teng GJ (2012) Aberrant resting-state corticostriatal functional connectivity in cirrhotic patients with hyperintense globus pallidus on T1-weighted MR imaging. PLoS One 7(11):e48886
Acknowledgment
We express our thanks for the grants from Natural Scientific Foundation of China (30700194, 81171313, 81322020, 81230032 to L.J.Z.) and Program for New Century Excellent Talents in University (NCET-12-0260 to L.J.Z.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L.J., Wu, S., Ren, J. et al. Resting-state functional magnetic resonance imaging in hepatic encephalopathy: current status and perspectives. Metab Brain Dis 29, 569–582 (2014). https://doi.org/10.1007/s11011-014-9504-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-014-9504-9