Abstract
Objectives
Protein kinase C (PKC) is a central enzyme in the regulation of growth and hypertrophy. Little was known on PKC isoform regulation in human heart. Goal of this study was to characterize the isoforms of protein kinase C in human heart, their changes during ontogenesis, and their regulation in myocardial hypertrophy and heart failure.
Methods
In left ventricular and atrial samples from adults with end-stage dilated cardiomyopathy (DCM), from adults with severe aortic stenosis (AS), from small infants undergoing repair of ventricular septal defects, and from healthy organ donors (CO), activity of protein kinase C and the expression of its isozymes were examined.
Results
In the adult human heart, the isoforms PKC-α, PCK-β, PKC-δ, PKC-ε, PKC-λ/-ι, and PKC-ζ were detected both on protein and on mRNA level. All isozymes are subjected to downregulation during ontogenesis. No evidence, however, exists for an isoform shift from infancy to adulthood. DCM leads to a pronounced upregulation of PKC-β. Severe left ventricular hypertrophy in AS, however, recruits a distinct isoform pattern, i.e., isoforms PKC-α, PKC-δ, PKC-ε, PKC-λ/-ι, and PKC-ζ are upregulated, whereas PKC-β is not changed under this condition.
Conclusion
This work gives evidence for a differential recruitment of human PKC isoforms in various forms of myocardial hypertrophy and heart failure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Protein kinase C (PKC) plays a major role in transmembrane signal transduction. For long time it is known as a key enzyme in the regulation of cell growth and differentiation (reviewed by [1, 2]) as well as in tumor promotion in many cell types (reviewed by [3]). PKC is also implicated in the regulation of cardiac function, resulting in inotropic and chronotropic effects [4–6], in the phosphorylation of several substrates [7], and in the regulation of calcium and other intracellular ion levels [4, 5]. Moreover, activation of protein kinase C modulates gene expression [8, 9]. Mounting evidence suggests that PKC is also involved in myocardial hypertrophy [10].
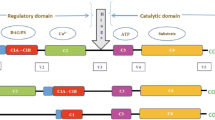
Molecular cloning studies have revealed that PKC consists of a large family of at least 13 different isoenzymes that can be divided into three major subgroups (reviewed by [11]). The first subfamily comprises the α, β1, β2, and γ isotype, with β1 and β2 arising via alternate splicing of the same gene transcript. The activity of this conventional group of PKC (cPKC) depends on Ca2+-binding to the regulatory domain and on the presence of phospholipids such as dioleoylglycerol (DAG) and phosphatidylserine (PS). The second, novel group of PKC isoforms (nPKC) consists of PKC-δ, -ε, -η, and -θ. These isoforms do not depend on Ca2+, but require DAG and PS for their activation. Both groups are activated by phorbol esters such as phorbol-12,13-myristate-acetate (PMA). The third, so-called atypical subfamily (aPKC-ζ, -λ/-ι and -μ) also lacks the sensitivity to calcium, but they also lack the sensitivity to DAG/PMA, whereas arachidonic acid and phosphatidylinositol 3,4,5-triphosphate may activate these isotypes.
Only few reports are available examining expression of PKC in normal and diseased human hearts. Consistently, the expression of the isoforms PKC-α, PKC-δ, PKC-β, and PKC-ε was shown [12–15]. One report showed the presence of other isoforms, i.e., PKC-ζ, PKC-λ, and PKC-ι [15]. These studies vary in the tissue used, since some groups examined right atrial tissue [14, 15], and other groups examined left ventricular samples [12, 13], and in the cardiac conditions examined, i.e., the stage of heart failure.
Goal of the present study was to characterize the isoforms of protein kinase C in human heart, their changes during ontogenesis, and their regulation in pressure overload hypertrophy and in heart failure due to dilated cardiomyopathy.
Materials and methods
Human heart samples and standards for PKC isoforms
For this study, myocardial samples were obtained from four groups of patients:
-
1.
Failing human adult heart samples (left ventricular and right atrial) were obtained from explanted hearts of patients undergoing heart transplantation due to end-stage heart failure caused by idiopathic dilated cardiomyopathy (n = 10, age, 53 ± 9 years, ejection fraction <25% in all patients).
-
2.
Small left ventricular and right atrial samples from adult patients with severe, symptomatic aortic stenosis (n = 6, age 74 ± 3 years, left ventricular ejection fraction 59 ± 10%, peak-to-peak transvalvular gradient, 80 ± 16 mmHg) undergoing aortic valve replacement were obtained. Only those patients were used where small amounts of left ventricular tissue had to be removed during the adaptation of the valve prosthesis. The right atrial appendages were routinely removed during the cannulation for extracorporeal circulation.
-
3.
Myocardial samples from small infants (age 6–27 months; n = 6) were from right atrial appendages routinely removed for cannulation during the repair of ventricular septal defects or persistent ductus arteriosus. For ethical reasons, no ventricular samples were obtained in this group.
-
4.
Furthermore, nonfailing human adult hearts (n = 3) were obtained from organ donors whose hearts could not be transplanted for reasons independent of this study.
The local ethics committee of the university approved the study. The investigation conforms to the principles outlined in the Declaration of Helsinki. All tissue samples were collected immediately in the operating theatre and rapidly frozen in liquid nitrogen until further use. All hearts were subjected to a comparable protocol for cardioplegia.
Tissue extracts (human skeletal muscle, rat skeletal muscle, rat brain) or cells (HeLa cells, Transduction Laboratories) known to contain the specific isoforms of PKC [11, 16] served as positive control for human isoforms of PKC not found in human heart. Where available, human recombinant PKC isoforms (Calbiochem) were used in addition.
RNA preparation, rt-PCR and Southern blot analysis
The extraction of total cardiac RNA was carried out according to a modification of the guanidine isothiocyanate ethanol precipitation method of Chirgwin, as described previously [17]. Total cardiac RNA was then reverse transcribed into cDNA and amplified using a modified protocol of RT-PCR [18] as described previously [17]. All PCR amplification products were separated on agarose gels and were vacuum blotted on a NY-13 nitrocellulose membrane. The blots were hybridized with 32P-labeled internal oligonucleotides (1 μg) to further confirm the specificity of the amplified PCR products.
TUNEL assay
Terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL) was performed using the “In situ cell death detection kit” (Roche, Mannheim, Germany) according to the manufacturer’s description.
Preparation of subcellular fractions
Subcellular fractions were prepared as reported previously [17]. Protein concentrations were determined according to the method of Bradford using bovine serum albumin as standard.
To rule out that tissue degradation may influence the results obtained, the protein yield of every preparation was monitored. The yield was 22.9 ± 0.9 mg protein/g tissue in the particulate fraction and 58.6 ± 5.5 mg protein/g tissue in the cytosol and did not differ significantly between the groups examined.
PKC assay
Maximally stimulatable PKC activity was determined in the presence of Calcium, phosphatidyl serine and dioleoyl glycerol using histone IIIs as substrate, as previously reported [17]. This assay has previously been shown to be reliable to determine maximally stimulated enzyme activity both of calcium-dependent and -independent isoforms [17].
Immunoblot analysis of the PKC Isoforms
Westernblot analysis was performed as reported previously [17]. Polyclonal antibodies were from Gibco BRL (anti-PKC-α, anti-PKC-βco, anti-PKC-βI, anti-PKC-βII, anti-PKC-γ, anti-PKC-δ, anti-PKC-ε) or Calbiochem (anti-PKC-δ, anti-PKC-ε, anti-PKC-η), and monoclonal antibodies were from Transduction laboratories (anti-PKC-ι, anti-PKC-λ, anti-PKC-μ, anti-PKC-θ). To ensure the specificity of the immunoreactive proteins, analyses were done in the presence and absence of competing immunizing peptide. Staining of the membranes with Ponceau-S was used to correct the specific bands by the protein loaded. Degradation of single isozymes was ruled out by the missing detection of further specific bands of lower molecular weight.
Density of the specific bands was analyzed by laser-densitometry analysis (ULTRO SCAN XL, Pharmacia LKB). The data are expressed as mean ± SEM. Two-tailed Student’s t-test for unpaired data were performed. p-values <0.05 were considered significant.
Results
To evaluate the expression of the PKC isoforms in human left ventricle, left ventricular samples from a healthy organ donor were subjected to Westernblot analysis. Results are shown in Fig. 1A. PKC-α, PKC-δ, PKC-ε, PKC-λ, and PKC-ζ stained as single, dominant band of the expected molecular weight. Specifity of the staining was demonstrated by addition of the immunogenic peptide (PKC-α, PKC-δ, PKC-ε, and PKC-ζ) when using polyclonal antibodies, or by excluding cross-reactivity against closely related isoforms (PKC-λ) when using monoclonal antibodies. PKC-β stained as a triple band, consisting of both splice variants (PKC-βI and PKCβII), as demonstrated by counterstaining with a specific antibody for PKC-βII. Staining for PKC-γ and PKC-θ demonstrated that those isoforms are not expressed in adult human heart. Staining for PKC-μ and -η gave inconstant results (not shown).
protein kinase C isoform expression in the adult human heart: (A) To detect PKC isoform protein expression in the adult human heart, samples obtained from the left ventricle of a donor heart (see methods) were homogenized, electrophoresed and subjected to Westernblot analysis using polyclonal antibodies (PKC-α, -δ, -ε, -β, -γ, -ζ) or monoclonal antibodies (PKC-λ, -μ, -θ). As positive control, tissues with known expression of the respective isoform (rat brain for PKC-α, -β, -ε, -λ, -ζ; skeletal muscle for PKC-θ), recombinant isoforms, or HeLa cell lysate (known to selectively express PKC-λ, but not PKC-ζ) was used. Competition experiments in the presence of the immunogenic peptide confirmed the specifity of the staining for the isoforms analyzed with polyclonal antibodies. Consecutive assays with antibodies for PKC-βco (showing a triple band) and -βII demonstrated the presence of both splice variants of PKC-β, i.e., -βI and -βII, in the heart. Using this technique, the presence of the isoforms -α (80 kDa), -βI (80 kDa), -βII (82 kDa), -δ (75 kDa), -ε (93 kDa), -λ (72 kDa), -ζ (72 kDa), but not of -γ or -θ, in the heart was shown. Staining for PKC-μ, -η, and -ι gave inconstant results (not shown). (B) Southernblot hybridization of PKC isoform-specific mRNA from human heart confirmed the presence of mRNA specific for PKC-α, PKC-β, PKC-δ, PKC-ζ, and PKC-ε in samples obtained from a donor heart. RNA from rat brain, which is known to contain these isoforms, is used as standard. RNAse digest precludes contamination with DNA
Using increasing concentrations of recombinant isoform protein as external standard, the expression of PKC-α was most abundant in the left ventricle (960 ng/mg total protein), whereas PKC-ε, PKC-δ, and PKC-ζ (242, 170, and 215 ng/mg total protein) were expressed to a lower extent.
To confirm the results obtained on protein level, isoform-specific rt-PCR was performed to evaluate the presence of PKC isoform-specific mRNA in human heart. Specifity of the amplificates was tested by southernblot hybridization (Fig. 1B). Using this technique, the presence of mRNA specific for PKC-α, PKC-β, PKC-λ, PKC-δ, PKC-ζ, and PKC-ε could be demonstrated.
The relative distribution of the PKC-isoforms in human atria and ventricle is shown in Fig. 2. The calcium-dependent isoforms, PKC-α and PKC-β, reside predominantly in the ventricular myocardium, whereas PKC-δ and PKC-ζ are mainly expressed in the atria. PKC-ε and PKC-λ are evenly distributed. No difference in isoform expression was found between samples obtained from the left and from the right ventricle (data not shown).
Expression of PKC isoforms in human adult ventricle and atria. To examine the relative distribution of PKC isoform expression between atria and ventricle, left ventricular and right samples of two donor hearts were compared. Samples were analyzed on the same blots for exact comparison. Whereas the Calcium-dependent isoforms PKC-α and PKC-βi/βII reside predominantly in the ventricle, PKC-δ and PKC-ζ are mainly expressed in the atria. PKC-ε and PKC-λ are evenly distributed in those compartments
PKC isoform regulation during ontogenesis in human hearts was examined by comparing isoform expression in the right atria of small infants with those of adults without severe heart failure (Fig. 3). During ontogenesis, down regulation of all isoforms occurs. The extent of this down regulation clearly varies: Expression of PKC-β is decreased by about 90%. The other isoforms are down regulated to a lesser extent. This is reflected by the maximally stimulatable enzyme activity in both fractions. Activity in the particulate clearly decreases during ontogenesis (152 ± 10 vs. 40 ± 8 pmol/mg protein/min, n = 5, p < 0.01), whereas activity in the cytosol is only marginally changed (163 ± 8 vs. 122 ± 18 pmol/mg protein/min, n = 5, p = n.s.). To quantify the relative distribution between cytosol and particulate in the isozymes, the cytosol-to-particulate ratio was analyzed for all samples (Table 1). In newborns, PKC-ε, and PKC-β reside mainly in the particulate fraction. During ontogenesis, a significant increase of the relative cytosolic isoform content was detectable for PKC-ε and PKC-α, suggesting a translocation and activation of these isoforms in infants, which is reversed in adults. In infants, no further isozymes of PKC could be detected compared with those detectable in adults.
Changes of PKC isoform expression during ontogenesis. Right atrial samples of small infants (age 6–27 months, n = 6) were compared with right atrial samples from adult patients without heart failure. Care was given only to include patients without pulmonary hypertension or right heart enlargement into this analysis. Representative Westernblot analyses (A) show that the predominant changes occur in the particulate fraction. Quantitative analysis (B) demonstrated a downregulation of all isoforms during ontogenesis, but to a quite different extent. Note that PKC-β expression is nearly completely switched off in adult nonfailing hearts
The regulation of PKC isoforms under distinct pathological conditions was examined in patients with pressure-induced, concentric hypertrophy of the left ventricle with preserved left ventricular function, i.e., aortic stenosis, and in patients with end-stage heart failure due to left ventricular dilatation and severely compromised left ventricular function, i.e., dilated cardiomyopathy. Results are shown in Figs. 4 and 5. Aortic stenosis leads to an upregulation of all PKC isoforms examined except PKC-β, which is even downregulated under this condition. Increased levels of isoform protein were detectable both in the particulate fraction (PKC-α, PKC-δ, PKC-ε, PKC-λ, PKC-ζ) and in the cytosol (PKC-λ, PKC-ζ). In contrast, PKC-β is highly upregulated in dilated cardiomyopathy, together with an increased expression of PKC-δ and PKC-ε. The other isoforms are not upregulated. Maximally stimulatable PKC enzyme activity is not significantly changed between these conditions, both in the cytosol and in the particulate fraction (data not shown). To clarify if isoform translocation may play a role in dilated cardiomyopathy or aortic stenosis, the cytosol-to-particulate ratio was analyzed in those samples (Table 2). In aortic stenosis, a significant translocation of PKC-α, PKC-δ, and PKC-ε was seen, whereas in dilated cardiomyopathy, only PKC-ε was significantly changed.
PKC isoform expression in aortic stenosis. Left ventricular samples from patients with severe aortic stenosis (i.e., pressure-induced left ventricular hypertrophy) and preserved left ventricular function were compared with healthy controls. Representative experiments (A) and the quantification of all experiments (B) show that during hypertrophy without heart failure, PKC-β-expression is decreased, whereas all other isoforms expressed are upregulated in this setting. *p < 0.05 versus control
PKC isoform expression in dilated cardiomyopathy. Left ventricular samples from patients with end-stage dilated cardiomyopathy (dCMP) with severely compromised left ventricular function were compared with those from healthy organ donors. Representative experiments (A) comparing the particulate fraction of both groups demonstrate a re-expression of PKC-β in dCMP. Analyses of cytosolic samples were nearly unchanged (not shown). Quantification (B) of this series of experiments showed a marked increase in the expression of PKC-β, PKC-δ, and PKC-ε, but not of PKC-α, PKC-λ, and PKC-ζ. *p < 0.05 versus control
To determine if the programmed cell death is activated in aortic stenosis or in dilated cardiomyopathy in our model, TUNEL assays were performed. In control samples, 0.72 ± 0.24 per 1000 nuclei were TUNEL-positive. Both aortic stenosis (1.7 ± 0.8 per 1000) and dilated cardiomyopathy (2.1 ± 1.2 per 1000) lead to an slightly increased number of TUNEL-positive cells. This increase, however, did not reach the level of significance.
Discussion
The salient findings of this study are that in human heart, six isoforms of PKC are expressed, i.e., PKC-α, PKC-β, PKC-δ, PKC-ε, PKC-λ, and PKC-ζ. PKC-β is expressed in both splicing variants, PKC-βI and PKC-βII. Other isoforms, namely PKC-γ, PKC-θ, PKC-η, and PKC-μ (to which some authors refer as PKD, what is more correct in terms of its enzymatic properties) were not detectable. PKC-ι, another isoform cited in some references, is identical to PKC-λ. Nomenclature of this isoform in human samples is inhomogeneous throughout the literature. The relative distribution between atria and ventricle is very variable for the isoforms. During ontogenesis, all isoforms are subjected to downregulation. Namely, PKC-β-expression is nearly totally switched off in normal cardiac tissue. Severe heart failure due to dilated cardiomyopathy leads to a reexpression of PKC-β and to an upregulation of PKC-δ and PKC-ε. Surprisingly, severe left ventricular hypertrophy with preserved left ventricular function recruited a quite distinct isoform pattern. Under this condition, upregulation of all isoforms except PKC-β, which was even downregulated, was seen.
The expression of PKC isoforms in cardiac tissue varies, depending on the species examined. In rats, many reports consistently demonstrate the expression of PKC-α, PKC-δ, PKC-ε, and PKC-ζ [17, 19, 20]. In rabbits, ten isoforms are detectable (PKC-α, PKC-βI/βII, PKC-γ, PKC-δ, PKC-ε, PKC-ζ, PKC-η, PKC-ι, PKC-λ, PKC-μ [21]). In human samples, many studies consistently found PKC-α, PKC-δ, PKC-ε, consistent with the findings demonstrated in this work [12–15]. Remarkably, the presence of PKC-β in healthy human hearts was clearly demonstrated by the studies examining left ventricular samples [12, 13] and could also be shown in this study, whereas studies only examining samples from right atria are inconsistent at this point [14, 15]. This difference is most likely due to the differences in isoform expression between atria and ventricle shown in this work. Namely PKC-β is predominantly expressed in the ventricle. The presence of PKC-ζ and PKC-λ in human heart, shown in this report, was reported before by some authors [13, 15]; other studies, however, did not test for these isoforms [14]. Consistency exists that PKC-γ and PKC-θ are not present in human heart [13, 15]. Together, the existing data is quite uniform about the expression pattern of PKC isoforms in human cardiac tissue.
The Westernblot analyses shown in this work use tissue extracts. In consequence, the cell types expressing the various isoforms of PKC cannot be clearly distinguished in these experiments. In rats, the isoforms PKC-α, PKC-δ, PKC-ε, and PKC-ζ are expressed in neonatal cardiac myocytes, whereas adult ventricular cardiac myocytes only express PKC-δ and PKC-ε [19, 22]. As a limitation of this study, we did not perform histochemical analyses.
To our knowledge, the change of PKC isoform expression in the human heart during ontogenesis has not been studied before. In rat heart extracts, an age-dependent decline in the expression of PKC-α, PKC-δ, PKC-ε, and PKC-ζ was shown, with a relatively uniform decrease of all isoforms from the neonate to the adult animal [22]. Expression of PKC-β in cardiac tissue was not detected in this study. Interestingly, in isolated rat adult ventricular myocytes, PKC-α and PKC-ζ were not detectable at all, and only traces of PKC-δ were found, whereas a robust expression of PKC-ε persisted in adult animals [22, 23]. Comparable results, with a decline of the isoforms during ontogenesis, were obtained in mice [24]. In the human heart, a downregulation of all isoforms found could be demonstrated in this study, with PKC-β being the isoform down regulated to the largest extent.
Two recent reports examined the expression of PKC isoforms in human heart failure. Bowling and coworkers examined 12 explanted hearts with end-stage heart failure, 6 due to dilated cardiomyopathy, and 6 due to ischemic heart disease, and compared them with ten nonfailing donor hearts. They could demonstrate an increased expression and activity of PKC-β and an increased expression of PKC-α in heart failure, whereas PKC-ε was unchanged. Differences between dilated cardiomyopathy and ischemic heart disease specimen were not reported [12]. The reactivation of PKC-β was discussed as “hypertrophic signaling” and “reactivation of a fetal gene program” [12, 25], and the latter fact is highly supported by the ontogenetic data shown in our study. Another report done by Shin and coworkers compared samples from seven patients with end-stage cardiomyopathy exclusively due to ischemic heart disease with five donor hearts [13]. They conclude “the same isoforms which are expressed in nondiseased heart are also present in this pathologic state, without evidence of an isoform switch.” They, however, only performed a qualitative analysis of the isoforms, since they could show that the detected amount of the isoforms is highly dependent on the sample collection and preparation. The representative analyses shown in this paper, however, point even to a downregulation of PKC-β, PKC-δ, and PKC-ε in heart failure, in some contradiction to the report of Bowling [13]. In the present study, extreme caution was given to prevent differences in the sample preparation. All samples were collected in the operating theatre and immediately frozen in liquid nitrogen. Sample collection and preparation was done by a single researcher (S. K. B.) to reduce variations. Using this approach, the data obtained in patients with dilated cardiomyopathy show a re-expression of PKC-β, comparable to Bowling’s data in humans [12].
Data on PKC isoforms in aortic stenosis in man have, to our knowledge, not been reported yet. Surprisingly, in contrast to the data obtained in end-stage heart failure, PKC-β is even down regulated in these severely hypertrophied, non-failing hearts. The other PKC isoforms examined are upregulated. This PKC isoform regulation in aortic stenosis is in agreement with data obtained in animal models: in aortic stenosis in rats, Braun and coworkers showed an upregulation of PKC-α and PKC-δ, but not of PKC-β [26]. Comparable data were obtained by two other groups [27, 28]. Thus, in our view, the reexpression of PKC-β seen in severe heart failure seems to occur rather late in the progression of this disease, but not during the process of left ventricular hypertrophy. The concept of PKC-β as one key player of “hypertrophic signaling” is clearly questioned by this data. In animal models, overexpression of PKC-β leads to a cardiomyopathic phenotype [29, 30], whereas a knockout of this isozyme does not influence the cardiac phenotype of mice and its response to various hypertrophic stimuli [31]. Thus, PKC-β seems to be the isoform re-expressed in heart failure in mice and man more than the isoform involved in hypertrophy. More data, however, is needed to support this hypothesis.
It is difficult to speculate on the role of the various isoforms of PKC in myocardial hypertrophy and heart failure. Data obtained in isolated cardiac myocytes [32] as well as in animal models of hypertrophy [33] suggest that PKC-α promotes myocyte hypertrophy, i.e., increased cell surface area and protein turnover, via activation of the ERK1/2 kinase and changes in calcium handling of the cells. In contrast, overexpression of PKC-ε in isolated cardiac myocytes induced cellular elongation and remodeling without increasing cellular protein content or cell surface area [32, 34], suggesting that this isoform has a distinct role in mediating the remodeling of cardiac myocytes in hypertrophy. However, another group could show that PKC-ε is no prerequisite for cardiac hypertrophy [35], so that the role of this isoform in hypertrophy remains not fully clarified. PKC-δ was reported to play a role in apoptosis induction under various conditions such as reperfusion, more than in the induction of hypertrophy or growth [36]. Activation of apoptosis was also found in this study together with an upregulation of PKC-δ. Overexpression of PKC-β in the heart may cause a cardiomyopathic phenotype [29]; another group, in contrast, could show that hypertrophy may develop independent of PKC-β. Little is known about the function of PKC-λ or PKC-ζ in the heart. Together, the function of the individual isoforms in hypertrophy and heart failure is not fully clarified, and research on this point is difficult since many recent data point to redundant effects of various isoforms in knock-out or overexpression models [10, 37].
In conclusion, this work gives evidence for a differential recruitment of human PKC isoforms in various forms of myocardial hypertrophy and heart failure.
References
Parker PJ, Kour G, Marais RM et al (1989) Protein kinase C–A family affair. Mol Cell Endocrinol 65:1–11
Farago A, Nishizuka Y (1990) Protein kinase C in transmembrane signalling. FEBS Lett 268:350–354
Nishizuka Y (1984) The role of protein kinase C in cell surface signal transduction and tumor promotion. Nature 308:693–698
MacLeod K, Harding SE (1991) Effects of phorbol ester on contraction, intracellular pH and intracellular Ca2+ in isolated mammalian ventricular myocytes. J Physiol 444:481–498
Capogrossi MC, Kaku T, Filburn CR et al (1990) Phorbol ester and dioctanoylglycerol stimulate membrane association of protein kinase C and have a negative inotropic effect mediated by changes in cytosolic Ca2+ in adult rat cardiac myocytes. Circ Res 66:1143–1155
Johnson JA, Mochly-Rosen D (1995) Inhibition of the spontaneous rate of contraction of neonatal cardiac myocytes by protein kinase C isozymes: A putative role for the ε isozyme. Circ Res 76:654–663
Kwiatkowska-Patzer B, Dománska-Janik K (1991) Increased 19 kDa protein phosphorylation and protein kinase C activity in pressure-overload cardiac hypertrophy. Basic Res Cardiol 86:402–409
Allo SN, McDermott PJ, Carl LL, Morgan HE (1991) Phorbol ester stimulation of protein kinase C activity and ribosomal DNA transcription. Role in hypertrophic growth of cultured cardiomyocytes. J Biol Chem 266:22003–22009
Allo SN, Carl LL, Morgan HE (1992) Acceleration of growth of cultured cardiomyocytes and translocation of protein kinase C. Am J Physiol 263:C319–C325
Dorn GW II, Force T (2005) Protein kinase cascades in the regulation of cardiac hypertrophy. J Clin Invest 115:527–537
Nishizuka Y (1988). The molecular heterogeneity of protein kinase C and its implications for cellular regulation. Nature 334:661–665
Bowling N, Walsh R, Song Q et al (1999) Increased protein kinase C activity and expression of Calcium sensitive isoforms in the failiing human heart. Circulation 99:384–391
Shin HG, Barnett JV, Chang P et al (2000) Molecular heterogeneity of protein kinase C expression in human ventricle. Cardiovasc Res 48:285–299
Hassouna AB, Matata M, Galinanes M (2004) PKC-ε is upstream and PKC-α is downstream of mito-KATP-channels in the signal transduction pathway of ischemic preconditioning of human myocardium. Am J Physiol 287:C1418–C1425
Erdbrugger W, Keffel J, Knocks M et al (1997) Protein kinase C isoenzymes in rat and human cardiovascular tissues. Br J Pharmacol 120:177–186
Osada S, Mizuno K, Saido TC et al (1992) A new member of the protein kinase C family, nPKC θ, predominantly expressed in skeletal muscle. Mol Cell Biol 12:3930–3938
Strasser RH, Simonis G, Schoen SP et al (1999) Two distinct mechanisms mediate a differential regulation of protein kinase C isozymes in acute and prolonged myocardial ischemia. Circ Res 85:77–87
Ponzoni M, Lucarelli E, Corrias MV, Cornaglia-Ferraris P (1993) Protein kinase C isoenzymes in human neuroblasts: Involvement of PKC? in cell differentiation. FEBS Lett 322:120–124
Steinberg SF, Goldberg M, Rybin VO (1995) Protein kinase C isoform diversity in the heart. J Mol Cell Cardiol 27:141–153
Kohout TA, Rogers TB (1993) Use of a PCR-based method to characterize protein kinase C isoform expression in cardiac cells. Am J Physiol 264:C 1350–C 1359
Ping P, Zhang J, Qiu Y et al (1997) Ischemic preconditioning induces selective translocation of protein kinase C isoforms ε and η in the heart of conscious rabbits without subcellular redistribution of total protein kinase C activity. Circ Res 81:404–414
Rybin VO, Steinberg F (1994) Protein kinase C isoform expression and regulation in the developing rat heart. Circ Res 74:299–309
Bogoyewitch MA, Parker PJ, Sugden PH (1993) Characterization of protein kinase C isotype expression in adult rat heart. Circ Res 72:757–767
Schreiber KL, Paquet L, Allen BG, Rindt H (2001) Protein kinase C isoform expression and activity in the mouse heart. Am J Physiol 281:H2062–H2071
Simpson PC (1999) β-protein kinase C and hypertrophic signalling in human heart failure. Circulation 99:334–337
Braun MU, La Rosée P, Schoen S et al (2002) Differential regulation of cardiac protein kinase C isozyme expression after aortic banding in rat. Cardiovasc Res 56:52–63
Gu X, Bishop SP (1994) Protein kinase C isozyme expression in the developing rat heart and changes in expression in response to pressure overload hypertrophy. J Mol Cell Cardiol 27:CLXVI
Bayer AL, Heidkamp MC, Patel N et al (2003) Alterations in protein kinase C isoenzyme expression and autophosphorylation during the progression of pressure overload-induced left ventricular hypertrophy. Mol Cell Biochem 242:145–152
Wakasaki H, Koya D, Schoen FJ et al (1997) Targeted overexpression of protein kinase C β2 isoform in myocardium causes cardiomyopathy. Proc Natl Acad Sci USA 94:9320–9325
Bowman JC, Steinberg SF, Jiang T et al (1997) Expression of protein kinase C ? in the heart causes hypertrophy in adult mice and death in neonates. J Clin Invest 100:2189–2195
Roman BB, Geenen DL, Leitges M, Buttrick PM (2001) PKC-β is not necessary for cardiac hypertrophy. Am J Physiol 280:H2264–H2270
Braz JC, Bueno OF, De Windt LJ, Molkentin JD (2002) PKCα regulates the hypertrophic growth of cardiomyocytes through extracellular signal-regulated kinase1/2 (ERK1/2). J Cell Biol 156:905–919
Muth JN, Bodi I, Lewis W et al (2001) A Ca2+-dependent transgenic model of cardiac hypertrophy: a role for protein kinase Cα. Circulation 103:140–147
Strait JB, Martin JL, Bayer A et al (2001) Role of protein kinase C ε in hypertrophy of cultured neonatal rat ventricular myocytes. Am J Physiol 280:H756–H766
Klein G., Schaefer A, Hilfiker-Kleiner D et al (2005) Increased collagen deposition and diastolic dysfunction but preserved myocardial hypertrophy after pressure overload in mice lacking PKCε. Circ Res 96:748–755
Sabri A, Steinberg SF (2003) Protein kinase C isoform-selective signals that lead to cardiac hypertrophy and the progression of heart failure. Mol Cell Biochem 251:97–101
Murphy S, Frishman WH (2005) Protein kinase C in cardiac disease and as a potential therapeutic target. Cardiol Rev 13:3–12
Acknowledgements
Prof. Thomas Dengler, M.D., helped with the characterization of the patients.
Author information
Authors and Affiliations
Corresponding author
Additional information
Gregor Simonis and Steffen K. Briem contributed equally to this work.
Rights and permissions
About this article
Cite this article
Simonis, G., Briem, S.K., Schoen, S.P. et al. Protein kinase C in the human heart: differential regulation of the isoforms in aortic stenosis or dilated cardiomyopathy. Mol Cell Biochem 305, 103–111 (2007). https://doi.org/10.1007/s11010-007-9533-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-007-9533-3