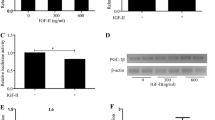
Abstract
Peroxisome proliferator-activated receptor γ (PPARγ) is a member of the nuclear receptor superfamily known to regulate adipocyte differentiation. However, its role in skeletal muscle differentiation is not known. To investigate possible involvement of PPARγ in skeletal muscle differentiation, we modulated its expression in C2C12 mouse skeletal muscle cells by stable transfection with sense or antisense plasmid constructs of PPARγ cDNA. Phenotypic observations and biochemical analysis of different myogenic markers showed that altered expression of PPARγ inhibited the formation of myotubes, as well as expression of muscle-specific myogenic proteins including myogenin, MyoD and creatine kinase activity. Together, we show that critical expression of PPARγ is required for skeletal muscle cells differentiation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Smith CK, Janney MJ, Allen RE (1994) Temporal expression of myogenic regulatory genes during activation, proliferation and differentiation of rat skeletal muscle satellite cells. J Cell Physiol 159:379–385
Ueda Y, Wang MC, Ou BR, Haung J, Elce J, Tanaka K, Ichihara A, Forsberg NE (1998) Evidence for the participation of the proteasome and calpain in early phases of muscle cell differentiation. Int J Biochem Cell Biol 30:679–694
Puri PL, Sartorelli V (2000) Regulation of muscle regulatory factors by DNA-binding, interacting proteins, and post transcriptional modifications. J Cell Physiol 185:155–173
Lassar AB, Skapek SX, Novitch B (1994) Regulatory mechanisms that coordinate skeletal muscle differentiation and cell cycle withdrawal. Curr Opin Cell Biol 6:788–794
Olson EN, Klein WH (1994) bHLH factors in muscle development. Dead lines and commitments, what to leave in and what to leave out. Genes Dev 8:1–8
Khurana A, Dey CS (2002) Subtype specific roles of mitogen activated protein kinases in L6L9 skeletal muscle cell differentiation. Mol Cell Biochem 238:27–39
Carbane C, Englaro W, Yeow K, Ragno M, Derijard B (2003) Regulation of C2C12 myogenic terminal differentiation by MKK3/p38α pathway. Am J Physiol Cell Physiol 284: C658–C666
Figuera JD, Hayman MJ (2004) Differential effects of the Ski-interacting protein (SKIP) on differentiation induced by transforming growth factors-β1 and bone morphogenetic protein 2 in C2C12 cells. Exp Cell Res 296:163–172
Dreyer C, Krey G, Keller H, Givel F, Helftenbein G, Wahli W (1992) Control of the perexisomal β-oxidation pathway by a novel family of nuclear hormone receptors. Cell 68:879–887
Evans RM, Barish GD, Wang YX (2004) PPARs and the complex journey of obesity. Nat Med 10:1–7
He W, Barak Y, Havener A, Olson P, Liao D, Le J, Nelson M, Ong E, Olefsky JM, Evans RM (2003) Adipose-specific peroxisome proliferator-activated receptor knockout causes insulin resistance in fat and liver but not in muscle. Proc Natl Acad Sci USA 100:15,712–15,717
Rosen ED, Sarraf P, Troy AE, Bradwin G, Moore K, Milstone DS, Spiegelman BM, Mortensen RM (1999) PPAR gamma is required for the differentiation of adipose tissue in vivo and in vitro. Mol Cell 4:611–617
Kruszynska YT, Mukherjee R, Jow L, Dana S, Paterniti JR, Olefsky JM (1998) Skeletal muscle peroxisome proliferator activated receptor gamma expression in obesity and non-insulin dependent diabetes mellitus. J Clin Invest 101:543–548
Zierath JR, Ryder JW, Doebber T, Woods J, Wu M, Ventre J, Li Z, McCrary C, Berger J, Zhang B, Moller DE (1998) Role of skeletal muscle in thiazolidinediones insulin sensitizer (PPARγ agonist) action. Endocrinol 139:5034–5041
Loviscach M, Rahman N, Carter L, Muudaliar S, Mohadeen P, Ciaraldi TP, Veerkamp JH, Henry RR (2000) Distribution of peroxisome proliferator activated receptors (PPARs) in human skeletal muscle and adipose tissues: relation to insulin action. Diabetologia 43:304–311
Hevener A, He W, Barak Y, Le J, Bandyopadhyay G, Olson P, Wilkes J, Evans RM, Olefsky J (2003) Muscle-specific Pparg deletion causes insulin resistance. Nat Med 9:1491–1497
Noris AW, Chen L, Fisher SJ, Szanto I, Ristow M, Jozsi AC, Hirshman MF, Rosen ED, Goodyear LJ, Gonzalez FJ, Spiegelman BM, Kahn CR (2003) Muscle-specific PPARγ-deficient mice develop increased adiposity and insulin resistance but respond to thiazolidinediones. J Clin Invest 112:608–618
Lehmann JM, Moore LB, Smitholiver TA, Wilkison WO, Willson TM, Kliewer SA (1995) An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma). J Biol Chem 270:12,953–12,956
Desvergne B, Wahli W (1999) Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocrine Rev 20:649–688
Picard F, Auwerx J (2002) PPARγ and glucose homeostasis. Annu Rev Nutr 22:167–197
Kubota N, Terauchi Y, Miki H, Tamemoto H, Yamauchi T, Komeda K, Satoh S, Nakano R, Ishii C, Sugiyama T, Eto K, Tsubamoto Y, Okuno A, Murakami K, Sekihara H, Hasegawa G, Naito M, Toyoshima Y, Tanaka S, Shiota K, Kitamura T, Fujita T, Ezaki O, Aizawa S, Nagai R, Tobe K, Kimura S, Kadowaki T (1999) PPARγ mediates high-fat diet-induces adipocyte hypertrophy and insulin resistance. Mol Cell 4:597–609
Miles PDG, Barak Y, Evans RM, Olefsky JM (2000) Improved insulin-sensitivity in mice heterozygous for PPAR-γ deficiency. J Clin Invest 105:287–292
Varley CL, Stahlschmidt J, Lee WC, Holder J, Diggle C, Selby PJ, Trejdosiewicz LK, Southgate J (2004) Role of PPARγ and EGFR signaling in the urothelial terminal differentiation programme. J Cell Sci 117:2029–2036
Solanes G, Pedraza N, Iglesias R, Giralt M, Villarroya F (2003) Functional relationship between MyoD and peroxisome proliferator receptor-activated receptor dependent regulatory pathways in the control of the human uncoupling protein-3 gene transcription. Mol Endocrinol 17:1944–1958
Kumar N, Dey CS (2002) Metformin enhances insulin signaling in insulin-dependent and -independent pathways in insulin resistant muscle cells. Brit J Pharmacol 137:329–336
Verma NK, Singh J, Dey CS (2004) PPARγ expression modulation insulin sensitivity in C2C12 skeletal muscle cells. Br J Pharmacol 143:1006–1013
Brunetti A, Goldfine ID (1990) Role of myogenin in myoblast differentiation and its regulation by fibroblast growth factor. J Biol Chem 265:5960–5963
Kitzmann M, Carnac G, Vandromme M, Primig M, Lamb NJC, Fernandez A (1998) The muscle regulatory factors MyoD and Myf-5 undergo distinct cell cycle-specific expression in muscle cells. J Cell Biol 142:1447–1459
Friday BB, Horsley V, Pavlath GK (2000) Calcineurin activity is required for the initiation of skeletal muscle differentiation. J Cell Biol 149:657–665
Andres V, Walsh K (1996) Myogenin expression, cell cycle withdrawal, and phenotypic differentiation are temporally separable events that precede cell fusion upon myogenesis. J Cell Biol 132:657–666
Sarbassov DD, Peterson CA (1998) Insulin receptor substrate-1 and phosphatidylinositol 3-kinase regulate extracellular signal-regulated kinase-dependent and -independent signaling pathways during myogenic differentiation. Mol Endocrinol 12:1870–1878
Kumar N, Dey CS (2003) Development of insulin resistance and reversal by thiazolidinediones in C2C12 skeletal muscle cells. Biochem Pharm 65: 249–257
Kumar N, Dey CS (2003) Metformin enhances insulin signaling in insulin-dependent and -independent pathways in insulin resistant muscle cells. Br J Pharmacol 137:329–336
Barak Y, Nelson MC, Ong ES, Jones YZ, Ruiz-Lozano P, Chein KR, Koder A, Evans RM (1999) PPARγ is required for placental, cardiac, and adipose tissue development. Mol Cell 4:585–595
Koutnikova H, Cock TA, Watanabe M, Houseten SM, Champy MF, Dierich A, Auwerex J (2003) Compensation by the muscle limits the metabolic consequences of lipidystrophy in PPARγ hypomorphic mice. Proc Natl Acad Sci USA 100:14,457–14,462
Imai T, Takakuwa R, Marchand S, Dentz E, Bornert JM, Messaddeq N, Wendling O, Mark M, Desvergne B, Wahli W, Chambon P, Metzger D (2004) Peroxisome proliferator-activated receptor γ is required in mature white and brown adipocytes for their survival in the mouse. Proc Natl Acad Sci USA 101:4543–4547
Rosen ED, Spiegelman BM (2001) PPARγ: a nuclear regulator of metabolism, differentiation, and cell growth. J Cell Biol 276:37,731–37,734
Mukherjee R, Hoener PA, Jow L, Bilakovics J, Klausing K, Mais DE, Faulkner A, Croston GE, Paterniti JR Jr (2000) A selective peroxisome proliferator activated receptor -γ (PPARγ) modulator blocks adipocute differentiation but stimulates glucose uptake in 3T3-L1 adipocytes. Mol Endocrinol 14:1425–1433
Mukherjee R, Jow L, Croston GE, Paterniti JR Jr (1997) Identification, characterization, and tissue distribution of human peroxisome proliferator activated receptor (PPAR) isoforms (PPARγ2 verses PPARγ1 and activation with retinoid × receptor agonists and antagonists. J Biol Chem 272:1075–1078
Mukherjee R, Jow L, Noonan D, McDonnell DP (1994) Human and rat peroxisome proliferator activated receptors (PPARs) demonstrate similar tissue distribution but different responsiveness to PPAR activators. J Steroid Biochem Mol Biol 51:157–166
Schoonjans K, Peinado-Onsurbe J, Lefebvre AM, Heyman RA, Briggs MR, Deeb S, Staels B, Auwerx J (1996) PPARα and PPARγ activators direct a distinct tissue-specific transcriptional response via a PPRE in the lipoprotein lipase gene. EMBO J 15:5336–5348
Rosen ED, Spiegelman BM (2001) PPARγ: a nuclear regulator of metabolism, differentiation, and cell growth. J Cell Biol 276:37,731–37,734
Tontonoz P, Hu E, Spiegelman BM (1994) Stimulation of adipogenesis in fibroblasts by PPAR gamma 2, a lipid-activated transactivation factor. Cell 79:1147–1156
Saladin R, Fajas L, Dana S, Halvorsen YD, Auwerx J, Briggs M (1999) Differential regulation of peroxisome proliferator activated receptor γ1 (PPARγ1) and PPARγ2 messenger RNA expression in the early stages of adipogenesis. Cell Growth Differ 10:43–48
He W, Barak Y, Havener A, Olson P, Liao D, Le J, Nelson M, Ong E, Olefsky JM, Evans RM (2003) Adipose-specific peroxisome proliferator-activated receptor knockout causes insulin resistance in fat and liver but not in muscle. Proc Natl Acad Sci USA 100:15,712–15,717
Koutnikova H, Cock TA, Watanabe M, Houseten SM, Champy MF, Dierich A, Auwerex J (2003) Compensation by the muscle limits the metabolic consequences of lipidystrophy in PPARγ hypomorphic mice. Proc Natl Acad Sci USA 100:14,457–14,462
Qi C, Zhu Y, Reddy JK (2000) Peroxisome proliferator-activated receptors, coactivators, and downstream targets. Cell Biochem Biophys 32:187–204
Chang JH, Lin HK, Chung HS, Chang YJ, Chi HC, Chen SL: Myogenic bHLH proteins regulate the expression of PPARγ coactivator-1α. Endocrinol Mar 9, doi:10.1210/en: 2005–1317, 2006
Puigserver P (2005) Tissue-specific regulation of metabolic pathways through the transcriptional coactivator PGC1-alpha. Int J Obes (Lond). Mar 29(Suppl 1):S5–S9
Corton JC, Brown-Borg HM (2005) Peroxisome proliferator-activated receptor gamma coactivator 1 in caloric restriction and other models of longevity. J Gerontol Biol Sci Med Sci 60:1494–1509
Acknowledgements
We thank Dr. C.L. Kaul, Ex-Director, NIPER, for his support in this work. Authors greatly acknowledge Dr. R.M. Evans, The Salk Institute for Biological Studies, San Diego, CA, USA for the generous gift of pCMX-mPPARg plasmid. We are thankful to Dr. N. Kumar and Dr. A. Khurana for his constant support. R. Singh for his assistance in the laboratory. J.K.S., B.K. and K.S.M. are recipients of NIPER fellowship and N.K.V. was research scientist of NIPER.
Author information
Authors and Affiliations
Corresponding author
Additional information
*These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Singh, J., Verma, N.K., Kansagra, S. et al. Altered PPARγ expression inhibits myogenic differentiation in C2C12 skeletal muscle cells. Mol Cell Biochem 294, 163–171 (2007). https://doi.org/10.1007/s11010-006-9256-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-006-9256-x