Abstract
Marine ascidians are considered as one of the richest sources of bioactive compounds. The extraction and utilization of marine peptides have been attracted much attention owing to their potential health benefits. Most of the bioactive compounds from marine ascidians are already in different phases of the clinical and preclinical pipeline. They can be used in different functional and nutraceutical values due to their antineoplastic, antihypertensive, antioxidant, and antimicrobial properties. The screening in vivo and in vitro bioassays are coupled to the purification process for the exploration of its biological interest which is of great value. The growing significance to study marine natural products results from the discovery of novel pharmacological tools including potent anticancer drugs and other drugs are in clinical/pre-clinical trials. The present review highlights the recent research progress in marine ascidians’ peptides and its prospects for the future pharmaceutical development.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ocean is the rich biodiverse and it has occupied 70% of the earth. The marine environment needs more adaptive changes in the organisms so that they form a special structure of the bioactive substances. Marine organisms are living in complex habitats and are exposed to extreme conditions, such as temperature, salinity, pressure, and illumination; thus, they could produce a wide variety of secondary metabolites (Hamed et al. 2015). Ascidians belongs to the phylum of Chordata, which encompasses all vertebrate animals, including mammals and, therefore, they represent the most highly evolved group of animals commonly investigated by marine natural products chemists (Menna et al. 2011). Recently marine peptides have opened a new perspective for novel and efficient drug developments. Cyclic and linear peptides are derived from marine ascidians have been increased our knowledge regarding novel cytotoxic, antimicrobial, anti-oxidant, antiviral and many other biological properties (Destoumieux et al. 1997; Li et al. 2014; Cheung et al. 2015). It has the advantages of less toxicity, high targeting specificity and effective biological activity. These bioactive peptides are extracted from sponges, ascidians, seaweeds, and mollusks and reported to have various pharmacological properties. The bioactive peptides often have 3 to 20 amino acid residues, and their biological activities are determined based on their sequence strength and amino acid composition (Pihlanto-Leppälä 2000; Bhat et al. 2015; Stoye et al. 2016). Recently, these peptides have been paid attention to unravel their structure, sequences and compositional properties (Lordan et al. 2011). The objectives of this review are to provide an overview of the available sources of marine bioactive peptides and their biological activities and its potential applications in functional foods and pharmaceuticals for further drug development.
Techniques for the Synthesis of Bioactive Peptides
The widely used protocols for the isolation of bioactive peptides are by chemical methods containing acid and alkaline hydrolysis, enzymatic hydrolysis is performing on the laboratory scale and Recombinant DNA technology (Guzmán et al. 2006; Magana et al. 2015; Lemes et al. 2016). The few other common methods were used by bioreactor systems or immobilized enzymes to increase product yield and fermentation technology. Recently, many advanced extraction techniques like supercritical fluid extraction, pressurized solvent extraction, microwave-assisted extraction, ultrasound-assisted extraction, pulsed electric field-assisted extraction and enzyme-assisted extraction are developed and most preferred for extracting bioactive peptides (Grosso et al. 2015). The acid hydrolysis is an important method for chemical modification that can significantly change the structure and functional properties of peptides and this method is preferred over other pretreatments because of its low cost and higher effectiveness (Lee and Jeffries 2011; Loow et al. 2016). The enzymatic methods of peptide bond formation are mediated by an enzyme (protease) in free or immobilized form and this method is especially useful in the synthesis of very short peptides (2–5 oligomers) as well in the condensation of large peptide fragments (So et al. 1998; Perez Espitia et al. 2012). The recombinant DNA technology offers modern methods of cloning and gene expression in microorganism and other organisms, allowing production of a several recombinant peptides (Sewald and Jakubke 2002). While comparing the all available methods for the synthesis of bioactive peptides, the recombinant DNA technology approach seems to be the most cost effective and very good alternative for large-scale peptide production (Li 2011).
Purification and Characterization of Peptides
The separation and purification of bioactive peptides are primarily based on their physical and chemical characteristics, such as their molecular size, charge, polarity, solubility, and specific covalent or noncovalent interactions (Jo et al. 2017). The separation strategies are mainly employed depending on the characteristics feature the target peptides. The different techniques used in the purification of peptides are ion-exchange chromatography, affinity chromatography, capillary electrophoresis, size exclusion chromatography and reverse-phase high-performance liquid chromatography (RP-HPLC). The isolated and purified peptides are characterized with different analytical equipment, majorly as LC-MS, LC/-MS/MS or LC-NMR (Martins et al. 2014; Pérez-Victoria et al. 2016; Ulrike 2016) and fast atom bombardment mass spectrometry (FAB-MS), electrospray ionization (ESI-MS), or matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS) (Jo et al. 2017). The accuracy of these new techniques helped to determine the molecular mass to charge ratio of the peptide, along with its chemical structure, with high resolution and sensitivity. In future, genome mining will be one of the most important aspects and will provide useful information. Generally, the silent genes/gene clusters, is the key factor to discover the interesting and new peptide compounds (Gross 2009).
Physiological Properties of Bioactive Peptides Derived from Ascidians
Bioactive peptides from ascidians has three types of structure including cyclic, acyclic (depsipeptides), and linear that have potential biological functions of antineoplastic, antiviral activity, antidiabetic, antioxidant activity, immunomodulatory properties (Schwartsmann et al. 2001). The Urochordata, also known as tunicates and ascidians, have emerged as a rich source of peptides and successfully preceded several drugs candidates including few under the clinical trials (Table 1). Lee et al. (1997 ) discussed that antimicrobial peptides of Clavanins A, B, C and D contain 23 amino acid residues were reported from Solitary tunicate, Styela clava and it has shown antibacterial activity against the three Gram-negative (E. coli, S. typhimurium, P. aeruginosa) and two Gram-positive strains (S. aureus, M. flavus) at 0.4–5 μM concentration (MIC).
Halocyamine A and B are two antimicrobial tetrapeptides isolated from the hemocytes of the ascidian Halocynthia roretzi (Table 2). It shows antimicrobial activities against several strains of bacteria and yeasts with cytotoxic activities of neuroblastoma N-18 cells at 160 µM and human hepatoma Hep-G2 cells at 100 µM (Azumi et al. 1990). The cytotoxic depsipeptides of Tamandarins A and B have been discovered from a marine ascidian of the family Didemnidae, which was evaluated against different human cancer cell lines such as pancreatic carcinoma (BX-PC3), prostate carcinoma (DU145) and the head and neck carcinoma (UMSCC10b). Tamandarin A showed a 50% reduction in overall cell survival (IC50) at a concentration of 1.79, 1.36, and 0.99 ng/mL, respectively (Hamad and Shioiri 2005; Vervoort et al. 2000). Styelin D, the antimicrobial peptides with a 32-residue C-terminus, extracted from ascidian Styela clava blood cells (hemocytes), contains two novel amino acids, dihydroxyarginine and dihydroxylysine (Vervoort et al. 2000). Lee et al. 2001 reported antimicrobial peptides were isolated from the solitary tunicate Halocynthia aurantium compound of Dicynthaurin is composed two 30-residue cynthaurin monomers containing only four lysines and no arginines or histidines and its show activity against the 5 bacterial pathogens (E. coli, P. aeruginosa, M. luteus, L. monocytogenes, S. aureus) at 140 μg/mL (MIC). Perez and Faulkner (2003) reported that bistratamides C–J, cyclic hexapeptides derived from Philippines ascidian Lissoclinum bistratum showed moderate cytotoxicity against HCT-116 cell line at varying concentrations of 7.9; 28; 5; 1.7; 9;1 μg/mL. Styelin D is effective against Gram-positive and Gram-negative bacteria, even in excessive salinity (200 mM NaCl). These antibiotic properties are used for the development of microbicides effective at high concentrations of NaCl or physiological conditions (Aneiros and Garateix 2004).
A new cytotoxic cyclic heptapeptide, Mollamide (Fig. 1c) obtained from the ascidian Didemnum molle, shows very good cytotoxicity effect against various cell lines with IC50 values of 1 μg/mL towards P388 murine leukemia line, 2.5 μg/mL against A549 human lung carcinoma and HT29 human colon carcinoma CV1 (monkey kidney fibroblast) cell lines with IC50 value of 2.5 μg/mL (Carroll et al. 1994). Donia et al. 2008 reported that two new mollamides B and C isolated from the Indonesian tunicate Didemnum molle. Mollamides B evaluated against different cell lines of non-small cell lung cancer cell line (H460), breast cancer cell line (MCF7) and CNS cancer cell line (SF-268) cytotoxicity activity at 100 μM. The antimalarial activity was assessed against Plasmodium falciparum (D6 clone and W2 clone) at IC50 values shows of 2.0 and 2.1 μg/mL. From Mollamides B showed marginal activity against Leishmania donoVani at IC50 and IC90 values of 18 and 35 μg/mL. The cytotoxicity against HIV-1 in human PBM cells at EC50 value of 48.7 μM in vitro condition. Mollamide C showed minor activity against the two leukemias, five solid tumors, murine, human normal cell and other few cell lines. Trunkamide A is one of cyclopeptide with a tiazoline ring similar to Mollamide isolated from ascidians of the genus Lissoclinum, where antitumor activities under pre-clinical trials are being demonstrated (Hamada and Shioiri 2005; Wipf et al. 1995).
Fu et al. 1998 isolated the four new cyclic peptides, patellamide G and ulithiacyclamides E–G, along with the known patellamides A–C and ulithiacyclamide B from the ascidian Lissoclinum patella collected in Pohnpei, Federated States of Micronesia and it has shown vinblastine against the drug resistant cells (CEM/VBL100) was IC50 at 90 nM and Patellamides B and C exhibited in vitro modulation of multidrug resistance in CEM/VBL100 cells. The bicyclic peptide, Vitilevuamide derived from two marine ascidians, Didemnum cuculiferum and Polysyncranton lithostrotum and showed cytotoxic activity in several human tumor cell lines with LC50 values of 6, 124, 311 and 311 nM were obtained for human colon tumor (HCT116), lung cancer (A549), malanoma tumor (SK Mel-5) and kidney cancer cell lines (A498), respectively. Vitilevuamide suppressed tubulin polymerization and it can arrest the cell cycle of target cells in the G2/M phase (Edler et al. 2002).
Two new antimicrobial peptides, Halocyntin and papillosin were derived from hemocytes of the solitary tunicate, Halocynthia papillosa and these molecules showed the antibacterial activity against Gram-positive and Gram-negative bacteria at 0.75–100 μM (MBC) (Galinier et al. 2009). Five new lipopeptides, peptidolipins B–F were derived from the associated organism of Nocardia sp. isolated from the ascidian Trididemnum orbiculatum and Peptidolipins B and E has shown the moderate antibacterial activity against MRSA and methicillin-sensitive Staphylococcus aureus (Wyche et al. 2012). Ko et al. 2016 discussed the antihypertensive Peptides of Ala-His-Ile-Ile-Ile were derived from the Tunicate Styela clava and showed Peptide increases glucose uptake via activation of the AMPK pathway and may potentially emerge as a novel target for the treatment of type II diabetes. Two cyclic Hexapeptides of Bistratamide M and N derived from the marine ascidian Lissoclinum bistratum were collected from Raja Ampat (Papua Bar, Indonesia) and it has shown the cytotoxic activity Human tumor cell lines of breast (MDA-MB-231), colon (HT-29), lung (A-549) and pancreas (PSN1) with LC50 values at ≥ 20 µm (Urda et al. 2017).
Recent Pharmacological Advances for Drug Development
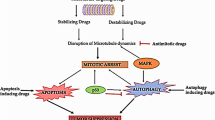
The success of biotechnology in pharmaceutical industry has been developed, now by the extensive number of scientific advances for the currently approved novel products for human health care. In this concept various techniques are involved in the drug development. The confluence of computational and high-throughput experimental methods for protein-engineering and platform technologies has ushered in unprecedented opportunities to develop safe, effective, and more convenient protein and peptide therapeutics and also Recombinant DNA technology allow the production of a wide range of peptides, proteins and biochemicals from naturally nonproducing cells (Kumar et al. 2006; Torchilin 2008; Carter 2011). This technology, now approximately 25 years old, is becoming one of the most important technologies for currently drug development. Intelligent coupling of cellular tests with omics technologies will help to identify interesting drug candidates, to clarify mode of action and to avoid late stage failure of compounds in the pipeline (Fig. 2). More attention should be given to early investigation of toxicological risks and to pharmacokinetic behavior of the drug candidates. Modern biotechnology and pharmaceutical analysis has evolved beyond this to encompass combination techniques, high-throughput technologies, chemo metrics, micro dosing studies, miniaturization and nanotechnology. These recent techniques are very useful to ascidians peptides for future drug development.
Conclusion
In conclusion, bioactive peptides derived from marine ascidians have a potential for human health care. The unique chemical classes of peptides found in the ascidians have promising biological activities which make them excellent candidates for drug design and development. The ascidians peptides are important for pharmaceutical studies and discovering new therapeutic treatment like anti-tumor/anti-cancer, anti-HIV, antimicrobial, and various diseases. In this review, we concluded Marine ascidians contains a wide range of secondary metabolites such as protein, peptides and minor metabolites might serve as vital role in future drug development.
References
Aneiros A, Garateix A (2004) Bioactive peptides from marine sources: pharmacological properties and isolation procedures. J Chromatogr B 803(1):41–53
Azumi K, Yokosawa H, Ishii S (1990) Halocyamines: novel antimicrobial tetrapeptide-like substances isolated from the hemocytes of the solitary ascidian Halocynthia roretzi. Biochemistry 29(1):159–165
Bhat ZF, Kumar S, Bha HF (2015) Bioactive peptides of animal origin: a review. J Food Sci Technol 52(9):5377–5392
Broggini M, Marchini SV, Galliera E, Borsotti P, Taraboletti G, Erba E, Sironi M, Jimeno J, Faircloth GT, Giavazzi R, D’Incalci M (2003) Aplidine, a new anticancer agent of marine origin, inhibits vascular endothelial growth factor (VEGF) secretion and blocks VEGF-VEGFR-1 (flt-1) autocrine loop in human leukemia cells MOLT-4. Leukemia 17:52–59
Carroll AR, Bowden BF, Coll JC, Hockless DC, Skelton BW, White AH (1994) Studies of Australian ascidians. IV. Mollamide, a cytotoxic cyclic heptapeptide from the compound ascidian Didemnum molle. Aust J Chem 47(1):616–619
Carter PJ (2011) Introduction to current and future protein therapeutics: a protein engineering perspective. Exp Cell Res 317(9):1261–1269. https://doi.org/10.1016/j.yexcr.2011.02.013
Cheung RCF, Ng TB, Wong JH (2015) Marine peptides: bioactivities and applications. Mar Drugs 13:4006–4043. https://doi.org/10.3390/md13074006
Destoumieux D, Bulet P, Loewi D, Dorsselaeri AV, Rodriguez J, Bachere E (1997) Penaeidins, a new family of antimicrobial peptides isolated from the shrimp Penaeus vannamei (Decapoda). J Biol Chem 272:28398–28406
Donia MS, Wang B, Dunbar DC, Desai PV, Patny A, Avery M, Hamann MT (2008) Mollamides B and C, cyclic hexapeptides from the Indonesian tunicate Didemnum molle. J Nat Prod 71(6):941–945
Edler MC, Fernandez AM, Lassota P, Ireland CM, Barrows LR (2002) Inhibition of tubulin polymerization by vitilevuamide, a bicyclic marine peptide, at a site distinct from colchicine, the vinca alkaloids, and dolastatin 10. Biochem Pharmacol 63(4):707–715
Fu X, Do T, Schmitz FJ, Andrusevich V, Engel MH (1998) New cyclic peptides from the ascidian Lissoclinum patella. J Nat Prod 61(12):1547–1551
Galinier R, Roger E, Sautiere PE, Aumelas A, Banaigs B, Mitta G (2009) Halocyntin and papillosin, two new antimicrobial peptides isolated from hemocytes of the solitary tunicate, Halocynthia papillosa. J Pept Sci 15(1):48–55
Gross H (2009) Genomic mining: a concept for the discovery of new bioactive natural products. Curr Opin Drug Discov Dev 12(2):207–219
Grosso C, Valentão P, Ferreres F, Andrade PB (2015) Alternative and efficient extraction methods for marine-derived compounds. Mar drugs 13(5):3182–3230
Guzmán F, Barberis S, Illanes A (2006) Peptide synthesis: chemical or enzymatic. Electron J Biotechnol 10(2):280–314
Hamada Y, Shioiri T (2005) Recent progress of the synthetic studies of biologically active marine cyclic peptides and depsipeptides. Chem Rev 105(12):4441–4482
Hamed I, Özogul F, Özogul Y, Hamed I, Özogul F, Özogul Y, Regenstein JM (2015) Marine bioactive compounds and their health benefits: a review. Compr Rev Food Sci Food Saf 14(4):446–465
Jo C, Khan FF, Khan MI, Iqbal J (2017) Marine bioactive peptides: types, structures, and physiological functions. Food Rev Int 33(1):44–61
Ko SC, Kim JI, Park SJ, Jung WK, Jeon YJ (2016) Antihypertensive peptide purified from Styela clava flesh tissue stimulates glucose uptake through AMP-activated protein kinase (AMPK) activation in skeletal muscle cells. Eur Food Res Technol 242(2):163–170
Kumar TR, Soppimath K, Nachaegari SK (2006) Novel delivery technologies for protein and peptide therapeutics. Curr Pharm Biotechnol 7(4):261–276
Lee JW, Jeffries TW (2011) Efficiencies of acid catalysts in the hydrolysis of lignocellulosic biomass over a range of combined severity factors. Bioresour Technol 102(10):5884–5890
Lee IH, Cho Y, Lehrer RI (1997) Effects of pH and salinity on the antimicrobial properties of clavanins. Infect Immun 65(7):2898–2903
Lee IH, Lee YS, Kim CH, Kim CR, Hong T, Menzel L, Boo LM, Pohl J, Sherman MA, Waring A, Lehrer RI (2001) Dicynthaurin: an antimicrobial peptide from hemocytes of the solitary tunicate, Halocynthia aurantium. Biochim Biophys Acta 1527(3):141–148
Lemes AC, Sala L, Ores JC, Braga ARC, Egea ME, Fernandes KF (2016) A review of the latest advances in encrypted bioactive peptides from protein-rich waste. Int J Mol Sci 17:950. https://doi.org/10.3390/ijms17060950
Li Y (2011) Recombinant production of antimicrobial peptides in Escherichia coli: a review. Protein Expr Purif 80(2):260–267
Li W, Tailhades J, O’Brien-Simpson NM, Separovic F, Otvos L Jr, Hossain MA, Wade JD (2014) Proline-rich antimicrobial peptides: potential therapeutics against antibiotic-resistant bacteria. Amino Acids 46(10):2287–2294
Lindequist U (2016) Marine-derived pharmaceuticals–challenges and opportunities. Biomol Ther 24(6):561
Loow YL, Wu TY, Jahim JM, Mohammad AW, Teoh WH (2016) Typical conversion of lignocellulosic biomass into reducing sugars using dilute acid hydrolysis and alkaline pretreatment. Cellulose 23(3):1491–1520
Lordan S, Ross RP, Stanton C (2011) Marine bioactives as functional food ingredients: potential to reduce the incidence of chronic diseases. Mar Drugs 9(6):1056–1100
Magana MD, Segura-Campos M, Dávila-Ortiz G, Betancur-Ancona D, Chel-Guerrero L (2015) ACE-I inhibitory properties of hydrolysates from germinated and ungerminated Phaseolus lunatus proteins. Food Sci Technol 35(1):167–174
Martins A, Vieira H, Gaspar H, Santos S (2014) Marketed marine natural products in the pharmaceutical and cosmeceutical industries: tips for success. Mar drugs 12(2):1066–1101
Menna M, Fattorusso E, Imperatore C (2011) Alkaloids from marine ascidians. Molecules 16(10):8694–8732
Palanisamy SK, Giacobbe S, Sundaresan U (2015) Marine ascidians potential source for new class of anti-cancer drugs. World J Pharm Pharm Sci 4(8):474–485
Perez LJ, Faulkner DJ (2003) Bistratamides E–J, modified cyclic hexapeptides from the Philippines ascidian Lissoclinum bistratum. J Nat Prod 66(2):247–250
Perez Espitia PJ, de Fátima Ferreira Soares N, dos Reis Coimbra JS, de Andrade NJ, Souza Cruz R, Medeiros A, Antonio E (2012) Bioactive peptides: synthesis, properties, and applications in the packaging and preservation of food. Compr Rev Food Sci Food Saf 11(2):187–204
Pérez-Victoria I, Martín J, Reyes F (2016) Combined LC/UV/MS and NMR strategies for the dereplication of marine natural products. Planta Medica 82(09/10):857–871
Pihlanto-Leppälä A (2000) Bioactive peptides derived from bovine whey proteins: opioid and ace-inhibitory peptides. Trends Food Sci Technol 11(9):347–356
Ribrag V, Caballero D, Fermé C, Zucca E, Arranz R, Briones J, Gisselbrecht C, Salles G, Gianni AM, Gomez H, Kahatt C (2013) Multicenter phase II study of plitidepsin in patients with relapsed/refractory non-Hodgkin’s lymphoma. Haematologica 98(3):357–363
Schmitz FJ, Bowden BF, Toth SI, Attaway DH, Zaborsky OR (1993a) Marine biotechnology, vol 1, pharmaceutical and bioactive natural products. Plenum Press, New York, p 277
Schmitz FJ, Bowden BF, Toth SI, Attaway DH, Zaborsky OR (1993b) Marine biotechnology, vol 1, pharmaceutical and bioactive natural products. Plenum Press, New York, p 270
Schwartsmann G, da Rocha AB, Berlinck RG, Jimeno J (2001) Marine organisms as a source of new anticancer agents. Lancet Oncol 2(4):221–225
Sewald N, Jakubke H (2002) Peptides: chemistry and biology. Wiley, Weinheim, p 543
So JE, Kang SH, Kim BG (1998) Lipase-catalyzed synthesis of peptides containing D-amino acid. Enzym Microb Technol 23(3):211–215
Stoye A, Nagalingam G, Britton WJ, Payne RJ (2016) Synthesis of norfijimycin A with activity against Mycobacterium tuberculosis. Aust J Chem 70(2):229–232
Suarez-Jimenez GM, Burgos-Hernandez A, Ezquerra-Brauer JM (2012) Bioactive peptides and depsipeptides with anticancer potential: sources from marine animals. Mar Drugs 10:963–986. https://doi.org/10.3390/md1005096
Taylor SW, Craig AG. Fisher WH, Park WH, Lehrer MRI, RI (2000) Styelin D, an extensively modified antimicrobial peptide from ascidian hemocytes. J Biol Chem 275:38417
Tincu JA, Menzel LP, Azimov R, Sands J, Hong T, Waring AJ, Taylor SW, Lehrer R (2003) Plicatamide, an antimicrobial octapeptide from Styela plicata hemocytes. J Biol Chem 278(15):13546–13553
Torchilin V (2008) Intracellular delivery of protein and peptide therapeutics. Drug Discov Today Technol 5(2–3):e95–e103. https://doi.org/10.1016/j.ddtec.2009.01.002
Ulrike L (2016) Marine-derived pharmaceuticals - Challenges and opportunities. Biomol Ther (Seoul) 24(6):561–571
Urda C, Fernández R, Rodríguez J, Pérez M, Jiménez C, Cuevas C (2017) Bistratamides M and N, oxazole-thiazole containing cyclic hexapeptides isolated from Lissoclinum bistratum interaction of zinc (II) with bistratamide K. Mar Drugs 15(7):209
Vervoort H, Fenical W, Epifanio RD (2000) Tamandarins A and B: new cytotoxic depsipeptides from a Brazilian ascidian of the family Didemnidae. J Org Chem 65(3):782–792
Wipf P, Miller CP, Venkatraman S, Fritch PC (1995) Thiolysis of oxazolines: a new, selective method for the direct conversion of peptide oxazolines into thiazolines. Tetrahedron Lett 36(36):6395–6398
Wyche TP, Hou Y, Vazquez-Rivera E, Braun D, Bugni TS (2012) Peptidolipins B–F, antibacterial lipopeptides from an ascidian-derived Nocardia sp. J Nat Prod 75(4):735–740
Acknowledgements
This study was funded by Department of Science and Technology-Science and Engineering Research board (DST-SERB), India (SB/YS/LS-374/2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants and animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Arumugam, V., Venkatesan, M., Ramachandran, S. et al. Bioactive Peptides from Marine Ascidians and Future Drug Development–A Review. Int J Pept Res Ther 24, 13–18 (2018). https://doi.org/10.1007/s10989-017-9662-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-017-9662-9