Abstract
Graph-based analysis is a promising approach for analyzing the functional and structural connectivity of landscapes. In human-shaped landscapes, species have become vulnerable to land degradation and connectivity loss between habitat patches. Movement across the landscape is a key process for species survival that needs to be further investigated for heterogeneous human-dominated landscapes. The common frog (Rana temporaria) was used as a case study to explore and provide a graph connectivity analysis framework that integrates habitat suitability and dispersal responses to landscape permeability. The main habitat patches influencing habitat availability and connectivity were highlighted by using the software Conefor Sensinode 2.2. One of the main advantages of the presented graph-theoretical approach is its ability to provide a large choice of variables to be used based on the study’s assumptions and knowledge about target species. Based on dispersal simulation modelling in potential suitable habitat corridors, three distinct patterns of nodes connections of differing importance were revealed. These patterns are locally influenced by anthropogenic barriers, landscape permeability, and habitat suitability. And they are affected by different suitability and availability gradients to maximize the best possible settlement by the common frog within a terrestrial habitat continuum. The study determined the key role of landscape-based approaches for identifying the “availability-suitability-connectivity” patterns from a local to regional approach to provide an operational tool for landscape planning.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In fragmented human-dominated landscapes, movements across the landscape matrix are fundamental for plant and animal species survival (Wiens et al. 1993). Depending on the type of pressures and related anthropogenic disturbances within the landscape matrix, the effects of fragmentation vary with the specific habitat needs of particular species. Responses to habitat loss depend both on how the species uses resources in space and on the spatial pattern of the habitat fragmentation (Fahrig 2003). Habitat fragmentation leads to concomitant processes of decreasing habitat suitability and increasing patch isolation of remaining habitat patches within the landscape matrix (Andrén 1994; Joly et al. 2003). Movements between remaining habitat patches are also impeded by landscape matrix permeability loss and anthropogenic barriers. Yet, landscape connectivity remains of fundamental importance for population stability (movement of genes, individuals, populations and species over multiple time scales) (Taylor et al. 1993; Minor and Urban 2008). From a conservation biology perspective functional connectivity depends on how biological flows are influenced by landscape features (Foll and Gaggiotti 2006; Gauffre et al. 2008) and on how landscape features are organized within the matrix in relation to landscape continuities or barriers (i.e. structural connectivity) (Taylor et al. 1993). For conservation and land management, knowing species’ responses to landscape connectivity loss in relation to changes in land cover and environmental conditions is important. Two types of approaches are usually used to assess connectivity: (1) evaluation of the strength of connections between patches and identification of patch isolation based on dispersal simulation within the landscape matrix, using methods such as least-cost modelling, circuit theory and individual based models (Adriaensen et al. 2003; McRae et al. 2008; Janin et al. 2009; Wang et al. 2009; Safner et al. 2010; Sawyer et al. 2011); and (2) identification of continuities on species population distribution, based on habitat suitability modelling (Richard and Armstrong 2009; Bollmann et al. 2011). These two approaches can be integrated within a graph theory framework (also known as network analysis) (Urban and Keitt 2001; Schadt et al. 2002; Pascual-Hortal and Saura 2008; Zetterberg et al. 2010; Pereira et al. 2011), particularly for landscape planning applications (Taylor et al. 1993; Marulli and Mallarach 2005; Opdam et al. 2006; Saura and Pascual-Hortal 2007; Minor and Urban 2008; Rayfield et al. 2010; Garcia-Feced et al. 2011; Pereira et al. 2011).
A graph provides a simple method of representing habitat patches within a landscape while allowing the integration of functional flows between patches to identify the most important connected nodes and related connectivity measures (Bodin and Saura 2010; Pereira et al. 2011). When graph theory is incorporated on an integrative approach considering species-specific functional and structural landscape connectivity aspects, the method becomes promising to efficiently explore and analyze ecological networks, landscapes and habitats (Urban and Keitt 2001; Pascual-Hortal and Saura 2006; Minor and Urban 2008; Saura and Torné 2009; Urban et al. 2009; Galpern et al. 2011).
Efforts are needed to make these graph-theoretical approaches operational within ecological assessments and integrative landscape planning programs. We focus on an example to test the most important aspects of the habitat network structure that need to be maintained within a decision-making planning exercise. We combined least-cost and habitat suitability modelling with a graph-based representation and analysis (Dale and Fortin 2010; Galpern et al. 2011) to provide an operational method for planning, design, and assessment applications. The breeding site locations of the common frog (Rana temporaria) in the French Alps provided a testing ground for the development of the operational approach. The study focused on a twofold integration: (1) effects of landscape matrix permeability as a constraint influencing home-range area available around ponds and connections between populations (i.e. for this study patches were set to represent annual home ranges) and (2) effects of forest patch distribution as a driver of habitat suitability. Pond-breeding amphibians in permanent water bodies represent a challenge for a patch-based analytical approach to connectivity (Fortuna et al. 2006; Hartel et al. 2010; Zetterberg et al. 2010; Curado et al. 2011) due to their life cycle involving annual migrations to and from aquatics and upland habitats. This dual capacity makes amphibians extremely dependent on the surrounding environment and more vulnerable to land degradation and anthropogenic pressures (Fahrig et al. 1995; Allentoft and O’Brien 2010). Consequently when combined with its restricted dispersal ability, a holistic landscape level approach is needed when planning for amphibian conservation measures and habitat management. In what follows, we propose a multi-scale hierarchical view of the landscape, represented by ponds, patches and clusters.
Materials and methods
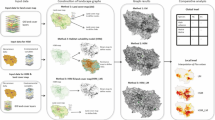
We only used breeding ponds site locations for the development of patch-based graphs (Galpern et al. 2011); these locations were described by the node attributes (i.e. patch quality) and links between nodes (i.e. connections). The graph was used for summarizing landscape organization and permeability relationships (Garcia-Feced et al. 2011; Galpern et al. 2011). Figure 1 shows the logical sequence of steps and the multi-scale hierarchical criteria used to build up the approach presented in this work as follows: (i) annual home-range area patch modelled as nodes based on least-cost surface modelling to simulate frogs’ annual mean dispersal around breeding sites (Safner et al. 2010; Zetterberg et al. 2010); (ii) node attribute assessment based on habitat suitability modelling; (iii) identification of potential node connections based on dispersal scenario; (iv) graph connectivity analysis based on indices computed using the software Conefor Sensinode (Saura and Torné 2009) and combining node attributes and connections.
Study area and common frog pond occupancy
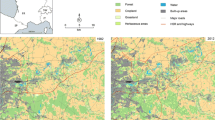
This study used the common frog (R. temporaria) as the focal species in the French Alps. This species is an explosive breeder that reproduces in this area in spring in various types of aquatic habitats. After the reproductive period, the frogs migrate to forest patches (distant up to 1,500 m) where hibernation occurs. The study area covers 4,067 km, within the regions of Isère and Savoie (Fig. 2), encompassing a densely populated central valley in the outer French Alps named Grésivaudan. The two main urban areas in the valley—Grenoble and Chambéry—with a population of approximately 500,000 inhabitants and 100,000 inhabitants respectively are undergoing increasing suburban sprawl (Fig. 2). The landscape matrix constitutes a complex gradient with different degrees of urban pressures surrounded by three mountainous ranges that present large forest patches acting as natural landscape units under the tree line (1,600 m).The northwest part of the area is composed of a dense agricultural network interlaced with small fragments of forest patches. A dense transportation infrastructure web is highly concentrated in the area with four main highways and several national and local minor roads (Fig. 2). Local populations of the common frog are threatened with isolation and a high mortality rate due to increasing forest fragmentation, urban sprawl, and pressures from road traffic. Because the subalpine belt, frog dispersal patterns are constrained by environmental variables such as climatic limiting factors, we focused on common frog populations occurring under the tree line. Thus, the study area elevation is between 200 and 1,600 m. We used 212 breeding sites (Fig. 2) from non-governmental organization surveys (LPO Isère and Avenir 38). The data used included potential breeding sites considered as stable patches where frogs were observed to be present and breeding during several years without discontinuities.
Annual home-range area patches as nodes based on least-cost surfaces around breeding sites
Cost-distance methods based on landscape matrix permeability (Ray et al. 2002; Adriaensen et al. 2003; Joly et al. 2003; Janin et al. 2009) were used to define annual home-range patches (Safner et al. 2010; Zetterberg et al. 2010). The patches were set to delineate annual home ranges, as the annually terrestrial surface of migration around a suitable pond based on landscape permeability. The patches are supposed to contain all the resources needed throughout the year for spawning, foraging and overwintering. The resulting patches are considered as centroids for the nodes of the spatial graph network representation. For the cost distance analysis, Spatial Analyst in the ArcGIS software was used (ESRI Inc, USA, 2008). The computation is based on the energy that individuals would spend during migration from ponds, followed by the additive costs surface of migration from breeding patches. In our simulations, individuals migrate along surface of least resistance and stopped when either they reached a barrier (e.g. highways) or reached the maximum cost of migration (Ray et al. 2002), defined as the minimum value of friction coefficient times the total migration distance. The matrix permeability was obtained from a friction map providing inputs in terms of species ability to cross the landscape matrix. The friction map was also calculated with ArcGIS software by applying resistance cost values (Safner et al. 2010) to hydrological and road networks derived from the French National Geographic Institute in tandem with CORINE Land Cover data raster layer (EEA 2007) (Table 1). CORINE Land Cover is a standardized land cover map for European Union countries with a minimum mapping unit size of 25 hectares. The friction map resolution was set to 5 m to fit the minimum landscape feature width used for the river and water networks. A maximum cumulative dispersal distance of 1,500 m corresponding to the maximum annual common frog dispersal distance (Kovar et al. 2009; Safner et al. 2010) was used for the least-cost surface analysis.
Node attributes based on habitat suitability
Habitat suitability considered here as a probability of occurrence in uplands within each annual home-range patch. It was considered as the attribute of each node of the resulting graph. As we only had presence data, a maximum entropy modelling approach was implemented to predict habitat suitability (Philips et al. 2006). The MaxEnt software approach (Philips and Dudik 2008) was applied to the 212 common frog breeding site locations (Fig. 2). Environmental predictors in relation to the species’ ecological requirements were computed and integrated in the analysis (Miaud et al. 1999; Pahkala et al. 2001; Palo et al. 2004): (1) distance to wetlands; (2) distance to forest patches crossed by a river; (3) slope; (4) elevation; (5) landscape indices as (largest forest patch index, forest mean and forest patch area, forest patch and densities) to describe the structure and organization of forest patch habitats for the common frog; and (6) land cover. Landscape indices were computed with Fragstats software (McGarigal et al. 2002) using a moving window of 1,500 m to match the terrestrial habitat area annually covered by the common frog. Landscape index calculation used the forest/non-forest European binary map provided by the Join Research Centre (Pekkarinen and Reithmaier 2009), in tandem with Corine Land Cover 2006 layer. The predictor resolution was set to 25 m. Seventy percent of the 212 presence data points were used as training data for model construction. The remaining 30% was randomly set as test data for assessing the model’s discriminative capabilities (ROC analysis) (Pearce and Ferrier 2000; Baldwin 2009) and omission rate (Anderson et al. 2003; Ward 2007). Fifty model replicates were processed to choose the best predictive model as calculated by the MaxEnt software. The model was selected according to the ROC analysis and omission rate with minimum training presence and tenth percentile training presence cut-off values (Philips and Dudik 2008; Baldwin 2009; Rödder et al. 2009; Morueta-Holme et al. 2010). The output raster map was used to calculate the mean occurrence probability in each annual home-range patch as a measure of habitat suitability.
Ecological network and potential connectivity structures
We tested the influence of potential flows on the independent annual home-range patch availability and their importance as connectivity providers by considering a connection scenario between patches. For the common frog, the maximum annual dispersal distance was assessed as 1,500 m (Safner et al. 2010). This distance was tested by radio tracking in previous studies within the area (Kovar et al. 2009; Janin et al. 2009; Safner et al. 2010). These studies showed that, some individuals may be observed far from any home-range patches centred on aquatic breeding sites, suggesting potential long distance dispersal. As a result, it was considered that some individuals may disperse by up to 3,000 m within a network of suitable areas corresponding to ‘suitable habitat corridors’ facilitating potential long-distance dispersal between annual home-ranges patches. A probability threshold to the probability of occurrence raster layer was applied to obtain a Boolean raster layer (Hu and Jiang 2010) by selecting the most suitable areas. Thus, a tenth percentile training presence threshold was chosen as the cut-off probability value (Rödder et al. 2009) for ‘suitable habitat corridor’ identification. A least-cost path simulation with a cumulative dispersal distance of 3,000 m was then applied to the ‘corridors’ based on the previously set resistance values. This dispersal scenario simulation served to identify connected or unconnected pairs of patches.
Spatial graph connectivity analysis
In order to find important structures within the ecological network, the connectivity of the resulting graph network was analyzed based on the Integral Index of Connectivity (IIC) (Pascual-Hortal and Saura 2006). This index is based on node attributes (i.e. the mean occurrence probability for each patch in our case) and unweighted links (i.e. the connection scenario) computed using the Conefor Sensinode software (Saura and Torné 2009). IIC index was chosen due to the unweighted nature of the node links (connected or unconnected) estimated with the connection scenario in comparison to the probability of connectivity index (PC) based on weighted links (Bodin and Saura 2010). One of the main advantages of the IIC index is the integration of habitat suitability and connectivity measures into a single value for the overall landscape connectivity. This calculated value provides an efficient indicator of patch availability according to permeability and habitat suitability depending on landscape matrix features. The same software tool was used to calculate the relative ranking of each patch within the network as connectivity providers according to their relevance to maintaining the overall connectivity of the graph network (dIIC). The dIIC index corresponds to the ranking of each patch according to how much the IIC value decreased when a given patch is removed. IIC and dIIC were computed for the connection scenario where home-range patches were allowed to be connected with a resistance cost determined in the prior section.
Results
Least-cost dispersal areas as a network of nodes
We simulated migration areas with a maximum dispersal distance of 1,500 m (see methods) from each of the 212 pond breeding sites. This resulted in 83 annual home range patches (Fig. 3), which represented the nodes in the spatial graph.
Breeding sites were lumped together in the same least cost surfaces if cost distances between sites were less than the maximum cost distance of dispersion. Two distinct patterns were observed (Fig. 3): (1) large and continuous annual home-range patches including more than one breeding site influenced by very permeable forest lands (e.g. areas A and B); and (2) small and scattered patches including only one isolated breeding site and influenced by human-dominated and forest-fragmented landscapes with urban fabric and roads as barriers (e.g. areas C and D, in Fig. 3).
Common frog’s occurrence prediction
The best model for predicting the frogs’ probability of occurrence among the fifty replicates provided a discriminative capability of 85.4% (AUC = 0.854). The omission rate was null at the minimum training presence threshold (logistic threshold of 0.117) and 7.4% at the tenth percentile training presence (logistic threshold of 0.251). The relative contributions of environmental variables to the best predictive model were as follows: (1) distance to wetlands (51.5%); (2) distance to forest patches crossed by a river (19.3%); (3) slope (8.2%); (4) elevation (6.5%); (5) largest forest patch index (5.7%); (6) forest patch density (4.6%); (7) land cover (3.9%); and (8) mean forest patch area (0.4%). The potential distribution of the common frog (Fig. 4) showed the effect of dense urbanized areas and highways as main barriers and unsuitable habitats. These distribution patterns showed scattered and isolated potential suitable areas concomitant with non-continuous forest patches distribution. The results showed that the species’ highest presence probability followed a wide central distribution range encompassing some western and eastern corridors, but the overall scattered distribution was constrained by main roads and suburban sprawl (Fig. 4).
Connection scenario between patches
The tenth percentile training presence threshold of 0.251 was applied to the distribution map of the common frog probability of occurrence (Fig. 4) to identify potential ‘suitable habitat corridors’ within the study area. The 3,000 m dispersal distance for the annual home-range patch connection scenario within the suitable habitat corridor networks highlighted several connectivity patterns but also the lack of connections influenced by landscape barriers. In sum, the 212 breeding ponds observations were lumped into 83 home-range patches (multiple ponds within a larger patch) and 16 of these were further connected at the 3,000 m distance (i.e. maximum distance observed by radio-tracking in previous studies) to create five clusters. From this, 67 home-range patches were totally unconnected, many were isolated by distance, while others were isolated by anthropogenic and natural barriers even if separated from another patch by less than 3,000 m (Fig. 5a). Five groups of more than two connected annual home-range patches were identified as clusters (A–E in Fig. 5a): seven connected patches for cluster A, five connected patches for cluster B, four nodes for clusters C and D and three connected nodes for cluster E. Clusters A and B are located within a natural landscape with large and homogeneous forests where the common frog has no constraints on movement between habitat patches. Most of the isolated patches were located in the most fragmented and dissected areas of the landscape matrix. Most of the unconnected patches or pairs of patches (e.g. F, H–K) were in a radius of less than 3,000 m from the closest patches, or clusters (Fig. 5a). The results highlight a pattern of discontinuities as a product of a heavily human-dominated landscape matrix with scattered forest fragments of different sizes.
Connection scenario and graph connectivity analysis: pairs of annual home-range patches and clusters of more than two connected patches within the habitat suitable corridors with a cumulative dispersal distance of 3,000 m (components are identified by letters A–O); mean occurrence probability for each patch as predicted by Maxent model, and patch importance for connectivity index (dIIC) (high, medium and low classes correspond to dIIC values of 2.6–6.8, 1–2.6 and 0–1%, respectively)
Graph connectivity analysis
Mean occurrence probability for each patch as predicted by the Maxent model (Fig. 5b) showed that 15 of the 83 patches were identified as the most suitable patches for the common frog with the highest mean occurrence probability (0.74) for the smallest patch (0.1 km²) (patch L1 on Fig. 5b). The largest patch of the graph (15.6 km²) presented a rather low occurrence probability (0.40), as a result of the unfavourable landscape permeability context. The inherent habitat values were high but the broader landscape permeability was low, as a consequence of the small size of migration areas.
When the connection scenario was applied, the dIIC index (Fig. 5c) ranked each patch as a connectivity provider in relation to its availability (Fig. 5a) and suitability (Fig. 5b). Clusters A and B (Fig. 5c) showed the highest cumulative connectivity importance (sum of the dIIC values for each patch in the cluster) with values of 39.71 and 30.78% respectively. These clusters showed a high importance for all their annual home-range patches (total area covered by the patches: 56 km² for cluster A and 38 km² for cluster B) (Fig. 5c). Clusters C, D and E showed the lowest cumulative connectivity importance values of 7.80, 5.57, and 4.18%, respectively, with a medium dIIC index value for each patch. Pair L presented a cumulative connectivity importance of 9.21% with both patches of high importance. The patches of pair L were also of higher importance than clusters C, D, and E and other pairs of patches, suggesting that pair L may be important in terms of connectivity conservation and local habitat suitability. In all, dIIC value for each annual home-range patches of the graph combined with the connection scenario provided some trends on the patch status for planning. Hence, we proceeded to a new dIIC computation by removing clusters A and B from the analysis, in order to highlight differences for each patch in terms of importance in clusters or pairs. These two clusters (A and B) are of limited interest in terms of prioritization since they are located within a natural landscape as compare to clusters and pairs located in urbanized and heterogeneous areas where rapid perturbations may occur. For landscape planning purposes, we can proceed to the removal of these clusters in order to highlight the hierarchical importance of patches within clusters of pairs of nodes (Fig. 5d). We can then obtain a final map of suitable habitats corridors between annual home-range patches of high importance. This type of output help to prioritize landscape planning to target areas, by maximizing and maintaining connectivity in particular important clusters and pairs of patches.
Discussion
How does landscape influence the dispersal and suitability within annual home-range patches?
The landscape feature distribution, permeability, and quality are considered the main drivers of dispersal patterns for the common frog in human-dominated landscapes, where rapid perturbations on connectivity and suitability occur due to suburban sprawl and increased development of the road network (Johansson et al. 2005; Safner et al. 2010). In this study, we first focused on how the species use the landscape around breeding sites, which we assessed by simulating annual home-range patch distributions based on least-cost modelling. As showed in other studies on amphibian populations (Ray et al. 2002; Janin et al. 2009; Wang et al. 2009; Safner et al. 2010), the approach allowed the identification of small and scattered patches with a single breeding site embedded within a human-dominated landscape as well as patches that contained multiple breeding sites as meta-populations. Nevertheless, a limited home range may only act as a surrogate of the local landscape pressures on amphibian dispersal abilities. In this context, the quantification of terrestrial habitat suitability within home-range patches highlights small and scattered patches with high potential habitat suitability, suggesting that these patches should be protected by limiting habitat disturbance from urbanization or intensive agriculture, as they serve as “stepping stones” connecting up the broader landscape.
How does landscape connectivity influence patch isolation or cluster organisation?
Assuming that inter-patch connectivity may exist (e.g. dispersal beyond annual home-range patches), we tested a connection scenario based on a dispersal of 3,000 m in potential suitable habitat corridors derived from the MaxEnt model. Based on this scenario, we then performed a landscape-based functional connectivity analysis Three distinct patterns of annual home-range patch organization as influenced by anthropogenic barriers, landscape permeability, and habitat suitability were found: isolated patches, pairs of patches, and clusters. The dIIC index ranked each patch as connectivity providers. The graph connectivity analysis also demonstrated the suitability versus availability and associated tradeoffs required for the settlement of the common frog in a terrestrial forested habitat continuum. Indicating that clusters of annual home-range patches are supposed to be more resilient than isolated patches, especially in landscapes where rapid changes may occur.
Even if we based our connection scenario on expert knowledge and radio tracking surveys, the quantification of links between patches is a key issue, especially at the regional level. As in previous studies applied to amphibians (Ray et al. 2002; Joly et al. 2003; Safner et al. 2010), least cost modelling was considered to be the most efficient approach for the identification of impermeable barriers no matter the dispersal distance. It must be noted that the aim of the study was not to explore other competing approaches for calculating the strength of the links between patches as movement models (e.g. cellular automata and individual based models) or circuit theory. Even so, despite the approach used, the key question still remains on the threshold value needed for the identification of connections (Lasso 2008). A genetic approach may be useful in the future to improve link estimation and weights (Safner et al. 2010). Genetic distance may help define the links between patches within a graph analysis scenario.
Toward an operational tool for patches prioritization
The modelling approach introduced in this study provides an operational approach for planning species connectivity; it is based on a spatial graph construction integrating the effects of the local landscape on the dispersal and occurrence of common frogs in each patch. This also provides information on the opposed effects of natural and human-dominated landscapes in relation to habitat suitability and patch size, this last one considered here as the dispersal area around ponds that depends on landscape permeability. The graph-theoretical approach is also a promising tool for the assessment of local landscape connectivity for the common frog. In all, Conefor Sensinode software provides a direct way for integrating habitat patch distribution, suitability issues and expert knowledge. Within the context of global amphibian decline (Allentoft and O’Brien 2010), the methodological framework proposed in this study may be useful for other landscape, where sensitive amphibian species may benefit from patch prioritization in terms of connectivity and habitat suitability. The connectivity analysis also suggests insights for further land planning perspectives. Scenarios of restoring connections between pairs and clusters of medium importance may be tested to identify the best suitable patches and clusters to improve landscape connectivity. Further analyses at the component level (network of connected ponds) may help to strengthen and quantify the importance of terrestrial habitat quality, availability and permeability between and within patches (intra and inter patch connectivity). All the more, the use of the betweenness centrality index in tandem with the previous indices should help to identify patches as stepping stones when considering biological flows at a broader scale (Bodin and Saura 2010).
Still we advice caution in the interpretation of results as many sources of uncertainties inherent to different phases of the approach needs to be considered. Albeit its limitations due to uncertainties, graph based analysis is useful also as a heuristic framework requiring a relatively low data input (Urban and Keitt 2001; Minor and Urban 2008).
Conclusion
The integrative and hierarchical approach used in this study provides insight on how to combine terrestrial habitat availability and suitability gradients influenced by landscape patterns for a functional connectivity analysis based only on presence data locations available at a regional level. Nevertheless, presence data should be treated with care when metapopulation dynamics are considered and when pond areas are dynamic. Potential habitat locations may also be considered as patches even if the species is not currently observed but habitat suitability conditions are high. In this context, the use of a population model and genetic information for each pond may help to identify different levels of population structure and help to provide relevant weighted links in order to complete the connectivity analysis.
The multi-scale hierarchical view of the landscape proposed in this study, represented by breeding ponds sites, patches and clusters allowed a holistic analysis of the landscape matrix particularly important for species that move from aquatic to terrestrial habitats. It is expected that effective management actions rely on maintaining acceptable levels of overall habitat connectivity considering levels of increasing anthropogenic pressures and barriers.
In this study we demonstrated the applied value of graph-based network analysis as a means to estimate parameters that measures different connectivity-related aspects of individual patches, which may be helpful, as a complement to other relevant ecological information, to optimize conservation efforts in the near future. Also, we hope that the approach used will help to further explore how the choice of links and node attributes (i.e. field data vs. predicted data) may influence graph connectivity analysis outputs.
References
Adriaensen F, Chardon JP, DeBlust GE, Swinnen E, Villalba S, Gulinck H, Matthysen E (2003) The application of least-cost modelling as a functional landscape model. Landsc Urban Plan 64:233–247
Allentoft ME, O’Brien J (2010) Global amphibian declines, loss of genetic diversity and fitness: a review. Diversity 2:47–71
Anderson RP, Lewc DA, Peterson T (2003) Evaluating predictive models of species’ distributions: criteria for selecting optimal models. Ecol Model 162:211–232
Andrén H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355–366
Baldwin RA (2009) Use of maximum entropy modeling in wildlife research. Entropy 11:854–866
Bodin Ö, Saura S (2010) Ranking individual habitat patches as connectivity providers: integrating network analysis and patch removal experiments. Ecol Model 221:2393–2405
Bollmann K, Graf RF, Suter W (2011) Quantitative predictions for patch occupancy of capercaillie in fragmented habitats. Ecography 34:276–286
Curado N, Hartel T, Arntzen JW (2011) Amphibian pond loss as a function of landscape change—a case study over three decades in an agricultural area of northern France. Biol Conserv. doi:10.1016/j.biocon.2011.02.011
Dale MRT, Fortin M-J (2010) From graphs to spatial graphs. Annu Rev Ecol Evol Syst 41:21–38
EEA (2007) CLC2006 technical guidelines. EEA technical report. European Environmental Agency, Copenhagen
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Fahrig L, Pedlar J, Pope S, Taylor P, Wegener J (1995) Effect of road traffic on amphibian density. Biol Conserv 73:177–182
Foll M, Gaggiotti OE (2006) Identifying the environmental factors that determine the genetic structure of populations. Genetics 174:875–891
Fortuna MA, Gomez-Rodriguez C, Bascompte J (2006) Spatial network structure and amphibian persistence in stochastic environments. Proc R Soc Lond B 273:1429–1434
Galpern P, Manseau M, Fall A (2011) Patch-based graphs of landscape connectivity: a guide to construction, analysis and application for conservation. Biol Conserv 144:44–55
Garcia-Feced C, Saura S, Elena-Rossello H (2011) Improving landscape connectivity in forest districts: a two-stage process for prioritizing agricultural patches for reforestation. For Ecol Manag 161:154–161
Gauffre B, Estoup A, Bretagnolle V, Cosson JF (2008) Spatial genetic structure of a small rodent in a heterogeneous landscape. Mol Ecol 17:4619–4629
Hartel T, Nemes S, Öllerer K, Cogalniceanu D, Moga CI, Arntzen JW (2010) Using connectivity metrics and niche modelling to explore the occurrence of the northern crested newt Triturus cristatus (Amphibia, Caudata) in a traditionally managed landscape. Environ Conserv 37:195–200
Hu J, Jiang Z (2010) Predicting the potential distribution of the endangered Przewalski’s gazelle. J Zool 282:54–63
Janin A, Léna J-P, Ray N, Delacourt C, Allemand P, Joly P (2009) Assessing landscape connectivity with calibrated cost-distance modelling: predicting common toad distribution in a context of spreading agriculture. J Appl Ecol 46:1–9
Johansson M, Primmer CR, Sahlsten J, Merilä JU (2005) The influence of landscape structure on occurrence, abundance and genetic diversity of the common frog, Rana temporaria. Glob Change Biol 11:1664–1679
Joly P, Morand C, Cohas A (2003) Habitat fragmentation and amphibian conservation: building a tool for assessing landscape matrix connectivity. C R Biol 326:132–139
Kovar R, Brabec M, Vita R, Bocek R (2009) Spring migration distances of some Central European amphibian species. Amphib-Reptil 30:367–378
Lasso E (2008) The importance of setting the right genetic distance threshold for identification of clones using amplified fragment length polymorphism: a case study with five species in the tropical plant genus Piper. Mol Ecol Resour 8:74–82
Marulli J, Mallarach JM (2005) A GIS methodology for assessing ecological connectivity: application to the Barcelona Metropolitan Area. Landsc Urban Plan 71:243–262
McGarigal K, Cushman SA, Neel MC, Ene E (2002) FRAGSTATS: Spatial pattern analysis program for categorical maps. Computer software program produced by the authors at the University of Massachusetts, Amherst. http://www.umass.edu/landeco/research/fragstats/downloads/fragstats_downloads.html
McRae BH, Dickson BG, Keitt TH, Shah VB (2008) Using circuit theory to model connectivity in ecology, evolution, and conservation. Ecology 89:2712–2724
Miaud C, Guyétant R, Elmberg J (1999) Variation in life-history traits in the common frog Rana temporaria (Amphibia: Anura): a literature reviews and new data from the French Alps. J Zool 249:61–73
Minor ES, Urban D (2008) A graph-theory framework for evaluating landscape connectivity and conservation planning. Conserv Biol 31:297–307
Morueta-Holme N, Fløjgaard C, Svenning J (2010) Climate change risks and conservation implications for a threatened small-range mammal species. PLoS One 5:1–12
Opdam P, Steingröver E, Van Rooij S (2006) Ecological networks: a spatial concept for multi-actor planning of sustainable landscapes. Landsc Urban Plan 75:322–332
Pahkala M, Laurila A, Merilä J (2001) Carry-over effects of ultraviolet-B radiation on larval fitness in Rana temporaria. Proc R Soc Lond B 268:1699–1706
Palo JU, Schmeller DS, Laurila A (2004) High degree of population subdivision in a widespread amphibian. Mol Ecol 13:2631–2644
Pascual-Hortal L, Saura S (2006) Comparison and development of new graph-based landscape connectivity indices: towards the priorization of habitat patches and corridors for conservation. Landscape Ecol 21:959–967
Pascual-Hortal L, Saura S (2008) Integrating landscape connectivity in broad-scale forest planning through a new graph-based habitat availability methodology: application to capercaillie (Tetrao urogallus) in Catalonia (NE Spain). Eur J For Res 127:23–31
Pearce J, Ferrier S (2000) Evaluating the predictive performance of habitat models developed using logistic regression. Ecol Model 133:225–245
Pekkarinen A, Reithmaier LPS (2009) Pan European forest/non-forest mapping with Landsat ETM+ and Corine land Cover 200 data. J Photogramm Remote Sens 64:173–183
Pereira M, Segurado P, Neves N (2011) Using spatial network structure in landscape management and planning: a case study with pond turtles. Landsc Urban Plan 100:67–76
Philips SJ, Dudik M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175
Philips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259
Ray N, Lehmann A, Joly P (2002) Modeling spatial distribution of amphibian populations: a GIS approach based on habitat matrix permeability. Biodivers Conserv 11:2143–2165
Rayfield B, Fortin M-J, Fall A (2010) The sensitivity of least-cost habitat graphs to relative cost surface values. Landscape Ecol 25:519–532
Richard Y, Armstrong DP (2009) The importance of integrating landscape ecology in habitat models: isolation-driven occurrence of north island robins in a fragmented landscape. Landscape Ecol 25:1363–1374
Rödder D, Kielgast J, Bielby J, Schmidtlein S, Bosch J, Garner T, Veith M, Walker S, Fisher M, Lötters S (2009) Global amphibian risk assessment for the panzootic chytrid fungus. Diversity 1:52–66
Safner T, Miaud C, Gaggiotti O, Decout S, Rioux D, Zundel S, Manel S (2010) Combining demography and genetic analysis to assess the population structure of an amphibian in a human-dominated landscape. Conserv Genet 12:161–173
Saura S, Pascual-Hortal L (2007) A new habitat availability index to integrate connectivity in landscape conservation planning: comparison with existing indices and application to a case study. Landsc Urban Plan 83:91–103
Saura S, Torné J (2009) Conefor Sensinode 2.2: a software package for quantifying the importance of habitat patches for landscape connectivity. Environ Model Softw 24:135–139
Sawyer SC, Epps CW, Brashares JS (2011) Placing linkages among fragmented habitats: do least-cost models reflect how animals use landscapes? J Appl Ecol 48:668–678
Schadt S, Knauer F, Kaczensky P et al (2002) Rule-based assessment of suitable habitat and patch connectivity for the Eurasian lynx. Ecol Appl 12:1469–1483
Taylor PD, Fahrig L, Henein K, Merriam NG (1993) Connectivity is a vital element of landscape structure. Oikos 68:571–573
Urban D, Keitt T (2001) Landscape connectivity: a graph-theoretic perspective. Ecology 82:1205–1218
Urban DL, Minor ES, Treml EA et al (2009) Graph models of habitat mosaics. Ecol Lett 12:260–273
Wang IJ, Savage WK, Shaffer HB (2009) Landscape genetics and least-cost path analysis reveal unexpected dispersal routes in the California tiger salamander (Ambystoma californiense). Mol Ecol 18:1365–1375
Ward DF (2007) Modelling the potential geographic distribution of invasive ant species in New Zealand. Biol Invasion 9:723–735
Wiens JA, Stenseth NC, Van Horne B, Ims RA (1993) Ecological mechanisms and landscape ecology. Oikos 66:369–380
Zetterberg A, Mörtberg UM, Balfors B (2010) Making graph theory operational for landscape ecological assessments, planning, and design. Landsc Urban Plan 95:181–191
Acknowledgments
Support for this work was partly provided from the EU-Interreg Alpine Space Program Econnect (reference number: 116/1/3/A) and the French Ministry of Ecology and Sustainable Development (DEB-ECOTRAM project). We would like to thank Santiago Saura for his advice on the subject and support in the use of the Conefor Sensinode software and Peter Vogt for constructive comments. Many thanks are also given to the members of the non-governmental organizations Avenir 38, LPO Isère, and CPN Savoie for the data provided for this study, and to the two anonymous referees for the constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Decout, S., Manel, S., Miaud, C. et al. Integrative approach for landscape-based graph connectivity analysis: a case study with the common frog (Rana temporaria) in human-dominated landscapes. Landscape Ecol 27, 267–279 (2012). https://doi.org/10.1007/s10980-011-9694-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-011-9694-z