Abstract
Biodiesel is susceptible to autoxidation if exposed to air, light and temperature, during its storage. Physic nut (Jatropha curcas L.) seeds show potential application for biodiesel production since its oil yields high quality biodiesel. This work aims to evaluate the thermal behavior of the physic nut oil and biodiesel, from several Brazilian crops, by means of thermoanalytical techniques. Thermogravimetry (TG) and pressurized-differential scanning calorimetry (PDSC) were used in order to determine the applicability of physic nut biodiesel as fuel. Results suggest that physic nut biodiesel is a practical alternative as renewable and biodegradable fuel able to be used in diesel motors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rising costs of the petroleum-derived fuels, appended to environmental pollution and global warming, has encouraged researches on alternative fuels and low-energy design processes. Nowadays, biodiesel became an important alternative due to its environmental benefits and easy industrial fabrication from renewable resources. New investments for industrial-scale plants in Europe, Asia, Australia, United States and Brazil have increased the production of biodiesel which the bigger advantage is the higher lubricity if compared to the mineral diesel; however, such vegetable oils based products present lower oxidation stability and processing temperature [1].
Transesterification reaction of oil or fatty with short chain alcohol, usually methanol and ethanol, leads to a mixture of corresponding mono-alkyl esters, which are defined as biodiesel. As biodiesel presents the same fatty acids compositions of its initial oils or fatties, characterized by a considerable amount of unsaturated fatty acids, its oxidative stability is a crucial property, especially during long-time period storage [2]. Ultraviolet irradiation, high temperature exposition and metal traces (contaminants) can reduce the overall stability of the biofuel, decreasing significantly its quality. Oxidative degradation can affect some properties of the biodiesel such as kinematic viscosity, cetane number, and acid value of the fuel [3]. On the other hand a number of researches groups are searching for substances that inhibit this oxidation process on biodiesel [2, 4].
Several vegetable oils obtained from soybean, castor, sunflower, cotton, corn, palm, and others, are widely applied on biodiesel production. Physic nut (Jatropha curcas L.) is a native species from Latin American. The interest in using the seeds of physic nut for the production of excellent quality biofuel, as a substitute for fossil fuels to reduce greenhouse gas emissions, is rapidly growing [5]. Furthermore, as consequence of its adaptability to different environmental and climates, the physic nut plants show high potential as oleaginous source for different regions of Brazil, mainly north and northeast [6–8].
Physic nut seeds composition shows approximately 30% of fatty (53% of this into the kernel), 24% of protein and 2% of fiber. Overall fatty acid composition is mixture of palmitic (16:0), estearic (18:0), oleic (18:1) and linoleic acids (18:2) [5, 8]. Depending on the location of the crop the proportions of these two last fatty acids are 50 and 30%, respectively. Samples from Indonesia are composed by 40% and 37% of oleic and linoleic acids [7, 9]. In Brazil, these amounts are 30 and 50% of these acids, indicating the predominance of the most unsaturated fatty acid and the higher susceptibility to oxidation [2]. In Mexico, it is found similar proportions of oleic and linoleic acids (40%) [6–8].
Pressurized-differential scanning calorimetry (PDSC) has been applied to evaluate oxidation process, polymerization, antioxidant effects and thermal degradation reactions. This technique is widely recommended to determinate oxidation of oils and biodiesels, in addition to thermogravimetric data [3, 9–11]. The thermogravimetry technique (TG) is based on the study of sample mass variation, as function either of time or temperature. The most important parameters of TG analysis are initial decomposition temperature, the maximum temperature of conversion and final decomposition temperature. The DTG curve is defined as the differentiate curve from TG, in which the sample mass loss is proportional to the area under the derivative peak [9, 12, 13].
Inserted on this context, this work aims to determine the oxidative stability and thermal decomposition of physic nut oil and its biodiesel, from several Brazilian crops, by means of TG/DTG and PDSC techniques.
Experimental
The physic nut biodiesel was synthesized by transesterification reaction with ethanol, using oils from seeds of different crops and homogeneous catalyst (KOH). Samples were named as 2005/2006, 2006/2007, 2007/A and 2007/B, been related to harvest period. A consequence, the 2005/2006 and 2006/2007 samples have the lower humidity, among all samples, since they have been stored for a longer time. The 2007/A and B samples differ between them for its storage process. In the first case, the sample was exposed to sun radiation (summer crop), while the other sample was stored under elevated environmental humidity (winter crop).
TG/DTG and PDSC experiments were used to obtain information on the temperature-controlled combustion of the biofuels synthesized. TG/DTG curves were obtained using a simultaneous analyzer SDT 2960 (TA Instruments), alumina pans, under dry air atmosphere (100 mL min−1), heating rate of 10 °C min−1 and temperature range from 10 to 700 °C [14]. Prior to the experiments, the equipment was calibrated by the TG mass (using standards masses and empty beam), DTA baseline and temperature adjust (evaluation of melting endotherms of purity zinc, tin and indium metals) [15].
PDSC experiments were carried out using a DSC 2920 thermal analyzer from TA Instruments, coupled with a high-pressure cell. As standard, approximately 10 mg of the sample was placed in platinum pan, under static dry air atmosphere, being pressurized in isobaric module (0.7 MPa). The isothermal temperature (110 °C), used for the data collection, was reached using a heating rate of 10 °C min−1. The oxidative induction time (OIT) was obtained from the PDSC curves. The PDSC module was calibrated by means of the baseline slope, cell constant and temperature adjust using indium as standard metal.
Results and discussion
Thermogravimetric analysis
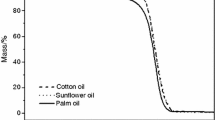
The TG/DTG curves of physic nut oil (Fig. 1) show three events of thermal decomposition assigned to the volatilization and/or combustion of triacylglycerides. The thermodynamic proprieties of physic nut oil and its biodiesel are summarized in Table 1.
The maximum decomposition temperatures (DTG peaks) were 354.0 (2005/2006), 348.9 (2006/2007), 377.4 (2007/A) and 380.5 °C (2007/B), with 52.5% (±3.1%) of mass loss attributed to the volatilization of unsaturated fatty acids from the triacylglycerides. The second event of the samples takes place at the average temperature of 382.7 °C (±10.8 °C) with 36.9% (±3.3%) of mass loss, which are due to the volatilization of saturated fatty acids. The last stage of thermal decomposition (474.8 ± 13.7 °C) corresponds to the carbonization of residual product [16].
The TG/DTG curves of biodiesel (Fig. 2a−d) show only two mass loss steps, indicating volatilization and/or combustion of ethyl esters (the first step), mainly the ethyl oleate and linoleate, once oleic and linoleic fatty acids are the most abundant components of the initial oils. Such results are in agreement with the literature, in which were observed two mass loss steps for biodiesel [17].
Table 1 shows the decomposition temperature, as well as, the maximum decomposition temperatures from DTG curves, for all biodiesel samples, corresponding to degradation of residuals mono-, di-, and triacylglycerides [16].
For the oil, the first decomposition temperature occurs at about 186 °C (±15 °C), however the yielded biodiesel occurs close to 98 °C (±2 °C), in average. The oil from 2005/2006 crop is less stable among all samples, since it shows the lower decomposition initial temperature 169 °C. The same behavior was observed to its yielded biodiesel (90 °C), being attributed to water content in the dried seeds. As 2005/2006 is the older sample, its humidity is the lowest among the others, as observed by the total mass loss percentage (TG curves).
Oxidative induction time
The results from PDSC analysis demonstrated similar behavior between the oils and biodiesels (Fig. 3) curves, except for 2007/B sample.
According to Berchamans et al. (2007), the quality of Jatropha curcas oils depends on how its seeds were dried, treated and stored. Since the 2007/A sample was dried at approximately 45 °C, water traces has remained in the raw seeds. As consequence, secondary hydrolysis and oxidation reactions has probably occurred to its oil, decreasing its thermal stability (Fig. 1). For the 2007/B sample, such phenomenon is not significant since its oil was treatment at 105 °C (Fig. 1). In general, the OITs present values of 13.1 min (±2.7 min) for both oils and biodiesels (Table 2), not including 2007/B sample that was 33.2 min (±1.3 min), due to the storage process of the oil. The oxidative stability can be used for evaluating the quality of oils, fats and biodiesels. Such property depends on chemical composition, the quality of the raw material, the conditions of refining processes (oils and fats), transesterification route (biodiesel), and the storage conditions [18–20]. According to Smouse [21], besides these factors the oxidative stability also depends on seeds storage. The pronounced oxidative stability for 2007/B oil can be attributed to its seeds storing under elevated environmental humidity (winter crop).
As result of the Brazilian physic nut oil composition (high linoleic fatty acid content) its oxidative stability is widely affected and its values consequently reduced. However, such fact can be solved though blending with others vegetable oils rich in oleic acid [22].
The lower oxidation stability for the majority of biodiesel samples, compared to the oils, can be explained by means of the natural antioxidants release, such as tocopherols and phospholipids, during the refine process of the crude oil [23, 24].
Conclusions
Physic nut oil and ethyl biodiesel from different crops were thermally stable until 203 °C (2007/B) and 108.9 °C (2007/A). The higher volatility of biodiesel, indicated by lower initial decomposition temperature, certifies the quality of physic nut biodiesel as biofuel. The oil and biodiesel from 2005/2006 crop were less stable than the others due to its seeds higher water content. In general, the OITs values were 13 min for both oils and biodiesels, except to 2007/B sample that was 33 min.
References
Moser BR, Sharma BK, Doll KM, Erhan SZ. Diesters from oleic acid: synthesis, low temperature properties, and oxidation stability. J Am Oil Chem Soc. 2007;84:675–80.
Knothe G. Some aspects of biodiesel oxidative stability. Fuel Process Technol. 2007;88:669–77.
Dunn RO. Oxidative stability of biodiesel by dynamic mode pressurized-differential scanning calorimetry (P-DSC). Am Soc Agric Biol Eng. 2006;49(5):1633–41.
Pinto AC, Guarieiro LLN, Rezende MJC, Ribeiro NM, Torres EA, Lopes WA, et al. Biodiesel: an overview. J Braz Chem Soc. 2005;16:1313–30.
Acten WMJ, Verchot L, Franken YJ, Mathijs E, Singh VP, Aerts R, et al. Jatropha bio-diesel production and use. Biomass an bioenergy. Biomass Bioenergy. 2008;32:1063–84.
Berchmans HJ, Hirata S. Biodiesel production from crude Jatropha curcas L. seed oil with a high content of free fatty acids. Bioresour Technol. 2008;99:1716–21.
Foidl N, Foidl G, Sanchez M, Mittelbach M, Hackel S. Jatropha curcas L. as a source of production of biofuel in Nicaragua. Bioresour Technol. 1996;58(1):77–82.
Zhou H, Lu H, Liang B. Solubility of multicomponent systems in the biodiesel production by transesterification of Jatropha curcas L. oil with methanol. J Chem Eng Data. 2006;51:1130–5.
Conceição MM, Candeia RA, Silva FC, Bezerra AF, Fernandes VJ Jr, Souza AG. Thermoanalytical characterization of castor oil biodiesel. Renew Sustain Energy Rev. 2007;11:964–75.
Wan Nik WB, Ani FN, Masjuki HH. Thermal stability evaluation of palm oil as energy transport media. Energy Convers Manag. 2005;46:2198–215.
Dunn RO. Effect of antioxidants on the oxidative stability of methyl soyate (biodiesel). Fuel Process Technol. 2005;86:1071–85.
Goodrum JW. Rapid measurements of boiling point and vapor pressure of short-chain triglycerides by thermogravimetric analysis. J Am Oil Chem Soc. 1997;74(8):947–50.
Mothé CG, Azevedo AD. Análise Térmica de Materiais. São Paulo, 2002;ch.3:32.
Kök MV, Acar C. Kinetics of crude oil combustion. J Therm Anal Calorim. 2006;83(2):445–9.
Kök MV, Pokol G, Keskin C, Madarász J, Bagci S. Light crude oil combustion in the presence of limestone matrix. J Therm Anal Calorim. 2004;75:781–6.
Moura KRM, Silva FC, Brandão KSR, Souza AG, Conceição MM. Estabilidade Térmica do Sebo Bovino e do Biodiesel Metílico e Caracterização Físico-química. In: I Congresso da Rede Brasileira de Tecnologia de Biodiesel, Brasília; 2006;1:207–12.
Dantas MB, Conceição MM, Fernandes VJ Jr, Santos NA, Rosenhaim R, Marques ALB, et al. Rosenhaim thermal and kinetic study of corn biodiesel and obtained by the methanol and ethanol. J Therm Anal Calorim. 2007;87(3):835–9.
Garcia-Mesa JA, Luque de Castro MD, Valcarcel M. Factors affecting the gravimetric determination of the oxidative stability of oils. J Am Oil Chem Soc. 1993;70(3):245–7.
Gutiérrez F. Determination of the oxidative stability of virgin olive oils: comparison of the active oxygen and the Rancimat methods. Grasas Aceites. 1989;40(1):1–5.
Hill SE, Perkins EG. Determination of oxidation stability of soybean oil with the oxidative stability instrument: operation parameter effects. J Am Oil Chem Soc. 1995;72(6):741–3.
Smouse TH. In: Warner K, Eskin NAM, editors. Methods to assess quality and stability of oils and fat-containing foods. Champaign, IL: AOCS; 1995. p. 17–8.
Frankel EN, Huang SW. Improving the oxidative stability of polyunsaturated vegetable oils by blending with high-oleic sunflower oil. J Am Oil Chem Soc. 1994;71(3):255–9.
Jung MY, Yoon SH, Min DB. Effects of processing stepson the contents of minor compounds and oxidation of soybean oil. J Am Oil Chem Soc. 1989;66(1):118–20.
Gordon MH, Rahman IA. Effect of processing on the composition and oxidative stability of coconut oil. J Am Oil Chem Soc. 1991;68(8):574–6.
Acknowledgements
The authors acknowledge CNPq, CAPES and FINEP for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Freire, L.M.S., Bicudo, T.C., Rosenhaim, R. et al. Thermal investigation of oil and biodiesel from Jatropha curcas L.. J Therm Anal Calorim 96, 1029–1033 (2009). https://doi.org/10.1007/s10973-009-0055-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0055-y