Abstract
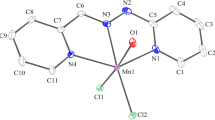
4-(4-ethoxy-phenylhydrazono)-1-phenyl-3-methyl-1H-pyrazolin-5(4H)-one (5a) (H-EMPhP) as ligand and its Cu(II), Co(II) and Ni(II) complexes 4(a-c) were synthesized and characterized by their thermal and spectral properties. The azocoupling product (H-EMPhP), able of azo-hydrazone tautomerism 5(a-d), act as a bidentate ligand involving in coordination the azogroup nitrogen of its common anion (7) and the oxygen atom that is bound to the pyrazole ring of the mentioned anion (7).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
W. Krohs and O. Hensel, Pyrazolone und Dioxopyrazolidine, Cantor/Aulendorf i. Württ, 1961, pp. 411-460.
R. Price, in ‘The Chemistry of Synthetic Dyes’, Editor K. Venkataraman, Vol. III Academic Press, London 1970, pp. 303-369.
H. Zollinger, Color Chemistry, 2nd Edn. VCH, Basel 1991, pp. 110-127, 134-136, 156-157.
M. M. Moustafa and R. M. Issa, Egypt. J. Chem., 42 (1999) 267; Chem. Abstr., 132 (2000) 69948b.
R. R. Rudalal, J. J. Patel and A. G. Mehta, Orient. J. Chem., 15 (1999) 559; Chem. Abstr., 132 (2000) 252441v.
J. J. Patel, G. H. Bhat and K. R. Desai, J. Inst. Chem. (India), 71 (1999) 185; Chem. Abstr., 133 (2000) 136740p.
I. V. Mur, J. Obsch. Khim., 26 (1956) 384.
F. A. Snavely and F. H. Suydam, J. Org. Chem., 24 (1959) 2039.
B. E. Zaitev, V. A. Zaiteva, A. K. Molodkin and E. S. Obraztova, Zh. Neorg. Khim., 24 (1979) 127.
F. A. Adam and M. T. El-Haty, Delta J. Sci., 11 (1987) 1089; Chem. Abstr., 111 (1989) 145745s.
I. Panea, A. Ghirisan, I. Cristea, R. Gropeanu and I. A. Silberg, Het. Commun., 7 (2001) 563.
R. Jones, A. J. Rian, S. Sternhell and S. E. Wright, Tetrahedron, 19 (1963) 1497.
L. M. Jackman and S. Sternhell, Application of Nuclear Magnetic Resonance Spectroscopy in Organic Chemistry, 2ndEdn. Pergamon Press, London 1969, p. 164.
J. Elguero, C. Marzin, A. R. Katritzky and P. Linda, The Tautomerism of Heterocycles, Academic Press, New York 1976, pp. 313-339.
H. Mustroph, Z. Chem., 27 (1987) 281.
Q. Peng, M. Li, K. Gao and L. Cheng, Dyes Pigm., 18 (1992) 271.
J. Oakes and P. Gratton, J. Chem. Soc., Perkin Trans., 2 (1998) 1857.
N. Ertan, Dyes Pigm., 44 (2000) 41.
I. Panea, A. Ghirişan, I. Bâldea, I. Silaghi-Dumitrescu, L. Crăciun and I. A. Silberg, Studia Univ. ‘Babeş-Bolyai’, Chemia, 48 (2003) 65.
S. A. Abdel-Latif and H. B. Hassib, J. Therm. Anal. Cal., 68 (2002) 983.
S. A. Abdel-Latif, O.M. El-Roudi and M. G. K. Mohamed, J. Therm. Anal. Cal., 73 (2003) 939.
S. L. Stefan, J. Thermal Anal., 42 (1994) 1299.
F. E. Mabbs and D. Colisson, Electron Paramagnetic Resonance of d Transition Metal Compound, Elsevier, Amsterdam 1992, p. 105.
G. Simeonov, A. Draganov and D. Ructschev, J. Thermal Anal., 41 (1994) 201.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bâtiu, C., Panea, I., Ghizdavu, L. et al. Divalent transition metal complexes. J Therm Anal Calorim 79, 129–134 (2005). https://doi.org/10.1007/s10973-004-0573-6
Issue Date:
DOI: https://doi.org/10.1007/s10973-004-0573-6