Abstract
Trimethylethoxysilane (TMES) has been recognized as a good co-precursor to increase the degree of hydrophobicity during the synthesis of a silica aerogel because of its methyl groups. Therefore, some physical properties of silica aerogels, including the contact angle and porosity, were investigated using TMES as a co-precursor at different molar ratios with the main precursor such as tetramethoxysilane (TMOS) or tetraethoxysilane (TEOS). In contrast to TMES, most silylating agents such as hexamethyldisilazane (HMDZ) and trimethylchlorosilane (TMCS) have been used for surface modification because of their ability to enhance the hydrophobicity of the aerogel surface. This work examines the silylation effect, which includes increasing hydrophobicity by TMES to determine the possibility of using it as an alternative silylating agent during ambient pressure drying in the synthesis of sodium silicate-based silica aerogel. In addition, the physical properties of sodium silicate-based silica aerogels with silylation under different TMES/TMCS volume ratio are investigated. The physical properties of sodium silicate-based aerogels can be changed by the TMES/TMCS volume ratio during the surface modification step. Aerogels with a high specific surface area (458 m2/g), pore volume (3.215 cm3/g), porosity (92.7%), and contact angle (131.8°) can be obtained TMES/TMCS volume ratio of 40/60.

Highlights
-
Sodium silicate-based silica aerogel was synthesized by APD using TMES/TMCS surface co-modifying agent.
-
TMES can act as an alternative surface modification agent with TMCS.
-
Hydrophobicity was not measured at the case of only TMES usage.
-
TMES/TMCS surface co-modifier for silylation enhances the physical properties of silica aerogel.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Silica aerogels can be synthesized using a sol–gel process followed by different drying methods [1], and these materials were given attention after Kistler’s invention [2]. It shows distinctive physical characteristics such as a high porosity (>90%), low thermal conductivity (<0.02 W/m·K), low dielectric constant (k < 1.7), low refractive index (<1.1), and low bulk density (~0.1 g/cm3) [2,3,4]. It is a promising candidate for a broad range of applications related to these properties, such as building insulation, interlayer dielectrics, drug delivery systems, oil absorbers, gas storage systems, sensors, aerospace applications, hydrogen storage media, capacitors, batteries, construction applications, and filters [1,2,3,4,5].
Various drying methods are used in synthesizing silica aerogel, including supercritical drying (SCD) and ambient pressure drying (APD) [1]. In particular, APD is an attractive drying method for synthesizing silica aerogel because it has a lower cost for production of silica aerogels [6] and is safer to handle than SCD [4]. A surface modification agent is generally introduced for a silylation process before APD to maintain the pore structure and increase the hydrophobicity of the silica aerogel [5]. Generally, trimethylchlorosilane (TMCS) and hexamethyldisilazane (HMDZ) are popular surface modification agents because of their high silylation tendencies [7]. APD is an affordable method that consumes less energy. Thus, it could be a potential route for silica aerogel commercialization [8]. It shows a synergy effect when synthesizing a silica aerogel with sodium silicate, which is a cheap precursor compared to metal alkoxides. Reports are available on silica aerogel prepared by APD with sodium silicate as the main precursor with different surface modification agents [7,8,9,10].
Many reports have been published on the effects of surface modification with some variables for silica aerogels. The effects of different functional groups of silylating agents on the physical properties of silica aerogels [11, 12], comparison between TMCS and HMDZ, which were used for silylation with different molar ratio of surface modification agent/Na2SiO3 [13], and various silylating agents other silylating agents on a silica aerogel have been investigated [14,15,16]. Furthermore, some researchers have reported the combined effect of two different silylating agents with similar molecular formulas.
In this present work, the effect of trimethylethoxysilane (TMES) as a surface modification agent was investigated. TMES has a molecular structure similar to that of TMCS. However, instead of the chlorine present in TMCS, an ethoxy group is attached to TMES, and it does not form HCl during the sylilation process. It has been reported that TMES can be used as a co-precursor to increase the hydrophobicity of a silica aerogel [17,18,19]. In the present study, the effect of a TMES/TMCS mixture on the physical properties of silica aerogels was studied.
2 Experimental procedure
Sodium silicate-based silica aerogels with various percentages of TMES and TMCS were synthesized using a two-step sol–gel process followed by the APD method [20, 21]. The following chemicals were used in the synthesis. Sodium silicate solution (53 wt%, Duksan Chemical Co. ltd., South Korea) was used as the main precursor. Methanol (99.8%, Duksan Chemical Co. ltd., South Korea) and n-hexane (95.0%, Duksan Chemical Co. ltd., South Korea) were used as aging solvents during aging process. Surface modification was performed using TMCS (pH ~1, 98.0%, Daejung Chemicals & Metals Co., ltd., South Korea) and TMES (pH ~8, ≥ 98.0%, skc silicone, South Korea). Acid and base catalysts such as hydrochloric acid (HCl, 35%, Daejung Chemicals & Metals Co., Ltd., South Korea) and ammonium fluoride (NH4F, 98.0%, Sigma-Aldrich, USA) were used for the sol–gel process of the sodium silicate. A water-glass solution with a specific gravity of 1.05 was prepared by mixing sodium silicate agent and distilled water while maintaining the pH value of the solution at approximately 11–12. The solution was stirred to form a homogeneous sodium silicate-based silica sol, and a NH4F solution was added to accelerate the hydrolysis for 4 h at room temperature. This led to the formation of silicic acid from sodium silicate, and NaF was formed as a salt during the hydrolysis step. Then, a hydrochloric acid solution was introduced for the condensation step. The molar ratio of Na2SiO3:H2O:NH4F:HCl was kept constant at 1:121.67:0.2467:4.144. After gelation, the gel was aged for 3 h in an oven at 50 °C. Then, the gel was cut into small pieces and washed five times with de-ionized water. These silica gel pieces were aged in methanol at 50 °C for 24 h. The surface modification process was performed by aging hydrogels in a mixture of methanol and n-hexane at same volume as 50 ml with variation in the volume ratio between TMCS and TMES mixture. In this study, TMES/TMCS volume ratios of 20/80, 40/60, 60/40, and 80/20 were used. In addition, samples silylated by different volume ratios of TMCS compared with the volume of n-hexane (20, 40, 60, 80, and 100 vol%) and TMES alone (50, 100, and 200 vol%) were prepared for comparison. The silica aerogel samples prepared with different surface modification conditions are summarized in Table 1. The reactor was sealed during the surface modification step, and put in the oven at 50 °C for 24 h. After that, the wet gels were washed five times with n-hexane to remove any residue. The aging of the gel was conducted with excess n-hexane at 50 °C in the oven for 24 h to strengthen the silica network. Finally, the silylated wet gels were successively dried at 50 °C for 1 h and 200 °C for 1 h. Figure 1 summarizes the complete experimental process in a flow chart.
The bulk density was calculated using the mass-to-volume ratio. The volume was determined from a cylindrical column of a specific volume, which was filled with aerogel powders [22, 23], and the mass was measured using an electronic microbalance with an accuracy of 10−5 g [22,23,24]. In addition, the porosity of each sample was calculated using the formula given below Eq. 1.
where ρb is the bulk density, ρs is the skeletal density of each sample, and the ρs values were determined using a helium pycnometer (Micromeritics, AccuPyc 1330, USA). The skeletal density values for all the samples were ~1.9 g/cm3. The chemical bonds in the water-glass-based silica aerogels were detected by means of Fourier transform infrared (FTIR) spectroscopy (Perkin Elmer, Model No. 760, USA) using a wavenumber range of 4000 to 400 cm−1. The contact angle (θ) was measured using a contact angle meter (GITsoft, UNI-CAM-M-Dimension, South Korea) to analyze the degree of hydrophobicity of each sample. The specific surface area was calculated using a Brunauer–Emmett–Teller (BET) analysis from the amount of N2 gas adsorbed with a relative pressure in the range of 0.01 < P/P0 < 1. In addition, the total pore volume, average pore diameter, and pore size distribution were analyzed by the Barrett–Joyner–Halenda (BJH) method using the BET surface analyzer (Quantachrome, autosorb-iQ, USA). A field emission scanning electron microscope (FESEM) and energy-dispersive X-ray spectroscopy (EDX) (JEOL, JSM-7001F, Japan) were used to investigate the microstructure and elements present in the silica aerogel surface. The optical transmittance of the prepared aerogel granules was measured in triplicate by using a UV–visible spectrophotometer (JASCO, V-570, USA). The average size of the granules was approximately 2 mm, and the thickness of the cell used to measure the optical transmittance was 10 mm (Cuvets Quartz glass precision cell; Hellma, 6040-UV, German) when using a wavelength of 200–800 nm [25].
3 Results and discussion
The silica aerogels were synthesized using a two-step acid–base catalyzed sol–gel process followed by a two-step acid–base catalyzed sol–gel process. These two processes are shown below in Eqs. 2 and 3.
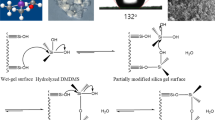
After the sol–gel process, silylation was performed to prevent gel shrinkage during drying. The reaction mechanism for the surface modification is explained in Eq. 4.
Both the sol–gel process and surface modification step influenced the physical properties of the final products [26]. Figure 2 shows the FTIR spectra of aerogel samples with different percentage of the TMCS and TMES silylating agents used to modify the silica aerogel surface. The strong Si–O–Si bonding is observed at 460 and 1100 cm−1, and the peak at 950 cm−1 shows Si-OH absorption, which confirms the formation of a silica network [27]. Further, Si–C stretching vibrations are shown at 850 and 1250 cm−1. In addition, the peak at 2900 cm−1 means C–H bond [28]. These peaks correspond to the silylation process used to modify the silica surface with hydrophobic groups. First, the peaks at 460, 1250, and 2900 cm−1 are shown at samples C100, C80, C60, C80E20, C60E40, C40E60, and C20E80 for different TMCS values, and TMCS and TMES volume percentages, as seen in Fig. 2(a, b). It indicates that the hydrophobicity can decrease with a decrease in the TMCS vol%, and introducing TMES with TMCS can affect the silylating reaction even if the TMCS vol% is lower. In contrast, samples C40, C20, E50, and E100 do not show any peaks related to the Si–C and C–H bonds. Therefore, the surface modification step was not very effective in their cases. In addition, it can be suggested that using TMES alone for the surface modification step could not produce the silylation effect. It is clear that samples E50, E100, and E200 do not show the C–H and Si–C peaks in Fig. 2(c). Therefore, TMES should be used with TMCS to produce the silylation effect. However, sample E200 showed a different phenomenon compared to the other samples, as shown in Fig. 2(c). It has a peak at 2900 cm−1, which indicates the C–H bond rather than the Si–C bond. It is supposed that the TMES introduced during the surface modification step was not completely washed away. Consequently, this peak is the result of the TMES.
The difference in the degree of hydrophobicity under different silylating conditions is shown in Fig. 3 by the contact angle measurements and Fig. 4. Contact angle decreases with a decrease in the TMCS vol%, as shown in Fig. 3(a). This was due to a decrease in the silylation effect with a decrease in the silylating agent vol% [29]. However, the combination of TMES and TMCS that maintained the silylating effect is shown in Fig. 3(b). In particular, the contact angle increased at a TMES/TMCS = 60/40 as volume ratio. Rapid silylation could increase the modification of the surface with hydrophobic methyl groups. Therefore, the inner part of the pores could not be silylated because the permeation of the silylating agent was interrupted as a result of the steric hindrance of the trimethylsilyl groups outside of the pores [30, 31]. In contrast, a lower TMCS vol% induced a lower rate of silylating reaction with an influence on the TMES as the surface modification agent. Hence, the samples with TMES and TMCS mixed silylating agents maintained their degree of hydrophobicity. Additionally, a stable silylating reaction caused a higher degree of hydrophobicity at a TMES/TMCS = 60/40 volume ratio. However, using TMES alone could not produce the surface modification. The TMES could not interact with the silanol groups, which prevented any degree of hydrophobicity. In other words, the sample with TMES alone had hydrophilicity. However, using only TMES up to a 200 vol% could increase the contact angle up to 76.2°. It was considered that under-washed residual TMES on the gel surface caused an increase in the contact angle. This explanation is related to the FTIR spectra shown in Fig. 2, which simply shows the C–H bond without the peaks of the Si–C bonds for samples with an excess TMES vol% introduced, such as 200 vol%. Thus, it was confirmed that the FTIR peak of the C–H bond and the increase in the contact angle were not derived from the surface modification under this condition.
Figure 5 shows the EDX spectra of the sodium silicate-based silica aerogels prepared under different silylation conditions. Silicon and oxygen peaks at approximately 0.5 and 1.7 keV, respectively, are shown for samples C100, C20E80, C40, and E200 [32]. However, a carbon peak at approximately 0.3 eV is shown for samples C100, C20E80, and E200, but not for sample C40 [33]. It gives evidence about whether or not the surface modification was produced because sample C40 underwent silylation with a 40 vol% of TMCS. However, sample C20E80 has the carbon element from the hydrophobic groups even though the TMCS vol% for sample C20E80 is lower than that for sample C40. Additionally, it can be suggested that the carbon peak is shown for sample E200 as a result of the residual TMES that remained at the gel surface in spite of the washing step. The FTIR data for sample E200 in Fig. 2 verifies this idea because it does not have peaks for the Si–C bonds. Therefore, the effects of the excess TMES and lower percentage of TMCS were shown by the EDX spectra and other measurements.
The optical transmittance at 550 nm is shown in Fig. 6 and Table 2. The optical transmittance at 550 nm is in the range of 6–79%, and the resultant images are given in Fig. 7. It can be confirmed that the optical transmittance differed in accordance with the different silylation conditions. Among these samples, sample C100 had the highest optical transmittance of approximately 79%. In addition, the tendency for the optical transmittance to decrease differed between the cases using the combination surface modification and TMCS alone. This was related to the change in the physical properties by the capillary pressure between the solvent and silanol groups during drying due to the lower hydrophobicity of the silica aerogel. In particular, the combination of TMCS and TMES resulted in less of a decrease in the optical transmittance of the silica aerogel, which can be observed by comparing sample C40 and sample C40E60. However, the cases with TMES had low optical transmittance values below 10%. This was similar to the results for the silica aerogels subjected to the silylation effect with a lower TMCS vol% of <40 vol%, as represented by samples C40 and C20. Therefore, it is evidence that TMES would not produce the surface modification when used alone as a silylation agent. As a result, the cases where TMES was used had the lowest optical transmission values of all the silica aerogels prepared under the different silylation conditions.
The variations in the bulk density and porosity under different silylating conditions are presented in Fig. 8. The porosity decreased and bulk density increased for the silica aerogel with decrease of TMCS vol% in surface modification step [34]. This was because the smaller amount of introduced silylating agent caused gel shrinkage during drying [35,36,37,38]. In contrast, the combination of TMES and TMCS produced constant porosity values greater than 89%. This was additional evidence that TMES could assist in the surface modification step with TMCS. Moreover, it was confirmed that the TMES alone could not produce a porosity greater than 85% or bulk density of <0.3 g/cm3. This was due to the influence of a higher capillary force during the drying step compared to other silylated silica aerogel samples, which had higher physical properties than samples E50, E100, and E200 [15].
The summarized information about the results for the specific surface area, pore volume, average pore diameter, bulk density, porosity, and contact angle for silica aerogels with various silylating agents is given in Table 2. This confirms that most of the results varied with the variation in the volume ratio of TMCS to TMES. In particular, Fig. 9 represents the variation of the pore volume with the vol% of TMCS and TMES/TMCS. The pore volume decreased with a decrease in the TMCS vol%. At 40 vol% of TMCS, a drastic decrease in the pore volume is observed from 2.83 cc/g to 1.57 cc/g. This suggests that silica aerogels with a lower degree of hydrophobicity caused a lower pore volume due to the capillary force between the solvent and silanol groups during the drying step [16]. However, the TMES and TMCS combination increased the pore volume of the final products above a volume ratio of TMES/TMCS = 20/80. This was due to a reduction of the drawbacks of the lower silylating reaction rate caused by the reaction of the silylating agent and greater silanol group surface modification [15]. Thus, a higher degree of hydrophobicity was caused, which prevented a decrease in pore volume [16]. Therefore, sample C60E40 had a high specific surface area (458 m2/g) and large pore volume (3.215 cc/g), as listed in Table 2.
Figure 10 shows the pore size distributions under the different surface modification conditions. It confirms that different surface modification conditions such as the vol% of TMCS and TMES/TMCS caused different physical properties for the sodium silicate-based silica aerogel, mainly as a result of homogeneous gel formation and a high degree of hydrophobicity during drying. It is observed that the average pore diameter dramatically decreased when TMCS was introduced at a vol% of <40 vol% for the TMCS alone, as shown in Fig. 10(a). This was related to the lower TMCS vol%, which caused more interaction between the solvent and silanol groups during drying [16]. Therefore, the average pore size decreased with a decrease in the degree of hydrophobicity. This can be confirmed by the information on the average pore diameter listed in Table 2. In addition, a decrease in the TMCS vol% resulted in a more homogeneous pore size when the TMCS vol% was above 60%. The silylation effect with the combination of TMES and TMCS is shown as Fig. 10(b). The most homogeneous pore size distribution is observed at a volume ratio of TMES/TMCS = 60/40. It can be supposed that the silylation proceeded at a stable reaction rate during the surface modification step with different amounts of reactants [15, 16, 39, 40]. However, it was confirmed that the pore size distribution became inhomogeneous when the volume ratio of TMES in the combination solution was lower than this condition for a TMCS 40 vol%. However, the samples that were not silylated had homogeneous pore size distributions, as shown in Fig. 10 (c). It was supposed that TMES showed a high aging effect similar to the aging for tetraethoxysilane (TEOS), which is one of the alkoxysilanes [33]. This phenomenon was also proven by the high specific surface areas for samples E50, E100, and E200.
Morphological images of the silica aerogels prepared under the different silylating conditions are shown in Fig. 11. It can be confirmed that the different silylating conditions can make influence on the microstructures of the prepared silica aerogel samples. It can be observed that the TMES and TMCS combination shows fine microstructure although TMES/TMCS volume ratio is high. In contrast, a lower volume ratio for TMCS (below 40 vol%) caused collapsed microstructure as a result of the lower silylation effect. In other words, a lower degree of hydrophobicity increased the interaction between the silanol groups and solvent, with less spring-back effect [41]. Hence, the network structure decreased with a decrease in the physical properties and hydrophobicity, as seen in samples C40 and C20 [41, 42]. However, it was confirmed that the network of the gel structure was coarse in the cases that used TMES alone for the surface modification step, as seen in samples E50, E100, and E200.
The mechanism of the TMES with TMCS is suggested in Fig. 12 to investigate the TMES reaction. It was important to understand the previous analyses and results in this research. The TMCS and silanol group reacted with each other. Then, hydrogen chloride was produced in both the liquid and vapor phases. Next, liquid HCl from the reaction between the TMCS and silanol group would react with TMES. A noncovalent electron pair at a chlorine atom in the HCl could interact with a silicon atom in TMES in a Si-SN2 reaction, which was similar to the case where hydroxyl anions attacked a silicon atom of the TMOS [43]. An oxygen atom in the TMES could also react with a hydrogen atom in the HCl. As a result, TMCS would be produced from the reaction of TMES and HCl. It should be the chain reactions that enhanced the surface modification with a uniform structure, lower density, and high degree of hydrophobicity [15]. This TMCS should cause a greater silylating effect, similar to the conventional TMCS in the mixed silylating solution [15, 16]. However, this effect had a restriction due to the low reactivity between the TMES and HCl. In addition, a considerable amount of HCl from this reaction would be transformed into vapor, which could not cause this phenomenon. As a result, using a low volume ratio of TMES/TMCS could not produce a high surface modification effect, and this combination reaction could not be an infinite cycle for reproducing TMCS. Thus, poor physical properties would be produced in the silica aerogel when using a combination of TMES and a lower TMCS vol%, such as in sample C20E80.
4 Conclusions
In summary, it is possible to decrease the TMCS vol% used in the silylation step by mixing it with TMES as a new silylating agent. The combination of TMES and TMCS provided a constant degree of hydrophobicity >128° compared with unmodified silica aerogels. Furthermore, it was confirmed that the physical properties of the silica aerogel were enhanced by using the combination of TMES and TMCS in the surface modification step. It could be supposed that a lower silylation rate caused a stable silylating reaction as a result of the lower volume ratio of TMCS. Specifically, a silica aerogel with a volume ratio of TMES/TMCS = 40/60 showed a high porosity (92.7%), high specific surface area (458 m2/g), high pore volume (3.215 cc/g), low density (0.139 g/cc), high degree of hydrophobicity (131.8°), and homogeneous pore size distribution compared to other sodium silicate-based silica aerogels. Therefore, physical properties could be improved for a sodium silicate-based silica aerogel when using a combination of TMCS and TMES in the surface modification step.
References
Parale VG, Mahadik DB, Kavale MS, Rao AV, Wagh PV, Gupta SC (2011) Potential application of silica aerogel granules for cleanup of accidental spillage of various organic liquids. Soft Nanosci Lett 1:97–104
Dorcheh AS, Abbasi MH (2008) Silica aerogel; synthesis, properties and characterization. J Mater Process Technol 199(1-3):10–26
Schmidt M, Schwertfeger F (1998) Applications for silica aerogel products. J Non-Cryst Solids 225:364–368
Parale VG, Jung H-N-R, Han W, Lee KY, Mahadik DB, Cho HH, Park HH (2017) Improvement in the high temperature thermal insulation performance of Y2O3 opacified silica aerogels. J Alloy Comp 727:871–878
Parale VG, Lee KY, Park HH (2017) Flexible and transparent silica aerogels: An overview. J Korean Ceram Soc 54(3):184–199
Schwertfeger F, Frank D, Schmidt M (1998) Hydrophobic waterglass based aerogels without solvent exchange or supercritical drying. J Non-Cryst Solids 225:24–29
Lee CJ, Kim GS, Hyun SH (2002) Synthesis of silica aerogels from waterglass via new modified ambient drying. J Mater Sci 37:2237–2241
Bhagat SD, Kim Y-H (2007) A cost-effective and fast synthesis of nanoporous SiO2 aerogel powders using water-glass via ambient pressure drying route. Solid State Sci 9:628–635
Prakash SS, Brinker CJ, Hurd AJ, Rao SM (1995) Silica aerogel films prepared at ambient pressure by using surface derivatization to induce reversible drying shrinkage. Nature 374(30):439–443
Shi F, Wang L, Liu J (2006) Synthesis and characterization of silica aerogels by a novel fast ambient pressure drying process. Mater Lett 60(29-30):3718–3722
Kim GS, Hyun SH (2003) Synthesis of window glazing coated with silica aerogel films via ambient drying. J Non-Cryst Solids 320(1-3):125–132
Mahadik SA, Pedraza F, Parale VG, Park HH (2016) Organically modified silica aerogel with different functional silylating agents and effect on their physico-chemical properties. J Non-Cryst Solids 453:164–171
Shewale PM, Rao AV (2008) Effect of different trimethyl silylating agents on the hydrophobic and physical properties of silica aerogels. Appl Surf Sci 254:6902–6907
Gurav JL, Rao AV, Rao AP, Nadargi DY, Bhagat SD (2009) Physical properties of sodium silicate based silica aerogels prepared by single step sol–gel process dried at ambient pressure. J Alloy Comp 476:397–402
Rao AP, Rao AV (2010) Modifying the surface energy and hydrophobicity of the low-density silica aerogels through the use of combinations of surface-modification agents. J Mater Sci 45(1):51–63
Rao AP, Rao AV (2009) Improvement in optical transmission of the ambient pressure dried hydrophobic nanostructured silica aerogels with mixed silylating agents. J Non-Cryst Solids 355:2260–2271
Rao AV, Kulkarni MM (2003) Synthesis and characterization of hydrophobic silica aerogels using trimethylethoxysilane as a co-precursor. J Sol-Gel Sci Technol 27:103–109
Rao AV, Kulesh RR, Amalnerkar DP, Seth T (2003) Synthesis and characterization of hydrophobic TMES/TEOS based silica aerogels. J Porous Mater 10:23–29
Hegde N, Hirashima H, Rao AV (2007) Two step sol-gel processing of TEOS based hydrophobic silica aerogels using trimethylethoxysilane as a co-precursor. J Porous Mater 14:165–171
Latthe SS, Imai H, Ganesan V, Rao AV (2009) Superhydrophobic silica films by sol–gel co-precursor method. Appl Surf Sci 256:217–222
Nah H-Y, Jung H-N-R, Lee K-Y, Ku YS, Park H-H (2017) Effect of acid catalyst kinds on the pore structural characteristics of water glass based silica aerogel. J Microelectron Packag Soc 24(3):13–18
Bangi UKH, Jung IK, Park CS, Baek S, Park HH (2013) Optically transparent silica aerogels based on sodium silicate by a two step sol–gel process and ambient pressure drying. Solid State Sci 18:50–57
Yu Y, Guo D, Fang J (2015) Synthesis of silica aerogel microspheres by a two-step acid–base sol–gel reaction with emulsification technique. J Porous Mater 22(3):621–628
Dervin S, Lang Y, Perova T, Hinder SH, Pillai SC (2017) Graphene oxide reinforced high surface area silica aerogels. J Non-Cryst Solids 465:31–38
Nah H-Y, Parale VG, Jung H-N-R, Lee K-Y, Lim C-H, Ku YS, Park H-H (2018) Role of oxalic acid in structural formation of sodium silicate-based silica aerogel by ambient pressure drying. J Sol-Gel Sci Technol 85:302–310
Hwang SW, Kim TY, Hyun SH (2008) Optimization of instantaneous solvent exchange/surface modification process for ambient synthesis of monolithic silica aerogels. J Colloid Interface Sci 322:224–230
Parale VG, Mahadik DB, Mahadik SA, Kavale MS, Rao AV, Wagh PB (2012) Wettability study of surface modified silica aerogels with different silylating agents. J Sol-Gel Sci Technol 63(3):573–579
Parale VG, Mahadik DB, Kavale MS, Mahadik SA, Rao AV, Mullens S (2013) Sol–gel preparation of PTMS modified hydrophobic and transparent silica coatings. J Porous Mater 20:733–739
Rao AP, Pajonk GM, Rao AV (2005) Effect of preparation conditions on the physical and hydrophobic properties of two step processed ambient pressure dried silica aerogels. J Mater Sci 40(13):3481–3489
Jal PK, Patel S, Mishra BK (2004) Chemical modification of silica surface by immobilization of functional groups for extractive concentration of metal ions. Talanta 62(5):1005–1028
Belyakova LA, Varvarin AM (1999) Surfaces properties of silica gels modified with hydrophobic groups. Colloids Surf A Physicochem Eng Asp 154(3):285–294
Bhagat SD, Kim YH, Suh KH, Ahn YS, Yeo JG, Han JH (2008) Superhydrophobic silica aerogel powders with simultaneous surface modification, solvent exchange and sodium ion removal from hydrogels. Microporous Mesoporous Mater 112(1-3):504–509
Zhou D, Seraphin S (1994) Production of silicon carbide whiskers from carbon nanoclusters. Chem Phys Lett 222:233–238
Wang LJ, Zhao SY, Yang M (2009) Structural characteristics and thermal conductivity of ambient pressure dried silica aerogels with one-step solvent exchange/surface modification. Mater Chem Phys 113:485–490
Mahadik DB, Rao AV, Rao AP, Wagh PB, Ingale SV, Gupta SC (2011) Effect of concentration of trimethylchlorosilane (TMCS) and hexamethyldisilazane (HMDZ) silylating agents on surface free energy of silica aerogels. J Colloid Interface Sci 356:298–302
Yokogawa H, Yokoyama M (1995) Hydrophobic silica aerogels. J Non-Cryst Solids 186:23–29
Sarawade PB, Kim JK, Hilonga A, Kim HT (2010) Production of low-density sodium silicate-based hydrophobic silica aerogel beads by a novel fast gelation process and ambient pressure drying process. Solid State Sci 12:911–918
Bhagat SD, Kim YH, Ahn YS, Yeo JG (2007) Rapid synthesis of water-glass based aerogels by in situ surface modification of the hydrogels. Appl Surf Sci 253:3231–3236
Wu G, Yu Y, Cheng X, Zhang Y (2011) Preparation and surface modification mechanism of silica aerogels via ambient pressure drying. Mater Chem Phys 129:308–314
Einarsrud M-A, Nilsen E (1998) Strengthening of water glass and colloidal sol based silica gels by aging in TEOS. J Non-Cryst Solids 226:122–128
Bangi UKH, Rao AV, Rao AP (2008) A new route for preparation of sodium-silicate-based hydrophobic silica aerogels via ambient-pressure drying. Sci Technol Adv Mater 9:1–10
Rao AP, Rao AV, Pajonk GM (2007) Hydrophobic and physical properties of the ambient pressure dried silica aerogels with sodium silicate precursor using various surface modification agents. Appl Surf Sci 253(14):6032–6040
Rao AV, Kulkarni MM (2002) Effect of glycerol additive on physical properties of hydrophobic silica aerogels. Mater Chem Phys 77(3):819–825
Acknowledgements
This work was supported by Nano-Convergence Foundation (www.nanotech2020.org) funded by the Ministry of Science and ICT (MSIT, Korea) & the Ministry of Trade, Industry and Energy (MOTIE, Korea) [Project Name: Commercialization development of super thermal insulation aerogel composite foam for cold insulation material]. This work was supported by the Center for Advanced Meta-Materials (CAMM) funded by the Ministry of Science, ICT and Future Planning as Global Frontier Project (CAMM-No. NRF-2014M3A6B3063716).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Nah, HY., Parale, V.G., Lee, KY. et al. Silylation of sodium silicate-based silica aerogel using trimethylethoxysilane as alternative surface modification agent. J Sol-Gel Sci Technol 87, 319–330 (2018). https://doi.org/10.1007/s10971-018-4729-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-018-4729-4