Abstract
Cefotaxime, a cephalosporin antibiotic, used to treat bacterial infections was investigated to label with 99mTc. Labeling was performed using sodium dithionite as a reducing agent at 100 °C for 10 min and radiochemical analysis involved ITLC and HPLC methods. The stability of labeled antibiotic was checked in the presence of human serum at 37 °C up to 24 h. The maximum radiolabeling yield was 92 ± 2%. Bacterial binding assay was performed with S. aureus and the in vivo distribution was studied in mice. Images showed minimal accumulation in non-target tissues, with an average target/non-target ratio of 2.89 ± 0.58.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A wide range of radiopharmaceuticals have been proposed to visualize infection and inflammation scintigraphically. Radiolabeled leukocytes and 67Ga-citrate are the most commonly applied radiopharmaceuticals [1, 2]. The radiolabeled leucocytes can be considered as “gold standard” that can visualize a majority of infectious and inflammatory lesions but it is labor-intensive and the in vitro labeling carries risks of handling potentially contaminated blood and also requires specialized equipment, taking approximately three hours [3–5].
The use of radiolabeled antibiotics is fast emerging as a promising diagnostic test for the detection of infective lesions. Antibiotics localize in the infectious focus, where they are frequently taken up and metabolized by microorganisms.
The majority of the various antibiotics studied so far are those of the quinolones group. Recently, a new radiopharmaceutical, 99mTc-ciprofloxacin (Infecton), has been developed which its microbiological activity is mediated by inactivation of bacterial DNA gyrase [6–8].
Second and third generation cephalosporines, including cefuroxime and ceftizoxime have been labeled with 99mTc previously [9, 10]. Ceftizoxime has a wide spectrum of activity to the beta lactamases, binds onto the bacterial wall, inhibits the synthesis of peptidoglycan and therefore inhibits the synthesis of bacterial wall, which drives to bacterial death. It has a half life of 1.7 h being almost totally eliminated within 24 h. Ceftizoxime and cefotaxime structural formulas differ only by an acetoxy group at position 3 on the beta-lactam ring.
Like other third-generation cephalosporines, cefotaxime has a broad spectrum activity against gram-positive and gram-negative bacteria. Cefotaxime is cleared more rapidly than ceftizoxime from the body and mainly metabolized in vivo to desacetyl cefotaxime, which also has intrinsic antibacterial activity; so acts synergistically with cefotaxime toward several pathogens. Total body clearance is significantly faster for cefotaxime [11].
In this paper, we describe the optimum condition for radiolabeling of cefotaxime with the most widely used imaging radionuclide, 99mTc. The radiolabeling was performed by the sodium dithionite as the reducing agent. In addition, radiochemical purity, stability in human serum albumin, bacterial binding assay and in vivo biodistribution in infected mice were investigated.
Experimental
Reagents and methods
All chemicals were purchased from Merck or Fluka. The chemicals and solvents were of the highest purity and analytical grade and used without further purification. 99mTc-pertechnetate was supplied by AEOI, as 99Mo/99mTc generator. Radioactivity measurements were carried out using Na(Tl) scintillation counter (ORTEC Model 4001 M Minibin & Power Supply).
Radiolabeling procedure
Cefotaxime (2.5 mg) was dissolved in 200 μL distilled water. To determine the optimal amount of reducing agent, sodium dithionate in different concentrations (4.25–8.25 mg/200 μL NaHCO3 0.5%) was added. Then 370–740 MBq freshly eluted pertechnetate solution (in maximum 1 ml of saline) was added into the vials in different pH ranges between 7.5 and 10.5. Finally the reaction mixture was heated in a boiling water bath at different times from 5 to 60 min.
Radiochemical and HPLC Analysis of 99mTc-cefotaxime
The labeling yield and radiochemical purity were determined by thin layer chromatography. The reaction product was spotted on silica gel ITLC-SG strips (Sigma Chemical Company, USA) (10 × 1.5 cm2 sheets) and developed in acetone and ACD (citrate–dextrose buffer solution) as the mobile phase. After developing, they were cut into 1 cm pieces and counted. Radiochemical purity of the complex has been checked by ITLC and HPLC. Analytical reverse phase high performance liquid chromatography (RP-HPLC) was performed on a JASCO 880-PU intelligent pump HPLC system equipped with a multi-wave length detector and a flow through RAY test-Gabi gamma detector. CC 250/4.6 Nucleosil 120-5 C18 column from Teknokroma was used for analytical HPLC. A mixed solvent containing 10% ethanol on 0.2 M phosphate buffer, pH 7.2 and flow rate 0.5 mL/min was used for analytical HPLC.
Stability of 99mTc-cefotaxime in human serum
The affinity of the labeled antibiotic to human serum proteins was examined by mixing 1 mL of labeled cefotaxime with activity between 5 and 20 mCi to 1 mL of human serum in a vial. The sample was incubated in 37 °C for 24 h and 100 μL of reaction mixture was placed on a Sephadex G25-column to evaluate the complex affinity to plasma protein. After washing the column with PBS or normal saline, activity bound to serum protein and labeled cefotaxime was measured with a well-type gamma counter.
To test the serum stability of 99mTc-cefotaxime complex, we added 1 mL of freshly prepared human serum to 100 μL of labeled antibiotic. The mixture was incubated in 37 °C for 24 h. Then the serum protein was denatured by mixing 100 μL of the solution with 100 μL absolute ethanol. After that, the mixture was centrifuged at 2000×g in 4 °C for 10 min. Radiochemical stability was determined by taking samples of 10 μL of supernatant at different times up to 24 h of incubation that were analyzed by ITLC.
In vitro binding of 99mTc-cefotaxime to bacteria
Binding of 99mTc-cefotaxime to bacteria was assessed by the method described previously [12–14]. Briefly, 0.1 mL 99mTc-cefotaxime (37 MBq) was transferred to a test tube. Then, 0.9 mL of 50% (v/v) 0.01 M acetic acid in phosphate buffer (Na–PB, pH 7.5) containing approximately 1 × 108 colony forming units (CFU) per mL viable S. aureus were added. The mixture was incubated for 1 h at 4 °C and thereafter the vials were centrifuged in a pre-cooled centrifuge for 5 min at 2000×g at 4 °C. The supernatant was removed, and the radioactivity in the bacterial pellet was gently re-suspended in 1 mL of Na–PB and re-centrifuged as above. The supernatant was removed and the radioactivity in the bacterial pellet was determined by gamma counter. The radioactivity related to bacteria was expressed in percent of the added 99mTc activity bounded to viable bacteria in regard to total 99mTc.
Partition coefficient (log P value)
About 100 μL of the labeled compound was mixed with 0.9 mL water and 1 mL of n-octanol on a vortex mixer for about 1 min. The two phases were allowed to separate. Equal aliquots of the organic and aqueous layers were withdrawn and measured for radioactivity to determine the partition coefficient (log P = activity in octanol/activity in water). The log P data are reported as an average plus the standard variation based on the results from the three independent measurements.
Animal biodistribution
Male Swiss mice, weighing 25–30 g were infected by injecting 0.1 mL of saline containing 1 × 108 CFU bacteria into right thigh muscle. After 24 h, they were injected under ether anesthesia with 74 MBq of 99mTc-cefotaxime in saline into the tail vein. At 1 h after injection, accumulation of the tracer in infected area was assessed by planar scintigraphy under ether anesthesia. For ex vivo counting, the mice were sacrificed after 1 h, 2 h and 24 h and the organs of interest were collected, weighed and radioactivity was measured in a γ-counter.
Statistical analysis
The calculations of means and standard deviations were made on Microsoft Excel. Student’s t-test was used to determine statistical significance. Differences at the 95% confidence level (p < 0.05) were considered significant.
Results and discussions
Radiolabeling and radiochemical purity
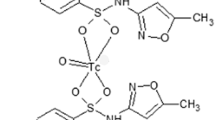
The chemical structure of cefotaxime is presented in Fig. 1. The various complexes of 99mTc may be formed by interactions between electron donor atoms and reduced technetium. In order to form bonds with technetium, the structure must contain electron donors such as oxygen, nitrogen and sulfur. Although the exact complex structure is not known, results showed that cefotaxime coordinated with 99mTc because of its electron donor atoms in its structure [15–17].
Sodium dithionate was chosen for the reduction of 99mTc from +7 to lower valence state, which facilitates its chelating by compounds of diagnostic impotence. The effects of sodium dithionite amount as a reducing agent are summarized in Fig. 2. The data showed that the radiochemical yield was dependent on the amount of sodium dithionite present in the reaction mixture. The effect of reducing agent was studied between 4.25 and 8.25 mg of sodium dithionite. The highest labeling efficiency was obtained by using 7.25 mg of sodium dithionite. On the other hand, labeling efficiency decreased at 8.25 mg sodium dithionite.
The effect of pH was shown in Fig. 3. The pH of the reaction medium was found to play an important role in the labeling process. The effect of pH on the radiolabeling yield was examined for pH 7.5–10. The highest labeling yield was obtained at pH = 8.5–9.
Heating time in 100 °C is an important factor as is clear in Fig. 4, heating up to 10 min produced 99mTc-cefotaxime with yield of 92% and by increasing heating time up to 60 min, the yield decreased to 58% due to heat decomposition of the produced complex. A short duration of heating may produce a small quantity of 99mTc-labeled cefotaxime decomposition products.
In radiochemical analysis study by ITLC-SG chromatography using acetone as the mobile phase, reduced 99mTc and 99mTc-cefotaxime remained at the point of spotting, while free 99mTcO4 − moved towards the solvent front. In using ACD solution as another mobile phase, 99mTc-cefotaxime and 99mTcO4 − moved to the front, where as reduced 99mTc remained at the point of spotting. The maximum radiolabeling yield of cefotaxime was 92 ± 2% using HPLC and also ITLC. The HPLC elution times were 588 ± 36 s for pertechnetate and 918 ± 12 s for 99mTc-cefotaxime. The complex is stable at least for 12 h. Fig. 5.
Serum study and bacterial binding assay
The affinity of the labeled antibiotic to human serum proteins after 24 h was about 25 ± 5% and labeled antibiotic was stable in human serum with radiochemical purity of about 85% after 24 h.
In vitro testing of 99mTc-cefotaxime to S. aureus showed 35% of radioactivity bound to bacteria. It should be mentioned that in the in vitro competition assay we observed the inhibition of binding of 99mTc-cefotaxime to S. aureus by unlabeled cefotaxime with maximal inhibition of approximately 80% when 100-fold excess of unlabeled antibiotic was used as competitor.
In vivo experiments
Based on data presented in Table 1, we conducted biodistribution analysis of 99mTc-cefotaxime complex in normal and abscess-bearing mice at different time intervals post injection. For non-targeted organs (liver, spleen, heart and lung) the uptake of the tracer was similar, to a large extent, to the normal mice. Accumulation of the complex in the infected tight muscles as indicated by T/NT ratio was 2.89 ± 0.58 one hour post injection. In most organs, the activity declined at 4-hour post injection. The kidney shows the maximal activity 1 h post injection and it shows the main route of excretion of the radiotracer. Typical scintigram of mice with infectious thigh muscle within 1 h after injection of radiotracer is shown in Fig. 6. The scan demonstrated, high uptakes of activity in infection site with no accumulation in non-infected muscle. Upon autopsy the bacterial infection had the same appearance as demonstrated on the Scintigraphic image.
The log P value of the complex was found to be −1.98 ± 0.21 reflecting its low lipophilisity. The low n-octanol/water partition coefficient is also associated with the lower and shorter retention in background tissues and blood [18]. Therefore, the nature of radiolabeled antibiotic might result in lower uptake in the normal tissues and faster clearance so as to obtain favorable target to non-target (tissue) ratios and these results are in accordance with the biodistribution results.
Infecton, a wide spectrum fluoroquinolone, has a biological half-life of 3.5–4.5 h. Approximately 60% of the injected activity has been recovered in urine by 24 h post injection [19, 20]. One possible advantage of 99mTc-cefotaxime over 99mTc-ciprofloxacine is lesser hepato-biliary excretion with the new radiotracer. Lower intestinal excretion would be advantageous in identifying infective intestinal lesions. On the other hand, cefotaxime has a half-life of 1.43 h, being almost totally eliminated within 24 h after administration. As opposed to Infecton, cefotaxime is more rapidly depurated from the organism, thus diminishing the circulating pool and favoring the specific capture by the infectious site.
Invitro experiments of cefuroxime axetile have been reported and it has depicted a high radiolabeling yield (92 ± 1% in the optimal condition of: pH = 3, reaction time of 20 min in room temperature and using SnCl2 as reducing agent) but its in vivo results have not reported yet [9].
Ceftizoxime, third generation cephalosporin, has a half life of 1.7 h, being almost totally eliminated within 24 h after administration. In comparison with ceftizoxime, the T/NT ratio of 99mTc-cefotaxime was 2.98 ± 0.58 which is higher than 99mTc-ceftizoxime (1.97 ± 0.31) [21–25].
Cefotaxime with the biological half life of 1.43 h (MW = 445.47) has a lower degree of interaction with plasma protein (25–30%). New radiotracer is eliminated mainly from the kidney and the amount of log P value proves this matter, which is the other advantage of this antibiotic over ceftizoxime. 99mTc-cefotaxime has a higher uptake in the septic abscess than the non-infected muscle (p < 0.05) and its uptake in the septic muscle remains stable within 1 h post injection. Finally, considering the criteria for obtaining an ideal radiopharmaceutical, 99mTc-cefotaxime fits the criteria better and may be applied for infection imaging.
Conclusion
In this study, labeling of Cefotaxime with 99mTc was performed by using sodium dithionite as a reducing agent. The labeling of 99mTc-cefotaxime was optimal in the following reaction mixture: cefotaxime (2.5 mg in 200 μL distilled water), 7.25 mg of Na2S2O4 in 200 μL NaHCO3 0.5%, pH: 8.5–9, reaction time: 10 min and reaction temperature: 100 °C. The resulting complex is quite stable and labeling of 92 ± 2% is maintained for up to 12 h. This antibiotic showed an improvement in excretion pathway from the liver to the kidney followed by an accumulation of radioactivity in infected areas. These promising characteristics make our new radiotracer a very suitable candidate for diagnostic of infectious foci in nuclear medicine.
References
Knight LC (2003) Q J Nucl Med 47:279
Weiner RE, Thakur ML (2001) Semin Nucl Med 31:296
Cardoso VN, Plaza P, Roca M, Armero F, Martin Comin J (2002) Nucl Med Commun 23:715
Wareham D, Michael J, Das S (2005) Braz Arch Biol Technol 48:145
Martin-Comin J, Cardoso VN, Plaza P (2002) Braz Arch Biol Technol 45:39
Hall AV (1998) J Clin Pathol 51:215
Vinjamuri SH, Hall AV, Solanki KK (1996) Lancet 347:233
Britton KE, Vinjamuri S, Hall AV, Solanki K, Siraj QH, Bomanji J, Das S (1997) Eur J Nucl Med 24:553
Lambrecht FY, Durkan K, Unak P (2008) J Radioanal Nucl Chem 275:161
Diniz SOF, Siqueria CF, Nelson DL, Martin-Comin J, Cardoso VN (2005) Braz Arch Biol Technol 48:89
Tavares W (2002) Manual de antibioticos e quimioterapicos antiinfecciosos. Atheneu, Sao Paulo
Gandomkar M, Najafi R, Shafiei M, Mazidi M, Goudarzi M, Mirfallah SH (2009) Nucl Med Biol 36:199
Gandomkar M, Najafi R, Mazidi M, Goudarzi M, Mirfallah SH, Iran (2008) J Nucl Med 16(1):25
Gandomkar M, Najafi R, Mazidi M, Mirfallah SH, Goudarzi M (2009) J Nucl Med Biol 7(2):97
Barreto VG, Iglesias F, Roca M, Tubau F, Martin Comin J (2000) Rev Esp Med Nucl 19:479
Roohi S, Mushtaq A, Jehangir M, Malik SA (2006) J Radioanal Nucl Chem 267:561
Vallee F, Lebel M (1991) J Antimicrob Agents Chemother 35:2057
McAfee JG, Gagne G, Subramanian G, Schneider RF (1991) J Nucl Med 32:2126
Li ZJ, Chu TW, Liu XQ, Wang XY (2005) Nucl Med Biol 32:225
Sampson CB (1996) Nucl Med Commun 17:648
Sonmezoglu K, Sonmezoglu M, Halak M, Akgun I, Turkmen C, Onsel C, Kanmaz B, Solanki K, Britton KE (2001) J Nucl Med 42:567
Yapar Z, Kibar M, Yapar AF, Togrul E, Kayaselcuk U, Sarpel Y, Eu (2001) J Nucl Med 28:822
Larikka MJ, Ahonen AK, Niemela O, Puronto O, Junila JA, Hamalainen MM, Britton KE, Syrajala HP (2002) Nucl Med Commun 23:655
Larikka MJ, Ahonen AK, Niemela O, Puronto O, Junila JA, Hamalainen MM, Britton KE, Syrajala HP (2002) Nucl Med Commun 23:167
Welling MM, Paulusma-Annema A, Balter HS, Pauwels EK, Nibbering PH (2000) Eur J Nucl Med 27:292
Acknowledgments
The authors wish to thank Mr. Mirfallah and Mr. Mazidi of the radioisotope department (AEOI) for providing sodium pertechnetate and assistance in quality control tests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirshojaei, S.F., Gandomkar, M., Najafi, R. et al. Radio labeling, quality control and biodistribution of 99mTc-cefotaxime as an infection imaging agent. J Radioanal Nucl Chem 287, 21–25 (2011). https://doi.org/10.1007/s10967-010-0826-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-010-0826-8