Abstract
Gel structure of ethane-bridged trialkoxysilsesquioxane has been investigated extensively. In contrast, only a few studies have focused on the characterization of soluble ethane-bridged silsesquioxane polymer (sEBSP) because of difficult preparation and isolation processes. The present study aims to characterize the sEBSP and investigate its mixing effect in an organic polymer. The sEBSP was prepared by the hydrolysis–condensation of bis(trimethoxysilyl)ethane under a nitrogen flow and characterized by nuclear magnetic resonance spectroscopy (NMR), Fourier-transform infrared (FTIR) spectroscopy, and gel permeation chromatography (GPC). Based on the NMR and FTIR results, sEBSP was characterized as a randomly structured polymer with hybridized linear, cyclic, branched, and bicyclic units. It was mixed with poly(methyl methacrylate) (PMMA) and poly(bisphenol A-co-epichlorohydrin) (PBE) to form PMMA–sEBSP and PBE–sEBSP, respectively, which were then characterized by FTIR spectroscopy. The results confirmed the formation of hydrogen bonds between sEBSP and the organic polymer. The thermal stabilities of PMMA–sEBSP were better than those of the pure polymers. PBE–sEBSP exhibited a lower 5% weight loss temperature (Td5) because of the transalkoxylation between PBE and sEBSP. The miscibility of organic polymers and sEBSP was confirmed by differential scanning calorimetry (DSC). Based on their glass transition temperature, PMMA–sEBSP and PBE–sEBSP were classified as a hybrid and nanocomposite, respectively. Therefore, sEBSP was miscible to PMMA and PBE and affected to thermal properties.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Organo-bridged alkoxysilane, [(RO)3Si]2R’, is an important silsesquioxane precursor because the functionality of the resulting polymers can be easily controlled by changing the physical and chemical properties of the bridging groups. These precursors are used as mesoporous materials [1], separation membranes [2, 3], flexible aerogels [4], adsorbents [5], photo-induced materials [6], and so on. These materials are synthesized using colloids and particles, which are prepared by the sol–gel reaction of organo-bridged alkoxysilanes. Their gelation time and structure can be changed by changing the structure of bridging groups [7, 8]. Low-molecular-weight compounds (dimers and trimers) have also been synthesized and isolated from hydrolysis–condensation reaction of organo-bridged alkoxysilane [9, 10]. The compounds, sols (colloids and hydrolyzed alkoxysilanes), and gels (particles and insoluble polymers) obtained using organo-bridged alkoxysilane have been well investigated. However, their soluble polymers are not understood completely because of their difficult preparation and isolation.
Yamamoto et al. synthesized soluble ethane-bridged silsesquioxane polymer (sEBSP) by the hydrolysis–condensation reaction of bis(triethoxysilyl)ethane (BTESE) under nitrogen flow [11]. Herein, we focus on bis(trimethoxysilyl)ethane (BTMSE) because the molecular weight of its polymers and the colloidal size can be controlled by controlling the hydrolysis rate of alkoxy groups [12,13,14]. However, the structural units of sEBSP have not been identified yet. Although the use of organic–inorganic composites containing silsesquioxanes as sols (colloids, particles) [15,16,17], polyhedral oligomeric silsesquioxanes (POSS) [18,19,20], and cyclic silsesquioxanes [21] has been investigated, not much information is available on organic–inorganic composites fabricated using soluble silsesquioxane polymers.
In this work, we investigate the preparation and characterization of sEBSP. We also prepared an organic–inorganic composite comprising sEBSP and poly(methyl methacrylate) (PMMA) and poly(bisphenol A-co-epichlorohydrin) (PBE). The sEBSP was characterized by nuclear magnetic resonance (NMR), Fourier-transform infrared (FTIR) spectroscopy, and gel permeation chromatography (GPC), while the organic polymer–sEBSP composite was characterized by FTIR spectroscopy and thermal investigation.
Experimental
Measurements
NMR spectra were recorded using a JEOL Resonance JNM-ECP 300 spectrometer (JEOL, Tokyo, Japan; 1H at 300.53 MHz and 29Si{1H} at 59.70 MHz). The chemical shifts were reported in ppm using chloroform-d (CDCl3; for 1H: 7.26 ppm in residual chloroform) and tetramethylsilane as internal standards (for 29Si{1H}: 0.00 ppm). To obtain the 29Si{1H} NMR spectra, Cr(acac)3 was added to the samples as a paramagnetic relaxation agent. The broadening factor (BF) was 20 Hz. GPC was performed using an HPLC system (LC-20AD, Shimadzu, Kyoto, Japan) attached to a PLgel 5 µm Mixed-D column. Tetrahydrofuran (THF) was used as the eluent (1 mL min−1), and RID-20A was used as the detector at 40 °C. The molecular weight was calculated using polystyrene standards. The FTIR spectra of sEBSP were recorded on an FTIR spectrophotometer (FT/IR-6100, JASCO, Tokyo, Japan) with a neat sample sandwiched between KBr aperture plates. The FTIR spectra of the composites were recorded using the same spectrophotometer with attenuated total reflectance (JASCO ATR PRO 0450-S, ZnSe prism). Thermogravimetric analysis (TGA) was performed using a TG–DTA analyzer (2000SE, Netzsch Japan, Kanagawa, Japan). The samples were heated to 1000 °C at a rate of 10 °C min−1 under air flow. Differential scanning calorimetry (DSC) was performed using DSC 3500 Sirius (Netzsch Japan). Both heating and cooling were performed at a rate of 5 °C min−1 under nitrogen flow. This process was performed three times.
Materials
Methanol and THF were purified by standard methods and stored over activated molecular sieves. BTMSE was purchased from Tokyo Chemical Industry Ltd. (Tokyo, Japan). A 6 mol L−1 HCl aq solution was purchased from FUJIFILM Wako Pure Chemical Corp. (Osaka, Japan). PMMA (Mw = 997,000 g mol−1) and PBE (Mw = 40,000 g mol−1) were purchased from Sigma-Aldrich (Tokyo, Japan). Water was purified using the PURELAB Flex 5 water purification system (ELGA, Marlow, UK).
Hydrolysis–condensation of BTMSE
BTMSE (1.35 g, 5.0 mmol) and methanol (1.61 g) were placed in a 100-mL four-necked flask equipped with nitrogen inlet and outlet tubes and a mechanical stirrer. The mixture was stirred at 150 rpm and cooled in an ice bath for 10 min. Then, 6 mol L−1 HCl aq solution (0.96 g, HCl/BTMSE = 0.05) and H2O was added. The resulting mixture was stirred in the ice bath for 10 min and then at 22 ± 3 °C for 10 min, followed by heating at 70 °C for 3 h. The hydrolysis–condensation reaction was carried out under a nitrogen flow of 360 mL min−1. The sEBSP was obtained as a colorless and viscous liquid.
Preparation of free-standing films
First, 0.12 g of organic polymer (PMMA or PBE) in THF (5 mL) was added to the sEBSP (13 mg) and stirred for 24 h at 22 ± 3 °C. The precursor was poured into a 50-mmϕ Teflon Petri dish followed by drying at 22 ± 3 °C for 1 day and curing at 120 °C for 1 day.
Results and discussion
Preparation and characterization of sEBSP
The sEBSP was prepared by the hydrolysis–condensation of BTMSE catalyzed by hydrochloric acid. The hydrolysis–condensation reaction was carried out under a nitrogen gas flow to remove the catalyst [13, 22, 23] and to accelerate the condensation rate by evapolating the solvent [22, 24]. The results of the hydrolysis–condensation of BTMSE are shown in Table 1. The yield of sEBSP and the content of methoxy group in sEBSP (determined by 1H NMR spectra) decreased when the amount of water is increased because the hydrolysis ratio depends on the amount of water used. Compared with BTESE, BTMSE was easily hydrolyzed and formed a gel (gelation point: H2O/BTMSE > 3.8, H2O/BTESE > 5.0 [11]). The sEBSP showed high solubility in acetone, THF, chloroform, and toluene and only a slight solubility in methanol, ethanol, and 2-propanol, similar to the BTESE polymer [11]. GPC analyses of sEBSP are summarized in Table 1 and Fig. 1. The number average molecular weight (Mn), weight average molecular weight (Mw), and polydispersity index (PDI) of sEBSP increased with the increasing ratio of H2O/BTMSE because the polycondensation rate was dependent on the hydrolysis ratio in our process [13, 24]. The PDI value was very large.
The 29Si{1H} NMR spectra of BTMSE and sEBSP are shown in Fig. 2. The 29Si{1H} spectrum of BTMSE showed a signal assigned to the T0 unit at − 42.2 ppm in CDCl3. The sEBSP contains no unreacted BTMSE, hydrolyzed monomers, [CH2Si(OMe)3−x(OH)x]2 (x = 0–3), or an intramolecularly cyclized monomer ([CH2Si(OMe)2]2O) because no signals were observed in the region from − 37 to − 42.2 ppm. Moreover, no signals of the T3 unit ([CH2Si(OSi)3]) were observed from − 64.5 to − 66.6 ppm [25,26,27]. For bis(triethoxysilyl)ethane, bicyclic T1–OEt and T2–OEt appeared at − 51 and − 53.3 ppm, respectively [28]. Because the chemical shift of Si–OMe is 2–4 ppm smaller than that of Si–OEt [26, 29], we assigned the signals at − 47.6 and − 51.4 ppm to bicyclic T1–OMe and T2–OMe, respectively. Loy et al. reported that a xerogel with a bicyclic structure showed a signal at − 60.5 ppm [8, 10]. The sEBSP exhibited a signal at − 60.6 ppm, which corresponds to the bicyclic T3 unit. The cyclic T2 unit appeared downfield compared with the branched T2 unit [30, 31]. Therefore, the signals at − 56.7 and − 54.4 ppm can be assigned to branched T2–OMe and cyclic T2–OMe, respectively. The signal at − 51.8 ppm is due to linear T1–OMe, and that at − 50.4 ppm might be due to cyclic T1–OMe. These assignments are summarized in Table 2. The end-capping-sEBSP (cap-sEBSP), prepared by the reaction of sEBSP (H2O/BTMSE = 3.4) with Me3SiCl, was measured to identify the silanol group (Tn–OH). The 29Si{1H} NMR spectrum of cap-sEBSP showed a new signal at 10.1 ppm. However, this signal intensity was very weak, implying that the amount of silanol is low in sEBSP. The structural units of sEBSP are illustrated in Scheme 1. The sEBSP has two types of cyclic units: I (number of Si ≥ 4) and II (number of Si = 3). However, the exact number of the units could not be determined.
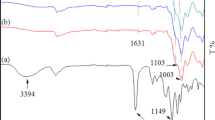
The FTIR spectra of BTMSE and sEBSP were measured for the sample sandwiched between KBr aperture plates (Fig. 3). Their vibration wavenumbers are assigned based on the vibrational assignments of silsesquioxanes [10, 32,33,34,35,36,37,38,39,40,41] and are summarized in Table 3. The sEBSP showed absorption bands formed by stretching vibration of siloxane bonding (Si–O–Si) at 1000–1100 cm−1. These bands overlapped with three absorption bands. In general, branched, cyclic, and linear Si–O–Si appear at 1080, 1050, and 1020 cm−1, respectively [36,37,38]. Moreover, bicyclic Si–O–Si exhibits a band at 1007 cm−1 [10]. The FTIR results show that the sEBSP was composed of a branched siloxane unit (at 1087 cm−1), a cyclic siloxane unit (at 1049 cm−1), and a linear/bicyclic siloxane unit (at 1011 cm−1). No difference was observed in the molar ratio of H2O/BTMSE for FTIR, which implies that the skeleton structure will be similar. The sEBSP has more methoxy groups than the ethane-bridged silsesquioxane colloid and gel [32, 34, 38]; hence, sEBSP can be isolated and obtained as a soluble polymer. The sEBSP showed a peak at 3410 cm−1 for silanol (Si–OH) and a shoulder peak at 913 cm−1. In contrast, the cap-sEBSP showed a decreasing intensity for the silanol band, which implies that it has no silanols.
Hence, the results suggest that (i) the sEBSP contained methoxy and silanol groups and (ii) its structure contained hybridized linear, branched, cyclic, and bicyclic siloxane units. Scheme 2 illustrates the hydrolytic condensation reaction of BTMSE in the initial stage. At least six types of structural units (linear, branched, cyclic I (SiOC and SiO rings), cyclic II, and bicyclic-type) were formed by cyclization, bicyclization, and crosslinking condensation from the first-stage unit. Therefore, the PDI of the sEBSP increase because of the complicated condensation reaction.
FTIR studies of organic polymer–sEBSP
Organic polymer–sEBSP was prepared by a solution–melt blending method. The free-standing films were characterized by FTIR spectra, as shown in Fig. 4. The spectrum of pure PMMA exhibited absorption bands due to νC = O at 1722 cm−1, δCH at 1386 cm−1, νC–O–C at 1142 cm−1, and skeletal mode of C–C at 749 cm−1 [42, 43]. The absorption bands assigned to the PMMA matrix of PMMA–cap-sEBSP correspond to those of pristine PMMA. In comparison, the FTIR of PMMA–sEBSP showed that the vibration wavenumbers of C = O (at 1728 cm−1) and C–O–C (at 1148 cm−1) shifted 6 cm−1 higher than that of pristine PMMA. The vibration wavenumbers of C–H and skeletal mode had the same values. These results show that the sEBSP will affect the carbonyl group of PMMA. In general, the vibration wavenumber of the hydrogen bond between the carbonyl group and silanol (νC = O∙∙∙HO–Si) is lower than that of the carbonyl group that forms a non-hydrogen bond [21, 44]. However, the carbonyl group of PMMA–sEBSP showed a higher wavenumber, which implies the absence of C = O∙∙∙HO–Si. To investigate the hydrogen bonding mode of carboxylic acid, the vibration wavenumber of the carboxyl group that forms hydrogen bonds with the hydroxy group (CH3O–H∙∙∙O(H)–C = O) was calculated and was observed to upshift (to 11 cm−1), compared with the carboxyl group that forms nonhydrogen bonds [45]. Based on these results, we suggest that hydrogen bonds (SiO–H∙∙∙O(Me)–C = O) are formed between PMMA and sEBSP, as shown in Fig. 5.
The FTIR spectrum of pure PBE exhibited absorption bands due to νC = C–H at 1506 cm−1, νC–O–H at 1413 cm−1, δCH2 at 1361 cm−1, and νC–C–O–C at 1228 cm−1 [46, 47]. The absorption bands due to the PBE matrix for PBE–cap-sEBSP corresponded to those of pristine PBE. In contrast, νC–C–O–C of PBE–sEBSP shifted to a higher wavenumber (1241 cm−1) than that of pristine PBE. Moreover, νC–O–H of PBE–sEBSP was observed at the same wavenumber as that of pristine PBE, which indicates the absence of any hydrogen bond with the hydroxy group. These results show that the PBE–sEBSP would form a hydrogen bond between silanol and the oxygen atom of the phenoxy ether moiety in PBE, as illustrated in Fig. 5. Transalkoxylation between PBE and sEBSP can be confirmed by the vibration stretching of Si–O–alkyl: νSi–O(–Me) at 699 cm−1 and νSi–O(–iPr) at 686 cm−1 [41]. Although νSi–O(–Me) was observed at 697 cm−1, νSi–O(–iPr) was not observed; therefore, no transalkoxylation between PBE and sEBSP occurred.
Thermal analyses of organic polymer–sEBSP
The TGA of organic polymer–sEBSP under air is shown in Fig. 6. The temperatures for 5% weight loss (Td5) are summarized in Table 4. Degradation of pure PMMA occurred through two steps: (i) scission of head-to-head linkages [48] or degradation by radical transfer to the vinyl chain end [49] at 150–200 °C, and (ii) cleavage of the main chain and oxidative degradation at 270 °C [50]. The PMMA–sEBSP was more thermally stable than pristine PMMA, probably because of the radical scavenging effect caused by groups such as metal oxides [51] or the restriction of the main chain [52]. This behavior was similar to that of PMMA–POSS [18], PMMA–organosilsesquioxane oligomer [21], and PMMA–titanium phosphonate cluster composites [53]. The Td5 value of the PBE–sEBSP was lower than that of pristine PBE. This tendency was similar to that of PBE–titanium phosphonate cluster hybrids [46, 54]. It is possible to eliminate methanol while forming Si–O–iCH(CH2)2 by transalkoxylation of sEBSP and PBE with heating.
The miscibility of organic polymers and sEBSP was confirmed by DSC analyses. If these composites are immiscible, the glass transition temperature (Tg) values will be similar to those of pristine organic polymers [55]. Moreover, the Tg values of organic–inorganic hybrids/composites depend on the type of the formation system. The following types of formation systems are known: (i) composites, in which organic and inorganic domains are combined over long lengths (at least at micrometer level); (ii) nanocomposites, in which composites are combined over the nanometer order; and (iii) hybrids, characterized by weak interactions and strong bond formation (from atomic level to 50 nm) [56]. For PMMA–inorganic domains, the Tg values decrease in the order of nanocomposites < composites ≈ pristine PMMA < hybrids [57,58,59]. PBE–inorganic domains showed a similar tendency [60, 61]. The DSC analyses of PMMA–sEBSP and PBE–sEBSP are shown in Fig. 7, and the Tg values are summarized in Table 4. The Tg values of pure PMMA and PBE were 115 and 98 °C, respectively. The Tg values of PMMA–cap-sEBSP and PMMA–sEBSP were slightly higher than those of pristine PMMA. In contrast, the Tg values of PBE–cap-sEBSP and PBE–sEBSP were lower than those of pristine PBE. Therefore, sEBSP was miscible in organic polymers, and PMMA–sEBSP and PBE–sEBSP were classified as a hybrid and nanocomposite, respectively. Hence, the sEBSP and cap-sEBSP exerted similar thermally stable effects on organic polymer matrices. No difference was observed in the thermal effect between sEBSP and cap-sEBSP because of the low content of silanol group in sEBSP.
Conclusions
The sEBSP was prepared by a hydrolysis–condensation reaction of BTMSE and showed solubility in organic solvents (acetone, THF, chloroform, and toluene). Its methoxy group content decreased, while its Mw and PDI decreased when the amount of water was increased. When H2O/BTMSE was 3.8, the ethane-bridged silsesquioxane was obtained as a gel. The structure of sEBSP was determined by NMR and FTIR. The sEBSP contained hydroxy and methoxy groups on the silicon atoms and was composed of linear, branched, cyclic, and bicyclic units.
The organic polymer–sEBSP composites were prepared by solution–melt blending of the organic polymer with sEBSP. The FTIR results showed that the PMMA–sEBSP can be formed by hydrogen bonding with the methoxy group in the PMMA matrix (SiO–H∙∙∙O(Me)–C = O). The PBE–sEBSP was formed by hydrogen bonding with the phenoxy moiety of PBE (SiO–H∙∙∙O(R)–C6H4). In contrast, the organic polymer–cap-sEBSP (with no silanol) was observed without any hydrogen bonding interaction. The Td5 values of PMMA–sEBSP and PMMA–cap-sEBSP increased in contrast to that of pristine PMMA. The PBE–sEBSP and PBE–cap-sEBSP had lower Td5 values than those of pure PBE. We proposed that transalkoxylation between the hydroxy group of PBE and the methoxy group of sEBSP involved the elimination of methanol. The sEBSP and cap-sEBSP exerted a similar thermally stable effect on organic polymers. The Tg value of PMMA–sEBSP was slightly higher than that of pristine PMMA, while that of PBE–sEBSP was lower than that of pristine PBE. The sEBSP showed miscibility in organic polymers. PMMA–sEBSP and PBE–sEBSP were classified as a hybrid and nanocomposite, respectively.
References
Waki M, Mizoshita N, Ohsuna T, Tani T, Inagaki S (2010) Crystal-like periodic mesoporous organosilica bearing pyridine units within the framework. Chem Commun 46:8163–8165. https://doi.org/10.1039/C0CC01944E
Yamamoto K, Ohshita J (2019) Bridged polysilsesquioxane membranes for water desalination. Polym J 51:1103–1116. https://doi.org/10.1038/s41428-019-0237-9
Ren X, Tsuru T (2019) Organosilica-based membranes in gas and liquid-phase separation. Membranes 9:107. https://doi.org/10.3390/membranes9090107
Aoki Y, Shimizu T, Kanamori K, Maeno A, Kaji H, Nakanishi K (2017) Low-density, transparent aerogels and xerogels based on hexylene-bridged polysilsesquioxane with bendability. J Sol-Gel Sci Technol 81:42–51. https://doi.org/10.1007/s10971-016-4077-1
Sainohira Y, Fujino K, Shimojima A, Kuroda K, Kaneko Y (2019) Preparation of CO2-adsorbable amine-functionalized polysilsesquioxanes containing cross-linked structures without using surfactants and strong acid or base catalysts. J Sol-Gel Sci Technol 91:505–513. https://doi.org/10.1007/s10971-019-05072-6
Liu N, Yu K, Smarsly B, Dunphy DR, Jiang YB, Brinker CJ (2002) Self-directed assembly of photoactive hybrid silicates derived from azobenzene-bridged silsesquoxane. J Am Chem Soc 124:14540–14541. https://doi.org/10.1021/ja027991w
Shea KJ, Loy DA (2001a) Bridged polysilsesquioxanes. Molecular-engineered hybrid organic–inorganic materials. Chem Mater 13:3306–3319. https://doi.org/10.1021/cm011074s
Shea KJ, Loy DA (2001b) A mechanistic investigation of gelation. The sol–gel polymerization of precursors to bridged polysilsesquioxanes. Acc Chem Res 34:707–716. https://doi.org/10.1021/ar000109b
Kuge H, Hagiwara Y, Shimojima A, Kuroda K (2008) Oligomeric alkoxysilanes with cagelike hybrids as cores: designed precursors of nanohybrid materials. Chem Asian J 3:600–606. https://doi.org/10.1002/asia.200700242
Loy DA, Carpenter JP, Alam TM, Shaltout R, Dorhout PK, Greaves J, Small JH, Shea KJ (1999) Cyclization phenomena in the sol–gel polymerization of α,ω-bis(triethoxysilyl)alkanes and incorporation of the cyclic structures into network silsesquioxane polymers. J Am Chem Soc 121:5413–5425. https://doi.org/10.1021/ja982751v
Yamamoto K, Ohshita J, Mizumo T, Tsuru T (2014) Polymerization behavior and gel properties of ethane, ethylene and acetylene-bridged polysilsesquioxanes. J Sol-Gel Sci Technol 71:24–30. https://doi.org/10.1007/s10971-014-3322-8
Gunji T, Hayashi Y, Komatsubara A, Arimitsu K, Abe Y (2012) Preparation and properties of flexible free-standing films via polyalkoxysiloxanes by acid-catalyzed controlled hydrolytic polycondensation of tetraethoxysilane and tetramethoxysilane. Appl Organomet Chem 26:32–36. https://doi.org/10.1002/aoc.1861
Gunji T, Iizuka Y, Arimitsu K, Abe Y (2004) Preparation and properties of alkoxy(methyl)silsesquioxanes as coating agents. J Polym Sci A: Polym Chem 42:3676–3684. https://doi.org/10.1002/pola.20233
Wang Z, Wang D, Qian Z, Guo J, Dong H, Zhao N, Xu J (2015) Robust superhydrophobic bridged silsesquioxane aerogels with tunable performances and their applications. ACS Appl Mater Interfaces 7:2016–2024. https://doi.org/10.1021/am5077765
Chiang CL, Ma CCM, Wu DL, Kuan HC (2003) Preparation, characterization, and properties of novolac-type phenolic/SiO2 hybrid organic–inorganic nanocomposite materials by sol–gel method. J Polym Sci A Polym Chem 41:905–913. https://doi.org/10.1002/pola.10624
Arkhireeva A, Hay JN, Lane JM, Manzano M, Masters H, Oware W, Shaw SJ (2004) Synthesis of organic-inorganic hybrid particles by sol-gel chemistry. J Sol-Gel Sci Technol 31:31–36. https://doi.org/10.1023/B:JSST.0000047956.24117.89
Zhang QG, Liu QL, Zhu AM, Xiong Y, Zhang XH (2008) Characterization and permeation performance of novel organic–inorganic hybrid membranes of poly(vinyl alcohol)/1,2-bis(triethoxysilyl)ethane. J Phys Chem B 112:16559–16565. https://doi.org/10.1021/jp807573g
Tanaka K, Adachi S, Chujo Y (2009) Structure–property relationship of octa-substituted POSS in thermal and mechanical reinforcements of conventional polymers. J Polym Sci A: Polym Chem 47:5690–5697. https://doi.org/10.1002/pola.23612
Mohamed MG, Kuo SW (2019a) Functional polyimide/polyhedral oligomeric silsesquioxane nanocomposites Polymers 11:26. https://doi.org/10.3390/polym11010026
Mohamed MG, Kuo SW (2019b) Functional silica and carbon nanocomposites based on polybenzoxazines. Macromol Chem Phys 220:1800306. https://doi.org/10.1002/macp.201800306
Sato Y, Hayami R, Miyase Y, Yamamoto K, Gunji T (2020) Preparation and properties of methyl- and cyclohexyl-silsesquoxane oligomers as organic–inorganic fillers. J Sol-Gel Sci Technol 95:474–481. https://doi.org/10.1007/s10971-020-05291-2
Abe Y, Gunji T (2004) Oligo- and polysiloxanes. Prog Polym Sci 29:149–182. https://doi.org/10.1016/j.progpolymsci.2003.08.003
Hayami R, Nishikawa I, Hisa T, Nakashima H, Sato Y, Ideno Y, Sagawa T, Tsukada S, Yamamoto K, Gunji T (2018) Preparation and characterization of stable DQ silicone polymer sols. J Sol-Gel Sci Technol 88:660–670. https://doi.org/10.1007/s10971-018-4839-z
Hayami R, Izumiya T, Kokaji T, Nakagawa H, Tsukada S, Yamamoto K, Gunji T (2019) 2-Triethoxysilylazulene derivatives: Syntheses and optical properties, and hydrolysis–condensation of 2-triethoxysilylazulene. J Sol-Gel Sci Technol 91:399–406. https://doi.org/10.1007/s10971-019-04991-8
Guan S, Inagaki S, Ohsuna T, Terasaki O (2001) Hybrid ethane–siloxane mesoporous materials with cubic symmetry. Micropor Mesopor Mater 44–45:165–172. https://doi.org/10.1016/S1387-1811(01)00181-0
Oviatt HW, Shea KJ, Small JH (1993) Alkylene-bridged silsesquioxane sol–gel synthesis and xerogel characterization. Molecular requirements for porosity Chem Mater 5:943–950. https://doi.org/10.1021/cm00031a012
Wahab MA, Imae I, Kawakami Y, Kim I, Ha CS (2006) Functionalized periodic mesoporous organosilica fibers with longitudinal pore architectures under basic conditions. Micropor Mesopor Mater 92:201–211. https://doi.org/10.1016/j.micromeso.2005.12.016
Ambati J, Rankin SE (2011) Reaction-induced phase separation of bis(triethoxysilyl)ethane upon sol–gel polymerization in acidic conditions. J Colloid Interface Sci 362:345–353. https://doi.org/10.1016/j.jcis.2011.06.064
Loy DA, Baugher BM, Baugher CR, Schneider DA, Rahimian K (2000) Substituent effects on the sol–gel chemistry of organotrialkoxysilanes. Chem Mater 12:3624–3632. https://doi.org/10.1021/cm000451i
Depla A, Lesthaeghe D, Van Erp TS, Aerts A, Houthoofd K, Fan F, Li C, Van Speybroeck V, Waroquier M, Kirschhock CEA, Martens JA (2011) 29Si NMR and UV–Raman investigation of initial oligomerization reaction pathways in acid-catalyzed silica sol–gel chemistry. J Phys Chem C 115:3562–3571. https://doi.org/10.1021/jp109901v
Mazúr M, Mlynárik V, Valko M, Pelikán P (1999) The time evolution of the sol-gel process: 29Si NMR study of hydrolysis and condensation reactions of tetramethoxysilane. Appl Magn Reson 16:547–557. https://doi.org/10.1007/BF03161950
Li YS, Lu W, Wang Y, Tran T (2009) Studies of (3-mercaptopropyl)trimethoxysilane and bis(trimethoxysilyl)ethane sol–gel coating on copper and aluminum. Spectrochim Acta A 73:922–928. https://doi.org/10.1016/j.saa.2009.04.016
Li YS, Wang Y, Tran T, Perkins A (2005) Vibrational spectroscopic studies of (3-mercaptopropyl)trimethoxysilane sol–gel and its coating. Spectrochim Acta A 61:3032–3037. https://doi.org/10.1016/j.saa.2004.11.031
Moriyama N, Nagasawa H, Kanezashi M, Ito K, Tsuru T (2018) Bis(triethoxysilyl)ethane (BTESE)-derived silica membranes: pore formation mechanism and gas permeation properties. J Sol-Gel Sci Technol 86:63–72. https://doi.org/10.1007/s10971-018-4618-x
Rocha MCG, Moraes LRC, Cella N (2017) Thermal and mechanical properties of vinyltrimethoxysilane (VTMOS) crosslinked high molecular weight polyethylene (HMWPE). Mater Res 20:1332–1339. https://doi.org/10.1590/1980-5373-mr-2016-0552
Chen MA, Lu XB, Guo ZH, Huang R (2011) Influence of hydrolysis time on the structure and corrosion protective performance of (3-mercaptopropyl)triethoxysilane film on copper. Corros Sci 56:2793–2802. https://doi.org/10.1016/j.corsci.2011.05.010
Park ES, Ro HW, Nguyen CV, Jaffe RL, Yoon DY (2008) Infrared spectroscopy study of microstructures of poly(silsesquioxane)s. Chem Mater 20:1548–1554. https://doi.org/10.1021/cm071575z
Guo M, Kanezashi M, Nagasawa H, Yu L, Yamamoto K, Gunji T, Ohshita J, Tsuru T (2019) Tailoring the microstructure and permeation properties of bridged organosilica membranes via control of the bond angles. J Membr Sci 584:56–65. https://doi.org/10.1016/j.memsci.2019.04.072
Capozzi CA, Pye LD, Condrate RA Sr (1992) Vibrational spectral/structural changes from the hydrolysis/polycondensation of methyl-modified silicates. I. comparisons for single monomer condensates. Mater Lett 15:130–136. https://doi.org/10.1016/0167-577X(92)90028-I
Liang Y, Anwander R (2004) Synthesis of pore-enlarged mesoporous organosilicas under basic conditions. Micropor Mesopor Mater 72:153–165. https://doi.org/10.1016/j.micromeso.2004.03.013
Newton WE, Rochow EG (1970) Vibrational spectra of some trialkoxysilanes. J Chem Soc A 1970:2664–2668. https://doi.org/10.1039/J19700002664
Tommasini FJ, Ferreira LC, Tienne LGP, Aguiar VO, Silva MHP, Rocha LFM, Marques MFV (2018) Poly(methyl methacrylate)-SiC nanocomposites prepared through in situ polymerization. Mater Res 21:e20180086. https://doi.org/10.1590/1980-5373-mr-2018-0086
Ismayil RV, Bhajantri RF, Praveena SD, Poojary B, Dutta D, Pujari PK (2010) Optival and microstructural studies on electron irradiated PMMA: A positron annihilation study. Polym Degrad Stab 95:1083–1091. https://doi.org/10.1016/j.polymdegradstab.2010.02.031
Chan CK, Peng SL, Chu IM, Ni SC (2001) Effects of heat treatment on the properties of poly(methyl methacrylate)/silica hybrid materials prepared by sol–gel process. Polymer 42:4189–4196. https://doi.org/10.1016/S0032-3861(00)00817-X
Nie B, Stutzman J, Xie A (2005) A vibrational spectral maker for probing the hydrogen-bonding status of protonated Asp and Glu residues. Biophys J 88:2833–2847. https://doi.org/10.1529/biophysj.104.047639
Hayami R, Nakamoto W, Sato Y, Yamamoto K, Gunji T (2019) Organic–inorganic hybrids based on poly(bisphenol A-co-epichlorohydrin) containing titanium phosphonate clusters. Polym J 51:1265–1271. https://doi.org/10.1038/s41428-019-0243-y
Nikolic G, Zlatkovic S, Cakic M, Cakic S, Lacnjevac C, Rajic Z (2010) Fast Fourier transform IR characterization of epoxy GY systems crosslinked with aliphatic and cycloaliphatic EH polyamine adducts. Sensors 10:684–696. https://doi.org/10.3390/s100100684
Kashiwagi D, Inaba A, Brown JE, Hatada K, Kitayama T, Masuda E (1986) Effects of weak linkages on the thermal and oxidative degradation of poly(methyl methacrylates). Macromolecules 19:2160–2168. https://doi.org/10.1021/ma00162a010
Manring LE (1989) Thermal degradation of poly(methyl methacrylate). 2. Vinyl-terminated polymer Macromolecules 22:2673–2677. https://doi.org/10.1021/ma00196a024
Peterson JD, Vyazovkin S, Wight CA (1999) Kinetic study of stabilizing effect of oxygen on thermal degradation of poly(methyl methacrylate). J Phys Chem B 103:8087–8092. https://doi.org/10.1021/jp991582d
Tong Y, Lunsford H (1991) Mechanistic and kinetic studies of the reactions of gas-phase methyl radicals with metal oxides. J Am Chem Soc 113:4741–4746. https://doi.org/10.1021/ja00013a005
Tanaka K, Yamane H, Mitamura K, Watase S, Matsukawa K, Chujo Y (2014) Transformation of sulfur to organic–inorganic hybrids employed by networks and their application for the modulation of refractive indices. J Polym Sci Part A: Polym Chem 52:2588–2595. https://doi.org/10.1002/pola.27274
Hayami R, Wada K, Sagawa T, Tsukada S, Watase S, Gunji T (2017) Preparation and properties of organic–inorganic hybrid polymer films using [Ti4(µ3-O)(OiPr)5(µ-OiPr)3(PhPO3)3]•thf. Polym J 49:223–228. https://doi.org/10.1038/pj.2016.108
Hayami R, Wada K, Nishikawa I, Sagawa T, Yamamoto K, Tsukada S, Gunji T (2017) Preparation and properties of organic–inorganic hybrid materials using titanium phosphonate clusters. Polym J 49:665–669. https://doi.org/10.1038/pj.2017.34
Aid S, Eddhahak A, Ortega Z, Froelich D, Tcharkhtchi A (2017) Experimental study of the miscibility of ABS/PC polymer blends and investigation of the processing effect. J Appl Polym Sci 134:44975. https://doi.org/10.1002/app.44975
Mammeri F, Bourhis EL, Rozes L, Sanchez C (2005) Mechanical properties of hybrid organic–inorganic materials. J Mater Chem 15:3787–3811. https://doi.org/10.1039/B507309J
Džunuzović E, Marinović-Cincović M, Vuković J, Jeremić K, Nedelijković JM (2009) Thermal properties of PMMA/TiO2 nanocomposites prepared by in-situ bulk polymerization. Polym Composites 30:737–742. https://doi.org/10.1002/pc.20606
Thomas P, Ernest Ravindran RS, Varma KBR (2014) Structural, thermal and electrical properties of poly(methyl methacrylate)/CaCu3Ti4O12 composite sheets fabricated via melt mixing. J Therm Anal Calorim 115:1311–1319. https://doi.org/10.1007/s10973-013-3500-x
Džunuzović E, Jeremić K, Nedelijković JM (2007) In situ radical polymerization of methyl methacrylate in a solution of surface modified TiO2 and nanoparticles. Eur Polym J 43:3719–3726. https://doi.org/10.1016/j.eurpolymj.2007.06.026
Varzeghani HN, Amraei IA, Mousavi SR (2020) Dynamic cure kinetics and physical-mechanical properties of PEG/nanosilica/epoxy composites. Int J Polym Sci 2020:7908343. https://doi.org/10.1155/2020/7908343
Hayami R, Wada K, Miyase Y, Sagawa T, Tsukada S, Yamamoto K, Gunji T (2018) Properties and surface morphologies of organic–inorganic hybrid thin films containing titanium phosphonate clusters. Polym J 50:1169–1177. https://doi.org/10.1038/s41428-018-0108-9
Acknowledgements
This work was supported by JSPS KAKENHI Grant Numbers JP18K14287 and JP19K05636.
Author information
Authors and Affiliations
Contributions
RH wrote original draft and performed the acquisition and analysis of data. YI performed the preparation and measurement of silsesquioxane polymer and composites (in original manuscript). YS prepared silsesquioxane polymer (in original and revised) and measured silsesquioxane polymer (in revised). HT performed the preparation and measurement of composites (in revised). KY was supervision. TG wrote for review and was supervision. KY and TG were funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10965_2020_2294_MOESM1_ESM.docx
Supplementary file1 (DOCX 179 kb)1
Rights and permissions
About this article
Cite this article
Hayami, R., Ideno, Y., Sato, Y. et al. Soluble ethane-bridged silsesquioxane polymer by hydrolysis–condensation of bis(trimethoxysilyl)ethane: characterization and mixing in organic polymers. J Polym Res 27, 316 (2020). https://doi.org/10.1007/s10965-020-02294-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-020-02294-z