Abstract
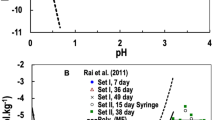
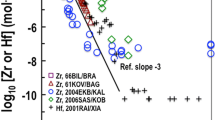
The solubility of SnO2(cassiterite) was studied at 23±2 °C as a function of time (7 to 49 days) and pH (0 to 14.5). Steady state concentrations were reached in <7 days. The data were interpreted using the SIT model. The data show that SnO2(cassiterite) is the stable phase at pH values of < ∼11.7. These extensive data provided a log 10 K 0 value of −64.39±0.30 for the reaction (SnO2(cassiterite) +2H2O⇄Sn4++4OH−) and values of 1.86±0.30, ≤−0.62, −9.20±0.34, and −20.28±0.34 for the reaction (\(\mathrm{Sn}^{4+} + n\mathrm{H}_{2}\mathrm{O} \rightleftarrows \mathrm{Sn}(\mathrm{OH})_{n}^{4 - n} + n\mathrm{H}^{+}\)) with values of “n” equal to 1, 4, 5, and 6 respectively. These thermodynamic hydrolysis constants were used to reinterpret the extensive literature data for SnO2(am) solubility, which provided a log 10 K 0 value of −61.80±0.29 for the reaction (SnO2(am)+2H2O⇄Sn4++4OH−). SnO2(cassiterite) is unstable under highly alkaline conditions (NaOH concentrations >0.003 mol⋅dm−3) and transforms to a double salt of SnO2 and NaOH. Although additional well-focused studies will be required for confirmation, the experimental data in the highly alkaline region (0.003 to 3.5 mol⋅dm−3 NaOH) can be well described with log 10 K 0 of −5.29±0.35 for the reaction Na2Sn(OH)6(s)⇄Na2Sn(OH)6(aq).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Weast, R.C.: Handbook of Chemistry and Physics, 53rd. Chemical Rubber Company, Cleveland (1972)
Amaya, T., Chiba, T., Suzuki, K., Oda, C., Yoshikawa, H., Yui, M.: Solubility of Sn(IV) oxide in dilute NaClO4 solution at ambient temperature. Mater. Res. Soc. Symp. Proc. 465, 751–758 (1997)
Lothenbach, B., Ochs, M., Wanner, H., Yui, M.: Thermodynamic data for the speciation and solubility of Pd, Pb, Sn, Sb, Nd, and Bi in aqueous solutions. JNC TN8400 99-011. Japan Nuclear Cycle Development Institute (now Japan Atomic Energy Agency) (1999)
Seby, F., Potin-Gautier, M., Giffant, E., Donard, O.F.X.: A critical review of thermodynamic data for inorganic tin species. Geochim. Cosmochim. Acta 65, 3041–3053 (2001)
Oda, C., Amaya, T.: Effects of ligands on the solubility of tin. Japanese Nuclear Cycle Development Institute (1998)
Feitknecht, W., Schindler, P.: Solubility constants of metal oxides, metal hydroxides and metal hydroxide salts in aqueous solution. Pure Appl. Chem. 1, 130–199 (1963)
Wagman, D.D., Evans, W.H., Parker, V.B., Schumm, R.H., Halow, I., Bailey, S.M., Churney, K.L., Nuttall, R.L.: The NBS tables of chemical thermodynamic properties: selected values for inorganic and C1 and C2 organic substances in SI units. J. Phys. Chem. Ref. Data 11(2), 1–392 (1982)
Barsukov, V.L., Klintsova, A.P.: Solubility of cassiterite in water and aqueous NaOH at 25 °C. Geokhimiya 10, 1268–1272 (1970)
Barybin, V.I., Sharygin, P.M., Gonchar, V.F., Moiseev, V.E.: Solubility of hydrated tin dioxide in water. Izv. Akad. Nauk SSSR, Neorg. Mater. 23, 1162–1165 (1988)
Dadze, T.P., Sorokhin, V.I., Nekrasov, I.Y.: Solubility of SnO2 in water and in aqueous solution of HCl HCl + KCl, and HNO3 at 200–400 °C and 101.3 MPa. Geokhimiya 10, 1482–1492 (1981)
Klintsova, A.P., Varsukov, V.L.: Solubility of cassiterite in water and in aqueous NaOH solution at elevated temperatures. Geokhimiya 5, 701–709 (1973)
Huey, C.S., Tartar, H.V.: The stannous-stannic oxidation-reduction potential. J. Am. Chem. Soc. 56, 2585–2588 (1934)
Rai, D., Moore, D.A., Felmy, A.R., Rosso, K.M., Bolton, H.J.: PuPO4(cr, hyd.) solubility product and Pu3+ complexes with phosphate and ethylenediaminetetraacetic acid. J. Solution Chem. 39, 778–807 (2010)
Rand, M.H., Fuger, J., Grenthe, I., Neck, V., Rai, D.: Chemical thermodynamics of thorium. Nuclear Energy Agency, Organization for Economic Co-Operation and Development (2008)
Nazarenko, V.A., Antonovich, V.P., Nevskaya, E.M.: Spectrophotometric determination of the hydrolysis constants of tin(IV) ions. Russ J. Inorg. Chem. 15, 980–982 (1971)
Macchi, G., Pettine, M.: Voltammetric characterization and chemical behavior of inorganic tin in natural waters. Environ. Sci. Technol. 14, 815–818 (1980)
Pourbaix, M.: Atlas of d’equilibres electrochimiques a 25 °C. Gauthier-Villars & Cie, Paris (1964)
Rai, D.: Solubility product of Pu(IV) hydrous oxide and equilibrium constants of Pu(IV)/Pu(V), Pu(IV)/Pu(VI), and Pu(V)/Pu(VI) couples. Radiochim. Acta 35, 97–108 (1984)
Felmy, A.R.: GMIN: A Computerized Chemical Equilibrium Model Using a Constrained Minimization of the Gibbs Free Energy. Pacific Northwest National Laboratory, Richland (1990)
Sterner, S.M., Felmy, A.R., Rustad, J.R., Pitzer, K.S.: Thermodynamic Analysis of Aqueous Solutions Using INSIGHT. Pacific Northwest National Laboratory, Richland (1997)
The Powder Diffraction File (PDF) International Center for Diffraction Data (ICDD). Newtown Square, PA (2011)
Fatouros, N., Rouelle, F., Chemla, M.: Influence de la formation de complexes chlorures sur la reduction electrochimique de SnIV en milieu perchloroique acide. J. Chim. Phys. Phys.-Chim. Biol. 75, 477–483 (1978)
Jackson, K.J., Helgeson, H.C.: Chemical and thermodynamic constraints on the hydrothermal transport and deposition of tin: I. Calculation of the solubility of cassiterite at high pressures and temperatures. Geochim. Cosmochim. Acta 49, 1–22 (1985)
Migdisov, A.A., Williams-Jones, A.E.: An experimental study of cassiterite solubility in HCl-bearing water vapour at temperatures up to 350 °C. Implications for tin ore formation. Chem. Geol. 217, 29–40 (2005)
Sherman, D.M., Ragnarsdottir, K.V., Oelkers, E.H., Collins, C.R.: Speciation of tin (Sn2+ and Sn4+) in aqueous Cl solutions from 25 °C to 350 °C: an in situ EXAFS study. Chem. Geol. 167, 169–176 (2000)
Lothenbach, B., Ochs, M., Hager, D.: Thermodynamic data for the solubility of tin(IV) in aqueous cementitious environments. Radiochim. Acta 88, 521–526 (2000)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rai, D., Yui, M., Schaef, H.T. et al. Thermodynamic Model for SnO2(cr) and SnO2(am) Solubility in the Aqueous Na+–H+–OH−–Cl−–H2O System. J Solution Chem 40, 1155–1172 (2011). https://doi.org/10.1007/s10953-011-9723-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-011-9723-1