Abstract
Aqueous calcium (Ca) concentrations are currently decreasing in many softwater lakes on the Boreal Shield. As the onset of these declines often pre-date direct monitoring programs, indirect techniques are required to examine the impacts of reduced Ca availability on aquatic communities with relatively high Ca demands such as the Cladocera (Class: Branchiopoda). Among the Cladocera, the family Daphniidae has been identified as a taxonomic group potentially useful for inferring past Ca concentrations due to their high Ca demands and preservation in lake sediments. Here, we use a “top/bottom” paleolimnological analysis to compare present-day cladoceran communities preserved in the surface sediments of 36 softwater lakes in south-central Ontario, Canada, which are potentially vulnerable to Ca decline (i.e. small headwater systems with present-day lakewater [Ca] < 3 mg L−1), with the communities present in lake sediments deposited prior to the onset of regional acid deposition. To distinguish the potential impacts of lake acidification from those of Ca availability (as Ca and pH trends are strongly correlated in this region), the study lakes were chosen to be evenly distributed about a present-day lakewater pH of 6 and Ca concentration of 1.5 mg L−1 (threshold values). Despite the importance of pH as an explanatory variable for the present-day assemblages, a comparison of the sedimentary remains from the two time periods indicate there have been large declines since pre-industrial times in the relative abundances of Ca-rich Daphnia spp. (particularly of the Daphnia longispina species complex), regardless of present-day pH, accompanied by increases in the Ca-poor species Holopedium glacialis. These observations suggest that recent declines in Ca concentration may have already fallen below baseline conditions, with marked implications for ecosystem function due to the differential responses among cladoceran taxa.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In recent decades, declines in the aqueous calcium (Ca) concentration of many softwater, boreal lakes in North America have been observed by long-term aquatic monitoring programs (Likens et al. 1998; Keller et al. 2001; Molot and Dillon 2008). These Ca declines are partly a long-term consequence of regional acid deposition enhancing the release of base cations (principally Ca) from catchment soils (Likens et al. 1996; Stoddard et al. 1999; Keller et al. 2001). As lakewater Ca concentration is dependent upon the Ca reservoirs available in catchment soils (Houle et al. 2006), Ca declines and reduced aqueous Ca availability are expected to persist in regions experiencing ongoing acid deposition, due to the continued depletion of these Ca reservoirs. In addition, these Ca declines may be exacerbated by Ca losses from the watershed associated with biomass removal (i.e. timber harvesting; Watmough and Aherne 2008). The potential of reduced Ca availability to both delay recovery from lake acidification (Stoddard et al. 1999), and act as an environmental stressor in its own right (Jeziorski et al. 2008), is of particular interest in softwater regions such as the Canadian Precambrian Shield. Many catchments within the Precambrian Shield have a limited ability to neutralize acid inputs due to their thin soils underlain by slow-weathering, granitic bedrock (a poor source of Ca minerals; Shaw et al. 1967). Although the observed declines in Ca are often small in absolute terms (e.g. 1–2 mg L−1), these changes can constitute large relative reductions in Ca availability in softwater lakes (e.g. from a survey of 3,700 Ontario lakes, 80 % had a [Ca] < 10 mg L−1; Neary et al. 1990), and declines of up to 40 % since the 1980s are widespread across Ontario, Canada (Jeziorski et al. 2008).
The regional extent of the Ca declines is raising concern regarding the ecological impacts of reduced Ca availability in terms of the direct consequences for aquatic organisms (Ashforth and Yan 2008; Cairns and Yan 2009), the indirect impacts upon aquatic food webs (e.g. changes among algal grazers due to Ca limitation facilitating increased algal production; Korosi et al. 2012), and the potential cascading impacts for terrestrial systems (St. Louis and Breebaart 1991; Watmough and Aherne 2008). The ability to predict the consequences of reduced aqueous Ca for aquatic ecosystems is of considerable interest for scientists, managers and policy makers; however, forecasting the future response of Ca-sensitive communities from direct monitoring records of Ca declines and concurrent ecological changes remains difficult. In part, this difficulty stems from the complexity of the site-specific mechanisms responsible for Ca declines (i.e. the dependence on local geology, weathering rates, acid inputs and timber harvesting; Watmough and Aherne 2008), but also from the lack of information regarding “baseline” Ca conditions (i.e. Ca concentrations prior to the onset of acid deposition).
To date, little information is available regarding either pre-impact Ca concentrations (i.e. prior to the onset of acid deposition and lake acidification in eastern North America during the early-mid twentieth century; Cogbill and Likens 1974; Charles et al. 1990), or the timing of the onset of the declines (i.e. the onset of Ca mobilization), which often pre-dates direct monitoring records (e.g. the Hubbard Brook Experimental Forest in New Hampshire, USA (Likens et al. 1998); the Experimental Lakes Area in northwestern Ontario, Canada (Blanchfield et al. 2009); and the Dorset Environmental Science Centre in south-central Ontario, Canada (Yan et al. 2008a)). Therefore, despite rising interest in the observations of decreasing lakewater Ca, the relationship with pre-impact concentrations is uncertain. In some instances, the decline may be a return from an anthropogenically elevated state that is induced by the accelerated release of base cations from watershed soils (i.e. a ‘recovery’; Watmough and Aherne 2008). Whether such ‘recoveries’ represent a return to pre-impact conditions, or alternatively, a continued decline below historical concentrations, remains unclear. The lack of historical perspective regarding lakewater Ca necessitates the use of indirect methods such as modeling (Watmough and Aherne 2008) and paleolimnological techniques (Keller et al. 2001; Smol 2008) to infer past Ca concentrations. However, before accurate predictive models of past Ca concentrations can be constructed from biological remains, suitable paleolimnological indicator species must be identified.
The development of paleolimnological models to infer past lakewater Ca concentrations has so far proven difficult due to the strong correlation between Ca concentration and pH. Commonly used paleolimnological indicator species such as diatoms and scaled chrysophytes have relatively low Ca requirements, and Ca-inference models using these taxa have proven insensitive to short-term variations in environmental conditions (likely due to concurrent changes in pH over the period of interest: Keller et al. 2001). In contrast, some members of the Cladocera (Crustacea, Branchipoda), particularly among the family Daphniidae, have been identified in recent laboratory and field surveys as especially vulnerable to Ca decline, due to their direct dependence on aqueous Ca availability (Ashforth and Yan 2008; Jeziorski and Yan 2006). Paleolimnological investigations utilizing the Cladocera have been used to examine a broad suite of environmental variables including climate variation (Kamenik et al. 2007), trophic status of lakes (Chen et al. 2010) and lake acidification (Paterson 1994). The application of these cladoceran paleolimnological techniques to softwater systems experiencing Ca declines may provide insight regarding historical Ca conditions, due to the differing Ca requirements among cladoceran taxa.
Similar to many other invertebrates, the Cladocera use Ca obtained from the surrounding environment as a structural material within their carapaces (Greenaway 1985). These Ca requirements are continuous throughout the animals’ life cycles (and studied in most detail amongst Daphnia spp.), due to the large amounts of Ca lost during each moult when the carapace is shed and subsequently regenerated (Alstad et al. 1999). The dependence of cladocerans upon aqueous Ca availability has been demonstrated in both field surveys and examinations of present-day sedimentary remains that have identified lakewater Ca concentration as a significant explanatory environmental variable for cladoceran communities (Wærvågen et al. 2002; DeSellas et al. 2008; Jeziorski et al. 2012). The importance of aqueous Ca is likely due to species-specific differences in Ca requirements and Ca content (i.e. % dry weight), which are most pronounced among planktonic taxa such as Daphnia spp. (Ca-rich), Bosmina spp. (Ca-poor), and Holopedium glacialis (Ca-poor; Wærvågen et al. 2002; Jeziorski and Yan 2006). However, similar differences have also been identified within the littoral cladoceran community with Alona rectangula and Disparalona spp. having much higher Ca content than Acroperus harpae, Alonella excisa, and Chydorus spp. (Shapiera et al. 2011). The species-specific differences in Ca content do not appear to be a simple function of body size, as larger taxa (e.g. A. harpae) can have a lower Ca content than their smaller peers (e.g. A. rectangula; Shapiera et al. 2011). Furthermore, ambient Ca concentration has been observed to influence daphniid Ca content (albeit to a lesser degree than species identity; Jeziorski and Yan 2006).
Among the Cladocera, the response to differences in ambient Ca availability has been most intensively studied among daphniid taxa. In laboratory analyses, the species Daphnia pulex has been identified as having a threshold response at low Ca concentrations (<1.5 mg L−1, Ashforth and Yan 2008). This may be related to a limited ability to regulate body Ca content (particularly influx rates; Tan and Wang 2009), and declines in daphniid abundance coincident with this threshold have been observed in paleolimnological case studies (Jeziorski et al. 2008).
The application of daphniids to questions regarding environmental Ca decline is complicated by the lack of taxonomic resolution among the sedimentary remains of daphniid species (Szeroczyńska and Sarmaja-Korjonen 2007; Korosi et al. 2011), and the apparent tolerance for low Ca concentrations of at least one species (i.e. D. catawba) among the members of the D. pulex species complex (Jeziorski et al. 2012; Shapiera et al. 2012). Similar findings have been reported from laboratory experiments, showing that inter-specific differences in Ca content within the genus Daphnia may not be a reliable indicator of inter-specific differences in Ca-sensitivity (Tan and Wang 2010). Despite these complications, examination of how cladoceran communities have changed through time in lakes sensitive to Ca decline (i.e. presently at or below Ca concentrations known to be detrimental to Ca-rich Cladocera) provide useful information that is applicable to many other lakes on the Precambrian Shield experiencing declines in Ca concentration.
Although lake Ca concentration can explain significant amounts of variation among cladoceran communities (DeSellas et al. 2011; Jeziorski et al. 2012), there is still a lack of knowledge regarding the response of these communities to long-term changes in Ca availability. Of particular interest is whether species other than the daphniids are potentially limited by Ca (e.g. among the littoral community that is typically well-represented in the sediment record). We address this topic here by examining cladoceran remains preserved in the sediments of 36 low [Ca] lakes located in the Muskoka region of south-central Ontario, using what is often referred to by paleolimnologists as the “top/bottom” approach (Smol 2008). The remains of present-day cladoceran communities (i.e. those preserved in surface or “top” sediments; sediment depths = 0.0–0.25 cm) were compared with the remains preserved in sediments at a depth corresponding to times that predate the onset of Ca decline in this geographic region (i.e. prior to the onset of acid deposition in eastern North America during the early-mid twentieth century; Cogbill and Likens 1974; Charles et al. 1990) and the accelerated mobilization of Ca from soil reservoirs (“bottom” sediments; sediment depths ~ 25.0–25.25 cm).
A prior analysis relating the cladoceran assemblages from the surface sediments of these lakes to their present-day environmental variables identified lake depth and pH as explaining significant amounts of variation in the present-day assemblages (Jeziorski et al. 2012). However, although Ca was not a significant explanatory variable for the modern cladoceran assemblages, many similar lakes in this region have experienced substantial declines in Ca concentration in recent decades (Jeziorski et al. 2008; Molot and Dillon 2008). Therefore, the cladoceran communities within these lakes may have already been impacted by Ca limitation; many of these lakes now have Ca concentrations shown to be detrimental to taxa with high Ca demands (the actual Ca concentrations of these lakes prior to the onset of regional acid deposition remain unknown). Here, we ask the following questions: (1) How do present-day daphniid sedimentary assemblages differ from those deposited prior to the onset of regional acid deposition? (2) Is there evidence that any of these lakes have either crossed critical Ca thresholds or are naturally low Ca systems? (3) How have changes among the daphniid and non-daphniid species differed over this time period? and (4) Are there other species among the Cladocera, other than daphniids, that are potentially reliable indicators of historical lake Ca concentration? Collectively, these data should improve the interpretation of changes in the sedimentary cladoceran record with respect to the growing and widespread problem of reduced Ca availability.
Materials and methods
Lake selection
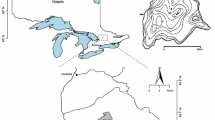
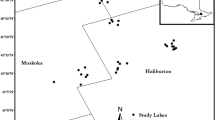
A subset of 36 softwater lakes from the Muskoka-Haliburton region of south-central Ontario (Fig. 1) was selected from the 300-lake chemistry database developed as part of the Canadian Aquatic Invasive Species Network (CAISN; Cairns et al. 2006). To focus our analysis on lakes that may have experienced ecologically important Ca declines, the subset was restricted to lakes that currently have Ca concentrations near levels known to impede the growth and survival of the Ca-rich cladoceran D. pulex (Ashforth and Yan 2008). Lakes were originally selected from the database according to the following criteria: Ca < 3.1 mg L−1, 5 < pH < 7, lake depth < 40 m, dissolved organic carbon (DOC) < 11 mg L−1, and lake surface area < 250 ha. The selection process reduced the data set to 162 lakes, from which the 36 study lakes (Table 1) were selected to be evenly distributed about important pH and Ca thresholds for cladoceran community composition (pH 6 and 1.5 mg Ca L−1; Holt et al. 2003; Ashforth and Yan 2008). The relationship between cladoceran sedimentary assemblages from surface sediments of these 36 lakes and present-day environmental variables have been analyzed previously (Jeziorski et al. 2012). Here, the analysis was extended to compare present-day sedimentary assemblages with those predating regional acid deposition in eastern North America.
Sediment cores were collected during the summers of 2007 and 2008 from the deepest basin of each lake using a Glew (1989) gravity corer and immediately sectioned on shore at a resolution of 0.25 cm using a Glew (1988) vertical extruder (this slice thickness typically represents the most recent ~1–3 years of accumulated sediment in this geographic region; Mills et al. 2009). A “top/bottom” analysis (Smol 2008) was performed using two intervals from each lake; a “top” slice (sediment depth = 0.0–0.25 cm) to represent present-day assemblages and a “bottom” slice (sediment depth ≈ 25.0–25.25 cm; Table 1) to represent assemblages from a period prior to the onset of lake acidification in eastern North America (i.e. pre-1920; Cogbill and Likens 1974; Charles et al. 1990). Radiometric 210Pb dating has demonstrated that sediment depths >25 cm from lakes of south-central Ontario are generally representative of times prior to European settlement in the region (i.e. pre-1850; Clerk et al. 2000; Little et al. 2000; Faulkenham et al. 2003; Shapiera et al. 2012), and is a depth commonly used in “top/bottom” analyses to represent the preindustrial period (e.g. Hall and Smol 1996; DeSellas et al. 2011). Between core collection and sample preparation, the sediments were transported to the Paleoecological and Environmental Assessment Research Laboratory (PEARL) at Queen’s University in Kingston, Ontario and stored in a cold room.
Sample preparation and cladoceran analysis
The preparation of slides followed standard cladoceran analysis methods (Korhola and Rautio 2001) with only minor deviations. A subsample of sediment (~1 g) from each interval was deflocculated in a 10 % KOH solution for ~30 min, then rinsed through a 38-μm sieve using deionized water. To aid identification, cladoceran remains were stained using a safranin-glycerin solution and several drops of ethanol were added to the slurry to prevent fungal growth, 50-μL aliquots of the slurry were then pipetted onto microscope slides and made semi-permanent using glycerin jelly.
For each interval a minimum of 90 cladocerans (a large enough subsample to provide a representative estimation of the sedimentary cladoceran assemblage; Kurek et al. 2010) were identified using a Leica DMRB light microscope with bright field optics (10–40X objective, 15X ocular lens). Individual cladoceran remains (carapaces, headshields, postabdominal claws, etc.) were tabulated separately and the minimum number of individuals present for each taxon was calculated from the most abundant remain (Frey 1986).
The primary taxonomic resources used for identification of the cladoceran subfossils included: Smirnov (1974, 1996), Sweetman and Smol (2006) and Szeroczyńska and Sarmaja-Korjonen (2007). Daphniid remains were attributed to a particular species complex based on the presence/absence of stout spines on the middle comb of the postabdominal claw (Korosi et al. 2011): in this geographic region the D. pulex complex (stout spines present) can include D. pulex, D. pulicaria, D. catawba and D. minehaha; and the D. longispina complex (stout spines absent) can include D. ambigua, D. mendotae, D. longiremis, D. dentifera, D. dubia, D. parvula and D. retrocurva (Hebert 1995; Yan et al. 2008b).
Statistical analysis
Changes among the cladoceran communities between the two time periods were identified with respect to individual species and genera, taxonomic groups (i.e. the daphniid species complexes and the family Sididae), as well as the complete assemblages. Prior to analysis, a pre-screening procedure was applied to the sedimentary assemblages to remove any taxa from the dataset that did not appear in at least two lakes with a relative abundance of 2 %; the resulting dataset contained 25 taxa or species complexes. The relative abundances were then square-root transformed in order to reduce the dominance of Bosmina spp. from obscuring any subtle trends in the data.
To determine whether the sedimentary assemblages differed between the two time periods, an Analysis of Similarity (ANOSIM) was performed using the software package PRIMER 6 (Clarke and Gorley 2006). The ANOSIM is a non-parametric multivariate test that examines a similarity matrix (of cladoceran relative abundances in our analysis) and compares a priori defined groups (“tops” vs. “bottoms”) of samples (lakes) by ranking them according to Bray-Curtis similarity, with minimal statistical assumptions (Clarke 1993). A test statistic (R) was calculated to determine whether average pair-wise similarity within groups was greater than average pair-wise similarity between groups, and the probability that such a value for the test statistic could be generated by chance was then determined. ANOSIM was used to test the null hypothesis that there was no difference between the subfossil assemblages from the “top” depth intervals versus those from the “bottom” intervals. The individual species that contributed most to any observed differences in community composition were identified using the SIMPER procedure also available in the PRIMER 6 software package (Clarke 1993).
To ascertain how the cladoceran communities from the 36 lakes dataset have changed through time relative to the environmental variables that best explain the present-day assemblages, the sedimentary assemblages were compared using the multivariate ordination methods available within the “vegan” package (Oksanen et al. 2010) for the R software environment (R Development Core Team 2010). An initial detrended correspondence analysis (DCA) of the dataset revealed the gradient lengths of the first two axes to be relatively short (1.13 and 1.17 standard deviation units) and that linear constrained ordination methods were appropriate for further analysis of the data set. The “bottom” assemblages were included as passive samples (as the preindustrial environmental conditions are unknown) in a redundancy analysis (RDA) of the 36 modern day assemblages constrained to pH and lake depth. This RDA analysis was used to determine how cladoceran communities have changed with respect to lake depth and pH, and whether the responses have differed between lakes with present-day pH above and below 6, a threshold value for crustacean zooplankton communities (including cladocerans; Holt et al. 2003).
In addition to examining the changes within the cladoceran communities between the two time periods with respect to pH, particular attention was also paid to changes possibly induced by reduced aqueous Ca availability, as the dataset is composed of softwater lakes potentially vulnerable to (and that may have experienced) substantive Ca declines (to concentrations that may be currently limiting for Ca-rich taxa) over the period of time between the deposition of the two sediment intervals. Responses to reductions in aqueous Ca availability will differ among cladoceran taxa according to species-specific requirements; therefore, within-lake changes in Ca-poor versus Ca-rich taxonomic groups (i.e. Ca-poor taxa such as Bosmina spp., H. glacialis and Alona rectangula vs. Ca-rich taxa such as Daphnia spp. and Chydorus spp.; Jeziorski and Yan 2006; Shapiera et al. 2011) were examined. More specifically, as laboratory analyses of Ca limitation within the Cladocera have largely been limited to daphniid species, daphniid changes over time were examined among the two species complexes (i.e. the D. longispina complex vs. the D. pulex complex).
Results
Comparison of the sedimentary remains of Cladocera between the present-day and preindustrial assemblages revealed that large changes over the intervening time period were common among the study lakes (Fig. 2). Although dominance of the sedimentary record by the remains of Bosmina spp. is universal for both time periods, there was a general increase in their dominance over time (“bottoms” 19–85 %, median 63 %; “tops” 29–92 %, median 68 %), accompanied by substantial changes among the wider community. Among the other planktonic taxa there have been notable declines in the relative abundances of the D. longispina species complex (“bottoms” 0–19 %, median 1 %; “tops” 0–8 %, median 2 %), substantial increases and decreases in the D. pulex species complex (“bottoms” 0–48 %, median 0 %; “tops” 0–20 %, median 1.3 %), and increases in H. glacialis (“bottoms” 0–11 %, median 0.9 %; “tops” 0–13 %, median 2.1 %). Within the diverse littoral communities of the study lakes, decreases were evident among the family Sididae (particularly Sida crystallina), whereas responses among the other taxa were subtle.
The ANOSIM identified significant differences between the two time periods (R = 0.08, p = 0.002). However, the calculated dissimilarity was quite low (i.e. the near zero value of the test statistic, R), indicative of the rarity of the complete elimination or introduction of taxa. Instead differences through time were primarily due to redistributions in relative abundances within the existing communities. The SIMPER analysis confirmed the principal sources of dissimilarity to be changes amongst the planktonic taxa, specifically, increases in the present-day samples of the D. pulex species complex, H. glacialis and Bosmina spp. and decreases in the D. longispina species complex.
The RDA of the sedimentary cladoceran remains revealed large directional changes between the present-day and historical assemblages, when constrained by the present-day environmental gradients (RDA axis 1—primarily associated with lake depth and strongly correlated with nutrients, and RDA axis 2—primarily associated with pH and strongly correlated with conductivity and ionic concentrations; Fig. 3). The observed changes between the two time periods were predominantly along RDA axis 2 and most pronounced among those lakes with present-day pH < 6 (Fig. 3). Among the planktonic taxa there have been broad increases in both Bosmina spp. and H. glacialis (Fig. 4). Furthermore, large declines of both daphniid species complexes have occurred within many of the lakes with historical daphniid populations, however the lakes with marked increases in daphniid relative abundances are often those from which daphniids were historically absent (Fig. 4). Among the littoral taxa, trends with respect to the time period and present-day pH were ambiguous, with the one exception being the general declines among the Sididae (primarily due to substantive declines of Sida crystallina; Fig. 4).
Redundancy analysis (RDA) results for the cladoceran sedimentary assemblages from the “top” (present-day) sediments of the 36 lakes (triangles). Only lake depth and pH were identified as significant environmental variables following forward selection. All other environmental variables and the “bottom” assemblages (squares) have been plotted passively. Dashed lines connect samples from the same lake. Symbol colours indicate present-day status; pH > 6 (white), pH < 6 (black)
A comparison of the trends within the relative abundances of Ca-rich Daphnia spp. revealed a differential response between the two species complexes (Fig. 5). Large daphniid declines have occurred in each lake that had a high daphniid relative abundance in the “bottom” sediments irrespective of species complex; however, the increases through time are principally due to the D. pulex species complex, whereas the several small increases in the relative abundance of the D. longispina complex were restricted to lakes where it was absent from the “bottom” sediment interval (Fig. 5). The 1.5 mg L−1 Ca threshold identified as detrimental to the survival of D. pulex (Ashforth and Yan 2008) is not reflected in the sedimentary assemblages, as there are high abundances, and even several increases since preindustrial times in the relative abundance of the D. pulex species complex in some of the lakes with present-day [Ca] < 1.5 mg L−1 (Fig. 5). Furthermore, when examining the changes with respect to present-day pH, although the lakes that have recorded the biggest changes in daphniid abundances are those with a present-day pH < 6, daphniid declines have also occurred (particularly among the D. longispina complex) in many of the lakes that are currently circumneutral (present-day pH 6–6.7; Table 1; Fig. 5).
Changes in the relative abundances of total daphniids between the pre-acid deposition (“bottoms”) and present-day (“tops”) sediment samples from the 36 study lakes arranged in ascending order (top to bottom) for: a present-day Ca concentration (dashed line indicates 1.5 mg L−1 Ca); and b present-day pH (dashed line indicates pH = 6.0)
Discussion
In our comparison of cladoceran communities preserved in the sediments of 36 softwater lakes within south-central, Ontario (Fig. 1), the assemblages preserved in the present-day sediments differed significantly from those in sediments deposited prior to the onset of acid precipitation in this region of North America (i.e. the pre-1920 “bottoms”; Fig. 2). Rather than radical community shifts, the changes were characterized by redistributions within the existing communities, evident in the low overall dissimilarity and taxonomic diversity (ANOSIM R = 0.08, p = 0.002; 25 taxa). Principally, the differences between the two time periods were due to increases in the present-day sediments of the relative abundances of Bosmina spp. and H. glacialis, decreases in the D. longispina species complex, as well as a variable response within the D. pulex species complex (Figs. 4, 5). Although both lake depth and pH have significant explanatory power with respect to the 36 present-day cladoceran assemblages (Jeziorski et al. 2012), there is little evidence of regional changes in lake depth between the two time periods (the importance of lake depth is likely related to a gradient of pelagic vs. littoral habitat within the study lakes), whereas the impacts of acid deposition upon the softwater lakes of south-central Ontario are a topic of intensive study (Hall and Smol 1996; Faulkenham et al. 2003; Holt et al. 2003; Yan et al. 2008a).
Among the Cladocera, environmental tolerances regarding changes in ambient pH (and related variables such as Ca concentration) have been best studied among planktonic taxa (especially Daphnia spp.), the ecological group that both dominated the sedimentary assemblages of all 36 lakes, and experienced the most marked changes through time (Fig. 2). Not surprisingly, the within-lake analysis of community changes through time, constrained to the significant explanatory variables for the present-day assemblages, found the principal direction of change to be along RDA Axis 2 (primarily associated with changes in pH and highly correlated with conductivity and ionic concentrations—including Ca; Fig. 3). Changes in lake chemistry with respect to pH in this geographic region have been demonstrated in diatom-inferred reconstructions of lakewater pH (Hall and Smol 1996). Although far removed from point sources of acid deposition, many lakes currently below pH 6.0 have acidified relative to their preindustrial state due to long-range, regional deposition (Hall and Smol 1996). A pattern seemingly related to acidification is apparent within our dataset, as the lakes with a present-day pH < 6.0 have exhibited the greatest changes in cladoceran species assemblages. However, the direction of these changes over time is somewhat counter-intuitive, as the present-day assemblages from many of the low pH lakes plot closer to the higher pH lakes than they did historically (Fig. 3). Thus, cladoceran communities from acidified and non-acidified lakes are more similar now than in the past, presenting an interesting contradiction that warrants a further examination of the individual taxa.
The present-day and historical cladoceran sedimentary assemblages differed primarily due to higher relative abundances of Bosmina spp. and H. glacialis within the present-day samples (the latter often absent from the preindustrial assemblages), concurrent with decreases among the daphniids (Daphnia spp.; Fig. 2). These changes among the major planktonic taxa appear indicative of increased lakewater acidity, as there is extensive field evidence of the sensitivity to low pH of members of Daphnia spp., in contrast to the relatively acid tolerant Bosmina spp. and H. glacialis (Malley and Chang 1986; Havens et al. 1993; Yan et al. 2008b). However, as mentioned above, potential decreases in lake pH alone provide an unsatisfactory explanation for the observed changes among the planktonic taxa, as these changes were not limited to lakes with a present-day pH < 6 (Fig. 4). Similarly, the present-day pH status appears to have little bearing on the changes among the prominent littoral taxa (Fig. 4). The inadequacy of variation in pH to explain community shifts along the ordination axis most strongly associated with pH (Axis 2; Fig. 3) suggests that the variation may be due to an environmental variable correlated with pH in the modern data set, specifically lakewater Ca concentration (r2 = 0.22, p = 0.004, n = 36).
The 36 study lakes were initially selected to examine cladoceran community change about a laboratory-determined Ca threshold for D. pulex (1.5 mg L−1; Ashforth and Yan 2008); however, this threshold did not extend to the broader cladoceran community (Jeziorski et al. 2012). Interestingly, Ca replaced pH as a significant explanatory variable when the lengths of the present-day Ca and pH gradients were extended (Ca from 1.0–3.1 mg L−1to 1.0–24 mg L−1and pH from 5.1–6.9 to 5.1–7.95), by expanding the data set to include the cladoceran sedimentary assemblages of 32 ecologically similar lakes from the same geographic region (DeSellas et al. 2008; Jeziorski et al. 2012).This importance of Ca to the cladoceran community composition of the broader 68 lake data set indicated that rather than capturing a threshold response, most of the lakes in the 36-lake dataset were below Ca concentrations disadvantageous to Ca-sensitive daphniid taxa due to the short length of the Ca gradient (the principal community differences between the two studies was the generally higher abundance of Daphnia spp. within the lakes analyzed in DeSellas et al. 2008). In contrast with the low relative abundance of daphniids among the 36 “top” sediments, daphniid abundance was much higher in the “bottoms” (Figs. 2, 4), suggesting the poor explanatory power of pH for the cladoceran changes in at least some of these lakes is due to Ca declines providing a buffer against aqueous pH change, while simultaneously reducing the competitiveness of Ca-sensitive taxa.
The Ca requirements and sensitivity to low ambient Ca concentration among the Cladocera have been best studied within the genus Daphnia, and the importance of Ca to daphniid regional distribution has been established in European and North American field surveys (Wærvågen et al. 2002: DeSellas et al. 2008). Due to the high Ca content of daphniids (as a proportion of their dry weight) relative to many of their competitors (Wærvågen et al. 2002; Jeziorski and Yan 2006), the daphniids are expected to be particularly vulnerable to regional Ca declines. Dramatic declines in the relative abundance of daphniid remains have been observed in the sediment record of several lakes that have recently fallen below the laboratory determined Ca threshold for D. pulex (Ashforth and Yan 2008; Jeziorski et al. 2008). The most conspicuous change among the 36 “top/bottom” samples was a dramatic reduction of daphniid relative abundance in the present-day samples (in some cases >20 %; Fig. 4). However, this response differed between the species complexes, and observed daphniid increases were typically due to replacement of the D. longispina species complex by members of the D. pulex complex (Fig. 4, 5). Although daphniid declines were common in lakes with present-day pH > 6, declines in the D. longispina complex also occurred in lakes where pH conditions were favourable for daphniids (Fig. 5). Conversely, several lakes with present-day pH > 6 (Beattie, ChubA, ChubH, Round, and Montgomery) have experienced large increases in the D. pulex complex, likely attributable to the acid-tolerant species D. catawba (Malley and Chang 1986). These inconsistencies in the daphniid response to both pH and Ca availability are due to the limited taxonomic resolution available when examining daphniid sedimentary remains.
Discrepancies between the trends in the relative abundances of daphniid sedimentary remains, and the expected responses based on present-day lake chemistry, are likely attributable to differing responses among the taxa composing the daphniid species complexes. Differences in Ca requirements among daphniids have been identified in laboratory analyses demonstrating that inter-specific Ca differences are a poor predictor of daphniid Ca sensitivity (Tan and Wang 2010). Furthermore, although field surveys of daphniid Ca optima and tolerances have found several other daphniid taxa to have similar Ca requirements to D. pulex, at least two species (D. ambigua and D. catawba) are tolerant of low Ca conditions (Cairns 2010). Therefore, from a paleolimnological perspective, at least one member of each species complex distinguishable in the daphniid sedimentary record is tolerant of low Ca conditions (D. ambigua from the D. longispina species complex and D. catawba from the D. pulex species complex). This ambiguity will hamper any paleolimnological interpretations of Ca declines based solely on changes in daphniid relative abundances, instead requiring the use of the broader cladoceran community, including taxa such as H. glacialis that may obtain a competitive advantage in low Ca conditions.
In addition to (and possibly related to) the known acid tolerance of Bosmina spp. and H. glacialis, both have a much lower Ca content than daphniids (Wærvågen et al. 2002; Jeziorski and Yan 2006), and presumably lower Ca requirements than the Ca-sensitive daphniid species. These Ca-rich and Ca-poor cladoceran taxa are often in direct competition as filter feeders with overlapping ranges of food particle size (Hessen 1985), and differ considerably in body size and other life history traits. For example, the polysaccharide mantle of H. glacialis has been identified as offering a competitive advantage over the Ca-rich daphniid carapace at Ca concentrations considerably higher than those examined in our dataset (Hessen et al. 1995). The increase in H. glacialis in many of the 36 study lakes, at the apparent expense of daphniids (Fig. 4), has occurred despite several limnological trends in south-central Ontario over recent decades that should have favoured daphniid taxa. Indeed, observed increases in daphniids have been reported in Ontario long-term monitoring programs in response to regional increases in lakewater pH (Havens et al. 1993), decreases in total phosphorus concentrations (Hall and Smol 1996), and with the spread of piscivorous bass (Yan et al. 2008b). Therefore, we suggest that the prevalence of daphniid declines (particularly of the D. longispina complex) regardless of lake pH status, concurrent with increases in the relative abundance of H. glacialis, indicate a trend of Ca limitation in many of these softwater lakes that warrants further investigation.
Distinguishing between the influences of lakewater pH and Ca on cladoceran communities remains difficult. In addition to being highly correlated, changes in these two variables have been accompanied by numerous concurrent stressors that have impacted south-central Ontario lakes over the past century, including climate warming, land-use changes, fish introductions, as well as changes in other chemical parameters such as total phosphorus and dissolved organic carbon (Yan et al. 2008b). Despite the multiple stressors involved and the limitations imposed by the “top/bottom” approach, large changes in relative abundance have occurred within the cladoceran sedimentary assemblages of the 36 study lakes since preindustrial times, which appear to be closely related to changes in pH and Ca availability. However, neither pH nor Ca alone provided an adequate explanation for the observed decreases in the relative abundance of acid-sensitive, Ca-rich daphniid species and increases of acid-tolerant, Ca-poor, non-daphniid planktonic species. Although substantial declines in the D. longispina species complex occurred in virtually every lake that had a preindustrial population, the D. pulex species complex response was mixed with substantial increases in both low pH (i.e. pH < 6) and low Ca (i.e. Ca < 1.5 mg L−1) lakes. The variable response of the D. pulex complex is likely an artifact of the taxonomic resolution available in the sediment record, and limits the use of daphniid remains in identifying lakes that have may have crossed Ca thresholds versus lakes with historically low Ca concentrations. Therefore, future paleolimnological investigations of changes in Ca availability will require understanding of the response of the broader cladoceran community to changing Ca concentrations.
We suggest that the Ca-poor species H. glacialis is a valuable indicator of changes in aqueous Ca concentrations of Precambrian Shield lakes, due to its sedimentary remains being identifiable to the species level, and its known tolerance to both low Ca and pH conditions. Increases in H. glacialis at the expense of Ca-rich daphniids provides a strong indication that Ca levels have fallen to concentrations of importance to the competitive interactions between these taxa, especially when accompanied by evidence of little pH change (e.g. information obtained from diatom-inferred pH). In addition, the littoral cladoceran community remains a large, untapped resource for Ca decline studies. Despite large differences in Ca content among some common littoral taxa (Shapiera et al. 2011), the Ca requirements of the majority of taxa found in softwater Shield lakes remain unknown, and continued laboratory and field determinations to identify other Ca-sensitive taxa will be valuable. In the meantime, attempts to tease apart the effects of Ca decline from those of acidification will benefit greatly from the adoption of a multi-proxy approach, in order to distinguish between acidified and naturally acidic low Ca lakes.
References
Alstad NEW, Skardal L, Hessen DO (1999) The effect of calcium concentration on the calcification of Daphnia magna. Limnol Oceanogr 44:2011–2017
Ashforth D, Yan ND (2008) The interactive effects of calcium concentration and temperature on the survival and reproduction of Daphnia pulex at high and low food concentrations. Limnol Oceanogr 53:420–432
Blanchfield PJ, Paterson MJ, Shearer JA, Schindler DW (2009) Johnson and Vallentyne’s legacy: 40 years of aquatic research at the experimental lakes area. Can J Fish Aquat Sci 66:1831–1836
Cairns A (2010) Field assessments and evidence of impact of calcium decline on Daphnia (Crustacea, Anomopoda) in Canadian Shield lakes. M.Sc. Thesis. York University, Canada
Cairns A, Yan ND (2009) A review of laboratory and field evidence of the influence of low ambient calcium concentrations on daphniids, gammarids and crayfish. Environ Rev 17:67–79
Cairns A, Elliott M, Yan ND, Weisz E (2006) Operationalizing CAISN project 1.V, Technical Report No. 1: lake selection. Technical report prepared for the Canadian Aquatic Invasive Species Network. Dorset Environmental Science Centre, Dorset, Ontario
Charles DF, Binford MW, Furlong ET, Hites RA, Mitchell MJ, Norton SA, Oldfield F, Paterson MJ, Smol JP, Uutala AJ, White JR, Whitehead DR, Wise RJ (1990) Paleoecological investigation of recent lake acidification in the Adirondcak Mountains, NY. J Paleolimnol 3:195–241
Chen G, Dalton C, Taylor D (2010) Cladocera as indicators of trophic state in Irish lakes. J Paleolimnol 44:465–481
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E, Plymouth
Clerk S, Hall R, Quinlan R, Smol JP (2000) Quantitative inferences of past hypolimnetic anoxia and nutrient levels from a Canadian Precambrian Shield lake. J Paleolimnol 23:319–336
Cogbill CV, Likens GE (1974) Acid precipitation in the northeastern United States. Water Resour Res 10:1133–1137
DeSellas AM, Paterson AM, Sweetman JN, Smol JP (2008) Cladocera assemblages from the surface sediments of south-central Ontario (Canada) lakes and their relationships to measured environmental variables. Hydrobiologia 600:105–119
DeSellas AM, Paterson AM, Sweetman JN, Smol JP (2011) Assessing the effects of multiple environmental stressors on zooplankton assemblages in Boreal Shield lakes since pre-industrial times. J Limnol 70:41–56
Faulkenham SE, Hall RI, Dillon PJ, Karst-Riddoch T (2003) Effects of drought-induced acidification on diatom communities in acid-sensitive Ontario lakes. Limnol Oceanogr 48:1662–1673
Frey DG (1986) Cladocera analysis. In: Berglund BE (ed) Handbook of holocene palaeoecology and palaeohydrology. Wiley, New York, pp 667–692
Glew JR (1988) A portable extruding device for close interval sectioning of unconsolidated core samples. J Paleolimnol 1:235–239
Glew JR (1989) A new trigger mechanism for sediment samplers. J Paleolimnol 2:241–243
Greenaway P (1985) Calcium balance and moulting in the Crustacea. Biol Rev 60:425–454
Hall RI, Smol JP (1996) Paleolimnological assessment of long-term water-quality changes in south-central Ontario lakes affected by cottage development and acidification. Can J Fish Aquat Sci 53:1–17
Havens KE, Yan ND, Keller W (1993) Lake acidification—effects on crustacean zooplankton populations. Environ Sci Technol 27:1621–1624
Hebert PDN (1995) The Daphnia of North America: an illustrated fauna—CD-ROM. Cyber Natural Software. University of Guelph
Hessen DO (1985) Filtering structures and particle size selection in coexisting Cladocera. Oecologia 66:368–372
Hessen DO, Faafeng BA, Andersen T (1995) Replacement of herbivore zooplankton species along gradients of ecosystem productivity and fish predation pressure. Can J Fish Aquat Sci 52:733–742
Holt CA, Yan ND, Somers KM (2003) pH 6 as the threshold to use in critical load modeling for zooplankton community change with acidification in lakes of south-central Ontario: accounting for morphometry and geography. Can J Fish Aquat Sci 60:151–158
Houle D, Ouimet R, Couture S, Gagnon C (2006) Base cation reservoirs in soil control the buffering capacity of lakes in forested catchments. Can J Fish Aquat Sci 63:471–474
Jeziorski A, Yan ND (2006) Species identity and aqueous calcium concentrations as determinants of calcium concentrations of freshwater crustacean zooplankton. Can J Fish Aquat Sci 63:1007–1013
Jeziorski A, Yan ND, Paterson AM, DeSellas AM, Turner MA, Jeffries DS, Keller B, Weeber RC, McNicol DK, Palmer ME, McIver K, Arseneau K, Ginn BK, Cumming BF, Smol JP (2008) The widespread threat of calcium decline in fresh waters. Science 322:1374–1377
Jeziorski A, Paterson AM, Smol JP (2012) Crustacean zooplankton sedimentary remains from calcium-poor lakes: complex responses to threshold concentrations. Aquat Sci 74:121–131
Kamenik C, Szeroczyńska K, Schmidt R (2007) Relationships among recent Alpine Cladocera remains and their environment: implications for climate-change studies. Hydrobiologia 594:33–46
Keller W, Dixit SS, Heneberry J (2001) Calcium declines in northeastern Ontario lakes. Can J Fish Aquat Sci 58:2011–2020
Korhola A, Rautio M (2001) Cladocera and other branchiopod crustaceans. In: Smol JP, Birks HJB, Last WM (eds) Tracking environmental change using lake sediments. Volume 4: Zoological indicators. Kluwer, Dordrecht, pp 4–41
Korosi JB, Jeziorski A, Smol JP (2011) Using morphological characters of subfossil daphniid postabdominal claws to improve taxonomic resolution within species complexes. Hydrobiologia 676:117–128
Korosi JB, Burke SM, Thienpont JR, Smol JP (2012) Anomalous rise in algal production linked to lakewater calcium decline through food web interactions. Proc R Soc B 279:1210–1217
Kurek J, Korosi JB, Jeziorski A, Smol JP (2010) Establishing reliable minimum count sizes for cladoceran subfossils sampled from lake sediments. J Paleolimnol 44:603–612
Likens GE, Driscoll CT, Buso DC (1996) Long-term effects of acid rain: response and recovery of a forest ecosystem. Science 272:244–246
Likens GE, Driscoll CT, Buso DC, Siccama TG, Johnson CE, Lovett GM, Fahey TJ, Reiners WA, Ryan DF, Martin CW, Bailey SW (1998) The biogeochemistry of calcium at Hubbard Brook. Biogeochemistry 41:89–173
Little JL, Hall RI, Quinlan R, Smol JP (2000) Past trophic status and hypolimnetic anoxia during eutrophication and remediation of Gravenhurst Bay, Ontario: comparison of diatoms, chironomids, and historical records. Can J Fish Aquat Sci 57:333–341
Malley DF, Chang PSS (1986) Increase in the abundance of cladocera at pH 5.1 in experimentally-acidified lake 223, experimental lakes area, Ontario. Water Air Soil Poll 30:629–638
Mills RB, Paterson AM, Blais JM, Lean DRS, Smol JP, Mierle G (2009) Factors influencing the achievement of steady state in mercury contamination among lakes and catchments of south-central Ontario. Can J Fish Aquat Sci 66:187–200
Molot LA, Dillon PJ (2008) Long-term trends in catchment export and lake concentrations of base cations in the Dorset study area, central Ontario. Can J Fish Aquat Sci 65:809–820
Neary BP, Dillon PJ, Munro JR, Clark BJ (1990) The acidification of Ontario lakes: an assessment of their sensitivity and current status with respect to biological damage. Ontario Ministry of the Environment, Dorset
Oksanen J, Blanchet FG, Kindt R, Legendre P, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Wagner H (2010) Vegan: community ecology package. R package version 1.17-4. http://CRAN.R-project.org/package=vegan
Paterson MJ (1994) Paleolimnological reconstruction of recent changes in assemblages of Cladocera from acidified lakes in the Adirondack Mountains (New York). J Paleolimnol 11:189–200
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org/
Shapiera M, Jeziorski A, Yan ND, Smol JP (2011) Calcium content of littoral Cladocera in three softwater lakes of the Canadian Shield. Hydrobiologia 678:77–83
Shapiera M, Jeziorski A, Paterson AM, Smol JP (2012) Cladoceran response to calcium decline and the subsequent inadvertent liming of a softwater Canadian lake. Water Air Soil Poll. doi:10.1007/s11270-011-1035-y
Shaw DM, Reilly GA, Muysson JR, Pattenden GE, Campbell FE (1967) An estimate of the chemical composition of the Canadian Precambrian Shield. Can J Earth Sci 4:829–853
Smirnov NN (1974) Fauna of the U.S.S.R., Crustacea. Vol 1: Chydoridae. Israel Program for Scientific Translations, Jerusalem
Smirnov NN (1996) Cladocera: the Chydorinae and Sayciinae (Chydoridae) of the world. SPB Academic Publishing, Amsterdam
Smol JP (2008) Pollution of lakes and rivers: a paleoenvironmental perspective, 2nd edn. Blackwell, Oxford
St. Louis VL, Breebaart L (1991) Calcium supplements in the diet of nestling tree swallows near acid sensitive lakes. The Condor 93:286–294
Stoddard JL, Jeffries DS, Lukewille A, Clair TA, Dillon PJ, Driscoll CT, Forsius M, Johnannessen M, Kahl JS, Kellogg JH, Kemp A, Mannio J, Monteith DT, Murdoch PS, Patrick S, Rebsdorf A, Skjelkvåle BL, Stainton MP, Traaen T, van Dam H, Webster KE, Wieting J, Wilander A (1999) Regional trends in aquatic recovery from acidification in North America and Europe. Nature 401:575–578
Sweetman JN, Smol JP (2006) A guide to the identification of cladoceran remains (Crustacea, Branchipoda) in Alaskan lake sediments. Arch Hydrobiol Suppl 151:353–394
Szeroczyńska K, Sarmaja-Korjonen K (2007) Atlas of subfossil Cladocera from central and northern Europe. Friends of the Lower Vistula Society, Świecie
Tan Q, Wang W (2009) The regulation of calcium in Daphnia magna reared in different calcium environments. Limnol Oceanogr 54:746–756
Tan Q, Wang W (2010) Interspecies differences in calcium content and requirement in four freshwater cladocerans explained by biokinetic parameters. Limnol Oceanogr 55:1426–1434
Wærvågen SB, Rukke NA, Hessen DO (2002) Calcium content of crustacean zooplankton and its potential role in species distribution. Freshw Biol 47:1866–1878
Watmough SA, Aherne J (2008) Estimating calcium weathering rates and future lake calcium concentrations in the Muskoka-Haliburton region of Ontario. Can J Fish Aquat Sci 65:821–833
Yan ND, Paterson AM, Somers KM, Scheider WA (2008a) An introduction to the Dorset special issue: transforming understanding of factors that regulate aquatic ecosystems on the southern Canadian Shield. Can J Fish Aquat Sci 65:781–785
Yan ND, Somers KM, Girard RE, Paterson AM, Keller B, Ramcharan CW, Rusak JA, Ingram R, Morgan GE, Gunn J (2008b) Long-term trends in zooplankton of Dorset, Ontario lakes: the probable interactive effects of changes in pH, TP, DOC and predators. Can J Fish Aquat Sci 65:862–877
Acknowledgments
This project was funded through a Natural Sciences and Engineering Research Council grant to JP Smol as well as through the Ontario Ministry of the Environment’s Best in Science Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeziorski, A., Paterson, A.M. & Smol, J.P. Changes since the onset of acid deposition among calcium-sensitive cladoceran taxa within softwater lakes of Ontario, Canada. J Paleolimnol 48, 323–337 (2012). https://doi.org/10.1007/s10933-012-9600-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10933-012-9600-y