Abstract
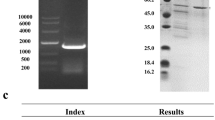
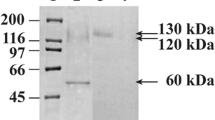
The gene encoding Lentinula edodes glucoamylase (GLA) was cloned into Saccharomyces cerevisiae, expressed constitutively and secreted in an active form. The enzyme was purified to homogeneity by (NH4)2SO4 fractionation, anion exchange and affinity chromatography. The protein had a correct N-terminal sequence of WAQSSVIDAYVAS, indicating that the signal peptide was efficiently cleaved. The recombinant enzyme was glycosylated with a 2.4% carbohydrate content. It had a pH optimum of 4.6 and a pH 3.4–6.4 stability range. The temperature optimum was 50°C with stability ≤50°C. The enzyme showed considerable loss of activity when incubated with glucose (44%), glucosamine (68%), galactose (22%), and xylose (64%). The addition of Mn++ activated the enzyme by 45%, while Li+, Zn++, Mg++, Cu+, Ca++, and EDTA had no effect. The enzyme hydrolyzed amylopectin at rates 1.5 and 8.0 times that of soluble starch and amylose, respectively. Soluble starch was hydrolyzed 16 and 29 times faster than wheat and corn starch granules, respectively, with the hydrolysis of starch granules using 10× the amount of GLA. Apparent K m and V max for soluble starch were estimated to be 3.0 mg/ml and 0.13 mg/ml/min (40°C, pH 5.3), with an apparent k cat of 2.9×105 min−1.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CHA:
-
β-cyclodextrin
- GLA:
-
glucoamylase
- YEP:
-
2% yeast extract, 1% bactopeptone
- YEPG:
-
2% yeast extract, 1% bactopeptone, 2% glycerol.
References
S. Ali Z. Hossain (1991) J. Appl. Bacteriol. 71 144–146 Occurrence Handle1:CAS:528:DyaK3MXlslOqsbw%3D
T. Ashikari N. Nakamura Y. Tanaka N. Kiuchi Y. Shibano T. Tanaka I. Amachi H. Yoshizumi (1986) Agric. Biol. Chem. 50 957–964 Occurrence Handle1:CAS:528:DyaL28XktF2ku7w%3D
L. Baracchini B. Spinelli T. M. Lourdes Polizeli Particlede H. F. Terenzi J. A. Jorge (1996) FEMS Microbiol. Lett. 138 173–177
R. S. Bhella I. Altosaar (1984) Anal. Biochem. 140 200–202 Occurrence Handle1:CAS:528:DyaL2cXks1yht70%3D Occurrence Handle10.1016/0003-2697(84)90154-4
E. Boel I. Hjort B. Svensson F. Norris K. E. Norris N. P. Fill (1984) EMBO J. 3 1097–1102 Occurrence Handle1:CAS:528:DyaL2cXksFSrsr0%3D
W. Brandani Silva Particleda R. M. Peralta (1998) Can. J. Microbiol. 44 493–497
R. Buettner R. Bode D. Birnbaum (1987) J. Basic Microbiol. 27 299–308 Occurrence Handle1:CAS:528:DyaL1cXhtVylsbw%3D
M. E. El-Zalaki M. A. Hamza (1979) Food Chem 4 203–211 Occurrence Handle1:CAS:528:DyaE1MXltlyrsL8%3D
W. M. Fogarty C. P. Benson (1983) Eur. J. Appl. Microbiol. Biotechnol. 18 271–278 Occurrence Handle1:CAS:528:DyaL2cXotFOhtg%3D%3D Occurrence Handle10.1007/BF00500491
Y. Hata K. Tsuchiya K. Kitamoto K. Gomi C. Kumagai G. Tamura S. Hara (1991) Gene 108 145–150 Occurrence Handle1:CAS:528:DyaK3sXhvVCnuw%3D%3D
T. Itoh I. Ohtsuki I. Yamashita S. Fukui (1987) J. Bacteriol. 169 4171–4176 Occurrence Handle1:CAS:528:DyaL1cXhslSmtA%3D%3D
S. Janecek J. Sevcik (1999) FEBS Lett 456 119–125 Occurrence Handle1:CAS:528:DyaK1MXlsFWhsLs%3D Occurrence Handle10.1016/S0014-5793(99)00919-9
A. Kimura J. F. Robyt (1995) Carbohydr. Res. 277 87–107 Occurrence Handle1:CAS:528:DyaK2MXpsV2jsbY%3D Occurrence Handle10.1016/0008-6215(95)00196-Z
D. E. Koshland SuffixJr. (1959) The Enzymes 1 305–346 Occurrence Handle1:CAS:528:DyaG1MXjsFWntg%3D%3D
C. C. Lee D. W. S. Wong G. H. Robertson (2001) FEMS Microbiol. Lett. 205 355–360 Occurrence Handle1:CAS:528:DC%2BD3MXpt1alsLk%3D Occurrence Handle10.1016/S0378-1097(01)00500-6
J. H. Nunberg J. H. Meade G. Cole F. C. Lawyer P. McCabe V. Schweickart R. Tal V.P. Wittman J. E. Flatgaard M. A. Innis (1984) Mol. Cell. Biol. 4 2306–2315 Occurrence Handle1:CAS:528:DyaL2MXktVymsg%3D%3D
Reilly, P. J. (2003). In: Whitaker et al. (ed.), Handbook of Food Enzymology, Marcel Dekker, New York
Robertson, G. H., and Wong, D. W. S. (2005). Enzymatic hydrolysis of Insoluble starch granules (In preparation).
J. Sambrook E. F. Fritsch T. Maniatis (1992) Molecular Cloning: A Laboratory Manual Cold Spring Harbor Laboratory Press Plainview, New York
A. Sanroman M. A. Murado J. M. Lema (1996) Appl. Biochem. Biotechnol. 59 329–336 Occurrence Handle1:CAS:528:DyaK28Xktlartr0%3D
I. Shibuya K. Gomi Y. Iimura K. Takahashi G. Tamura S. Hara (1990) Agric. Chem. Biol. 54 1905–1914 Occurrence Handle1:CAS:528:DyaK3cXmtVKis7w%3D
M. P. Silvanovich R. D. Hill (1976) Anal. Biochem. 73 430–433 Occurrence Handle1:CAS:528:DyaE28XksVaksbo%3D Occurrence Handle10.1016/0003-2697(76)90191-3
J. S. Smith D. R. Lineback (1976) Die Starke 28 243–249 Occurrence Handle1:CAS:528:DyaE28Xlt1Khs78%3D
P. J. Stone A. J. Makoff J. H. Parish A. Radford (1993) Curr. Genet. 24 205–211 Occurrence Handle1:STN:280:DyaK2c%2FjslWntg%3D%3D Occurrence Handle10.1007/BF00351793
T. Takahashi Y. Tsuchida M. Irie (1978) J. Biochem. 84 1183–1194 Occurrence Handle1:CAS:528:DyaE1MXjsFCruw%3D%3D
T. Takahashi N. Inokuchi M. Irie (1981) J. Biochem. 89 125–134 Occurrence Handle1:CAS:528:DyaL3MXpvVGhug%3D%3D
H. P. Tao D. D. Kasarda (1989) J. Exp. Bot. 40 1015–1020 Occurrence Handle1:CAS:528:DyaK3cXltVyiug%3D%3D
R. B. Wallace M. J. Johnson T. Hirose T. Miyake E. H. Kawashima K. Itakura (1981) Nucl. Acids. Res. 9 879–894 Occurrence Handle1:CAS:528:DyaL3MXhvVyitLw%3D Occurrence Handle10.1093/nar/9.4.879
O. P. Ward M. Moo-Young (1988) Biotechnol. Adv. 6 39–69 Occurrence Handle1:CAS:528:DyaL1cXksFSmsLk%3D Occurrence Handle10.1016/0734-9750(88)90573-3
D. W. S. Wong (1995) Food Enzymes: Structure and Mechanism Chapman & Hall New York
D. W. S. Wong S. B. Batt G. H. Robertson (2002a) J. Protein Chem. 20 619–623
D. W. S. Wong S. B. Batt C. C. Lee G. H. Robertson (2002b) J. Protein Chem. 21 419–425 Occurrence Handle1:CAS:528:DC%2BD38XovVClsrY%3D Occurrence Handle10.1023/A:1021186601208
D. W. S. Wong S. B. Batt C. C. Lee G. H. Robertson (2003) Protein Peptide Lett. 10 459–468 Occurrence Handle1:CAS:528:DC%2BD3sXnslylsL8%3D
J. Zhao Y. H. Chen H. S. Kwan (2000) Appl. Environ. Microbiol. 66 2531–2535 Occurrence Handle1:CAS:528:DC%2BD3cXjvFWns7k%3D
Y. Yamasaki Y. Suzuki (1978) Agric. Biol. Chem. 42 971–980 Occurrence Handle1:CAS:528:DyaE1cXksFGgtbo%3D
Y. Yamasaki Y. Suzuki J. Ozawa (1977) Agric. Biol. Chem. 41 2149–2161 Occurrence Handle1:CAS:528:DyaE1cXjtlKltA%3D%3D
I. Yamashita K. Suzuki S. Fukui (1985) J. Bacteriol. 161 567–573 Occurrence Handle1:CAS:528:DyaL2MXhs1WgtLg%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wong, D.W.S., Batt, S.B., Lee, C.C. et al. Characterization of Active Lentinula edodes Glucoamylase Expressed and Secreted by Saccharomyces cerevisiae . Protein J 24, 455–463 (2005). https://doi.org/10.1007/s10930-005-7641-z
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10930-005-7641-z