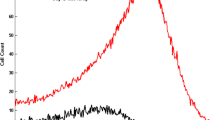
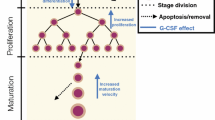
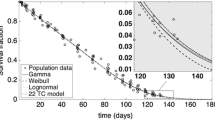
Indirect pharmacodynamic response (IDR) models were developed for agents which alter the generation of cell populations with arbitrary lifespan distributions. These models extend lifespan based IDR models introduced previously [J. Pharmacokinet. Biopharm. 27: 467, 1999] for cell populations with the same lifespan (“delta” distribution). Considered are cell populations exhibiting time-invariant lifespan distributions described by the probability density function ℓ(τ). It is assumed that cell response (R) is produced at a zero-order rate (k in(t)) and is eliminated from the population when the cell lifespan expires. The cell loss rate is calculated as k in*ℓ(t), where ‘*’ denotes the convolution operator. Therapeutic agents can stimulate or inhibit production rates according to the Hill function: 1 ± H(C(t)) where H(C(t)) contains the capacity (S max) and potency (SC 50) parameters and C(t) is a pharmacokinetic function. The production rate is k in(t)=k in· [ 1±H(C(t))]. The operational model is dR/dt = k in(t)−k in*ℓ(t) with the baseline condition R 0 = k in· T R , where T R is the mean lifespan. Single populations as well as populations with precursors were examined by simulation to establish the role of lifespan distribution parameters (mean and standard deviation) in controlling the response vs. time profile. Estimability of parameters was assessed. Numerical techniques of solving differential equations with the convolution integral were proposed. In addition, the models were applied to literature data to describe the stimulatory effects of single doses of recombinant human erythropoietin on reticulocytes in blood. The estimates of S max and SC 50 for these agents were obtained along with means and standard deviations for reticulocyte lifespan distributions. The proposed models can be used to analyze the pharmacodynamics of agents which alter natural cell production yielding parameters describing their efficacy and potency as well as means and standard deviations for cell lifespan distributions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Harker L.A., Roskos L.K., Marzec U.M., Carter R.A., Cherry J.K., Sundell B., Cheung E.N., Terry D., Sheridan W. (2000). Effects of megakaryocyte growth and development factor on platelet production, platelet lifespan, and platelet function in healthy human volunteers. Blood 95:2514–2522
Friberg L.E., Freijs A., Sandstrom M., Karlsson M.O. (2000). Semiphysiological model for the time course of leukocytes after varying schedules of 5-fluorouracil in rats. J. Pharmacol. Exp. Ther. 295:734–740
McKendrick A.G. (1926). Application of mathematics to medical problems. Proc. Edinburgh Math. Soc.44:98–130
Von Foerster H. (1959). Some remarks on changing populations. In: Stohlman F. (eds). The Kinetics of Cellular Proliferation. Grune & Stratton, New York
Belair J., Mackey M.C., Mahaffy J.M. (1995). Age-structured and two-delay models for erythropoiesis. Math. Biosci. 128:317–346
Krzyzanski W., Ramakrishnan R., Jusko W.J. (1999). Basic models for agents that alter production of natural cells. J. Pharmacokin. Biopharm. 27:467–489
Veng-Pedersen P., Chapel S., Schmidt P.R.L., Al-Huniti N.H., Cook R.T., Widness J.A. (2002). An integrated pharmacodynamic analysis of erythropoietin, reticulocyte, and hemoglobin responses in acute anemia. Pharm. Res. 19:1630–1634
Berlin N.I., Berk P.D. (1975). The biological life of the red cell. In: Surgenor DM. (eds). The Red Blood Cells, vol. II, Academic Press, New York
Paulus J.M. (1971). Measuring mean life span, mean age and variance of longevity in platelets. In: Paulus J.M. (eds). Platelet Kinetics, Radioisotopic, Cytological, Mathematical, and Clinical Aspects. North-Holland Publishing Company, Amsterdam
Dornhorst A.C. (1951). The interpretation of red cell survival curves. Blood 6:1284–1292
Bergner P.-E.E. (1962). On stochastic interpretation of cell survival curves. J. Ther. Biol. 2: 279–295
Branehog I., Ridell B., Weinfeld A. (1977). On the analysis of platelet survival curves and the calculation of platelet production and destruction. Scand. J. Haematol. 19:230–241
ICSH (1980). Recommended methods for radioisotope red-cell survival studies. Br. J. Haematol. 45:659–666
Paulus J.M. (1971). Platelet Kinetics, Radioisotopic, Cytological, Mathematical, and Clinical Aspects. North-Holland Publishing Company, Amsterdam
Murphy E.A., Francis M.E. (1969). The estimation of blood platelet survival. I. General principles of the study of cell survival. Thrombos. Diathes. Haemorrh. (Stuttg.), 22:281–295
Murphy E.A., Francis M.E. (1971). The estimation of blood platelet survival. II. The multiple hit model. Thrombos. Diathes. Haemorrh. (Stuttg.), 25:53–80
Uehlinger D.E., Gotch F.A., Sheiner L.B. (1992). A pharmacodynamic model of erythropietin therapy for uremic anemia. Clin. Pharmacol. Ther. 51:76–89
Dayneka N.L., Garg V., Jusko W.J. (1993). Comparison of four basic models of indirect pharmacodynamic responses. J. Pharmacokinet. Biopharm. 21:457–478
Veng-Pedersen P. (1977). Curve fitting and modeling in pharmacokinetics and some practical experiences with NONLIN and a new program FUNFIT. J. Pharmacokinet. Biopharm. 5:513–531
Cheung W.K., Goon B.L., Guilfoyle M.C., Wacholtz M.C. (1998). Pharmacokinetics and pharmacodynamics of recombinant human erythropoietin after single and multiple subcutaneous doses to healthy subjects. Clin. Pharmacol. Ther. 64:412–423
D’Argenio D.Z., Schumitzky A. (1979). A program package for simulation and parameter estimation in pharmacokinetics. Comp. Prog. in Biomed. 9:115–134
Cramer H. (1999). Mathematical Methods of Statistics. Princeton University Press, Princeton
Sheiner L.B., Beal S.L. (1981). Some suggestions for measuring predictive performance. J. Pharmacokin. Biopharm. 9:503–512
Sowade O., Sowade B., Brilla K., Franke W., Stephan P., Gross J., Scigalla P., Warnke H. (1997). Kinetics of reticulocyte maturity fractions and indices and iron status during therapy with epoetin beta (recombinant human erythropoietin) in cardiac surgery patients. Am. J. Hematol. 55:89–96
Akaike H. (1974). A new look at the statistical model identification. IEEE Trans Automat Control AC 19:716–723
Sun Y.-N., Jusko W.J. (1998). Transit compartments vs. gamma distribution function to model signal transduction process in pharmacodynamics. J. Pharm. Sci. 87:732–737
Krzyzanski W., Dmochowski J., Matsushima N., Jusko W.J. (2003). Assessment of intra-individual variability in estimation of parameters for basic indirect response models. AAPS PharmSci. 5(4):T2349
Ruixo J.J.P., Kimko H.C., Chow A.C., Piotrovsky V., Krzyzanski W., Jusko W.J. (2005). Population cell lifespan models for effects of drugs following indirect mechanism of action. J. Pharmacokin. Pharmacodyn. 32:767–793
Kimmel M., Grossi A., Amuasi J., Vannucchi A.M. (1990). Non-parametric analysis of platelet lifespan. Cell Tissue Kinet. 23:191–202
Ramakrishnan R., Cheung W.K., Wacholtz M.C., Minton N., Jusko W.J. (2004). Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after single and multiple doses in healthy volunteers. J. Clin. Pharmacol. 44:991–1002
Krzyzanski W., Jusko W.J., Wacholtz M.C., Minton N., Cheung W.K. (2005). Pharmacokinetic and pharmacodynamic modeling of recombinant human erythropoietin after multiple subcutaneous doses in healthy subjects. Eur. J. Pharm. Sci. 26:295–306
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported in part by Grant No. GM 57980 from the National Institute of General Medical Sciences, National Institutes of Health.
Rights and permissions
About this article
Cite this article
Krzyzanski, W., Woo, S. & Jusko, W.J. Pharmacodynamic Models for Agents that Alter Production of Natural Cells with Various Distributions of Lifespans. J Pharmacokinet Pharmacodyn 33, 125–166 (2006). https://doi.org/10.1007/s10928-006-9007-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-006-9007-3