Abstract
For group-living animals the choice of whether to join aggregations or initiate their own is influenced by potential benefits such as group protection and reduced energetic expenditure, as well as costs such as competition for food and mates. The bark beetle Ips grandicollis is an invasive pest species that colonises recently felled timber in Australian pine (Pinus spp.) plantations. Male beetles initiate colonies by burrowing under the bark of trees and emitting an aggregation pheromone which attracts conspecifics, including a harem of females with whom they mate. We predicted that males that initiated colonies, or who arrived early, would have larger harems than later arrivals (due to decreased competition for females). However, we found the opposite effect with early-arriving males actually associated with fewer females than later arriving males, although this may have resulted from some females leaving harems as they get older. We conclude that pioneering does not improve male likelihood of attracting females in Ips grandicollis, at least initially, but it may provide advantages for offspring when competing for food during development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Group living individuals are faced with numerous decisions, including the question of which groups to join and when. When aggregations are associated with procuring a resource (habitat, mates, food), the costs and benefits of joining a group may vary depending on the order in which an individual arrives at the group. Initiating an aggregation (pioneering) may allow individuals to secure better quality territories and/or procure an increased proportion of the resource (Bensch and Hasselquist 1991; Candolin and Voigt 2003; Smith and Moore 2005), but may incur costs such as greater energetic expenditure, and mortality or injury from prey defences (Heinsohn and Packer 1995; Pekar et al. 2005). Individuals that join established groups (‘joiners’) may avoid these costs, but could also experience higher competition and/or a decreased share of the resource (Mangel 1990; Giraldeau and Beauchamp 1999).
Many bark beetle species (subfamily: Scolytinae) live and feed in aggregations beneath the bark of coniferous trees (Wood 1982). In many species aggregation is necessary to overcome tree defences and the fitness of the individuals is contingent on the size of the aggregation (Raffa and Berryman 1983). In the genus Dendroctonus, individuals that initiate aggregations or arrive early in the colonisation have higher mortality and lower reproductive success because they have to deal with the toxic effects of tree defences at their strongest (Pureswaran et al. 2006; Latty and Reid 2009). Bark beetles of the genus Ips, however, typically attack non-living Pinus material (such as freshly windblown trees or cut log billets), which should remove this cost associated with pioneering. Indeed, early arrival may even be beneficial because of reduced competition for mates and food resources (Raffa 2001).
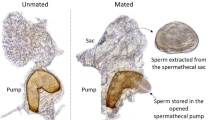
Ips bark beetles have a harem polygynous mating system (Kirkendall 1983). Adult males search for fallen logs, burrowing into the phloem and carving a nuptial chamber where they begin releasing an aggregation pheromone (Wood 1982). This attracts other males to the log, and up to 8 females to the specific boreholes occupied by the males (Kirkendall 1983; Schlyter and Zhang 1996; Latty et al. 2009). Each female carves her own tunnel radiating outward from the central nuptial chamber (Fig. 1). The female then lays eggs in individual niches along the sides of the tunnel. Harem size is related to reproductive success such that males with larger harems produce more offspring than males with small harems (Robertson 1998).
We examined the effect of male arrival order on their subsequent harem size in a South Australian population of the invasive eastern five-spined bark beetle Ips grandicollis. Early arriving males may face less competition for females; we therefore tested the hypothesis that these males will have larger harems. We also examined whether time of arrival and harem size were linked to male body size.
Methods and Materials
We examined natural colonisation by Ips grandicollis bark beetles of log billets in a 39 year-old Pinus radiata plantation at Wirrabara State Forest, South Australia (138° 16′ E, 33° 1′ S) approximately 250 km north of Adelaide. The experiments were carried out in summer 2007, from the 13th to 23rd February. Daily maximum temperatures ranged from 32–41°C and weather conditions were fine and sunny during the course of the work.
We cut 50 cm-long log billets from freshly felled Pinus radiata trees. Log billet diameter ranged from 9 to 23 cm. The billets were put together in 20 piles of three (=60 billets), in order to increase the likelihood of colonisation by beetles. Each pile was spaced 10 m apart along a transect.
We monitored colonisation by examining the billets every day for fresh Ips grandicollis boreholes (easily identified by the presence of orange frass on the outside of the log). New boreholes were marked by placing a date-labelled flat-topped push-pin into the log next to the hole. Some logs are colonised earlier than others, so the push-pin data allowed us to record not only the arrival order of each male to the log, but also the age of the borehole when its internal characteristics (see below) were measured. We measured arrival order as the day on which the borehole appeared in each log subsequent to the first borehole appearing (i.e. the first boreholes to appear in each log were assigned a day of arrival = 1, the boreholes that appeared the next day had a day of arrival = 2, and so on).
As colonisation continues, and the phloem is progressively consumed and degraded, the patterns of galleries become difficult to distinguish making accurate measurement of male harem size increasingly problematic. Hence, we concentrated on the effects of arrival order on reproductive success in the early stages of colonisation. Whilst this restricts our ability to discuss ultimate effects on survival and success of offspring, it does allow more accurate assessment of the initial costs or benefits of pioneering. Consequently, after 8–10 days, logs were carefully stripped of bark with a chisel, to uncover the nuptial chamber and galleries associated with each borehole. In South Australia, it takes male Ips grandicollis 4 days to attract their full complement of females (Morgan 1967). Therefore, we restricted our analysis to males who had been in the logs for at least 5 days. We noted the number of beetles observed under each borehole and the number of galleries. This provided us with two measures of harem size—one based on number of galleries and one based on actual number of females observed. Every borehole was assumed to have one male beetle, with the remaining beetles assumed to be female (Kirkendall 1983).
A subset of males that were physically undamaged during the removal of the bark were collected in labelled plastic vials and stored in the freezer for later body size measurements. Males were generally recognisable in the field as being the one beetle in the gallery system that was not at the far end of a gallery. However, in cases where this was not clear we collected all the beetles from each gallery system, and sexed them under a microscope in the lab using the criteria of Lanier and Cameron (1969). The body length and body width (across the base of the thorax) of each individual (male or female) was measured in the lab using callipers. Because Ips bark beetles are essentially cylindrical in shape we used length and width measures to estimate body volume:
Relationships between arrival order and both measures of harem size were examined using hierarchical mixed modelling to account for the non-independence and hierarchical structure of the data. Three trees were used as a source of the logs in the experiment. However, source tree did not account for significant variation in response variables so was not included in statistical models.
For analysis, arrival order was calculated in the context of the log pile (days from first borehole in any of the three logs in each pile) because females would most likely detect male aggregation pheromones based on the signal coming from a log pile rather than either individual logs within each pile or the entire population. Nevertheless, we also calculated male arrival order relative to individual log and the whole population to check our results. Analysis was performed using MLwiN 2.02 (Rasbash et al. 2004).
Results
In total we collected data from 95 harems across 43 logs in 19 log-piles. Mean (± s.e.) harem size was 3.57 ± 0.16 galleries or 2.96 ± 0.15 females. The analysis of arrival order at all three levels (i.e. relative to log, log pile and population) produced qualitatively the same results. Although trends were stronger (and in the case of the relationship with number of females more highly significant) in the analyses at the log and population levels, we present only the results from the analysis at the log pile level (see justification in the Methods and Materials above).
In 58 harems (61%), the number of galleries equalled the number of females, however in 35 harems (37%), there were fewer females than there were galleries. We found a significant relationship between arrival order and the proportion of females to gallery number (b = −0.36 ± 0.15 s.e.; P = 0.02) indicating that females were more likely to be ‘missing’ from galleries the longer the harem had been established. This proportion of females to galleries was not significantly related to the number of galleries in the harem (b = −0.13 ± 0.09 s.e.; P = 0.16).
The relationship between arrival order and harem size differed according to the measure of harem size used (Fig. 2). We did not find a significant association between arrival order and number of galleries (b = 0.14 ± 0.17 s.e.,; P = 0.43). However, there were fewer females in the harems of males that arrived earliest (b = 0.38 ± 0.16 s.e.; P = 0.02).
Model predictions of harem size in relation to the arrival order of males within log stack. Harem size is shown as both number of galleries and number of females. Male arrival order within each log stack is expressed relative to the arrival day of the first male to colonise the log stack. Predicted effect estimates are shown with 95% confidence interval. The relationship with number of females is statistically significant (p = 0.02), and is non-significant with number of galleries (p = 0.43) Only males arriving within 5 days of the first male were included (n = 95 males colonising 19 log stacks)
There was no significant relationship between male body size (volume) and arrival order (b = −0.13 ± 0.13 s.e.; P = 0.29), nor was there any association between male body size and harem size (b = 0.11 ± 0.12 s.e.; P = 0.39).
Discussion
Contrary to our prediction, early arriving males did not attract more females than those that arrived later in the aggregation. Indeed, using number of associated females as our measure of harem size, we found that early arriving males were associated with fewer females than were late arriving males. However, no relationship between arrival order and harem size was apparent when using number of galleries as the measure. The discrepancy in results using our two measures of harem size stems from the fact that 37% of harems in our study had fewer females than galleries, with the proportion of females to galleries declining in older harems. Therefore the relationship between fewer females and early arrival by males is likely explained by the increase in ‘missing’ females from older boreholes. This loss of females may have one of two explanations. The first is that individual females abandon harems. Re-emergence is a relatively common phenomenon in bark beetles, and is often related to declines in resource quality or increases in density and competition (Kirkendall 1983; Byers 1989; Anderbrant 1989). However, female densities in logs were low in our study (mean = 0.70 ± 0.09 s.e. females per dm2), and there were not proportionately more females missing from larger harems which makes it seem unlikely that competition is driving re-emergence here. A second possibility explaining the ‘missing’ females is not that they are missing but that individual females carve out more than one gallery, a phenomenon which is also known to occur in Ips species (Reid 1999). If this is the case, then our results suggest that early males may actually be losing out in terms of attracting females. However, because we cannot rule out either possibility it is most appropriate to conclude that there is no benefit to males of arriving early, at least in terms of harem size.
As with previous work on harem size in Ips (Schlyter and Zhang 1996; Latty et al. 2009) we found no evidence of an effect of male size on harem size, contrary to what is typically found in vertebrate harem polygynous systems (e.g. Webster 1992; Lindenfors et al. 2002) where larger body size enables males to defend their harems. The lack of a relationship between body size and arrival order likewise suggests that the latter is unrelated to male quality.
Early arrival may, however, have benefits that we could not examine. Most obvious of these is the main theoretical benefit of pioneering: that it reduces the amount of intraspecific larval competition for the offspring of pioneers. Numerous studies of Ips have demonstrated negative density effects on larval growth and survival (e.g. Zhang et al. 1992; Lawson et al. 1995; Robins and Reid 1997; Steed and Wagner 2004; Sallé and Raffa 2007). Our experiments only considered the initial stages of the attack, and not the ultimate outcome in terms of offspring survival. It is therefore possible that the offspring of early arriving males experience lower levels of larval competition, and as a result, have higher offspring survivorship. This could result in early arriving males ultimately having a greater number of offspring than late arriving males.
In summary, we have shown that pioneering behaviour in Ips grandicollis males is not beneficial to them in terms of the number of females they can attract. Further research into the effects of arrival order on larval survivorship would be needed to ascertain whether arrival order is ultimately an important predictor of male reproductive success in Ips grandicollis.
References
Anderbrant O (1989) Reemergence and second brood in the bark beetle Ips typographus. Holarct Ecol 12:494–500
Bensch S, Hasselquist D (1991) Territory infidelity in the polygynous great reed warbler Acrocephalus arudinaceus: the effect of variation in territory attractiveness. J Anim Ecol 60:857–871
Byers JA (1989) Behavioral mechanisms involved in reducing competition in bark beetles. Holarct Ecol 12:466–476
Candolin U, Voigt HR (2003) Size-dependent selection on arrival times in sticklebacks: why small males arrive first. Evolution 57:862–871
Giraldeau LA, Beauchamp G (1999) Food exploitation: searching for the optimal joining policy. Trends Ecol Evol 14:102–106
Heinsohn R, Packer C (1995) Complex cooperative strategies in group-territorial African lions. Science 269:1260–1262
Kirkendall LR (1983) The evolution of mating systems in bark and ambrosia beetles (Coleoptera: Scolytidae and Platypodidae). Zool J Linn Soc 77:293–352
Lanier GN, Cameron EA (1969) Secondary sexual characteristics in the North American species of the genus Ips (Coleoptera: Scolytidae). Can Entomol 101:862–870
Latty TM, Reid ML (2009) First in line or first in time? Effects of settlement order and arrival date on reproduction of a group-living beetle, Dendroctonus ponderosae. J Anim Ecol 78:549–555
Latty TM, Magrath MJL, Symonds MRE (2009) Harem size and oviposition behaviour in a polygynous bark beetle. Ecol Entomol 34:562–568
Lawson SA, Furuta K, Katagiri K (1995) Effect of tree host and beetle density on reproduction and survival of Ips typographus japonicus Niijima (Col., Scolytidae), in Hokkaido, Japan. J Appl Entomol 119:383–390
Lindenfors P, Tullberg BS, Biuw M (2002) Phylogenetic analyses of sexual selection and sexual size dimorphism in pinnipeds. Behav Ecol Sociobiol 52:188–193
Mangel M (1990) Resource divisibility, predation and group formation. Anim Behav 39:1163–1172
Morgan FD (1967) Ips grandicollis in South Australia. Austral For 31:137–155
Pekar S, Hruskova M, Lubin Y (2005) Can solitary spiders (Araneae) cooperate in prey capture? J Anim Ecol 74:63–70
Pureswaran DS, Sullivan BT, Ayres MP (2006) Fitness consequences of pheromone production and host selection strategies in a tree-killing bark beetle (Coleoptera: Curculionidae: Scolytinae). Oecologia 148:720–728
Raffa KF (2001) Mixed messages across multiple trophic levels: the ecology of bark beetle chemical communication systems. Chemoecology 11:49–65
Raffa KF, Berryman AA (1983) The role of host plant resistance in the colonization behavior and ecology of bark beetles (Coleoptera: Scolytidae). Ecol Monogr 53:27–49
Rasbash J, Steele F, Browne W, Prosser B (2004) A user’s guide to MLwiN version 2.0. Institute of Education, London
Reid ML (1999) Monogamy in the bark beetle Ips latidens: ecological correlates of an unusual mating system. Ecol Entomol 24:89–94
Robertson IC (1998) Paternal care enhances male reproductive success in pine engraver beetles. Anim Behav 56:595–602
Robins GL, Reid ML (1997) Effects of density on the reproductive success of pine engravers—is aggregation in dead trees beneficial? Ecol Entomol 22:329–334
Sallé A, Raffa KF (2007) Interactions among intraspecific competition, emergence patterns, and host selection behaviour in Ips pini (Coleoptera: Scolytinae). Ecol Entomol 32:162–171
Schlyter F, Zhang QH (1996) Testing avian polygyny hypotheses in insects: harem size distribution and female egg gallery spacing in three Ips bark beetles. Oikos 76:57–69
Smith RJ, Moore FR (2005) Arrival timing and seasonal reproductive performance in a long-distance migratory landbird. Behav Ecol Sociobiol 57:231–239
Steed BF, Wagner MR (2004) Importance of log size on host selection and reproductive success from Ips pini (Coleoptera: Scolytidae) in ponderosa pine slash of northern Arizona and western Montana. J Econ Entomol 97:436–450
Webster MS (1992) Sexual dimorphism, mating system and body size in new world blackbirds (Icterinae). Evolution 46:1621–1641
Wood SL (1982) The bark and ambrosia beetles of North and Central America (Coleoptera: Scolytidae), a taxonomic monograph. Great Basin Nat Mem 6:1–1359
Zhang QH, Byers JA, Schlyter F (1992) Optimal attack density in the larch bark beetle, Ips cembrae (Coleoptera: Scolytidae). J Appl Ecol 29:672–678
Acknowledgements
Forestry SA allowed us access to the Wirrabara plantations. We thank Charlma Phillips (Forestry SA) for making the arrangements for us to carry out the fieldwork, and the forester Bruno Turrini for logistical assistance and his generous help and enthusiasm. Emile van Lieshout and Kath McNamara provided comments on an earlier draft of this manuscript. This research was funded by a Discovery Project Grant from the Australian Research Council, a Canadian Natural Sciences and Research Council Graduate Scholarship, and travel grants from the Department of Zoology, University of Melbourne and the Western Forest Genetics Association (Canada and USA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Symonds, M.R.E., Magrath, M.J.L. & Latty, T.M. Reproductive Consequences of Male Arrival Order in the Bark Beetle, Ips grandicollis . J Insect Behav 25, 401–407 (2012). https://doi.org/10.1007/s10905-011-9308-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-011-9308-2