Abstract
The Allee effect is a positive relationship between any component of individual fitness and the density of conspecifics. Theoretical models predict that monogamy, where males and females are constrained from having multiple mates, is more susceptible to the Allee effect. In most termite species, including Reticulitermes spp., found new colonies by monogamous pairs (i.e., primary king and queen); however, little is known about the effects of alate density and flight timing on pairing and colony foundation success. In this study, a positive relationship between alate density and pairing success was observed in the subterranean termite, Reticulitermes speratus. Upon release of dealates (individuals after shedding wings) every 3 days for 9 days at 10, 20, 40, or 80 pairs/m2/day in a semi-natural field, no pairs were observed for the 10 pairs/m2/day treatment after 3 months. However, 7.5, 13.8, and 18.1% of dealates formed pairs in the 20, 40, and 80 pairs/m2/day groups, respectively. Most pairs (78.7%) comprised dealates released simultaneously, and 17.3 and 4% comprised dealates released 3 and 6 days apart, respectively. R. speratus also preferred brown rotten pine for colony foundations to white rotten oak. This study provides important new insights into the density effects and nest-site preference for termite colony foundation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Individuals of many species benefit from the presence of conspecifics. The inverse density dependence at low density, which is broadly referred to as the Allee effect (1931), has gained considerable attention in population and community ecology (Courchamp et al. 1999), conservation biology (Lande 1988; Groom 1998; Stephens and Sutherland 1999), and invasion biology (Liebhold and Bascompte 2003; Taylor and Hastings 2005; Tobin et al. 2011; Wang et al. 2011; Luque 2013). There are a lot of mechanisms that can cause the inverse density dependence in plants and animals including increased risk of predation (Andrewartha and Birch 1954; Clark 1974), inbreeding depression, loss of heterozygosity (Frankel and Soulé 1981; Charlesworth and Charlesworth 1987; Lamont et al. 1993), and the lack of cooperation (Courchamp et al. 1999; Stephens and Sutherland 1999; Stephens et al. 1999). The most notable cause of the Allee effect is the difficulty among sexually reproducing species in finding mates in low-density populations (Andrewartha and Birch 1954; Dennis 1989; McCarthy 1997; Kuussaari et al. 1998; Courchamp et al. 1999; Boukal and Berec 2002).
Allee effects play an important role in the evolution of mating systems and reproductive phenology (Stephens et al. 1999; Calabrese and Fagan 2004), and theoretical models predict that monogamy is more susceptible to the Allee effect, where males are constrained from having more than one mate (Legendre et al. 1999; Bessa-Gomes et al. 2003). In termites, monogamous pairs of primary reproductives (primary king and queen) found colonies and form lifetime partnerships (Nutting 1969; Nalepa and Jones 1991; Shellman-Reeve 1997). The phenomenon of a general synchronized flight from colonies has been reported for multiple termite species (Weesner 1960). Such synchronized flights reduce predation due to predator satiation (Nutting 1979), increase the likelihood of mate finding (Thorne 1983), and increase breeding between members of different colonies (Wynne-Edwards 1962; Luykx 1986). In the life cycle of termites, alates are at greatest risk of predation during the period from swarming to colony foundation, as they are away from nest-associated soldier defenses (Sheppe 1970; Deligne et al. 1981). After landing, males and females are also exposed to high predation risks from ants, resulting in limited mate-searching periods on the ground. Therefore, termite alates are highly susceptible to the Allee effect. However, little is known about this phenomenon in respect to termite mating and colony foundation.
Reticulitermes speratus Kolbe is the most common and widespread termite species in Japan. In Western Japan, R. speratus alates swarm during the day in May, and although the complete dispersal term lasts 1 week in each specific area, a massive synchronized flight from multiple colonies occurs on sunny days following rain (Matsuura 2002, 2006). An alate breaks off its wings upon alighting, and the resulting dealate travels randomly until it encounters a partner with whom it forms a tandem running pair. The pair then relocates to a suitable nest site, with the male following the female (Matsuura and Nishida 2001). Both heterosexual and homosexual tandems are formed (Matsuura et al. 2002b, 2004); female–female pairs are able to found new colonies by parthenogenesis (Matsuura and Nishida 2001; Matsuura et al. 2002a) and male–male pairs promote survivorship and provide the chance to replace a male in an incipient colony after colony fusion (Mizumoto et al. 2016).
In this study, we investigated the effects of flight timing and alate density on pairing and colony foundation success in R. speratus by a release–recapture experiment in a semi-natural field. The effects of alate density on nest-site preference were also assessed by providing different types of rotten wood at different depths within each experimental section.
Materials and methods
Termite collection
The nest wood of four mature R. speratus colonies (A, B, C, and D) were collected from late April to early May 2014 from pine forests at Hieidaira, Takaragaike, Okamotoguchi and Daigo, in Kyoto, Japan. The colonies were maintained primarily at 20 °C, and the temperature was subsequently raised to 25 °C to control alate maturation and flight timing. Following the emergence of alates from the wood, they were separated by sex and maintained in Petri dishes containing moist filter paper. Wings were removed at the basal structure to mimic dealation (wing shedding) behavior (Matsuura and Nishida 2002). Alates were isolated from the wood just before experiment and used in the experiment within 8 h.
Colony foundation experiment under semi-natural conditions
In an open-air greenhouse (3.0 × 7.2 m, plastic roof and fine mesh walls), four 1.0 × 1.0-m experimental sections (I, II, III, and IV) were established consisting of three layers of humus and soil, which was covered by litter (Fig. 1). Section bottoms and surrounding walls were covered by polyvinyl sheets, and the seams were sealed with waterproof tape. Adhesives were also applied to the outer wall to prevent ants and other predatory arthropods from entering the field. Thirty-six wood pieces consisting of brown rotten pine Pinus densiflora (100 × 100 × 30 mm), and thick (60-mm diameter × 150 mm), medium-sized (30-mm diameter × 150 mm), and thin branches (15-mm diameter × 150 mm) of white rotten oak Quercus serrate were arranged within each of the three layers. The wood species were arranged in a 6 × 6 lattice in each of the three layers in patterns based on random numbers generated by R software (Fig. 1).
Diagram and photograph of the experimental setup. a A diagram showing the setup from above (left), accompanied by a photo image (right). Density sections (I, II, III, and IV) are arranged in order; 10, 20, 40, and 80 pairs of alates, respectively, were released at each time. b Vertical arrangement of the experimental field, consisting of three layers of humus and soil covered by litter. The upper and middle layers consisted of humus, and the bottom layer of soil. Thirty-six wood samples were arranged in each layer. The seams of a polyvinyl sheet and the plastic board were sealed using waterproof tape to prevent termites from escaping the field. Similarly, to prevent ants and other predatory arthropods from entering the field, adhesive sealed the outer wall. c The arrangement of the four types of wood within each layer. Pieces of brown rotten pine and three size classes of white rotten oak were arranged in a 6 × 6 lattice in each of the three layers following random numbers generated by the R program
To investigate the relationship between release timing and pairing probability, dealates were released from four different colonies at 3-day intervals. Sib and non-sib dealates showed no difference in pair formation or in their time to excavation (Matsuura and Nishida 2001). On the day of the first release, 10, 20, 40, and 80 pairs of dealates from colony A were released into sections I, II, III, and IV, respectively. Male and female dealates were scattered throughout the entire surface of each section. Three days later, 10, 20, 40, and 80 pairs of dealates from colony B were released in the same manner. The dealates from colonies C and D were also released at 3-day intervals in the same manner. In total, 40 males and 40 females were released into section I (10-pair section), 80 males and 80 females into section II (20-pair section), 160 males and 160 females into section III (40-pair section), and 320 males and 320 females into section IV (80-pair section). One month following the last release, all males and females were collected from each wood pieces (Table S1). Males and females were extracted from the same nest so that all members of an incipient colony were preserved together in a 2.0-ml tube and then used for colony identification by microsatellite analysis.
Microsatellite genotyping for colony identification
DNA was extracted from ten alates from each original colony and from all males and females isolated from the wood pieces. Multiple microsatellite loci were analyzed, including Rf21-1, Rf24-2, Rf6-1 (Vargo 2000), and Rs15 (Dronnet et al. 2004), and compared among samples from the original (Table S2) and incipient colonies (Table S3). One primer from each pair was fluorescent-labeled for detection purposes. Termite DNA was extracted from alate legs using a modified Chelex extraction protocol (Walsh et al. 1991). Polymerase chain reaction (PCR) amplifications were performed in 15.25-μl reactions containing 0.3 μl of 25 mM MgCl2, 0.3 μl of 10 mM dNTPs, 1.5 μl of 10X PCR Buffer, 0.2 μl of 5 U/μl Taq DNA Polymerase (Qiagen, Valencia, CA, USA), 5 pmol of each of the multiplex primers, and 1 μl of the DNA template. Amplification consisted of initial denaturation at 95 °C for 5 min, followed by 35 cycles consisting of denaturation at 98 °C for 12 s, annealing at 60 °C for 30 s, and an extension at 72 °C for 1 min. Following amplification, 1 μl of product was combined with 0.5 μl of GS-600 (LIZ) size standard and 10 μl of HI-DI formamide. Samples were denatured at 95 °C and snap-cooled on ice for 5 min. Sample detection was performed using an Applied BioSystems 3500 Genetic Analyzer. Raw data were analyzed using GeneMapper 5.0 software (Applied Biosystems, Inc., Foster City, CA, USA).
Data analysis
To investigate whether alate density had an effect on pairing success (binary variable: success or failure), a generalized linear model (GLM) with a binomial distribution and logit link function was performed using STATISTICA 10 (StatSoft Inc., Tulsa, OK, USA).
In all models, colony of origin, sex and alate density were treated as fixed factors and the effects of the fixed factors were tested using the likelihood ratio test. Similarly, alate density effect on survival (binary variable: dead or alive) was tested using a GLM with binomial distribution and logit link function. To examine the effects of release timing on pairing probability, the proportion of the combinations of monogamous pairs formed by the alates released on the same day (0-day gap) was assessed and compared to those with 3-, 6-, and 9-day gaps. The proportions were then compared to the null hypothesis, i.e., that the proportion of pairs formed would be the same as that expected from random pairings. A three-way ANOVA was used to investigate nest-site preference, and the effects of alate density, layer, and wood type on the number of males and females extracted from each piece of wood were examined. For these analyses, the density section was included as a categorical variable, and the distribution of individuals among wood types was compared using χ2 tests.
Results
Allee effect on pairing success and survivorship
Pairing efficiency was low at low alate densities. In the 10-pairs/m2/day section, two single males and two single females were recovered from the wood, and no successful pairs were observed (Fig. 2). Alate density was positively correlated with pairing success (general linear model (GLM): estimate ± SE = 0.016 ± 0.0030, likelihood ratio χ 2 = 18.93, P < 0.0001; Fig. 3a). The colony of origin also had a significant effect on pairing success (likelihood ratio χ 2 = 20.83, P < 0.0001). There was no significant difference in pairing success between the sexes (likelihood ratio χ 2 = 0.021, P = 0.88). The survival rates of alates were also significantly affected by alate density (estimate ± SE = 0.016 ± 0.0038, likelihood ratio χ 2 = 29.63, P < 0.0001; Fig. 3b) and colony (likelihood ratio χ 2 = 33.50, P < 0.001), whereas sex had no significant effect (likelihood ratio χ 2 = 0.91, P = 0.34).
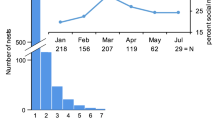
Composition of surviving units extracted from the wood. The number of units observed in each density section is shown in parenthesis. F, single female; M, single male; MF, male–female pair; MM, male–male pair; FF, female–female pair; MMF, a female with two males; MFF, a male with two females; MMM, three males
Comparison of pairing success rates (a) and survival rates (b) among the sections receiving 10, 20, 40, and 80 pairs/m2/day. The means of four original colonies released at 3-day intervals were shown. Bars denote standard errors. The alate density had significant effect on pairing success (likelihood ratio χ 2 = 18.93, P < 0.0001) and on survival rates (likelihood ratio χ 2 = 29.63, P < 0.0001)
Flight timing and pairing
The Rf21-1, Rf24-2, Rf6-1 (Vargo 2000), and Rs15 (Dronnet et al. 2004) microsatellites were allele-rich, containing four to six alleles each (Table S2). Given that the first three loci exhibited sufficient diversity for source colony identification, Rf21-1, Rf24-2, and Rf6-1 were used for profiling (Table S3).
The timing of alate release significantly affected pairing probability, with pair combinations being significantly different from expectations of random pairing regardless of release timing (χ 2 = 127.74, df = 3, P < 0.0001). Among 75 monogamous pairs recovered from wood, 59 (78.7%) contained males and females released on the same day (0-day gap), 13 (17.3%) showed the 3-day gap, 3 (4.0%) showed the 6-day gap, and no pairs showed the 9-day gap (Fig. 4). The density effects on pairing success and alate survival rate remained highly significant even when the data of 3- and 6-day-gap pairs were excluded from the analyses (pairing success: likelihood ratio χ 2 = 11.05, P < 0.001; survival rate: likelihood ratio χ 2 = 21.94, P < 0.0001).
Nest-site preference
Males and females were found in the upper and middle nest layers, but none was found in the bottom layer (Fig. 5). The number of individuals extracted from each piece of wood was significantly influenced by alate density, layer, and wood type (density: F 3,384 = 10.29, P < 0.0001; layer: F 2,384 = 10.53, P < 0.0001; wood type: F 3,384 = 4.06, P < 0.01; Fig. 5). An association between density and layer was also observed (F 6,384 = 6.51, P < 0.0001 by three-way ANOVA), indicating that layer preference differed according to density. The observed distribution among wood types differed from a random distribution (χ 2 = 103.02, P < 0.0001), and also differed according to wood size (χ 2 = 97.70, P < 0.0001). In general, termites preferred rotten brown pine for colony foundation to rotten white oak.
Discussion
A positive density-dependent relationship was observed between the number of alates released in each section and the rate of pairing, indicating the Allee effect. Surprisingly, however, no pairs were observed in the 10-pair/m2/day density section, where 10 males and 10 females were released at four different times (i.e., 40 males and 40 females in total) in a 1.0 × 1.0-m area. These data suggest that termites have low pairing efficiencies at low densities. Since this experiment was performed without predators such as ants and spiders, the Allee effect observed may be conservative.
The Allee effect could select for specific termite behaviors during swarming and colony foundation, as it is known to influence dispersal in other animals (Serrano et al. 2005; Fowler 2009; Travis and Dytham 2002). The intercolonial synchrony of swarming would increase alate density, and thus should limit the Allee effect. In addition, synchronized flight would also increase the chance for mating with an alate from a different colony of origin and therefore increase the chance for outbreeding. This study revealed that most pairs were formed by males and females released on the same day (Fig. 4). This result suggests that a colony which disperses alates at a different timing from the synchronous flight of the other colonies would have a reduced fitness. Therefore, the Allee effect may be a selective force behind the maintenance of synchronous flight. The Allee effect may also reduce selection for mate choice because alates have little or no opportunity to choose a mate at low density (Møller and Legendre 2001), although it is known that males prefer larger females in laboratory experiments (Matsuura and Nishida 2001).
The finding that termites are susceptible to the Allee effect following dispersal enhances our understanding of termite invasion. Although alate flight is considered limited, alates aided by the wind can travel long distances, such as between islands, and be introduced to new regions (Nutting 1969). In such situations, however, it is unlikely that alates will successfully locate partners. Our empirical data support earlier studies, suggesting termite invasions are mediated by human transportation of infested timber and that the capacity to generate new colonies from colony fragments facilitates the spread of subterranean termites (Dronnet et al. 2005; Vargo and Husseneder 2009). Indeed, the introduced populations of R. flavipes in Europe exhibit a particular colony breeding structure that is characterized by hundreds of inbreeding neotenic reproductives (Perdereau et al. 2015). In introduced populations or peripheral areas, neotenic-headed colonies are more common than primary-headed colonies of Reticulitermes (Vieau 1996) and Coptotermes (Lenz and Barrett 1982). Strong Allee effects in conjunction with low population-level genetic diversity must diminish the advantage of alate production during invasion. Therefore, as suggested by theoretical models (Travis and Dytham 2002) and empirical studies (Dronnet et al. 2005; Vargo and Husseneder 2009; Lenz and Barrett 1982; Husseneder et al. 2012), termites should exhibit reduced dispersal rates and lower rates of spread during invasion and early range expansion, while human transportation would mediate range expansion. Once established, swarm densities increase rapidly with the additional mature colonies founded in a given location, which would contribute to weaken the Allee effect.
In addition to monogamous pairs, other types of surviving units were observed, including single males, single females, male–male pairs, female–female pairs, a female with two males, a male with two females, and three males (Fig. 2). Although female-only units can reproduce by parthenogenesis (Matsuura and Nishida 2001), single males and male-only units cannot reproduce. However, fusion of the units may provide an opportunity to reproduce, as multiple units were observed in individual wood pieces (Table S1). For example, single males and single females may pair following wood excavation, or fusion of male–male and female–female pairs may also lead to monogamous relationships. Among the 75 monogamous pairs, three pairs showed the 6-day gap of release timing (Fig. 4). Thus, it is unlikely that those males and females stayed in a specific test site for 6 days. These data suggest that pair formation is rare following excavation, but not impossible.
In this study, major alate predators, including ants, spiders, and lizards, were eliminated. Thus, the Allee effect observed may be conservative. Additional studies are needed to evaluate the influence of predators on the Alee effect. Additionally, the sex ratio of alates released into each section was balanced throughout the study. Controversy remains over the prediction that monogamy is more susceptible to the Allee effect than other mating systems are. Although some theoretical models predict that monogamy is more susceptible to the Allee effect (Legendre et al. 1999; Bessa-Gomes et al. 2003), one recent study claimed that susceptibility to the Allee effect is not a general property of a given mating system but depends largely on the sex ratio (Engen et al. 2003; Bessa-Gomes et al. 2004; Lee et al. 2011). The alate sex ratio varies among Reticulitermes spp., such that species with an asexual queen succession (AQS) system (Matsuura et al. 2009; Vargo et al. 2012; Luchetti et al. 2013) have female-biased sex ratios, whereas non-AQS species have equal sex ratios (Kobayashi et al. 2013). Therefore, susceptibility to the Alee effect differs between AQS and non-AQS species in Reticulitermes termites. It must also be noted that sex ratios involves other unidentified factors in other termite taxa. In conclusion, termites provide an ideal system to further understand the relationship between the Allee effect and sex ratio.
References
Allee W (1931) Animal aggregations: a study in a general sociology. University of Chicago Press, Chicago
Andrewartha H, Birch L (1954) The distribution and abundance of animals. University of Chicago Press, Chicago
Bessa-Gomes C, Danek-Gontard M, Cassey P, Møller AP, Legendre S, Clobert J (2003) Mating behaviour influences extinction risk: insights from demographic modelling and comparative analysis of avian extinction risk. Ann Zool Fenn 40:231–245
Bessa-Gomes C, Legendre S, Clobert J (2004) Allee effects, mating systems and the extinction risk in populations with two sexes. Ecol Lett 7:802–812. doi:10.1111/j.1461-0248.2004.00632.x
Boukal DS, Berec L (2002) Single-species models of the Allee effect: extinction boundaries, sex ratios and mate encounters. J Theor Biol 218:375–394. doi:10.1006/jtbi.2002.3084
Calabrese J, Fagan W (2004) Lost in time, lonely, and single: reproductive asynchrony and the Allee effect. Am Nat 164:25–37. doi:10.1086/421443
Charlesworth D, Charlesworth B (1987) Inbreeding depression and its evolutionary consequences. Annu Rev Ecol Syst 18:237–268. doi:10.1146/annurev.es.18.110187.001321
Clark CW (1974) Possible effects of schooling on the dynamics of exploited fish populations. J Conseil 36:7–14
Courchamp F, Clutton-Brock T, Grenfell B (1999) Inverse density dependence and the Allee effect. Trends Ecol Evol 14:405–410. doi:10.1016/S0169-5347(99)01683-3
Deligne J, Quennedey A, Blum MS (1981) The enemies and defense mechanisms of termites. In: Hermann HR (ed) Social insects, vol 2. Academic Press, New York, pp 1–76
Dennis B (1989) Allee effects: population growth, critical density, and the chance of extinction. Nat Resour Model 3:481–538
Dronnet S, Bagneres AG, Juba TR, Vargo EL (2004) Polymorphic microsatellite loci in the European subterranean termite, Reticulitermes santonensis Feytaud. Mol Ecol Notes 4:127–129
Dronnet S, Chapuisat M, Vargo EL, Caroline L, Bagnères A-G (2005) Genetic analysis of the breeding system of an invasive subterranean termite, Reticulitermes santonensis, in urban and natural habitats. Mol Ecol 14:1311–1320. doi:10.1111/j.1365-294X.2005.02508.x
Engen S, Lande R, Sæther B-E (2003) Demographic stochasticity and Allee effect in populations with two sexes. Ecology 84:2378–2386. doi:10.1890/02-0123
Fowler MS (2009) Density dependent dispersal decisions and the Allee effect. Oikos 118:604–614. doi:10.1111/j.1600-0706.2008.17321.x
Frankel O, Soulé ME (1981) Conservation and evolution. Cambridge University Press, Cambridge
Groom MJ (1998) Allee effects limit population viability of an annual plant. Am Nat 151:487–496. doi:10.1086/286135
Husseneder C, Simms DM, Delatte JR, Wang C, Grace JK, Vargo EL (2012) Genetic diversity and colony breeding structure in native and introduced ranges of the Formosan subterranean termite, Coptotermes formosanus. Biol Invasions 14:419–437. doi:10.1007/s10530-011-0087-7
Kobayashi K, Hasegawa E, Yamamoto Y, Kawatsu K, Vargo EL, Yoshimura J, Matsuura K (2013) Sex ratio biases in termites provide evidence for kin selection. Nat Commun 4:2048. doi:10.1038/ncomms3048
Kuussaari M, Saccheri I, Camara M, Hanski I (1998) Allee effect and population dynamics in the Glanville fritillary butterfly. Oikos 384–392. doi:10.2307/3546980
Lamont BB, Klinkhamer PG, Witkowski E (1993) Population fragmentation may reduce fertility to zero in Banksia goodii—a demonstration of the Allee effect. Oecologia 94:446–450. doi:10.1007/bf00317122
Lande R (1988) Genetics and demography in biological conservation. Science 241:1455–1460
Lee AM, Sæther B-E, Engen S (2011) Demographic stochasticity, Allee effects, and extinction: the influence of mating system and sex ratio. Am Nat 177:301–313. doi:10.1086/658344
Legendre S, Clobert J, Møller AP, Sorci G (1999) Demographic stochasticity and social mating system in the process of extinction of small populations: the case of passerines introduced to New Zealand. Am Nat 153:449–463. doi:10.1086/303195
Lenz M, Barrett R (1982) Neotenic formation in field colonies of Coptotermes lacteus (Froggatt) in Australia, with comments on the roles of neotenics in the genus Coptotermes (Isoptera: Rhinotermitidae). Sociobiology 7:47–59
Liebhold A, Bascompte J (2003) The Allee effect, stochastic dynamics and the eradication of alien species. Ecol Lett 6:133–140. doi:10.1046/j.1461-0248.2003.00405.x
Luchetti A, Velonà A, Mueller M, Mantovani B (2013) Breeding systems and reproductive strategies in Italian Reticulitermes colonies (Isoptera: Rhinotermitidae). Insect Soc 60:203–211. doi:10.1007/s00040-013-0284-8
Luque GM, Giraud T, Courchamp F (2013) Allee effects in ants. J Anim Ecol 82:956–965. doi:10.1111/1365-2656.12091
Luykx P (1986) Termite colony dynamics as revealed by the sex- and caste-ratios of whole colonies of Incisitermes schwarzi banks (Isoptera: Kalotermitidae). Insect Soc 33:221–248. doi:10.1007/bf02224243
Matsuura K (2002) Sociobiology of the termite Reticulitermes speratus. Doctoral thesis, Kyoto University, Kyoto
Matsuura K (2006) Early emergence of males in the termite Reticulitermes speratus (Isoptera: Rhinotermitidae): protandry as a side effect of sexual size dimorphism. Ann Entomol Soc Am 99:625–628. doi:10.1603/0013-8746(2006)99[625:EEOMIT]2.0.CO;2
Matsuura K, Nishida T (2001) Comparison of colony foundation success between sexual pairs and female asexual units in the termite Reticulitermes speratus (Isoptera: Rhinotermitidae). Popul Ecol 43:119–124. doi:10.1007/pl00012022
Matsuura K, Nishida T (2002) Mechanism, induction factors, and adaptive significance of dealation in the subterranean termite Reticulitermes speratus (Isoptera, Rhinotermitidae). Insect Soc 49:241–244. doi:10.1007/s00040-002-8308-9
Matsuura K, Fujimoto M, Goka K, Nishida T (2002a) Cooperative colony foundation by termite female pairs: altruism for survivorship in incipient colonies. Anim Behav 64:167–173. doi:10.1006/anbe.2002.3062
Matsuura K, Kuno E, Nishida T (2002b) Homosexual tandem running as selfish herd in Reticulitermes speratus: novel antipredatory behavior in termites. J Theor Biol 214:63–70. doi:10.1006/jtbi.2001.2447
Matsuura K, Fujimoto M, Goka K (2004) Sexual and asexual colony foundation and the mechanism of facultative parthenogenesis in the termite Reticulitermes speratus (Isoptera, Rhinotermitidae). Insect Soc 51:325–332. doi:10.1007/s00040-004-0746-0
Matsuura K, Vargo EL, Kawatsu K, Labadie PE, Nakano H, Yashiro T, Tsuji K (2009) Queen succession through asexual reproduction in termites. Science 323:1687. doi:10.1126/science.1169702
McCarthy M (1997) The Allee effect, finding mates and theoretical models. Ecol Model 103:99–102. doi:10.1016/S0304-3800(97)00104-X
Mizumoto N, Yashiro T, Matsuura K (2016) Male same-sex pairing as an adaptive strategy for future reproduction in termites. Anim Behav 119:179–187. doi:10.1016/j.anbehav.2016.07.007
Møller AP, Legendre S (2001) Allee effect, sexual selection and demographic stochasticity. Oikos 92:27–34. doi:10.1034/j.1600-0706.2001.920104.x
Nalepa CA, Jones SC (1991) Evolution of monogamy in termites. Biol Rev 66:83–97. doi:10.1111/j.1469-185X.1991.tb01136.x
Nutting W (1969) Flight and colony foundation. In: Krishna K, Weesner F (eds) Biology of termites. Academic Press, New York, pp 233–282
Nutting W (1979) Termite flight periods: strategies for predator avoidance? Sociobiology 4:141–151
Perdereau E, Bagnères AG, Vargo EL, Baudouin G, Xu Y, Labadie P, Dupont S, Dedeine F (2015) Relationship between invasion success and colony breeding structure in a subterranean termite. Mol Ecol 24:2125–2142. doi:10.1111/mec.13094
Serrano D, Oro D, Ursua E, Tella JL (2005) Colony size selection determines adult survival and dispersal preferences: Allee effects in a colonial bird. Am Nat 166:E22–E31. doi:10.1086/431255
Shellman-Reeve JS (1997) Advantages of biparental care in the wood-dwelling termite, Zootermopsis nevadensis. Anim Behav 54:163–170. doi:10.1006/anbe.1996.0412
Sheppe W (1970) Invertebrate predation on termites of the African savanna. Insect Soc 17:205–218
Stephens PA, Sutherland WJ (1999) Consequences of the Allee effect for behaviour, ecology and conservation. Trends Ecol Evol 14:401–405. doi:10.1016/S0169-5347(99)01684-5
Stephens PA, Sutherland WJ, Freckleton RP (1999) What is the Allee effect? Oikos 87:185–190. doi:10.2307/3547011
Taylor CM, Hastings A (2005) Allee effects in biological invasions. Ecol Lett 8:895–908. doi:10.1111/j.1461-0248.2005.00787.x
Thorne B (1983) Alate production and sex ratio in colonies of the Neotropical termite Nasutitermes corniger (Isoptera; Termitidae). Oecologia 58:103–109. doi:10.1007/BF00384548
Tobin PC, Berec L, Liebhold AM (2011) Exploiting Allee effects for managing biological invasions. Ecol Lett 14:615–624. doi:10.1111/j.1461-0248.2011.01614.x
Travis JM, Dytham C (2002) Dispersal evolution during invasions. Evol Ecol Res 4:1119–1129
Vargo EL (2000) Polymorphism at trinucleotide microsatellite loci in the subterranean termite Reticulitermes flavipes. Mol Ecol 9:817–820. doi:10.1046/j.1365-294x.2000.00915.x
Vargo EL, Husseneder C (2009) Biology of subterranean termites: insights from molecular studies of Reticulitermes and Coptotermes. Annu Rev Entomol 54:379–403. doi:10.1146/annurev.ento.54.110807.090443
Vargo EL, Labadie PE, Matsuura K (2012) Asexual queen succession in the subterranean termite Reticulitermes virginicus. Proc Biol Sci 279:813–819. doi:10.1098/rspb.2011.1030
Vieau F (1996) Seasonal variation in caste proportions of the termite Reticulitermes santonensis Feytaud (Isoptera: Rhinotermitidae) in an environment of western France. Ann Soc Ent Fr (NS) 32:207–216
Walsh PS, Metzger DA, Higuchi R (1991) Chelex 100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. Biotechniques 10:506–513
Wang W, Liu H, Li Z, Guo Z, Yang Y (2011) Invasion dynamics of epidemic with the Allee effect. Biosystems 105:25–33. doi:10.1016/j.biosystems.2011.03.001
Weesner FM (1960) Evolution and biology of the termites. Annu Rev Entomol 5:153–170
Wynne-Edwards VC (1962) Animal dispersion in relation to social behaviour. Oliver & Boyd, Edinburgh
Acknowledgements
We thank K. Kobayashi, Y. Namba, R. Tanaka, C. Iwata, T. Nozaki, K. Takeuchi, Y. Miyaguni for their assistance with conducting the experiment; and N. Mizumoto and S. Dobata for valuable discussion and their assistance with data analysis. We also thank two anonymous reviewers for valuable comments on the manuscript. This study was supported by the Japan Society for the Promotion of Science (Kiban Kenkyu S: 25221206 to KM).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kusaka, A., Matsuura, K. Allee effect in termite colony formation: influence of alate density and flight timing on pairing success and survivorship. Insect. Soc. 65, 17–24 (2018). https://doi.org/10.1007/s00040-017-0580-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-017-0580-9