Abstract
A coumarin-based fluorescent chemosensor 1 for Zn2+ was designed and synthesized. Compound 1 exhibits lower background fluorescence due to intramolecular photoinduced electron transfer. However, upon mixing with Zn2+ in 30% (v/v) aqueous ethanol, a “turn-on” fluorescence emission is observed. The fluorescence emission increases linearly with Zn2+ concentration in the range 0.5–10 μmol L−1 with a detection limit of 0.29 μmol L−1. No remarkable emission enhancement was, however, observed for other metal ions. The proposed chemosensor was applied to the determination of Zn2+ in water samples with satisfactory results.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Zinc(II) ion, the second most abundant transition-metal ion in the human body, plays critical roles in many biological activities, such as gene expression, apoptosis, enzyme regulation and neurotransmission [1–3]. In addition, elevated levels of Zn2+ ions in water could become an environmental problem [4]. Therefore, the design and development of efficient fluorescent chemosensors selective to Zn2+ are of considerable interest. Although several fluorescence-based chemosensors for Zn2+ have been developed, some of them have shortcomings for practical application such as cross-sensitivities toward other metal ions, susceptibility to pH, difficult synthesis and non-aqueous media requirement [5–14]. It is therefore necessary to develop new chemosensors for Zn2+ with high selectivity and sensitivity at physiological pH.
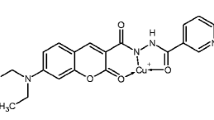
Coumarin dyes are important class of fluorescent compounds and have been widely used to construct a variety of fluorescent probes and chemosensors owing to their excellent photophysical properties [15–24]. Though coumarin-derived chemosensor for Zn2+ have been reported by Xi et al. [25], it can work only at CH3CN solution and is unpractical to real sample analysis. Herein, we report a new coumarin Schiff-based fluorescence chemosensor 1, which exhibits a high selectivity toward Zn2+ ion by utilizing a chelation-enhanced fluorescence (CHEF) mechanism (Scheme 1). The sensor 1 was developed by incorporating a semicarbazide at the 8 position of coumarin ring by a “C=N” bond, which would quench the excited state emission of the coumarin fluorophore via photoinduced electron transfer (PET). Because Zn(II) has a closed-shell d10 electronic configuration, we anticipated that its coordination would alleviate PET and provide fluorescence “turn-on.” As we expected, upon mixing with Zn2+ in aqueous ethanol solution, the fluorescence of 1 is largely enhanced. The fluorescent increase is linear with Zn2+ concentration in the range 0.5–10 μmol L−1 with a detection limit of 0.29 μmol L−1. The fluorescence sensing process is proved to be reversible. The proposed binding mechanism of Zn2+ with 1 was studied and proved in a 1:1 binding mode.
Experimental
Reagents
Unless otherwise stated, all reagents were purchased from commercial suppliers and used without further purification. Twice-distilled water was used throughout the experiments. 7-Hydroxycoumarin-3-carboxylic acid ethyl ester (2) was obtained from J&K CHEMICAL LTD. The solution of 1 (0.5 mmol L−1) was prepared by dissolving 16.2 mg of 1 in 100 mL DMF. Zn2+ solution (1.0 mmol L−1) was prepared by dissolving appropriate amount of ZnSO4 with water. Tris–HCl buffer solutions of different pH were prepared by using proper amount of Tris and HCl under adjustment by a pH meter.
Synthesis of 1
Compound 1 was synthesized in two steps as shown in Scheme 2.
Ethyl 7-hydroxy-8-formyl-coumarin-3-carboxylate (3)
To a stirred TFA (20 mL) was added compound 2 (3.28 g, 14 mmol) and hexamine (2.94 g, 21 mmol). This solution was refluxed for 20 h. Then, 40 mL of water was added to the solution, and the mixture was warmed at 60°C whilst stirring for another 30 min. Upon cooling on ice a yellow solid was precipitated out from the solution, which was collected by filtration and washed several times with water to give the desired product 3 as a yellow solid (2.5 g, 68% yield). IR (KBr, cm−1): 3431, 3045, 1742, 1700, 1659, 1595, 1480, 1445, 1304, 1236; MS (MALDI-TOF): m/z = 262.20 (M+); M+ calculated 262.22; 1H NMR (400 MHz, DMSO-d 6): 1.28–1.31 (t, 3H, CH3), 4.25–4.30 (q, 2H, CH2), 7.01 (q, 1H, ArH), 8.06 (t, 1H, ArH), 8.75 (s,1H, ArH), 10.40 (s, 1H, CHO), 12.10–12.50(1H, OH);
Compound 1
Compound 1 was synthesized by the condensation reaction between compound 3 and semicarbazide. Briefly, semicarbazide hydrochloride (52 mg, 0.46 mmol) was dissolved in 5 mL of ethanol, and the solution was neutralized to pH 7.0 with 1.0 mol L−1 of NaOH. The above solution was then mixed with the ethanol solution of compound 2 (60 mg, 0.23 mmol) and refluxed for 4 h. After cooling, the resulting solution was added dropwise into saturated brine. The precipitate was filtered off, washed with water several times and dried to give the desired product as light-yellow solid (30 mg, 43% yield). IR (KBr, cm−1): 3442, 3363, 3265, 1758, 1680, 1615, 1590, 1534, 1461, 1370, 1324, 1299, 1239, 1208, 1092, 1036, 797, 696. MS (MALDI-TOF): m/z = 320.59 (M+H)+; M+ calculated, 320.28;. 1H NMR (400 MHz, DMSO-d 6): 1.29–1.32 (t, 3H, CH3), 4.25–4.31 (q, 2H, CH2), 6.48 (s, 2H, NH2), 6.98 (q, 1H, ArH), 7.80 (t, 1H, ArH), 8.51 (s, 1H, ArH), 8.73 (s,1H, NH), 10.60 (s, 1H, C=NH), 11.99(1H, OH).
Apparatus
Absorption spectra were determined with a Shimadzu UV-1700 spectrophometer (Tokyo, Japan). The fluorescence spectra and relative fluorescence intensity were performed on a Sanco CRT-970 spectrofluorimeter (Shanghai, China) with a 10 mm quartz cuvette. Unless specific noted, the excitation and emission wavelength bandpasses were both set at 10 nm. The pH was measured with a Model pHs-3B meter (Shanghai, China). Infrared (IR) were taken in KBr pellets on a Bruker Tensor 27 FTIR spectrometer. Mass spectra were obtained with AXIMA-CFR plus MALDI-TOF Mass Spectrophotometer. 1H NMR spectra were recorded on an INOVA-400 spectrometer (Varian Unity), using tetramethylsilane(TMS)as internal standard. All of the measurements were operated at room temperature at about 298 K.
Procedure
Typically, to a 10-mL test tube containing 3.0 mL ethanol, 1.0 mL of Tris-HCl buffer (0.1 mol L−1, pH = 7.4) and 0.1 mL of compound 1 (1.0 mmol L−1), an appropriate aliquot of Zn2+ was added and the reaction mixture was diluted to 10.0 mL with water. The resulting solution was allowed to stand at room temperature for 5 min, and then the absorption and emission spectra were recorded. For fluorescence intensity measurements, the excitation and emission wavelengths were at 400 nm and 457 nm, respectively.
Results and discussion
Spectral characteristics
It is well-known that coumarin dyes are strongly fluorescent. In this study, we formylated compound 2 at its 8 position via Duff reaction [26, 27] and found that its fluorescence was slightly decreased and the emission maximum shifted from 446 to 452 nm. However, upon coupling with semicarbazide, 3’s fluorescence was dramatically quenched (Fig. 1). This quenching is due to a PET process from the electron-donating semicarbazide moiety to the coumarin ring. Experimentally, it was observed that the fluorescence of compound 1 is largely enhanced with the addition of Zn2+, differentiated from other metal ions, indicating that it is a selective chemosensor for Zn2+.
As shown in Fig. 2, compound 1 exhibits an absorption maximum at 416 nm, which undergoes a hypsochromic shift to 407 nm upon Zn2+ binding. Furthermore, to gain more insight into the chemosensing properties of 1 toward Zn2+, a fluorescence titration with Zn2+ in 30% aqueous ethanol solution were carried out. In the absence of Zn2+, the fluorescence emission maximum of 1 is centered at about 483 nm. Upon the gradual addition of the Zn2+ from 0 to 10 equiv, the λ em undergoes a blue shift to 457 nm, and the fluorescence emission at 457 nm increases with increasing Zn2+ concentration, which forms the base of fluorimetric determination of Zn2+ (Fig. 3). The blockage of PET process from the semicarbazide amine to coumarin ring induced by Zn2+ coordination is responsible for Zn2+-induced emission enhancement. Additionally, similar results are obtained when divalent zinc salts, such as ZnSO4, ZnCl2, Zn(NO3)2 and Zn(Ac)2 are used (Fig. S1). Therefore, it appears that counter anions have no perceptible effects on the fluorescence response of the proposed chemosensor.
Binding of 1 with Zn2+
Before the application of 1 in the detection of Zn2+, the binding interaction of 1 with Zn2+ was investigated. First, EDTA, a very strong cation chelating agent, was added to the solution of 1-Zn2+, and it was observed that the fluorescence intensity almost decreased to its original intensity of free ligand (Fig. S2), indicating that the coordination of 1 with Zn2+ is chemically reversible. Next, Job’s method for the emission changes at 457 nm was applied to study the binding stoichiometry of 1 and Zn2+ (Fig. 4) [28–30]. The data of Job’s plots using a total concentration of 20 μmol L−1 1 and Zn2+ in 30% (v/v) aqueous ethanol solution exhibited a maximum fluorescence signal when the molecular fraction of compound 1 was close to 50%, indicating a 1: 1 complexation between Zn2+ and 1 (Scheme 1). The binding constant of 1 to Zn2+ was calculated by nonlinear least-squares analysis (Fig. S3) to be (9.15 ± 3.50) × 105 L mol−1 with a good correlation (r = 0.9979) [31, 32].
Selectivity of 1
The selectivity of 1 toward Zn2+ was investigated by treating 1 with other ions such as Cu2+, Ni2+, Co2+, Fe2+, Mn2+, K+, Na+, Ba2+, Li+, Cd2+, Hg2+, Cr3+, Al3+, Pb2+, Ag+, Mg2+, and Ca2+ in 30% aqueous ethanol buffered at pH 7.4 (Fig. 5). The addition of Mn2+, K+, Na+, Ba2+, Li+, Hg2+, Cr3+, Al3+, Pb2+, Ag+, Mg2+ and Ca2+ gave no significant changes to the fluorescence spectra of 1, while Cu2+, Co2+, Ni2+, Fe2+ caused a dramatic fluorescence quenching. The observation that the fluorescence emission of 1 is slightly enhanced with the addition of Cd2+, which is due to Cd2+ and Zn2+ are in the same group of the periodic table and they have many similar properties. By contrast, the addition of Zn2+ to the solution of 1 results in a drastic fluorescence emission change. The fluorescence peak at 483 nm blue-shifts to 457 nm with a marked fluorescence intensity enhancement.
Furthermore, the competition experiments were carried by adding Zn2+ to the solution of 1 in the presence of Cu2+, Ni2+, Co2+, Mn2+, K+, Na+, Li+, Cd2+, Al3+, Pb2+, Ag+, Mg2+, and Ca2+, respectively, and the results were shown in Fig. 6. It was observed that the presence of some metal ions such as K+, Na+, Al3+, Ag+, Mn2+, Cd2+, Pb2+, Li+, Ca2+, Mg2+ does not interfere with the Zn2+-enhanced fluorescence. When 1 equiv of Zn2+ was added to the solution of 1 in the presence of 1 equiv of Cu2+, Co2+, Ni2+, however, no fluorescence response was observed, possibly because the formed complex between these metal ions and 1 is too stable to be replaced by Zn2+, which indicates that the signaling of Zn2+ by 1 is significantly interfered by these metal ions.
Fluorescence response of 1 (10 μmol L−1) in the presence of Zn2+ (10 μmol L−1) and / or various metal ions in ethanol–water (30/70, v/v) at pH 7.4 (Tris–HCl, 10 mmol L−1). All metal ions were evaluated at one equiv to Zn2+ except Na+, K+, which were used at 1 mmol L−1. White bars represent the addition of the appropriate metal ion to the solution of 1. Black bars represent the subsequent addition of Zn2+ to the solution
Effect of pH
The effect of pH on the fluorescence response of 1 to Zn2+ was studied in 10 mmol L−1 Tris-HCl buffer solution (30% water/ethanol, v/v), and the results were shown in Fig. 7. It can be observed that the fluorescence emission of 1-Zn(II) and free 1 are both increased with increasing pH from 3.6 to 7.4. However, when the pH is higher than 7.4, the fluorescence of 1-Zn2+ is decreased but the free 1 remained stable. In order to obtain a higher signal-to-noise, Tris-HCl buffer (pH 7.4, 10 mmol L−1) was employed for Zn2+ assay throughout the experiment.
Analytical characteristics of 1 for Zn2+ ion
Under the selected optimized conditions, the fluorescence enhancement (ΔF) is linear with Zn2+ concentration (C) in the range 0.5–10.0 µmol L−1 (Fig. S4). The linear regression equation was determined to be ΔF = 1.44 × 107C [mol L−1] + 9.92 (n = 7, r = 0.9989). According to IUPAC, the detection limit was determined from three times the standard deviation of the blank signal (3σ) as 0.29 µmol L−1. The relative standard deviation (R.S.D) for eight repeated measurements of 5.0 μmol L−1 of Zn2+ is 2.3%.
Preliminary application of 1 for Zn2+ assay in water samples
Synthesized water (by adding Zn2+ and other metal ions to tap water) and Rongshi Drinking Mineralized Water obtained from the local supermarket were analyzed by the proposed chemosensor under optimized conditions (Table 1). From the above results, it can be seen that 1 can measure the concentration of Zn2+ in water samples with good recovery results. Therefore, compound 1 can be employed for Zn2+ assay in a water setting.
Conclusion
In summary, a coumarin-based chemosensor 1 was designed and synthesized. Upon treatment with Zn2+ in 30% aqueous ethanol buffer at 7.4, the sensor showed a “turn-on” fluorescence response and the emission blue-shifted from 483 nm to 457 nm. The significantly enhanced fluorescence of 1 with the addition of Zn2+ is due to the formation of a 1:1 complex 1-Zn2+ in which the PET from a semicarbazide amine to coumarin fluorophore is hindered. The high selectivity of 1 for Zn2+ is evidenced as its exceptional fluorescence enhancement from various metal ions. Moreover, compound 1 can be prepared from cheap starting materials with easy preparation in organic synthesis, which is important for practical application.
References
Berg JM, Shi Y (1996) The galvanization of biology: a growing appreciation for the roles of zinc. Science 271:1081–1085. doi:10.1126/science.271.5252.1081
Vallee BL, Falchuk KH (1993) The biochemical basis of zinc physiology. Physiol Rev 72:79–118
Weiss JH, Sensi SL, Koh JY (2000) Zn2+ a novel ionic mediator of neural injury in brain disease. Trends Pharmacol Sci 21:395–401. doi:10.1016/S0165-6147(00)01541-8
Rensing C, Maier RM (2003) Issues underlying use of biosensors to measure metal bioavailability. Ecotoxicol Environ Saf 56:140–147. doi:10.1016/S0147-6513(03)00057-5
Fahrni CJ, Halloran TVO (1999) Aqueous coordination chemistry of quinoline-based fluorescence probes for the biological chemistry of zinc. J Am Chem Soc 121:11448–11458. doi:10.1021/ja992709f
Zhang X-A, Hayes D, Smith SJ, Friedle S, Lippard SJ (2008) New strategy for quantifying biological zinc by a modified zinpyr fluorescence sensor. J Am Chem Soc 130:15788–15789. doi:10.1021/ja807156b
Shiraishi Y, Ichimura C, Hirai T (2007) A quinoline-polyamine conjugate as a fluorescent chemosensor for quantitative detection of Zn(II) in water. Tetrahedron Lett 48:7769–7773. doi:10.1016/j.tetlet.2007.09.032
Kiyose K, Kojima H, Urano Y, Nagano T (2006) Development of a ratiometric fluorescent zinc ion probe in near-infrared region, based on Tricarbocyanine Chromophore. J Am Chem Soc 128:6548–6549. doi:10.1021/ja060399c
Joshi BP, Cho W-M, Kim J, Yoon J, Lee KH (2007) Design synthesis, and evaluation of peptidyl fluorescent probe for Zn2+ in aqueous solution. Bioorg Med Chem Lett 17:6425–6429. doi:10.1016/j.bmcl.2007.10.008
Xu ZC, Kim G-H, Han SJ, Jou MJ, Lee C, Shin I, Yoon J (2009) An NBD-based colorimetric and fluorescent chemosensor for Zn2+ and its use for detection of intracellular zinc ions. Tetrahedron 65:2307–2312. doi:10.1016/j.tet.2009.01.035
Wang H-H, Gan Q, Wang X-J, Xue L, Liu S-H, Jiang H (2007) A water-soluble, small molecular fluorescent sensor with femtomolar sensitivity for zinc ion. Org Lett 9(24):4995–4998. doi:10.1021/ol702192m
Liu ZP, Zhang CL, Li YL, Wu ZY, Qian F, Yang XL, He WJ, Gao X, Guo ZJ (2009) A Zn2+ fluorescent sensor derived from 2-(Pyridin-2-yl)benzoimidazole with ratiometric sensing potential. Org Lett 11(4):795–798
Lin WY, Feng JB, Yuan L, Tan W (2009) 4-(2, 2-Bipyridine-5-yl)benzaldehyde as a novel fluorescent reagent for Zn2+ with emission in the near-infrared region. Sens Actuat B 135:512–515. doi:10.1016/j.snb.2008.09.036
Li ZF, Xiang Y, Tong AJ (2008) Ratiometric chemosensor for fluorescent determination of Zn2+ in aqueous ethanol. Anal Chim Acta 619:75–80. doi:10.1016/j.aca.2007.12.037
Yao J, Dou W, Qin W, Liu W (2009) A new coumarin-based chemosensor for Fe3+ in water. Inorg Chem Commun 12:116–118. doi:10.1016/j.inoche.2008.11.012
Lin WY, Yuan L, Feng JB (2008) A dual-channel fluorescence-enhanced sensor for aluminum ions based on photoinduced electron transfer and excimer formation. Eur J Org Chem 3821–3825. doi:10.1002/ejoc.200800327
Lin WY, Yuan L, Feng JB, Cao XW (2008) A fluorescence-enhanced chemodosimeter for Fe3+ based on hydrolysis of bis(coumarinyl) schiff base. Eur J Org Chem 2689–2692. doi:10.1002/ejoc.200800077
Roussakis E, Pergantis SA, Katerinopoulos HE (2008) Coumarin-based ratiometric fluorescent indicators with high specificity for lead ions. Chem Commun (Camb.) 6221–6223. doi:10.1039/b811569a
Du LP, Li MY, Zheng SL, Wang BH (2009) Rational design of a fluorescent hydrogen peroxide probe based on the umbelliferone fluorophore. Tetrahedron Lett 49:3045–3048. doi:10.1016/j.tetlet.2008.03.063
Lin WY, Yuan L, Cao XW (2008) A rational approach to emission ratio enhancement of chemodosimeters via regulation of intramolecular charge transfer. Tetrahedron Lett 49:6585–6588. doi:10.1016/j.tetlet.2008.09.029
Jung HS, Kwon PS, Lee JW, Kim JI, Hong CS, Kim JW, Yan S, Lee JY, Lee JH, Joo T, Kim JS (2009) Coumarin-derived Cu2+-selective fluorescence sensor: synthesis, mechanisms, and applications in living cells. J Am Chem Soc 131:2008–2012. doi:10.1021/ja808611d
Ray D, Bharadwaj PK (2008) A coumarin-derived fluorescence probe selective for magnesium. Inorg Chem 47:2252–2254. doi:10.1021/ic702388z
Wu J-S, Liu W-M, Zhuang X-Q, Wang F, Wang P-F, Tao S-L, Zhang X-H, Wu S-K, Lee S-T (2007) Fluorescence turn on of coumarin derivatives by metal cations: a new signaling mechanism based on C=N isomerization. Org Lett 9(1):33–36. doi:10.1021/ol062518z
Lin WY, Yuan L, Cao ZM, Feng YM, Long LL (2009) A sensitive and selective fluorescent thiol probe in water based on the conjugate 1, 4-addition of thiols to a, b-unsaturated ketones. Chem Eur J 15:5096–5103. doi:10.1002/chem.200802751
Li H-Y, Gao S, Xi Z (2009) A colorimetric and “turn-on” fluorescent chemosensor for Zn(II) based on coumarin Shiff-base derivative. Inorg Chem Commun 12:300–303. doi:10.1016/j.inoche.2009.01.013
Ferguson LN (1946) The synthesis of aromatic aldehydes. Chem Rev 38(2):227–254. doi:10.1021/cr60120a002
Duff JC, Bills EJ (1932) Reactions between hexamethylenetetramine and phenolic compounds. Part I. A new method for the preparation of 3- and 5-aldehydosalicylic acids. J Chem Soc 1987–1988. doi:10.1039/jr9320001987
Vosburgh WC, Cooper GR (1941) Complex ions I. The identification of complex ions in solution by spectrophotometric measurements. J Am Chem Soc 63:437–442. doi:10.1021/ja01847a025
Xiang Y, Tong AJ, Jin PY, Ju Y (2006) New fluorescent rhodamine hydrazone chemosensor for Cu(II) with high selectivity and sensitivity. Org Lett 8:2863–2866. doi:10.1021/ol0610340
Zeng L, Miller EW, Pralle A, Isacoff EY, Chang CJ (2006) A selective turn-on fluorescent sensor for imaging copper in living cells. J Am Chem Soc 128:10–11. doi:10.1021/ja055064u
Bourson J, Pouget J, Valeur B (1993) Ion-responsive fluorescent compounds. 4. effect of cation binding on the photophysical properties of a coumarin linked to monoaza- and diaza-crown ethers. J Phys Chem 97:4552–4557. doi:10.1021/j100119a050
Liu WM, Xu LW, Sheng RL, Wang PF, Li HP, Wu SK (2007) A water-soluble “switching on” fluorescent chemosensor of selectivity to Cd2+. 9(19):3829–3832
Acknowledgements
This work was supported by the Science & Technology Department (No. SJ08B05) and the Education Department of Shaanxi Province of China (No. 09JK787). Zhao and Qi were supported by the NWU Graduate Experimental Research Funds (08YSY08, 09YSY19). Chen was supported by the NWU Graduate Cross-discipline Funds (09YJC21).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 463 kb)
Rights and permissions
About this article
Cite this article
Su, Z., Chen, K., Guo, Y. et al. A Coumarin-Based Fluorescent Chemosensor for Zn2+ in Aqueous Ethanol Media. J Fluoresc 20, 851–856 (2010). https://doi.org/10.1007/s10895-010-0628-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-010-0628-y