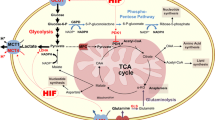
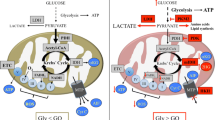
Abstract
Metabolic aberrations in the form of altered flux through key metabolic pathways are primary hallmarks of many malignant tumors. Primarily the result of altered isozyme expression, these adaptations enhance the survival and proliferation of the tumor at the expense of surrounding normal tissue. Consequently, they also expose a unique set of targets for tumor destruction while sparing healthy tissues. Despite this fact, development of drugs to directly target such altered metabolic pathways of malignant tumors has been under-investigated until recently. One such target is the ultimate step of glycolysis, which, as expected, presents itself as a metabolic aberration in most malignant tumors. Termed “aerobic glycolysis” due to abnormal conversion of pyruvic acid to lactic acid even under normoxia, the altered metabolism requires these tumors to rapidly efflux lactic acid to the microenvironment in order to prevent poisoning themselves. Thus, exposed is a prime “choke-point” to target these highly malignant, frequently chemo- and radio- resistant tumors. This review will focus on current outcomes in targeting lactate efflux in such tumors using glioma as a model, an ongoing project in our laboratory for the past half-decade, as well as supporting evidence from recent studies by others on targeting this “tail-end” of glycolysis in other tumor models.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- MCT:
-
monocarboxylate transporter
- LDH:
-
lactate dehydrogenase
- siRNA:
-
small intefering RNA
- miRNA:
-
microRNA
- CD:
-
cluster of differentiation
References
Argiles JM, Lopez-Soriano FJ (1990) Med Hypotheses 32:151–155
Baggetto LG (1992) Biochimie 74:959–974
Berg JM, Tymoczko JL, Stryer L (2006) Biochemistry. 6th ed. New York, WH Freeman
Carpenter L, Halestrap AP (1994) Biochem J 304(Pt 3):751–760
Chi SL, Pizzo SV (2006) Cancer Res 66:875–882
Coady MJ, Chang MH, Charron FM, Plata C, Wallendorff B, Sah JF, Markowitz SD, Romero MF, Lapointe JY (2004) J Physiol 557:719–731
Colen CB, Seraji-Bozorgzad N, Marples B, Galloway MP, Sloan AE, Mathupala SP (2006) Neurosurgery 59:1313–1324
Coss RA, Messinger JA, Wahl ML, Wachsberger PR, Leeper DB, Owen CS (1997) Int J Hyperthermia 13:325–336
Coss RA, Storck CW, Daskalakis C, Berd D, Wahl ML (2003) Mol Cancer Ther 2:383–388
Das B, Mondragon MO, Sadeghian M, Hatcher VB, Norin AJ (1994) J Exp Med 180:273–281
Fang J, Quinones QJ, Holman TL, Morowitz MJ, Wang Q, Zhao H, Sivo F, Maris JM, Wahl ML (2006) Mol Pharmacol 70:2108–2115
Fantin VR, St Pierre J, Leder P (2006) Cancer Cell 9:425–434
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Nature 391:806–811
Gatenby RA, Gawlinski ET (2003) Cancer Res 63:3847–3854
Gatenby RA, Gawlinski ET, Gmitro AF, Kaylor B, Gillies RJ (2006) Cancer Res 66:5216–5223
Gatenby RA, Gillies RJ (2004) Nat Rev Cancer 4:891–899
Gillies RJ, Martinez-Zaguilan R, Martinez GM, Serrano R, Perona R (1990) Proc Natl Acad Sci USA 87:7414–7418
Halestrap AP, Denton RM (1974) Biochem J 138:313–316
Halestrap AP, Meredith D (2004) Pflugers Arch 447:619–628
Halestrap AP, Price NT (1999) Biochem J 343(Pt 2):281–299
Juel C, Halestrap AP (1999) J Physiol 517(Pt 3):633–642
Kasischke KA, Vishwasrao HD, Fisher PJ, Zipfel WR, Webb WW (2004) Science 305:99–103
Kirk P, Wilson MC, Heddle C, Brown MH, Barclay AN, Halestrap AP (2000) EMBO J 19:3896–3904
Lee AH, Tannock IF (1998) Cancer Res 58:1901–1908
Lee RC, Feinbaum RL, Ambros V (1993) Cell 75:843–854
Lim LP, Glasner ME, Yekta S, Burge CB, Bartel DP (2003) Science 299:1540
Mathupala SP, Ko YH, Pedersen PL (2006) Oncogene 25:4777–4786
Mathupala SP, Parajuli P, Sloan AE (2004) Neurosurgery 55:1410–1419
Owen CS, Pooler PM, Wahl ML, Coss RA, Leeper DB (1997) J Cell Physiol 173:397–405
Pedersen PL (1978) Prog Exp Tumor Res 22:190–274
Pellerin L, Magistretti PJ (2004) Science 305:50–52
Poole RC, Halestrap AP (1993) Am J Physiol 264:C761–C782
Rotin D, Tannock IF (1984) Int J Radiat Oncol Biol Phys 10:1595–1598
Spencer TL, Lehninger AL (1976) Biochem J 154:405–414
Stubbs M, McSheehy PM, Griffiths JR (1999) Adv Enzyme Regul 39:13–30
Stubbs M, Rodrigues L, Howe FA, Wang J, Jeong KS, Veech RL, Griffiths JR (1994) Cancer Res 54:4011–4016
Volk C, Kempski B, Kempski OS (1997) Neurosci Lett 223:121–124
Wahl ML, Owen JA, Burd R, Herlands RA, Nogami SS, Rodeck U, Berd D, Leeper DB, Owen CS (2002) Mol Cancer Ther 1:617–628
Warburg O, Dickens F, Kaiser Wilhelm-Institut für Biologie B (1930) The metabolism of tumours: investigations from the Kaiser-Wilhelm Institute for Biology. Berlin-Dahlem, London, Constable
Wilson MC, Meredith D, Fox JE, Manoharan C, Davies AJ, Halestrap AP (2005) J Biol Chem 280:27213–27221
Yamagata M, Hasuda K, Stamato T, Tannock IF (1998) Br J Cancer 77:1726–1731
Zamore PD, Tuschl T, Sharp PA, Bartel DP (2000) Cell 101:25–33
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mathupala, S.P., Colen, C.B., Parajuli, P. et al. Lactate and malignant tumors: A therapeutic target at the end stage of glycolysis. J Bioenerg Biomembr 39, 73–77 (2007). https://doi.org/10.1007/s10863-006-9062-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-006-9062-x