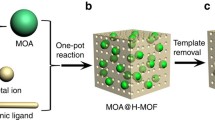
Abstract
Hierarchically porous MOFs (HP-MOFs) are commonly prepared by means of hydrothermal synthesis. Nonetheless, its relatively long crystallization time and harsh synthesis conditions have strongly obstructed the enhancement of HP-MOFs space–time yields (STYs) and the decrease in energy consumption. Herein, a simple and versatile method for preparing various HP-MOFs at room temperature was demonstrated, which had introduced surfactant as the template, whereas zinc oxide (ZnO) has been used as an accelerant. The resulting HP-MOFs showed multimodal hierarchical porous structures and excellent thermal stability. More importantly, the synthesis time was reduced dramatically to 11 min, with a maximal HP-MOFs STY of as high as 2575 kg m−3 d−1. Furthermore, the rapid formation process of HP-MOFs was examined through quantum chemistry calculation, and a feasible synthesis mechanism was also proposed. Notably, our synthesis strategy had shown a versatility, since other surfactants could also be used as the templates for the rapid room-temperature fabrication of diverse stable HP-MOFs. Importantly, the porosity of the HP-MOFs could be readily tuned through controlling the type of template. Moreover, gas adsorption measurement of HP-MOFs revealed high CH4 uptake capacity at 298 K due to the increase in surface area and pore volume. Our findings suggest that such method is applicable for the rapid synthesis of a wide variety of HP-MOFs on an industrial scale.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Metal–organic frameworks (MOFs), self-assembled from metal ions (or metal clusters) and organic ligands, have attracted considerable attention during the last two decades owing to their fascinating physical and chemical properties, such as high specific surface areas, tunable structures and porosities, as well as designable functionality [1]. Therefore, MOFs, which serve as the functional materials, have exhibited huge potential applications in diverse areas [2, 3], including gas storage [4], adsorption/separation [5], supercapacitor [6], drug delivery [7], sensing [8] and catalysis [9]. Notably, porosity is an essential feature in MOFs, which plays a vital role in functionalization like the diffusion and mass transfer of substrates/products [10, 11]. However, the pore sizes of the majority of MOFs have been reported to be within a micropore range (pore size < 2 nm) during the last few decades [4, 12, 13]. Consequently, the primary application of MOFs has focused on small molecules, which has prevented the large molecules to approach or leave the interior active sites owing to the confinement of pore size [14].
In recent years, tremendous efforts have been dedicated to develop efficient methods to synthesize hierarchically porous MOFs (HP-MOFs) containing micropores and mesopores (or micropores, mesopores and macropores) [15]. Specifically, the increased length of the bridging ligands is an apparent approach to obtain large pores in the resulting MOFs [16]. However, it is inevitably associated with certain difficulties, such as extended ligands synthesis, smaller mesopore size (< 10 nm), reduced porosity, interpenetration, disintegration and instability of crystal frameworks [17, 18]. On the other hand, HP-MOFs can also be synthesized through introducing mesopores and/or macropores into the structures of MOFs via the template strategy [19, 20] and post-synthetic modification [21]. Besides, a couple of new methods are also available, such as ionic liquids assisted synthesis [22], modulator-induced-defect-formation [23] and a spray-drying technology [24]. Typically, HP-MOFs synthesized by these methods are characterized by the superiorities of tunable mesoporosity and abundant active sites [25]. However, these approaches mostly require harsh experimental conditions and tedious downstream treatments (such as a long reaction time at high temperature and pressure and/or special apparatus) [26, 27], which is energy-consuming and may lead to pollution, a risk of explosion and a low production rate [28,29,30]. For instance, the space–time yields (STYs, kg m−3 d−1) are usually below 300 kg m−3 d−1 for products synthesized within several hours [28].
Recently, Huo groups reported the room-temperature synthesis of HP-MOFs (HKUST-1) with a high STY (2035 kg m−3 d−1) by controlling either the synthesis time or the copper source, for the first time [31]. However, it remains challenging to extend this method to prepare other HP-MOFs under facile synthesis conditions [12]. In our previous work, two HP-MOFs including HKUST-1 and ZIF-8 had been successfully prepared under facile synthesis conditions using a modified template strategy [32]. Nevertheless, several key problems have not been solved yet. For instance, (1) the type of surfactants and the produced HP-MOFs are limited; (2) the synthesis time (30 min) is still relatively long, and the STYs (1123 kg m−3 d−1) are still relatively low for the production of commercially available HP-MOFs; (3) the mechanism of the rapid room-temperature preparation of HP-MOFs has not been illustrated; and (4) the application of H-MOFs has not been explored. To our delight, the synthesis time had been shortened to 11 min after optimizing the synthesis procedure. More strikingly, the resulting HP-MOFs exhibited multimodal hierarchical structures with micropores, mesopores and macropores, together with excellent thermal stability. Notably, the STY of HP-MOFs was up to 2575 kg m−3 d−1, which is much higher than previously reported (1123 kg m−3 d−1) due to the reduction in synthesis time. In addition, the mechanism of the rapid synthesis of HP-MOFs had been elaborated by means of quantum chemistry (QC) calculations at the molecular level. As anticipated, the introduced amine surfactant could form the template micelles through self-assembly, as well as the addition of zinc oxide (ZnO) and metal ions to form a hydroxy double salt (HDS) intermediate, which could drive the fast growth of MOF crystals. In addition, four other surfactants could also be utilized as templates to rapidly synthesize HP-MOFs. Importantly, this strategy could also be generalized to rapidly synthesize other HP-MOFs, such as hierarchically porous ZIF-8 and ZIF-61. The resultant HP-MOFs showed enhanced CH4 uptake capacity in comparison with the microporous MOFs.
Experimental
Rapid room-temperature synthesis of hierarchically porous Cu–BTC
All chemicals were commercially available and used as received without further treatment. The HP-MOFs were synthesized with previously reported approaches with only a minor amend [32, 33]; 0.293 g of zinc oxide (ZnO) powder was dispersed in 18 mL N, N-dimethylmethanamide (DMF) and 8 mL deionized water (H2O) to form nanoslurry through using sonication for 30 min (denoted as solution A). 7.2 mmol of copper nitrate trihydrate (Cu(NO3)2·3H2O) was added to 18 mL of H2O (denoted as solution B), and then, the solution B was added to solution A under fast magnetic stirring to form (Zn, Cu) hydroxy double salt (HDS) (denoted as solution C). Then, 1.0 mL of surfactant (N,N-dimethyloctadecylamine, DOE) and 4 mmol of 1, 3, 5-benzenetricarboxylic acid (H3BTC) were added to 16 mL of methanol (denoted as solution D). After stirring for 30 min, solution C was added to solution D under fast magnetic stirring for 1 min, and then stand 10 min. After that, the glaucous solid product was collected by filtered. To remove the guest molecules such as surfactant, the solid product was immersed in ethanol at 60 °C with a total duration of 48 h (four times), and then dried overnight in a vacuum oven at 120 °C. The resulting product is denoted as HP–Cu–BTC. Similarly, HP-MOFs synthesized using other surfactants as templates are named as HP–Cu–BTC_X (X = B, C, D and E, where X represents the type of surfactants, B: hexadecyldimethylamine; C: N,N-dimethyltetradecylamine; D: n-butyl alcohol; and E: lauric acid). For comparison, conventional Cu–BTC was synthesized by using a solvothermal method following previous report [34], and labeled as C–Cu–BTC.
Rapid room-temperature synthesis of hierarchically porous ZIF-8
(Zn, Zn) HDS was prepared at room temperature and pressure using previously reported method [32, 35]. Specially, 0.406 g of ZnO was dispersed in 10 mL of H2O (denoted as solution A), and 1.098 g of zinc acetate dihydrate (Zn(CH3CO2)2·2H2O) was dissolved in 5 mL DMF (denoted as solution B). Mixed up these two solutions and stirred for 24 h, the generation of gel-like viscous fluid indicates the formation of (Zn, Zn) HDS. Next, 0.493 g of 2-methylimidazole (2Im) and 1.0 mL of DOE were dispersed in 9 mL DMF (denoted as solution C). 3 mL of (Zn, Zn) HDS suspension was added to solution C under magnetic stirring for 1 min, and then stand 10 min. After that, the obtained product was immediately filtered, activated and dried at 120 °C for overnight. The resulting product is denoted as HP-ZIF-8.
Rapid room-temperature synthesis of hierarchically porous ZIF-61
Similar to the above experiment procedures; 0.222 g of 2Im and 0.275 g of imidazole (Im) were dissolved in 10 mL methanol under fast magnetic stirring (denoted as solution A), and 1.0 mL of DOE was added to solution A. After stirring for 30 min, 3 mL of (Zn, Zn) HDS suspension was added to solution A under magnetic stirring for 1 min, and then stand 10 min, then immediately filtered, activated and dried overnight at 120 °C. The resulting product is denoted as HP-ZIF-61.
Materials characterization
X-ray diffraction (XRD) patterns of the HP-MOF samples were recorded on a diffractometer system (D8 ADVANCE, Bruker AXS) using a Cu sealed tube (40 kV, 40 mA, wavelength λ = 0.15418 nm) at room temperature and pressure. Fourier transform infrared (FT-IR) spectra of HP-MOFs were recorded on an FT-IR spectrometer (Vector 33, Bruker Corporation) with a resolution of 4 cm−1. Nitrogen (N2) adsorption–desorption measurements at 77 K were performed on an ASAP 2020 or 2460 (Micromeritics), and all samples were outgassed for overnight at 393 K before measurement to remove the guest molecules trapped in the pores. The morphologies of the samples were characterized by using scanning electron microscope (SEM; ZEISS Ultra 55, Carl Zeiss) at a low loading energy and voltage of 5.0 kV and transmission electron microscopy (TEM, JEM-2100HR, JEOL). Thermogravimetric analysis (TGA) data were obtained using a TG analyzer (TG 209, Tarsus, NETZSCH) and heated from 298 to 873 K in N2 atmosphere at a rate of 5 K min−1. The CH4 adsorption curve was obtained on a RUBOTHERM magnetic suspension balance.
Calculation
We applied density functional theory (DFT) method to calculate electrostatic properties of the surfactant so as to investigate the difference between amines and halogenated alkane in the rapid formation process of HP-MOFs [36]. Geometry optimization was performed at the B3LYP level with 6-311+G(d, p) basis set in the Gaussian 09 [37]. The chosen basis set offers the basis functions for a variety range of organic compounds with fair accuracy [38]. The molecular electrostatic potential (MEP) of surfactant molecules was calculated at B3LYP/6-31+G* level.
Calculation of STY
Space–time yield (STY) is an important parameter for the industrial production of the product [33] and can be calculated by Eq. (1)
where mMOF represents the powder mass (g) of the HP-MOFs, Vsolution represents the total volume (cm3) of the mixture, and τ represents the reaction time (min).
Results and discussion
Figure 1a shows the wide-angle XRD patterns of the HP–Cu–BTC sample synthesized with a cooperative template method and the C–Cu–BTC, as well as the simulated XRD pattern of Cu–BTC single-crystal. It can be clearly observed from Fig. 1a that the XRD patterns of the as-synthesized HP–Cu–BTC are consistent with those of C–Cu–BTC and the simulated Cu–BTC, indicating that the obtained product is topologically identical to Cu–BTC crystal [29]. Moreover, the sharp XRD peaks of HP–Cu–BTC confirm fine crystallinity [37]. The crystal structure of HP–Cu–BTC was further characterized using FT-IR spectroscopy. As shown in Fig. 1b, the FT-IR spectra of HP–Cu–BTC reveal that the carboxylate group ν(OCO) of ligand (H3BTC) was coordinated to metal ions (Cu2+) [39]. The absorption bands of HP–Cu–BTC at ca. 1560, 1450 and ~ 1370 cm−1 were clearly identified as pure Cu–BTC [40], verifying again that the obtained product is crystalline Cu–BTC. Furthermore, the characteristic C–N stretching vibration of template at around 1230–1030 cm−1 was not observed, confirming that HP–Cu–BTC is free of template, which can be attributed to the introduced template was removed easily by the treatment of activation and drying [41]. Therefore, the FT-IR results indicate the coordination between Cu2+ and ligand (H3BTC) in the framework.
The porosities of the attained samples were measured by N2 adsorption–desorption method after all samples were treated. As shown in Fig. 2a, the N2 adsorption–desorption isotherms of C–Cu–BTC exhibit a type I mode with a sharp increased uptake at the initial stage followed by a flatland, which is a characteristic of microporous materials [37]. Nevertheless, the N2 adsorption–desorption isotherms of the HP–Cu–BTC show an intermediate mode between type I and type IV with a high N2 uptake at low relative pressure region of P/Po < 0.01, indicating the presence of abundant micropores [32]. Moreover, a large type-H4 hysteresis loop can be observed at high relative pressure (P/Po > 0.8), related to capillary condensation of N2 in mesopores [42], confirming the formation of mesopores in HP–Cu–BTC sample, similar behavior has been observed in a previous report [18, 43]. In addition, it should be pointed out that the adsorption capacity of HP–Cu–BTC is inferior to that of C–Cu–BTC at low relative pressure (P/Po < 0.01), which can be attributed to the HP–Cu–BTC has a lower Brunauer–Emmett–Teller (BET) surface area than that of C–Cu–BTC [44].
More detail regarding the porosity properties of these samples can be gained from the pore size distributions (calculated from nonlocal density functional theory (NL-DFT) analysis). As shown in Fig. 2b, the C–Cu–BTC only possesses micropores with pore size distribution centered at around 0.86 nm, which is consistent with previous report [34]. However, for the HP–Cu–BTC sample, other than the intrinsic micropores, it contains a broad pore size distribution from 30 to over 100 nm (inset of Fig. 2b), including mesoporous and macroporous regions. These analyses confirm that the as-synthesized HP–Cu–BTC product possesses hierarchical pores with micropores, mesopores and macropores. As shown in Table 1, the attained HP–Cu–BTC shows a lower Brunauer–Emmett–Teller (BET) surface areas (SBET = 907 m2 g−1) than those of the C–Cu–BTC (SBET = 1425 m2 g−1) and previously reported Cu–BTC [33], as confirmed by the N2 adsorption–desorption isotherms (Fig. 2a). The lower SBET can be ascribed to the pore impenetration [45]. However, HP–Cu–BTC has a much larger mesopore volume (Vmeso = 0.24 cm3 g−1) and the mesopore surface area (Smeso) to micropore surface area (Smicro) (Smeso/Smicro = 0.25) than that of C–Cu–BTC (Vmeso = 0.06 cm3 g−1, Smeso/Smicro = 0.09), respectively. These results further verify the formation of mesoporosity in the HP–Cu–BTC.
SEM and TEM were applied to track the morphology and particle size of product [46, 47]. As illustrated in Fig. 3a, the as-synthesized C–Cu–BTC crystal exhibits a typical octahedron in shape with crystal dimension about 10 μm. Moreover, smooth surfaces can be clearly observed from the Fig. 3a–c, respectively, consistent with previous reports [34]. However, HP–Cu–BTC consists of nanoparticles with particle size of 500 nm (Fig. 3b). The smaller crystals in HP–Cu–BTC can be attributed to the rapid growth of frameworks [33, 48]. In addition, the high-dispersive nanoparticles are randomly stacked together and form abundant pore voids, as shown in Fig. 3b–d, respectively. Similar to previously reported that hierarchical pores were formed through the aggregation or packing of nanoparticles [49, 50]. Moreover, the scan mapping shows that the presence of elemental C, O and Cu with uniform distribution in the HP-MOF particles (Fig. S1) further verifies the chemical composition of the crystal [32]. Excellent stability of materials is necessary for the practical applications [51]. Therefore, the thermal stability of the as-synthesized HP-MOFs was investigated by TGA under a N2 atmosphere. As shown in Fig. S2, the TGA curve of HP–Cu–BTC indicates that the product undergoes three stages of weight loss; in the initial stage of weight loss occurred at 90 °C corresponding to the removal of trapped guest molecules (e.g., H2O) [52]. The second stage of weight loss which takes place at approximately 150 °C may be ascribed to the evaporation of template and DMF [53]. Subsequently, the TGA curve of HP–Cu–BTC exhibits a drop sharply after 320 °C, corresponding to the decomposition of framework [29]. By comparison, the collapse temperature of the sample HP–Cu–BTC (320 °C) is similar to that of C–Cu–BTC (320 °C) during the same thermal treatment, indicating that the introduction of mesopores and macropores does not degrade the thermal stability of the HP-MOFs [32].
In our previous study, amine surfactant (e.g., DOE) could be used as structure-directing agents (SDAs) to prepare HP-MOFs such as Cu–BTC and ZIF-8 under hydrothermal conditions, indicating the introduced DOE played a role of template during the synthesis [18]. Additionally, the Tyndall scattering effect appeared in the colloidal suspension of DOE indicated that the supramolecular micelles were formed through self-assembly [54], as shown in Fig. S3. However, in our most recent work, the selected halogenated alkenes such as 1-bromohexadecane cannot individually act as template to prepare HP-MOFs under same synthesis conditions owing to its weak interactions with ligands [32]. In order to explore the difference between the amine and halogenated alkane for the rapid formation process of HP-MOFs, theoretical calculation was applied to study the molecular property of surfactants. The optimized molecular geometries of two surfactants (DOE and 1-bromohexadecane) are demonstrated in Fig. S4. Figure 4 shows the molecular electrostatic potential (MEP) maps of the two surfactants, where the negative regions (red) of MEP maps relate to electrophilic reactivity and the positive regions (blue) to nucleophilic reactivity [29]. The negative regions (red) of the two surfactant molecules are mainly located on the amine and bromine groups, respectively, which indicates the most susceptible sites for electrophilic attacks [55]. The electrostatic potential of DOE located at amine group is lower than bromine group of 1-bromohexadecane, suggesting stronger electrophilic affinity of amines [56]. These theoretical calculations may explain the experimental results that the amines can be applied as template instead of halogenated alkane.
Based on the above experiments and theoretical calculations, a feasible mechanism for the usage of amine surfactant and ZnO to rapidly synthesize HP-MOFs is proposed and illustrated in Scheme 1. During the synthesis system, on the one hand, the introduced DOE forms supramolecular micelles through self-assembly in the ligand solution, and then the micelles surrounded by the ligands through an appropriate interaction [57, 58]. On the other hand, the introduced ZnO can react with Cu2+ to form (Cu, Zn) HDS (an intermediate that was dissolved completely in solution), which exhibits a high rate of anion exchange in the ligand solution and drives the rapid formation of MOF crystal at room temperature and pressure [33, 59]. Thus, ZnO played the role of an accelerant. Upon the (Cu, Zn), HDS was added to the mixture of the ligand and micelles, the ligand will react with HDS to form MOF frameworks on the surface of micelles [19, 60]. Subsequently, mesopores and macropores are formed after removing template [19].
For the HP–Cu–BTC_X (X = B and C), as shown in Fig. S5, the XRD patterns and FT-IR spectra of HP–Cu–BTC_X are in close agreement with the simulated Cu–BTC or C–Cu–BTC, respectively, confirming the formation of crystalline Cu–BTC. The N2 adsorption–desorption isotherms and electron microscopy (SEM and TEM) images demonstrate that the attained HP–Cu–BTC_X possesses hierarchical pores (see Figs. S6–7 in the Supporting Information for details). Moreover, as shown in Table 1, the prepared HP-Cu-BTC_X exhibits high Vmeso and Smeso/Smicro, further confirming the formation of mesopores. Notably, the mesoporosity of HP-MOFs depends on the type of templates (Table 1), which allows us control the porosity of HP-MOFs, such as SBET, Vt and Vmeso. Furthermore, all HP–Cu-BTC_X samples have excellent thermal stabilities (Fig. S2). More importantly, our synthesis strategy has a highly versatile due to other HP-MOFs such as ZIF-8 and ZIF-61 were also synthesized at room temperature (denoted as HP-ZIF-8 and HP-ZIF-61, respectively, see experiment “Rapid room-temperature synthesis of hierarchically porous ZIF-8 and Rapid room-temperature synthesis of hierarchically porous ZIF-61” sections). As shown in Fig. S8a, all diffraction peak positions of the HP-ZIF-8 sample well agreed with the simulation result, confirming the product obtained from rapid synthesis is crystalline ZIF-8. The FT-IR spectra of HP-ZIF-8 sample are in agreement with that of conventional ZIF-8 (C-ZIF-8) (Fig. S8b), which is clearly identified as pure ZIF-8 [61]. The N2 adsorption–desorption isotherms of the HP-ZIF-8 sample show an intermediate mode between type I and type IV (Fig. 5a), which is related to the coexistence of mesopores and micropores [62]. By comparison, the isotherms of C-ZIF-8 exhibit a type I mode (Fig. S9a), which is related to microporous materials [63]. Moreover, we can acquire more information concerning porosity from the pore size distributions [18]. As shown in Fig. 5b, in addition to the intrinsic micropores, HP-ZIF-8 sample also embraces the mesopore and macropore regions, which is absent in the C-ZIF-8 (Fig. S9b). Furthermore, SEM and TEM images (Fig. S10) displayed that HP-ZIF-8 product has an apparent mesovoid phase, which indicates that HP-ZIF-8 possesses mesoporous structures. Moreover, the as-synthesized HP-ZIF-8 exhibits good thermostability (Fig. S11). Similarly, HP-ZIF-61 sample prepared from the rapid synthesis also possesses a hierarchical porous structure (see Figs. S12–13). In addition to organic amines, other surfactants such as n-butyl alcohol and lauric acid can also be used as templates to rapidly fabricate HP-MOFs (HP-Cu-BTC_D and HP-Cu-BTC_E) (see Figs. S14–15 in the Supporting Information for details). These results indicate that our template method developed in this work is well versatile.
The introduction of mesopores and macropores into microporous MOFs can effectively improve adsorption performance of guest molecules [17]. To evaluate the adsorption properties of the HP-ZIF-8, single-component gas such as CH4 was selected as probe reactant. Figure 6 exhibits the isotherms of CH4 on HP-ZIF-8 and C-ZIF-8 measured at a broad range of high pressures at 298 K. It can be seen that the adsorption isotherm of CH4 on the HP-ZIF-8 is always higher than that on C-ZIF-8, implying the former has higher uptake capacities of CH4 than the later, i.e., at 3 MPa and 273 K, the CH4 uptake of HP-ZIF-8 is 68.4 mg/g, having an increase of 63.6% compared to that of C-ZIF-8 (41.8 mg/g). The significantly increase in CH4 uptake capacity can be attributed not only to an enhancement in the surface area of HP-ZIF-8, but also to the increase in the pore volume [64].
In addition to the porosity properties, energy consumption and production rate are two essential factors for the large-scale preparation of HP-MOFs due to them determine the environmental influence (e.g., carbon footprint) and manufacturing cost [12, 65]. In comparison with conventional template method for synthesizing HP-MOFs requires a long reaction time at high temperature and pressure, our strategy developed in this work realizes the rapid room-temperature synthesis of various HP-MOFs by the simultaneous introduction of surfactant and ZnO, which satisfies with the environmental-friendly and low-cost demands for the production of HP-MOFs. STY of a reaction is essential consideration to assess the viability of scale up, which should be as high as possible [31, 51]. For 11 min reactions, the STY of HP–Cu–BTC is as high as 2575 kg m−3 d−1, which is one order of magnitude higher than that of the commercial product Basolite C300 (225 kg m3 per day) [65], and also surpasses the previous report (1123 kg m−3 d−1) [32]. A plausible explanation of such high STY of HP-Cu-BTC may be attributed to the reduction in synthesis time [28]. Moreover, the STY of HP-Cu-BTC synthesized with tonne scale (> 1 × 103 kg m−3 d−1) is comparable to that of an Al-MOFs with large-scale production [66]. Therefore, our synthesis strategy may open up a pathway for the commercialized preparation of HP-MOFs which by means of conventional hydrothermal methods were not accessible on an industrial level before.
Conclusions
In summary, three stable HP-MOFs (Cu-BTC, ZIF-8 and ZIF-61) have been prepared using a simple and straightforward room-temperature synthesis strategy. Specifically, the synthesis conditions have been optimized, which can shorten the synthesis time to 11 min. Additionally, the obtained products exhibited multimodal hierarchical porous structures with micropores, mesopores and macropores. The STY of the produced HP–Cu–BTC is up to 2575 kg m−3 d−1, which is much higher than previously reported (1123 kg m−3 d−1). Moreover, theoretical calculations have illustrated the differences in surfactant type for the rapid formation of HP-MOFs. Consequently, a novel synthesis mechanism is proposed. The resulting HP-MOFs have much higher gas uptake capacity than that of conventional MOFs. Among them, the maximum CH4 uptake of HP-MOFs is 68.4 mg/g, which is 63.6% higher than that of microporous MOFs. Importantly, our synthesis strategy has a versatility due to other surfactants can also be used as templates, while other HP-MOFs can also be synthesized rapidly under mild synthesis conditions. The method developed in this work represents a versatile industrial method for the large-scale production of a wide variety of HP-MOFs.
References
Yang Q, Xu Q, Jiang H-L (2017) Metal–organic frameworks meet metal nanoparticles: synergistic effect for enhanced catalysis. Chem Soc Rev 46:4774. https://doi.org/10.1039/C6CS00724D
Dolgopolova EA, Rice AM, Martin CR, Shustova NB (2018) Photochemistry and photophysics of MOFs: steps towards MOF-based sensing enhancements. Chem Soc Rev. https://doi.org/10.1039/C7CS00861A
Wang H, Lustig WP, Li J (2018) Sensing and capture of toxic and hazardous gases and vapors by metal–organic frameworks. Chem Soc Rev. https://doi.org/10.1039/C7CS00885F
Zheng B, Huang L, Cao X et al (2018) A highly porous acylamide decorated MOF-505 analogue exhibiting high and selective CO2 gas uptake capability. CrystEngComm 20:1874. https://doi.org/10.1039/C8CE00103K
Qian X, Ren Q, Wu X, Sun J, Wu H, Lei J (2018) Enhanced water stability in Zn-doped zeolitic imidazolate Framework-67 (ZIF-67) for CO2 Capture Applications. ChemistrySelect 3:657. https://doi.org/10.1002/slct.201702114
Zhao Y, Liu J, Horn M, Motta N, Hu M, Li Y (2018) Recent advancements in metal organic framework based electrodes for supercapacitors. Sci China Mater 61:159. https://doi.org/10.1007/s40843-017-9153-x
Gao X, Wang Y, Ji G, Cui R, Liu Z (2018) One-pot synthesis of hierarchical-pore metal–organic frameworks for drug delivery and fluorescent imaging. CrystEngComm 20:1087. https://doi.org/10.1039/C7CE02053H
Yang Y, Liu X, Yan D, Deng P, Guo Z, Zhan H (2018) Europium ion post-functionalized zirconium metal–organic frameworks as luminescent probes for effectively sensing hydrazine hydrate. RSC Adv 8:17471. https://doi.org/10.1039/C8RA03049A
Tian P, He X, Li W et al (2018) Zr–MOFs based on Keggin-type polyoxometalates for photocatalytic hydrogen production. J Mater Sci. https://doi.org/10.1007/s10853-018-2476-0
Ma L, Abney C, Lin W (2009) Enantioselective catalysis with homochiral metal–organic frameworks. Chem Soc Rev 38:1248. https://doi.org/10.1039/B807083K
Lin S, Bediako JK, Cho C-W et al (2018) Selective adsorption of Pd(II) over interfering metal ions (Co(II), Ni(II), Pt(IV)) from acidic aqueous phase by metal–organic frameworks. Chem Eng J 345:337. https://doi.org/10.1016/j.cej.2018.03.173
Julien PA, Mottillo C, Friscic T (2017) Metal–organic frameworks meet scalable and sustainable synthesis. Green Chem 19:2729. https://doi.org/10.1039/C7GC01078H
Yin W, C-a Tao F, Wang J Huang, Qu T, Wang J (2018) Tuning optical properties of MOF-based thin films by changing the ligands of MOFs. Sci China Mater 61:391. https://doi.org/10.1007/s40843-017-9143-5
Kayal S, Chakraborty A (2018) Activated carbon (type Maxsorb-III) and MIL-101(Cr) metal organic framework based composite adsorbent for higher CH4 storage and CO2 capture. Chem Eng J 334:780. https://doi.org/10.1016/j.cej.2017.10.080
Szuromi P (2018) Mesoporous metal–organic frameworks. Science 359:172. https://doi.org/10.1126/science.359.6372.172-i
Ma S, Sun D, Simmons JM, Collier CD, Yuan D, Zhou H-C (2008) Metal–organic framework from an anthracene derivative containing nanoscopic cages exhibiting high methane uptake. J Am Chem Soc 130:1012. https://doi.org/10.1021/ja0771639
Gu X, Lu Z-H, Xu Q (2010) High-connected mesoporous metal–organic framework. Chem Commun 46:7400. https://doi.org/10.1039/C0CC02808H
Duan C, Li F, Zhang H, Li J, Wang X, Xi H (2017) Template synthesis of hierarchical porous metal–organic frameworks with tunable porosity. RSC Adv 7:52245. https://doi.org/10.1039/C7RA08798E
Qiu LG, Xu T, Li ZQ et al (2008) Hierarchically micro- and mesoporous metal–organic frameworks with tunable porosity. Angew Chem 47:9487. https://doi.org/10.1002/anie.200803640
Xueqing Y, Wei C, Haidong B et al (2018) Synthesis of mesoporous ZIF-8 nanoribbons and their conversion into carbon nanoribbons for high-performance supercapacitors. Chem Eur J. https://doi.org/10.1002/chem.201801869
Kim Y, Yang T, Yun G et al (2015) Hydrolytic transformation of microporous metal–organic frameworks to hierarchical micro-and mesoporous MOFs. Angew Chem Int Edit 54:13273
Zhao Y, Zhang J, Han B, Song J, Li J, Wang Q (2011) metal–organic framework nanospheres with well-ordered mesopores synthesized in an ionic liquid/Co2/surfactant system. Angew Chem Int Edit 50:636
Cai G, Jiang H-L (2017) A modulator-induced defect-formation strategy to hierarchically porous metal–organic frameworks with high stability. Angew Chem Int Edit 56:563. https://doi.org/10.1002/anie.201610914
Carne-Sanchez A, Imaz I, Cano-Sarabia M, Maspoch D (2013) A spray-drying strategy for synthesis of nanoscale metal–organic frameworks and their assembly into hollow superstructures. Nat Chem 5:203. https://doi.org/10.1038/nchem.1569
Chou L-Y, Hu P, Zhuang J et al (2015) Formation of hollow and mesoporous structures in single-crystalline microcrystals of metal–organic frameworks via double-solvent mediated overgrowth. Nanoscale 7:19408. https://doi.org/10.1039/c5nr06532a
He X, Yang C, Wang D, Gilliland Iii SE, Chen D-R, Wang W-N (2017) Facile synthesis of ZnO@ZIF core-shell nanofibers: crystal growth and gas adsorption. CrystEngComm 19:2445. https://doi.org/10.1039/C7CE00368D
Chen Y, Ni D, Yang X, Liu C, Yin J, Cai K (2018) Microwave-assisted synthesis of honeycomblike hierarchical spherical Zn-doped Ni-MOF as a high-performance battery-type supercapacitor electrode material. Electrochim Acta 278:114. https://doi.org/10.1016/j.electacta.2018.05.024
Stock N, Biswas S (2012) Synthesis of metal–organic frameworks (MOFs): routes to various MOF topologies, morphologies, and composites. Chem Rev 112:933. https://doi.org/10.1021/cr200304e
Duan C, Li F, Luo S, Xiao J, Li L, Xi H (2018) Facile synthesis of hierarchical porous metal–organic frameworks with enhanced catalytic activity. Chem Eng J 334:1477. https://doi.org/10.1016/j.cej.2017.11.086
Fu Z, Chuan-Ling Z, Huang-Yong P, Huai-Ping C, Hai-Sheng Q (2016) Near-infrared photocatalytic upconversion nanoparticles/TiO2 nanofibers assembled in large scale by electrospinning. Part Part Syst Char 33:248. https://doi.org/10.1002/ppsc.201600010
Huo J, Brightwell M, El Hankari S, Garai A, Bradshaw D (2013) Industrially relevant, aqueous room temperature synthesis of HKUST-1 with high space–time yield. J Mater Chem A 1:15220. https://doi.org/10.1039/C3TA14409G
Duan C, Li F, Li L et al (2018) Hierarchically structured metal–organic frameworks assembled by hydroxy double salt-template synergy with high space–time yields. CrystEngComm 20:1057. https://doi.org/10.1039/C7CE01843F
Zhao J, Nunn WT, Lemaire PC et al (2015) Facile conversion of hydroxy double salts to metal–organic frameworks using metal oxide particles and atomic layer deposition thin-film templates. J Am Chem Soc 137:13756. https://doi.org/10.1021/jacs.5b08752
Chui SS-Y, Lo SM-F, Charmant JP, Orpen AG, Williams ID (1999) A chemically functionalizable nanoporous material [Cu3(TMA)2(H2O)3] n. Science 283:1148
Li Z, Huang X, Sun C, Chen X, Hu J, Stein A, Tang B (2017) Thin-film electrode based on zeolitic imidazolate frameworks (ZIF-8 and ZIF-67) with ultra-stable performance as a lithium-ion battery anode. J Mater Sci 52:3979. https://doi.org/10.1007/s10853-016-0660-7
Singha Deb AK, Dwivedi V, Dasgupta K, Musharaf Ali S, Shenoy KT (2017) Novel amidoamine functionalized multi-walled carbon nanotubes for removal of mercury(II) ions from wastewater: combined experimental and density functional theoretical approach. Chem Eng J 313:899. https://doi.org/10.1016/j.cej.2016.10.126
Chongxiong D, Hang Z, Anguo P et al (2018) Synthesis of hierarchically structured metal−organic frameworks by a dual-functional surfactant. ChemistrySelect 3:5313. https://doi.org/10.1002/slct.201800571
Duan C, Li F, Xiao J, Liu Z, Li C, Xi H (2017) Rapid room-temperature synthesis of hierarchical porous zeolitic imidazolate frameworks with high space–time yield. Sci China Mater 60:1205. https://doi.org/10.1007/s40843-017-9136-y
Zhu C, Zhang Z, Wang B et al (2016) Synthesis of HKUST-1#MCF compositing materials for CO2 adsorption. Micropor Mesopor Mat 226:476. https://doi.org/10.1016/j.micromeso.2016.02.029
Kim S-Y, Kim A-R, Yoon JW, Kim H-J, Bae Y-S (2018) Creation of mesoporous defects in a microporous metal–organic framework by an acetic acid-fragmented linker co-assembly and its remarkable effects on methane uptake. Chem Eng J 335:94. https://doi.org/10.1016/j.cej.2017.10.078
Bradshaw D, El-Hankari S, Lupica-Spagnolo L (2014) Supramolecular templating of hierarchically porous metal–organic frameworks. Chem Soc Rev 43:5431
Shi Z, Li L, Xiao Y et al (2017) Synthesis of mixed-ligand Cu–MOFs and their adsorption of malachite green. RSC Adv 7:30904. https://doi.org/10.1039/C7RA04820C
Sun L-B, Li J-R, Park J, Zhou H-C (2011) Cooperative template-directed assembly of mesoporous metal–organic frameworks. J Am Chem Soc 134:126
Gregg SJ, Sing KSW, Salzberg H (1967) Adsorption surface area and porosity. J Electrochem Soc 114:279C
Peng L, Zhang J, Xue Z et al (2014) Highly mesoporous metal–organic framework assembled in a switchable solvent. Nat Commun 5:4465. https://doi.org/10.1038/ncomms5465
Wang Z, Hu S, Yang J et al (2018) Nanoscale Zr based MOFs with tailorable size and introduced mesopore for protein delivery. Adv Funct Mater. https://doi.org/10.1002/adfm.201707356
Wang X, Zhan C, Ding Y et al (2017) Dual-core Fe2O3@Carbon structure derived from hydrothermal carbonization of chitosan as a highly efficient material for selective adsorption. ACS Sustain Chem Eng 5:1457. https://doi.org/10.1021/acssuschemeng.6b02034
Gross AF, Sherman E, Vajo JJ (2012) Aqueous room temperature synthesis of cobalt and zinc sodalite zeolitic imidizolate frameworks. Dalton T 41:5458. https://doi.org/10.1039/C2DT30174A
Cao Y, Ma Y, Wang T, Wang X, Huo Q, Liu Y (2016) Facile fabricating hierarchically porous metal–organic frameworks via a template-free strategy. Cryst Growth Des 16:504. https://doi.org/10.1021/acs.cgd.5b01559
Hirai K, Furukawa S, Kondo M, Uehara H, Sakata O, Kitagawa S (2011) Sequential functionalization of porous coordination polymer crystals. Angew Chem Int Edit 50:8057. https://doi.org/10.1002/anie.201101924
Bian C, Zhang C, Pan S et al (2017) Generalized high-temperature synthesis of zeolite catalysts with unpredictably high space–time yields (STYs). J Mater Chem A 5:2613. https://doi.org/10.1039/C6TA09866E
Cravillon J, Münzer S, Lohmeier S-J, Feldhoff A, Huber K, Wiebcke M (2009) Rapid room-temperature synthesis and characterization of nanocrystals of a prototypical zeolitic imidazolate framework. Chem Mater 21:1410
Aarti S Bhadauria, Nanoti A et al (2016) [Cu3(BTC)2]-polyethyleneimine: an efficient MOF composite for effective CO2 separation. RSC Adv 6:93003. https://doi.org/10.1039/C6RA10465G
Singh A, Ojha AK, Jang HM (2018) Strategic design and utilization of molecular flexibility for straddling the application of organic superbases: a DFT Study. ChemistrySelect 3:837. https://doi.org/10.1002/slct.201702912
Wan Y, Zhao D (2007) On the controllable soft-templating approach to mesoporous silicates. Chem Rev 107:2821
Roy DR, Parthasarathi R, Padmanabhan J, Sarkar U, Subramanian V, Chattaraj PK (2006) Careful scrutiny of the philicity concept. J Phys Chem A 110:1084. https://doi.org/10.1021/jp053641v
Wan Y, Shi Y, Zhao D (2008) Supramolecular aggregates as templates: ordered mesoporous polymers and carbons. Chem Mater 20:932. https://doi.org/10.1021/cm7024125
Moliner M, Rey F, Corma A (2013) Towards the rational design of efficient organic structure-directing agents for zeolite synthesis. Angew Chem Int Edit 52:13880. https://doi.org/10.1002/anie.201304713
Zhao J, Kalanyan B, Barton HF, Sperling BA, Parsons GN (2017) In situ time-resolved attenuated total reflectance infrared spectroscopy for probing metal–organic framework thin film growth. Chem Mater 29:8804. https://doi.org/10.1021/acs.chemmater.7b03096
Meyn M, Beneke K, Lagaly G (1993) Anion-exchange reactions of hydroxy double salts. Inorg Chem 32:1209
Wang X, Liu J, Leong S et al (2016) Rapid construction of ZnO@ZIF-8 heterostructures with size-selective photocatalysis properties. ACS Appl Mater Inter 8:9080. https://doi.org/10.1021/acsami.6b00028
Yang X, Wu S, Wang P, Yang L (2018) Hierarchical 3D ordered meso-/macroporous metal–organic framework produced through a facile template-free self-assembly. J Solid State Chem 258:220. https://doi.org/10.1016/j.jssc.2017.10.020
Xukun Q, Qingbao R, Xiaofei W, Jing S, Hongyu W, Jun L (2018) Enhanced water stability in Zn-doped zeolitic imidazolate framework-67 (ZIF-67) for CO2 capture applications. ChemistrySelect 3:657. https://doi.org/10.1002/slct.201702114
Furukawa H, Ko N, Go YB et al (2010) Ultrahigh Porosity in metal–organic frameworks. Science 329:424. https://doi.org/10.1126/science.1192160
Czaja AU, Trukhan N, Muller U (2009) Industrial applications of metal–organic frameworks. Chem Soc Rev 38:1284. https://doi.org/10.1039/B804680H
Gaab M, Trukhan N, Maurer S, Gummaraju R, Müller U (2012) The progression of Al-based metal–organic frameworks—from academic research to industrial production and applications. Micropor Mesopor Mat 157:131. https://doi.org/10.1016/j.micromeso.2011.08.016
Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 21576094), SRFDP (No. 20130172110012) and Guangdong Natural Science Foundation (2017A030313052).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Duan, C., Huo, J., Li, F. et al. Ultrafast room-temperature synthesis of hierarchically porous metal–organic frameworks by a versatile cooperative template strategy. J Mater Sci 53, 16276–16287 (2018). https://doi.org/10.1007/s10853-018-2793-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-018-2793-3