Abstract
Increasing fragmentation of forests worldwide by timber and industrial development makes it important to understand the edge effects of common anthropogenic disturbances on forest fauna. We collected ground-active spiders along transects across the edge of logging clearcuts, gravel roads and gas pipelines in the boreal forest of Alberta, sampling on the disturbance (10 m from forest edge), and 10, 45, and 200 m into the forest. We asked whether the three disturbances were associated with edge effects on spider communities, and whether the extent of their associated edge effects were equivalent. The spider community at the edges of clearcuts was distinct from interior and on-disturbance communities 10 m into the forest from the clearcut edge, showing an edge effect of between 10 and 45 m from clearcut edges, while no edge effects were apparent at road and pipeline edges. Edge effects therefore differ at linear and non-linear openings in the boreal forest, which suggests that small linear openings may be associated with minimal edge effects compared to large polygonal forest openings. This result has important consequences for forest management, where clearcuts and other non-linear openings are likely to cause edge effects on spider communities that are between 10 and 45 m in their extent. The small size of clearcuts as practiced in the public forests of Canada, and their dense and broad application across the landscape, makes this edge effect of broad spatial significance in protecting biodiversity in managed landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The study of animal distributions across habitat edges has been a persistent topic of concern in ecology since the introduction of the idea by Leopold (1933), who recommended that edges be used as a tool for land managers to increase concentrations of game animals. Over the past several decades, evidence has accumulated that edge effects may be detrimental to forest species that require interior habitat to thrive (Laurance et al. 2007; Murcia 1995). For some of these species, sensitivity to the forest edge may be strong enough to void some smaller forest patches completely of interior habitat conditions, rendering them non-habitat and unable to support viable populations of interior species (Ewers and Didham 2008; Woodroffe and Ginsberg 1998). Although edge effects can vary widely in their direction and magnitude in a species-specific fashion (Ries et al. 2004), understanding the depth of edge influence (Harper and Macdonald 2002) for particular taxa can aid land managers in making management decisions that maintain sufficient forest interior habitat for those taxa. As forests worldwide become increasingly fragmented by logging, deforestation and other industrial development (Hansen et al. 2010), the necessity to understand the magnitude of edge effects associated with common anthropogenic disturbances in these habitats becomes ever more pressing (Harper et al. 2005; Harrison and Bruna 1999; Laurance and Curran 2008).
Although there is a rich literature of edge effect studies on vegetation, vertebrates, and insects (Duelli et al. 2002; Gates and Gysel 1978; Heliola et al. 2001; Laurance et al. 1997, 2002; Magura et al. 2001; Mullen et al. 2003; Ries and Sisk 2008; Taboada et al. 2004; Tylianakis et al. 2005), very little has been documented for edge effects on spiders (c.f. Larrivee et al. 2008; Pearce et al. 2005). Spiders (Arachnida: Araneae) are an abundant and diverse taxon in northern forests (Buddle et al. 2000) and are dominant generalist predators of arthropods of all forest strata (Clarke and Grant 1968; Turnbull 1973). Spiders represent a large portion of forest arthropod communities in their abundance and total biomass, making them important elements of the community as a significant source of food for their arthropod, avian and mammalian predators (Jansson and von Bromssen 1981; Pearce and Venier 2006).
Spiders can be expected to respond strongly to edge effects from forest disturbance. Their activity and habitat selection is strongly constrained by microclimatic conditions such as soil moisture and temperature (e.g., Vlijm and Kessler-Geschiere 1967; Ziesche and Roth 2008), which are greatly influenced in forests by proximity to anthropogenic edges (Chen et al. 1999). Spiders are also highly dependent on habitat structure (Langellotto and Denno 2004; Turnbull 1973). Web-building spiders require suitable surrounding structure for points of web attachment (Robinson 1981), and most hunting spiders sense their environment and their prey predominantly through mechanoreception, which is mediated through structural features in the environment (Uetz 1991).
Habitat structure and microclimate are altered within forest edge zones by both primary and secondary responses (sensu Harper et al. 2005) of the forest edge to the contrasting environments of forest interior and open disturbance. Primary responses include increased tree mortality (Lopez et al. 2006) and additional coarse woody debris deposited on the forest floor in the edge zone (Chen et al. 1992; Harper and Macdonald 2002). Increased wind and solar radiation at exposed forest edges cause higher fluxes in humidity and soil moisture at the edge than in the forest interior (Chen et al. 1995; Matlack 1993). Secondary responses to forest edge include changes in vegetation structure and composition that are influenced by available resources and stressors at the edge (Murcia 1995. Regenerating forest edges may create a dense “sidewall” of vegetation (Didham and Lawton 1999; Duelli et al. 2002; Matlack 1994), and herbaceous growth has been shown to increase at anthropogenic edges (Harper and Macdonald 2002).
Forest edge structure and any resulting edge effect can depend greatly on the nature of the edge origin and its maintenance (Didham and Lawton 1999; Larrivee et al. 2008). In particular, linear forest openings, such as roads or powerlines, have been shown in tropical environments to exhibit edge dynamics that are very different from openings of polygonal shape (Pohlman et al. 2007). Narrow linear openings experience less incident wind and solar radiation than polygonal openings, being sheltered by adjacent forest (Forman 1995). The edges associated with clearcut logging are particularly abrupt, relative to the ragged edges associated with fire, the primary natural disturbance in boreal forests (Harper et al. 2004; McRae et al. 2001). To date, there has not been a comparison of the edge effects of linear and non-linear openings in the boreal forest. Because boreal forests are currently subject to several classes of anthropogenic disturbances that differ in key characteristics such as configuration and maintenance regime, it is important to examine whether these disturbances are equivalent in their edge effects on forest populations.
With this study, we compare the edge effects associated with three common anthropogenic disturbances in the boreal forest of Alberta: logging clearcuts, gravel roads, and buried gas pipelines. We use comparisons between sites located at varying distances from the disturbance edge and sites located within interior forest to detect edge effects on ground-dwelling spider communities, which we explore both in terms of individual species abundances across the edge and in terms of community composition. The widespread clearcuts, roads and pipelines in the Alberta landscape provide the opportunity to compare edge effects based on differences in their configuration (i.e., linear or non-linear), and also in their maintenance: while the gravel roads are used regularly by vehicles, pipeline right-of-ways are only very rarely disturbed by vehicles and are seeded with grass (Tera Environmental Consultants 2003). Therefore our objectives are to detect and compare the extent of edge effects at clearcuts, roads and pipelines on (1) individual ground-dwelling spider species, and on (2) ground-dwelling spider communities.
Methods
Study location and sampling design
Spiders were collected at study sites in Kananaskis Country, Alberta (approximately 50°58′N, 114°43′W) in the summers of 2004 and 2008. Sites were located in mixedwood coniferous forest, dominated by lodgepole pine (Pinus contorta). Other important tree species included white spruce (Picea glauca), aspen (Populus tremuloides) and subalpine fir (Abies lasiocarpa). Mature trees at study sites were roughly 20 m tall. Elevation of study sites ranged from 1,375 to 1,650 m. Transects were set up through edges of differing orientations at logging clearcuts, roads, and gas pipelines (Table 1).
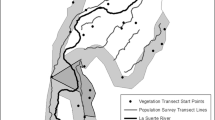
To account for regional differences in vegetation, elevation and topography, a blocked design was used where six blocks each contained one site for each of the three disturbance types (road, logging, or pipeline; Fig. 1). Blocks were usually at least 1 km from a nearest neighbor, and within a block, sites were between 200 and 800 m from a site of the nearest alternative disturbance type. Each site contained four distance levels (“stations”) arranged in a linear transect orthogonal to the disturbance edge: one 10 m into the disturbed area from the forest edge (hereafter the “0 m” station for convenience); one 10 m into the forest; one 45 m into the forest; and a final level 200 m from the disturbance edge. The edge was delineated as the line where mature trees gave way to cleared land or young regenerating trees, and was easy to distinguish. In the case of roads, because sampling directly on the road was impractical, traps at 0 m stations were placed approximately 1 m from the edge of the gravel, on the road verge.
Map of the study area showing the arrangement of clearcuts, pipelines and roads where sites were located. Grey shaded areas indicate overlap of road and buried pipeline. Numbered ovals show the locations of study blocks (six total), where each block contained a series of pitfall traps laid out in a distance transect from a clearcut edge, a pipeline edge, and a road edge. The areas shown in the two separate panes are separated by roughly 3 km. Bottom-right pane shows the location of the study area in Alberta, Canada
Each station consisted of five uncovered yellow (R: 230, G: 240, B: 80) pitfall traps, 15 cm in diameter and 10 cm deep. Yellow, uncovered traps were used so that a greater diversity of arthropods (including flower-visiting insects that are best sampled with pan traps) could be captured and included for analysis in separate studies. The traps therefore were doubly efficient as yellow pan traps (Leong and Thorp 1999) and as large pitfall traps (Work et al. 2002). To avoid detrimental effects to the many large vertebrates active in the study area, we filled traps halfway with a saturated salt solution and a few drops of detergent to reduce surface tension. The five traps within a station were spaced roughly 1 m apart in a linear row orthogonal to the disturbance edge. Eight additional control stations, each consisting of a line of five pitfall traps, were established at random points in undisturbed forest at least 200 m from any of the three disturbances. These control stations were designed to allow comparison with the 200 m site stations, in case the effects of disturbances extended beyond 200 m. Thus each disturbance-distance combination had six replicates for each year of sampling.
Traps were set twice for 9–11 days at a time between July 3 and July 13 and between July 21 and July 31 of both 2004 and 2008. Although spider species may peak in abundance at widely disparate points during the season, our sampling was timed to approximately coincide with that point during the season when species richness of ground-dwelling spiders is highest (Buddle and Draney 2004; Niemela et al. 1994). The contents of the two trapping periods during each year, and the five samples at each site, were pooled at the station level for analysis (i.e., pooling 10 samples, yielding n = 80 stations for each year).
Roughly 1 week after sampling during both summers, percent cover of plant species and ground-cover types was estimated in a 1 m2 quadrat around each pitfall trap. Because the pipeline in this area was built in the winter of 2004–2005, sampling on the pipeline disturbance was only conducted in 2008. Sampling in 2004 included sites along the proposed pipeline location (treated in analysis as control sites, or omitted) that were sampled again after pipeline construction in 2008. All other sites (control, clearcut and road edge sites) were sampled identically in 2004 and 2008.
All adult spiders were identified to species or morphospecies by external genitalia using published keys (Dondale and Redner 1978, 1982, 1990, 2003; Platnick and Dondale 1992). Identifications were made in consultation with reference collections at the University of Calgary, the University of Alberta, the Denver Museum of Nature and Science and, in some cases, with assistance from experts in spider taxonomy. Nomenclature followed Platnick (2010). Due to the difficulty of assigning most juvenile spiders to species, juveniles (which represented 12% of 12,796 individuals) were excluded from analysis.
Statistical methods
Except where otherwise noted, analysis was conducted in JMP v.8.0.2 (SAS Institute 2009). Because of some trap losses in the field and slightly unequal trapping times between sites, station-level abundances were standardized for sampling effort to 105 trap-days. We validated this ratio-based adjustment by confirming that total spider abundance was positively and significantly predicted by number of trap-days (F 1,158 = 44, P < 0.0001), and that the intercept of the regression line was zero (t 158 = 0.00, P = 0.99). To better meet model requirements for normal and homogeneous residuals, abundances were transformed by ln(x + 1). Because pipeline sites in 2004 were sampled before pipeline construction, we treated them as control sites with identical, arbitrary distances.
Our statistical approach considered edge effects of the three disturbances on individual species; on community metrics such as total spider abundance, species richness, and evenness; and on spider community composition. First, to identify spider species that were predominant within certain levels of the study design, we used indicator analysis (Dufrene and Legendre 1997) in the labdsv package for R v.2.8.1 (R Development Core Team 2008; Roberts 2007). This analysis identified spider species that were significantly associated with disturbance classes, i.e. disturbance type-distance combinations. We tested the statistical significance of indicator values with 1,000 permutations.
To assess whether the three disturbances had a significant edge effect on the abundances of individual species, we analyzed the abundance of the ten most abundant species in a mixed-model ANOVA with the following categorical X factors: block (random), year, disturbance type, distance, and disturbance type by distance interaction. We also confirmed the lack of a significant year by distance interaction in a model we do not report here. Because the pipeline was constructed in 2005, the unbalanced nature of our design made it impossible to test a year by disturbance type interaction. We compared 10 and 45 m distance levels to the 200 m level within disturbances using Tukey’s HSD, to determine the extent of edge effect at each disturbance type, if any. Where there were significant disturbance type by distance interactions, we compared distance levels for each disturbance type using Tukey’s HSD to test for edge effects occurring within each disturbance type.
We used an identical model to examine community-level measures: total spider abundance, species richness (rarefacted to a common sample size of 20 individuals), and evenness (using the ∆1 metric of Olszewski (2004)) at disturbance types and distance levels.
We used PERMANOVA (Anderson 2001), part of the PRIMER-E v.6 package (Clarke and Gorley 2006), to carry out a similar MANOVA-style analysis on spider community assemblages. PERMANOVA partitions multivariate variation on the basis of disturbance factors and analyzes interactions, random, and nested fixed effects. The analysis is performed on a distance matrix and significance of F ratios is assessed through permutation, which allowed for inclusion in the model of species whose residuals from model fits were not normal and homogeneous. We used the Bray–Curtis similarity index (Legendre and Legendre 1998) to analyze ln(x + 1)-transformed spider species abundances so that the model included the X factors year (fixed), block (random), disturbance type, and distance (fixed; nested within disturbance type).
We again used pairwise comparisons of distance levels within disturbance types to examine at what distance from the edge of each disturbance the spider community became statistically indistinguishable from “interior” communities. Because we were unable to compare specific distance levels (e.g., Logging-0 m) to control sites explicitly, we chose to instead compare distance levels within disturbance types to each other and to consider 200 m sites as equivalent to control, or forest interior. Subsequent ordination of spider communities confirmed that communities at 200 m levels within each disturbance were very similar to control communities. We used Hochberg’s (1988) procedure to control the false discovery rate in multiple comparisons.
Although PERMANOVA provides a powerful way to statistically measure the effect of different disturbance factors on community composition, it cannot be graphically displayed. Thus to provide a visual complement to PERMANOVA analysis we used non-metric multidimensional scaling (NMDS), using the Bray–Curtis similarity index, to ordinate spider community composition within different disturbance classes. We calculated centroids and 95% confidence intervals from individual sampling stations within each disturbance–distance combination. Analysis was performed with the vegan package in R v.2.8.1 (Oksanen et al. 2009; R Development Core Team 2008). To decrease the effect of rare species on analysis in PERMANOVA and NMDS, only those species occurring in at least 5% of traps over both years were included (McCune and Grace 2002).
Similar multivariate community-level analyses were carried out on the plants; percent cover records (ln(x + 1)-transformed) were combined into an overall percentage across all pitfall traps for each sampling station. We used PERMANOVA on the Bray–Curtis similarity index to explore differences in plant community composition between disturbances and distances.
Results
A total of 12,796 spiders were collected. Of these, 9,091 were adult spiders that could be identified to the species level, comprising 131 species in 16 families (for complete species list see “Appendix”, Table 6). Wolf spiders (Lycosidae), by far the most abundant family, accounted for 53% of individuals identified; individuals in Linyphiidae, the second-most abundant family, accounted for 28% of the total. Some spiders could not be identified and were excluded from analysis: morphospecies that could not be assigned to a species (n = 1,350 spiders in 121 morphospecies, nearly all in the family Linyphiidae) were excluded from analysis, except in the case of 63 individuals which were resolved to a single genus and retained as one category (Linyphiidae: Walckenaeria spp.) and 52 individuals in the genus Agyneta (Linyphiidae) which were separated from other Agyneta spp. and retained as a morphospecies (Agyneta #1).
Edge effects on individual species
Out of 127 candidate species, indicator analysis identified 16 species as significant indicators of at least one disturbance-distance class (Table 2). While 13 of these species were indicative of on-disturbance sites (5 for Logging 0 m, 6 for Pipeline 0 m, and 2 for Road 0 m), no species were characteristic of forest interior classes (i.e., 45 or 200 m from a disturbance edge). Three wolf spider species were characteristic of “edge” habitat 10 m from disturbances: Pardosa dorsuncata (Lowrie and Dondale 1981) was significantly associated with clearcut edges, while P. mackenziana (Keyserling 1877) and P. uintana (Gertsch 1933) were indicative of pipeline edges (Table 2). Because abundance within each specified class is incorporated in the indicator analysis algorithm, it is not surprising that many of the indicator species identified were among the most abundant species overall.
The abundances of the ten most abundant spider species were strongly significantly affected by distance from the disturbance edge (Table 3). The abundances of four species differed between years, and of two species were affected by disturbance type. One spider species displayed a significant disturbance type × distance interaction: Pardosa moesta (Banks 1892) was significantly more abundant on clearcuts than within the forest (Fig. 2). Most species were clearly more abundant either on-disturbance or in the forest interior, with the exception of P. uintana, which declined in abundance moving from the disturbance edge into the disturbance (Fig. 2), and showed a declining trend in the direction of the forest interior (regression F 1,88 = 4.54, P = 0.036). P. mackenziana also showed a significant edge effect, peaking in abundance at sites 10 m from the disturbance edge. None of the other abundant species showed a significant edge effect in terms of its abundance at distance levels.
Least square mean (±2SE) abundance of the most abundant species at distance (0, 10, 45, 200 m) levels. Abundances of all spider species were ln(x + 1) transformed. Capital letters indicate significant differences between levels as determined by Tukey’s HSD at P < 0.05. Pardosa moesta showed a significant interaction of distance with disturbance type, so for this species means are plotted for each disturbance type (circle = logging, cross = pipeline, diamond = road) and capital letters refer to distances within logging, the only disturbance for which distance levels differed from each other. For all other species, the distance by disturbance type interaction was not significant (Table 3). Sample size for each distance level = 30. Sample sizes for interactions as follows: all logging distance classes = 12; pipeline distance classes = 6; road distance classes = 12
Edge effects on spider communities
Spider community metrics showed generally strong distance effects (Table 3). Rarefacted species richness was significantly affected by distance from the disturbance edge but not by disturbance type (Table 3). Abundance and evenness both showed significant disturbance type by distance interactions, such that distance explained abundance only in clearcuts, and explained evenness only on pipelines (Fig. 3). Only species richness displayed a significant edge effect: richness dipped to a minimum level at 10 m from disturbance edges, and was significantly lower at 10 m sites than at sites in the forest interior (45 and 200 m).
Least square mean (±2 SE) abundance, richness and evenness of spiders (ln(x + 1)-transformed) at distance levels (0, 10, 45, 200 m). Capital letters indicate significant differences between levels as determined by Tukey’s HSD at P < 0.05. Where there was a significant disturbance type × distance interaction, separate plots are given for distance levels within each disturbance type. Sample sizes as follows: Control = 40; all logging distance classes = 12; pipeline distance classes = 6; road distance classes = 12
Viewed multivariately, spider community composition was significantly affected by year, block, disturbance type and distance (nested within disturbance type), and there were no significant interactions between year and disturbance type or distance (PERMANOVA, Table 4). Pairwise comparisons within the PERMANOVA showed a clear decay of differences in spider communities along distance transects with increasing distance from disturbance; within all disturbance types, 0 m sites were strongly different from 200 m sites. In the case of roads and pipelines, 10 and 45 m sites were not significantly different from 200 m; only in the case of logging transects was there a significant difference between distance levels that persisted to 10 m. Logging sites at 10 m were also significantly different from on-disturbance sites.
To visualize these contrasts, unconstrained ordination showed that sampling sites were separated primarily between on- and off-disturbance sites (Fig. 4). Ninety-five percent confidence intervals of centroids for distances of 10, 45 and 200 m within all disturbance types overlapped substantially with those of the control, although the centroid for Logging-10 m diverged from control along NMDS axis 3. Centroids for Logging-45 and 200 m scored higher on NMDS axis 2 than the other within-forest sites. The three on-disturbance centroids (i.e., Logging-0 m, Pipeline-0 m and Road-0 m) showed some separation along NMDS axis 2, although the confidence intervals of the Road-0 m centroid overlapped substantially with those of both Logging-0 m and Pipeline-0 m.
Three dimensional non-metric multidimensional scaling ordination of the first and second (top) and first and third (bottom) axes in an analysis of species abundance using the Bray–Curtis similarity index. Only species that appeared in at least 5% of total sampling sites were included (n = 57 species), and abundances were ln(x + 1)-transformed. Stress = 15.6. Points shown are centroids calculated from mean axis scores of disturbance type–distance combination replicates, so that each centroid depicts a disturbance type–distance combination (e.g., a filled circle represents Pipeline-200 m). Ninety-five percent confidence intervals were calculated from replicates with sample sizes as follows: control = 16; all logging distance classes = 12; pipeline distance classes = 6; road distance classes = 12
Spider species with the highest scores on NMDS axes were different from those selected by indicator analysis, and included a high proportion of linyphiid spiders (Table 5). Linyphiid species (# 3–9) occupied the positive end of axis 1 and the negative end of axis 2, while wolf spiders and crab spiders (Lycosidae, Philodromidae and Thomisidae, # 10–14) clustered together near zero on axis 2 (Fig. 5). High-scoring species were also significantly less abundant on average than indicator species (NMDS species mean ± SD: 41 ± 41 individuals; indicator species 474 ± 492 individuals; F 1,26 = 11.5, P = 0.002). Hence, indicators of distance classes were not necessarily important descriptors of community-level changes in species abundance across edges.
Spider species loadings on NMDS axes. The five species with the highest loadings on each of the three axes are shown here; species numbers correspond to Table 5. Stress = 15.6
Edge effects on vegetation
The vegetation in the on-disturbance sites was distinct from corresponding sites at 200 m from the disturbance edge, as determined by pairwise contrasts (Logging: t 5 = 3.0, P = 0.012; Pipeline: t 5 = 4.31, P = 0.002; Road: t 5 = 2.64, P = 0.004). On-disturbance sites in general were dominated by grass and bare ground (average ± SD percent cover, grass: 40 ± 28%; bare ground: 25 ± 30%), while vegetation at 200 m sites was largely composed of moss (21 ± 22%) and needles (20 ± 26%).
At clearcuts, sites 10 m from the forest edge had high levels of wood (15 ± 6%) and bare ground (8 ± 13%) compared to sites 10 m from the edges of pipelines and roads, which both had high levels of needles (Pipeline: 40 ± 26%; Road: 17 ± 21%). Clearcut 10 m sites showed a marginally significant (i.e., 0.05 < P < 0.1) difference compared to sites 200 m from the edge (10 m vs. 200 m: t 5 = 1.65, P = 0.07), while sites at 10 and 45 m from pipeline and road edges were very similar to 200 m sites (Pipeline 10 m vs. 200 m: t 5 = 0.725, P = 0.69; 45 m vs. 200 m: t 5 = 0.732, P = 0.76; Road 10 m vs. 200: t 5 = 1.173, P = 0.25; 45 m vs. 200 m: t 5 = 1.31, P = 0.19). Overall, some edge effects on vegetation were apparent at clearcut edges, while edge effects on vegetation at road and pipeline edges were negligible.
Discussion
Our objectives were to detect and compare the depth of edge influence at logging clearcuts, roads, and pipelines on ground-dwelling spider species and communities. We have shown that edge effects do not extend beyond 45 m into interior forest, but in some cases do extend up to 10 m from the disturbance edge. We have also shown that edge effects are more common at the edge of clearcuts than at roads or pipelines.
Indicator analysis, which considered only the abundances of spider species within disturbance classes and did not account for effects such as year and block, identified three species that were indicative of edge habitat. These three species were significantly associated with clearcut and pipeline edges at 10 m from the respective disturbance, suggesting that some edge effects were present at both of these disturbances. However, when we examined the abundances of ten species across edges in a full model including year and block effects, edge effects on individual species were rare (only 2 of 10 species tested). Two spider species (P. mackenziana, P. uintana) displayed edge effects up to 10 m from disturbance edges, peaking in abundance at 10 m from the edges of all disturbance types. Thus we did not find strong evidence that any of the abundant species that we examined experienced differing edge effects at the three disturbance types.
Using three univariate community metrics, we did not detect strong edge effects. The strongest was with species richness, which for all disturbances was lower at 10 m from the disturbance edge than within forest interiors. Evenness declined at 10 m from the disturbance edge in the case of pipelines, although this decline was only statistically significant relative to the disturbance. Total spider abundance was significantly higher on-disturbance than at forest interior sites for all three disturbance types, and while spider abundance remained high at 10 m into the forest from clearcuts and pipelines, it was not significantly higher than interior forest levels.
While we found little evidence of differing edge effects at roads, pipelines or clearcuts using abundance of individual spider species or simple community metrics (abundance, richness, evenness), we found that spider community composition viewed multivariately did discriminate between disturbance types. Spider assemblages at clearcut edges remained distinct from those of forest interior sites to a distance of at least 10 m, while at roads and pipelines the spider community became indistinguishable from that at interior sites within 10 m of the edge. Spider assemblages at 10 m from clearcut edges were significantly different from on-clearcut assemblages, suggesting that at 10 m from a clearcut there is a distinct “edge community.” Visualization of these patterns using NMDS ordination showed that spider assemblages at 10 m in the forest were nevertheless close in composition to control sites and sites at further distances from the clearcut edge, suggesting that the edge community at 10 m is the result of faunal mixing between on-disturbance and interior communities.
The penetration of edge influence of between 10 and 45 m, shown here both by an individual species across all disturbances and by community composition at clearcuts, is substantially smaller than many that have been reported for other invertebrates (Haskell 2000; Ries et al. 2004), but it is consistent with Larrivee et al.’s (2008) estimate of a 30 m edge effect on spider communities from a clearcut edge. It is possible that more edge effects would have been detected had sampling included distances that were smaller than 10 m from the forest edge. Edge effects in the boreal forest have been shown to be shorter in general than those reported in temperate or tropical forests (Harper and Macdonald 2002; Lopez et al. 2006).
It should be noted that only ground-active spiders were considered here and that edge effects may be different for spiders active in aboveground vegetation. Spiders that build webs in above-ground vegetation, because of their affinity with particular physical structure (Uetz 1991), could be expected to respond to changes in vegetation at forest edges to an even greater extent than those that hunt or build their webs on the forest floor. To date, studies that consider edge effects on spiders (e.g., Larrivee et al. 2008; Pajunen et al. 1995; Pearce et al. 2005) have exclusively used pitfall traps and thus are confined in application to ground-active spiders.
Although the study design makes it impossible to attribute differences in edge effects between the three disturbances to any one characteristic exclusively, for example shape, age or maintenance regime, evidence suggests that disturbance shape may be most important. Disturbance shape is important in determining the abiotic effects that are incident on forest edges. Pohlman et al. (2007) found that microclimatic gradients at the edges of linear openings in tropical forests dissipated within 25 m of the edge, which is much shorter than the penetration of microclimatic effects reported for edge effects at non-linear openings in similar forest (Laurance et al. 2002). The open space next to non-linear edges such as clearcuts leaves them more vulnerable to winds that increase in speed over the open area (Forman 1995), and they are exposed to incident solar radiation at more points during the day than are linear openings (Laurance et al. 2009). Although we did not measure abiotic features, we did find that forest edges at 10 m from clearcuts were more different from the forest interior than edges at 10 m from roads and pipelines in terms of their ground vegetation. Thus it seems possible that the greater exposure of forest edges at non-linear openings to abiotic effects causes greater penetration of edge effects on spider communities.
Apart from disturbance shape, the date of the edge origin could also contribute to different edge effects between disturbances. In eastern deciduous forests, edge effects at cut forests have been shown to change greatly over time, following succession and “aging” of the edge (Didham and Lawton 1999; Matlack 1993, 1994). The three disturbances considered here are unequal in age, the clearcuts and roads representing older, regenerating disturbances (12–19 years post-harvest) and the pipelines being much newer (within 2 years). The clearcuts and roads, constructed over the span of 8–10 years, also varied much more in age than the pipeline. Thus although the modulating effect of edge age cannot be explicitly accounted for, the fact that clearcuts and roads had similar average ages but different edge effects, and that roads and pipelines had different average ages but similar edge effects, suggests that disturbance shape is more important than age.
Like several studies of other invertebrates across forest/open space edges, we found that most spider species were clearly associated with either on-disturbance, or forest interior sites; all spider species showed strongly significant distance effects. A rich literature dealing with edge effects on carabid beetles in boreal forests suggests that most carabid species do not penetrate the edge but are likely to adhere either to the forest or the open area (Niemela et al. 2007), and this appears to be true of spiders as well. In addition, several authors have suggested that forest-associated spiders (Galle and Torma 2009; Pearce et al. 2005) and carabid beetles (Yu et al. 2010) “drift” into adjacent open spaces to a greater extent than open-associated species may drift into adjacent forests. This is consistent with our finding that species richness was significantly lower at the disturbance edge, suggesting that forest-associated species avoid the edge.
We have shown here that three common anthropogenic disturbances in the Alberta boreal forest are associated with significant impacts on spider communities. Not only do these disturbances themselves host markedly different spider communities from those that reside in interior forest, the disturbances are associated with edge effects that magnify the spatial extent of the disturbance effects on the landscape. Our finding that clearcuts are more strongly associated with edge effects than are narrow linear openings suggests that modified forestry practices could minimize the impact of tree harvest on forest spider communities. Our data indicate that silvicultural techniques such as variable retention harvest and partial cutting, especially when cut blocks are narrow, may minimize the detrimental impacts of logging on spider communities (Buddle and Shorthouse 2008).
As forests worldwide become increasingly vulnerable to fragmentation through development and deforestation, it is important to understand the ways that these human activities affect forest biota (Ewers and Didham 2008; Spence et al. 2008). The boreal forests of Alberta are subject to many sources of forest loss and disturbance due to their multiple uses as timber sources, oil and gas resources, and recreation (Schneider 2002). Compared to edges produced following forest fire, the edges that accompany clearcutting have been increasing in their extent on the landscape (Perera and Baldwin 2000). In Alberta, clearcuts are applied on the landscape in a checkerboard pattern of roughly evenly spaced 20–30 ha blocks, producing a between-cutblock forest habitat of similar size and shape to the cutblocks. The practiced pattern of imposing many small logging disturbances on the landscape, each with a large proportion of perimeter or edge, suggests that the relatively modest edge effects detected in this study are of broad significance. This study has shown that although spider communities in clearcuts, pipeline right-of-ways, and recreational roads do differ from spider communities in the forest, clearcuts are associated with the most marked edge effect of at least 10 m into undisturbed forests. In addition, this study presents some evidence that the configuration of open-space anthropogenic disturbances may have important consequences for their edge effects on spiders in boreal forests.
References
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26(1):32–46
Buddle CM, Draney ML (2004) Phenology of linyphiids in an old-growth deciduous forest in central Alberta, Canada. J Arachnol 32(2):221–230
Buddle CM, Shorthouse DP (2008) Effects of experimental harvesting on spider (Araneae) assemblages in boreal deciduous forests. Can Entomol 140(4):437–452
Buddle CM, Spence JR, Langor DW (2000) Succession of boreal forest spider assemblages following wildfire and harvesting. Ecography 23(4):424–436
Chen JQ, Franklin JF, Spies TA (1992) Vegetation responses to edge environments in old-growth Douglas-fir forests. Ecol Appl 2(4):387–396
Chen J, Franklin JF, Spies TA (1995) Growing-season microclimatic gradients from clearcut edges into old-growth Douglas-fir forests. Ecol Appl 5(1):74–86
Chen JQ, Saunders SC, Crow TR, Naiman RJ, Brosofske KD, Mroz GD, Brookshire BL, Franklin JF (1999) Microclimate in forest ecosystem and landscape ecology: variations in local climate can be used to monitor and compare the effects of different management regimes. Bioscience 49(4):288–297
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial, 6th edn. PRIMER-E Ltd, Plymouth
Clarke RD, Grant PR (1968) An experimental study of the role of spiders as predators in a forest litter community. Part 1. Ecology 49(6):1152–1154
Didham RK, Lawton JH (1999) Edge structure determines the magnitude of changes in microclimate and vegetation structure in tropical forest fragments. Biotropica 31(1):17–30
Dondale CD, Redner JH (1978) The crab spiders of Canada and Alaska (Araneae: Philodromidae and Thomisidae), vol 5. The insects and arachnids of Canada. Agriculture Canada, Ottawa
Dondale CD, Redner JH (1982) The sac spiders of Canada and Alaska (Araneae: Clubionidae and Anyphaenidae), vol 9. The insects and arachnids of Canada, vol 9. Agriculture Canada, Ottawa
Dondale CD, Redner JH (1990) The wolf spiders, nurseryweb spiders, and lynx spiders of Canada and Alaska (Araneae: Lycosidae, Pisauridae, and Oxyopidae), vol 17. The insects and arachnids of Canada. Agriculture Canada, Ottawa
Dondale CD, Redner JH (2003) The orb-weaving spiders of Canada and Alaska (Araneae: Uloboridae, Tetragnathidae, Araneidae, Theridiosomatidae), vol 23. The insects and arachnids of canada, vol 23. NRC Research Press, Ottawa
Duelli P, Obrist MK, Fluckiger PF (2002) Forest edges are biodiversity hotspots: also for neuroptera. Acta Zool Acad Sci Hung 48:75–87
Dufrene M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67(3):345–366
Ewers RM, Didham RK (2008) Pervasive impact of large-scale edge effects on a beetle community. PNAS 105(14):5426–5429. doi:10.1073/pnas.0800460105
Forman RTT (1995) Land mosaics: the ecology of landscapes and regions. Cambridge University Press, Cambridge
Galle R, Torma A (2009) Epigeic spider (Araneae) assemblages of natural forest edges in the Kiskunsag (Hungary). Community Ecol 10(2):146–151. doi:10.1556/ComEc.10.2009.2.2
Gates JE, Gysel LW (1978) Avian nest dispersion and fledging success in field-forest ecotones. Ecology 59(5):871–883
Hansen MC, Stehman SV, Potapov PV (2010) Quantification of global gross forest cover loss. PNAS 107:8650–8655
Harper KA, Macdonald SE (2002) Structure and composition of edges next to regenerating clear-cuts in mixed-wood boreal forest. J Veg Sci 13(4):535–546
Harper KA, Lesieur D, Bergeron Y, Drapeau P (2004) Forest structure and composition at young fire and cut edges in black spruce boreal forest. Can J For Res/Rev Can Rech For 34(2):289–302. doi:10.1139/x03-279
Harper KA, Macdonald SE, Burton PJ, Chen JQ, Brosofske KD, Saunders SC, Euskirchen ES, Roberts D, Jaiteh MS, Esseen PA (2005) Edge influence on forest structure and composition in fragmented landscapes. Conserv Biol 19(3):768–782
Harrison S, Bruna E (1999) Habitat fragmentation and large-scale conservation: what do we know for sure? Ecography 22(3):225–232
Haskell DG (2000) Effects of forest roads on macroinvertebrate soil fauna of the southern Appalachian mountains. Conserv Biol 14(1):57–63
Heliola J, Koivula M, Niemela J (2001) Distribution of carabid beetles (Coleoptera, Carabidae) across a boreal forest-clearcut ecotone. Conserv Biol 15(2):370–377
Hochberg Y (1988) A sharper Bonferroni procedure for multiple tests of significance. Biometrika 75(4):800–802
Jansson C, von Bromssen A (1981) Decline of spiders and insects in Spruce (Picea abies) and its relation to predation by birds. Holarctic Ecol 4(2):82–93
Langellotto GA, Denno RF (2004) Responses of invertebrate natural enemies to complex-structured habitats: a meta-analytical synthesis. Oecologia 139(1):1–10. doi:10.1007/s00442-004-1497-3
Larrivee M, Drapeau P, Fahrig L (2008) Edge effects created by wildfire and clear-cutting on boreal forest ground-dwelling spiders. For Ecol Manag 255(5–6):1434–1445
Laurance WF, Curran TJ (2008) Impacts of wind disturbance on fragmented tropical forests: a review and synthesis. Austral Ecol 33(4):399–408. doi:10.1111/j.1442-9993.2008.01895.x
Laurance WF, Laurance SG, Ferreira LV, RankindeMerona JM, Gascon C, Lovejoy TE (1997) Biomass collapse in Amazonian forest fragments. Science 278(5340):1117–1118
Laurance WF, Lovejoy TE, Vasconcelos HL, Bruna EM, Didham RK, Stouffer PC, Gascon C, Bierregaard RO, Laurance SG, Sampaio E (2002) Ecosystem decay of Amazonian forest fragments: a 22-year investigation. Conserv Biol 16(3):605–618
Laurance WF, Nascimento HEM, Laurance SG, Andrade A, Ewers RM, Harms KE, Luizao RCC, Ribeiro JE (2007) Habitat fragmentation, variable edge effects, and the landscape-divergence hypothesis. PLoS One 2(10):1017
Laurance WF, Goosem M, Laurance SGW (2009) Impacts of roads and linear clearings on tropical forests. Trends Ecol Evol 24(12):659–669. doi:10.1016/j.tree.2009.06.009
Legendre P, Legendre L (1998) Numerical Ecology, Second English edn. Elsevier, Amsterdam
Leong JM, Thorp RW (1999) Colour-coded sampling: the pan trap colour preferences of oligolectic and nonoligolectic bees associated with a vernal pool plant. Ecol Entomol 24(3):329–335
Leopold A (1933) Game management. Charles Scribner’s Sons, New York
Lopez LEM, Harper KA, Drapeau P (2006) Edge influence on forest structure in large forest remnants, cutblock separators, and riparian buffers in managed black spruce forests. Ecoscience 13(2):226–233
Magura T, Tothmeresz B, Molnar T (2001) Forest edge and diversity: carabids along forest-grassland transects. Biodivers Conserv 10(2):287–300
Matlack GR (1993) Microenvironment variation within and among forest edge sites in the eastern United States. Biol Conserv 66(3):185–194
Matlack GR (1994) Vegetation dynamics of the forest edge: trends in space and successional time. J Ecol 82(1):113–123
McCune B, Grace J (2002) Analysis of ecological communities. MJM Software Design, Gleneden Beach
McRae DJ, Duchesne LC, Freedman B, Lynham TJ, Woodley S (2001) Comparisons between wildfire and forest harvesting and their implications in forest management. Environ Rev 9:223–260
Mullen K, Fahy O, Gormally M (2003) Ground flora and associated arthropod communities of forest road edges in Connemara, Ireland. Biodivers Conserv 12(1):87–101
Murcia C (1995) Edge effects in fragmented forests: implications for conservation. Trends Ecol Evol 10(2):58–62
Niemela J, Pajunen T, Haila Y, Punttila P, Halme E (1994) Seasonal activity of boreal forest-floor spiders (Araneae). J Arachnol 22(1):23–31
Niemela J, Koivula M, Kotze DJ (2007) The effects of forestry on carabid beetles (Coleoptera: Carabidae) in boreal forests. J Insect Conserv 11(1):5–18. doi:10.1007/s10841-006-9014-0
Oksanen J, Kindt R, Legendre P, O’Hara B, Simpson GL, Solymos P, Stevens MHH, Wagner H (2009) Package vegan R: community ecology package. http://www.cran.r-project.org/
Olszewski TD (2004) A unified mathematical framework for the measurement of richness and evenness within and among multiple communities. Oikos 104(2):377–387. doi:10.1111/j.0030-1299.2004.12519.x
Pajunen T, Haila Y, Halme E, Niemela J, Punttila P (1995) Ground-dwelling spiders (Arachnida: Araneae) in fragmented old forests and surrounding managed forests in southern Finland. Ecography 18(1):62–72. doi:10.1111/j.1600-0587.1995.tb00119.x
Pearce JL, Venier LA (2006) The use of ground beetles (Coleoptera: Carabidae) and spiders (Araneae) as bioindicators of sustainable forest management: a review. Ecol Indic 6(4):780–793
Pearce JL, Venier LA, Eccles G, Pedlar J, McKenney D (2005) Habitat islands, forest edge and spring-active invertebrate assemblages. Biodivers Conserv 14(12):2949–2969
Perera AH, Baldwin DJB (2000) Spatial patterns in the managed forest landscape of Ontario. In: AH P, DL E, ID T (eds) Ecology of a managed terrestrial landscape: patterns and processes in forest landscapes of Ontario. University of British Columbia Press, Vancouver, pp 74–99
Platnick NI (2010) The World Spider Catalogue, Version 10.5. The American Museum of Natural History, Washington DC
Platnick N, Dondale CD (1992) The ground spiders of Canada and Alaska (Araneae: Gnaphosidae), vol 19. The insects and arachnids of Canada. Agriculture Canada, Ottawa
Pohlman CL, Turton SM, Goosem M (2007) Edge effects of linear canopy openings on tropical rain forest understory microclimate. Biotropica 39(1):62–71
R Development Core Team (2008) R v.2.8.1: a language and environment for statistical computing. 2.8.1 edn. R Foundation for Statistical Computing, Vienna
Ries L, Sisk TD (2008) Butterfly edge effects are predicted by a simple model in a complex landscape. Oecologia 156(1):75–86
Ries L, Fletcher RJ, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Ann Rev Ecol Evol Syst 35:491–522
Roberts D (2007) Package labdsv R: ordination and multivariate analysis for ecology. http://www.cran.r-project.org/
Robinson JV (1981) The effect of architectural variation in habitat on a spider community: an experimental field study. Ecology 62(1):73–80
SAS Institute (2009) JMP 8.0.2. Cary, NC
Schneider RR (2002) Alternative futures: Alberta’s boreal forest at the crossroads. Alberta Centre for Boreal Research, Edmonton AB
Spence JR, Langor DW, Jacobs JM, Work TT, Volney WJA (2008) Conservation of forest-dwelling arthropod species: simultaneous management of many small and heterogeneous risks. Can Entomol 140(4):510–525
Taboada A, Kotze DJ, Salgado JM (2004) Carabid beetle occurrence at the edges of oak and beech forests in NW Spain. Eur J Entomol 101(4):555–563
Tera Environmental Consultants (2003) Environmental protection plan. Shell Canada Limited Moose Mountain Interconnect Pipeline Project, Calgary
Turnbull AL (1973) Ecology of true spiders (Araneomorphae). Annu Rev Entomol 18:305–348
Tylianakis JM, Klein AM, Tscharntke T (2005) Spatiotemporal variation in the diversity of Hymenoptera across a tropical habitat gradient. Ecology 86(12):3296–3302
Uetz GW (1991) Habitat structure and spider foraging. In: McCoy ED, Bell SA, Mushinsky HR (eds) Habitat structure: the physical arrangement of objects in space. Chapman & Hall, pp 325–348
Vlijm L, Kessler-Geschiere A (1967) Phenology and habitat of Pardosa monticola, P. Nigriceps and P. Pullata (Araneae: Lycosidae). J Anim Ecol 36(1):31–56
Woodroffe R, Ginsberg JR (1998) Edge effects and the extinction of populations inside protected areas. Science 280(5372):2126–2128
Work TT, Buddle CM, Korinus LM, Spence JR (2002) Pitfall trap size and capture of three taxa of litter-dwelling arthropods: Implications for biodiversity studies. Environ Entomol 31(3):438–448
Yu XD, Luo TH, Zhou HZ (2010) Distribution of ground-dwelling beetle assemblages (Coleoptera) across ecotones between natural oak forests and mature pine plantations in north China. J Insect Conserv 14(6):617–626
Ziesche TM, Roth M (2008) Influence of environmental parameters on small-scale distribution of soil-dwelling spiders in forests: what makes the difference, tree species or microhabitat? For Ecol Manag 255(3–4):738–752
Acknowledgments
Many thanks to those who assisted with the identification of spider specimens (Jaime Pinzon, Dr. Rob Bennett, Joey Slowik and Dr. Wayne Maddison), fieldwork and arthropod sorting (Dayna Chetek, Stu Crawford, Amy Darling, Portia Lloyd, Sierra Love, Natasha Myers, April Matisz, Chelsea Matisz, and Gennifer Meldrum). We also thank Dr. John Swann for collections support; Canbra Foods of Lethbridge for materials donations; and the University of Calgary Faculty of Graduate Studies, Shell Canada Limited, Husky Oil, Alberta Ingenuity, and NSERC Discovery Grants for funding support. Shell Canada Limited and Husky Oil funded the bulk of the research.
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
About this article
Cite this article
Kowal, V.A., Cartar, R.V. Edge effects of three anthropogenic disturbances on spider communities in Alberta’s boreal forest. J Insect Conserv 16, 613–627 (2012). https://doi.org/10.1007/s10841-011-9446-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-011-9446-z