Abstract
Carbon dioxide (CO2) recovery with high alkalinity microalgal culture is expected to be an energy-efficient and environmentally friendly process. To increase the CO2 recovery efficiency, selection of rapidly growing alkalihalophilic microalgae is necessary. This study optimized the culture conditions of three species of alkalihalophilic microalgae, Arthrospira platensis, Dunaliella salina, and Euhalothece sp., and compared their CO2 fixation potential. Although D. salina tolerated relatively high dissolved inorganic carbon (DIC; 0.50 mol L−1), its carbon fixation rate was found to be slower than the other two species. The two cyanobacteria, A. platensis and Euhalothece sp., favored high pH (9.8–10) and high DIC (0.23–1.1 mol L−1). Euhalothece sp. grew in the highest alkalinity, resulting in the strongest pH buffer against acidification during CO2 absorption. However, the carbon fixation properties of A. platensis and Euhalothece sp. under the same light condition were found to be similar (33 and 35 mmol L−1 day−1). These results indicate that the carbon fixation potential per medium inorganic carbon was higher in A. platensis than in the others. Arthrospira platensis was found to be favorable in a CO2 recovery process unless extremely high pH stability is needed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The effects of global warming on earth’s ecology, weather, and geology have become more apparent, and immediate actions need to be taken. One of the intensively studied countermeasures is carbon dioxide (CO2) recovery at CO2 point sources such as thermal power plants, steel mills, and chemical plants. Although conventional chemical absorption and pressure swing absorption processes have high CO2 recovery efficiencies, the high energy demand for the regeneration of their absorption reagent is not favorable as a sustainable process (González-López et al. 2012). Algal CO2 recovery processes, on the other hand, are considered as relatively less energy-intensive processes, since they can utilize renewable solar energy. In addition, application of algal biomass such as for feed, raw materials, and biofuel is expected to reduce the total CO2 emissions (Walsh et al. 2015), and algal bioproducts could potentially support profitable operations (e.g. Radmer and Parker 1994; Spolaore et al. 2006; Rosenberg et al. 2008, 2011).
Despite the various advantages of algal CO2 recovery processes, there are still some challenges to overcome. Firstly, since common algal species prefer neutral to slightly basic pH with relatively low alkalinity, the medium possesses low CO2 absorption capacity. Secondly, absorption of CO2 into the medium reduces the medium pH, inhibiting the growth of algae and reducing the CO2 recovery efficiency. In order to solve these issues, the use of alkaline medium with high dissolved inorganic carbon (DIC; bicarbonate and carbonate) has been proposed (Chi et al. 2011; González-López et al. 2012). The increased alkalinity under such medium condition increases both CO2 solubility and pH stability. These studies propose a liquid circulating system between photobioreactor and CO2 absorption unit, since CO2-enriched absorbing reagent is regenerated through photosynthetic utilization of DIC, and pH of both reactors can be maintained at a constant value by a constant circulation.
In this circulating algal CO2 recovery processes, the amount of CO2 fixed by algae and the amount of CO2 supplied to the system should be kept in balance. In this term, improvement of the volumetric algal carbon fixation rate is necessary to realize an efficient algal CO2 recovery system. Since the bicarbonate/carbonate process applies medium with high alkalinity, fast-growing alkalihalophilic microalgal species need to be selected. Arthrospira (Spirulina) platensis is one of the most widely studied alkalihalophilic microalgae (Lee 1997; Borowitzka 1999), and it is characterized by high growth rate, easiness of harvest, and ease of cultivation (Vonshak 1997). Dunaliella salina is a halophilic green microalga commercialized for carotenoid production and noted for its high tolerance against heat and alkaline conditions when sodium chloride (NaCl) concentration is high (Ben-Amotz et al. 2009). Euhalothece sp. is a recently isolated alkalihalophilic cyanobacterium with high productivity (Mikhodiuk et al. 2008; Gerasimenko and Mikhodyuk 2009; Chi et al. 2013). To date, only few studies have compared the productivity of alkalihalophilic microalgae after optimization of culture conditions.
For the comparison of different algal species, the difference between specific growth rate (day−1) and biomass productivity (g L−1 day−1) needs to be considered. The specific growth rate is an indicator of how often the algal cells can divide, which does not depend on the initial cell density, but merely on the culture conditions such as light intensity, temperature, and salinity. The specific growth rate is therefore a suitable parameter for the optimization of culture conditions. On another hand, it is necessary to compare the biomass production in the view of CO2 capture capacity of an algal species, because it is a direct measure of the amount of carbon the algae have assimilated. The biomass productivity of a batch algal culture can be evaluated during the linear growth phase, where the growth is not limited by nutrients or inorganic carbon, but by light (Lee et al. 2013). By comparing the biomass productivities in the linear growth phase of different microalgal species under the same irradiance, algal intrinsic potential of carbon fixation can be measured. The comparison during linear growth phase is especially relevant to the practical application of algal CO2 recovery, in which algae should be cultivated under a continuous process to obtain stable carbon fixation rates. During the steady-state of a continuous process, light availability determines the biomass productivity, as with the case in the linear growth phase of a batch process. Thus, to evaluate the potential use of the three species for CO2 recovery, (1) culture condition should be optimized for specific growth rate, and (2) maximum biomass productivity should be compared under the same light conditions.
Therefore, in this study, the culture conditions of three alkalihalophilic microalgae, A. platensis, D. salina, and Euhalothece sp., were optimized using the response surface methodology, and their carbon fixation characteristics were compared under the optimum conditions.
Materials and methods
Algal strains, growth media, and culture conditions
Three alkalihalophilic microalgae, Arthrospira platensis NIES-39 (cyanobacterium), Dunaliella salina NIES-2257 (green alga), and Euhalothece sp. Z-M001 (cyanobacterium), were used. Arthrospira platensis was cultured in SOT medium (Ogawa and Terui 1970), D. salina in Ramaraj medium (Sathasivam et al. 2013), and Euhalothece sp. in M medium (Mikhodiuk et al. 2008). To avoid precipitation during autoclaving, the media were separated into two batches; the first consisting of NaHCO3, Na2CO3, and K2HPO4, and the second of the other medium components. Both batches were autoclaved at 121 °C for 20 min and mixed after cooling to room temperature. The algae were pre-cultured in Erlenmeyer flasks under cool-white fluorescent light at 160 μmol photons m−2 s−1. Arthrospira platensis and Euhalothece sp. were cultured at 35 °C, and D. salina was cultured at 25 °C. The media were stirred with a magnetic stirrer continuously at 350 rpm to avoid flocculation. Algal cells at a late exponential growth phase were used as inoculum for the experiments.
Growth optimization
A batch growth optimization study was conducted based on central composite design (CCD). It was aimed to clarify the effect and the optimal values of temperature, pH, DIC, and NaCl concentration. In a preliminary experiment, the range of DIC and NaCl concentrations for the growth of each microalga were determined. The results suggested that relatively low DIC concentration was suitable for the growth of A. platensis and D. salina. Therefore, face-centered design (α = 1) was adopted for the two species, because it allows optimization within the entire tested range, unlike rotatable design (α ≥ 1.414) which requires reference points outside the test range (NIST/SEMATECH 2015). In face-centered design, each factor has three levels, while rotational design has five levels (Table 1), and the range of optimization was set based on previous studies (Vonshak 1997; Rodrigues et al. 2010) and the above-mentioned preliminary experiments. For each strain, a total of 30 runs were carried with six replicates for the center point.
Experimental procedure
Media with respective pH, DIC, and NaCl concentrations were inoculated, and 8 mL was dispensed into 16-mL glass test tubes and incubated at various temperatures (Table 1) for 7 days. In order to exclude the effect of pH change with algal growth, pH was adjusted once a day with 1 or 10 N HCl and NaOH. The optical density at 750 nm (OD750) was measured immediately before the pH adjustments as a proxy of algal cell density. Specific growth rates (day−1) at log-growth phase were used for the response surface analysis.
For the fitting of the result, the following quadratic model was selected:
where Y is the predicted response, β i are the coefficients, and x i is the coded levels of variable i. The software Design-Expert 9 (Stat-Ease Inc., USA) was used for construction and analysis of the model.
Growth evaluation in batch cultures
The three microalgae were cultured in 500-mL Erlenmeyer flasks under the optimized conditions to evaluate the carbon fixation properties. The initial pH was set to the optimized values with 1 N NaOH. The DIC concentrations were adjusted with NaHCO3. The same light condition, 160 μmol photons m−2 s−1 with 24 h light, was adopted for all the cultures. The average growth rates at linear growth phase were evaluated. Each microalga was cultured in triplicate. Since the pH of A. platensis was raised to an inhibitive level (>11) during preliminary culture, it was controlled by automatic pH control with CO2 addition.
Analytical procedures
Optical density (OD) was measured with a portable spectrophotometer (Model DR2800 Spectrophotometer, Hach, USA) in the culture optimization study. For algal cell dry weight (DW) measurement, algal suspensions were filtered through pre-weighed glass fiber filters with pore size of 0.7 μm (GF/F, Whatman, USA) and washed with distilled water three times. Filters were dried in an oven at 60 °C for over 24 h and then cooled to room temperature in a desiccator before weighing. The relations between OD750 and DW were evaluated for each species, and OD values were converted into DW using (c.f., Online Resource Fig. S1). Specific growth rate (μ; day−1) during log-growth phase was calculated using the following equation:
where x i is the biomass concentration (gDW L−1) at time t i . Algal biomass productivity (P x ; gDW L−1 day−1) was calculated with the following equation:
Statistical analysis
Results are expressed as means ± standard deviations of the mean, where available. The correlation coefficients were obtained using simple regression analysis (Excel software). Multiple comparison between average biomass productivity was made by Tukey-Kramer test. Differences with P < 0.05 were considered significant.
Results and discussion
Growth optimization
Culture optimization with central composite design (CCD) was successful in creating significant models (P < 0.001 for all strains) that effectively predicted specific growth rate using A-temperature, B-pH, C-DIC, and D-NaCl concentration (Table 2).
Arthrospira platensis NIES-39
The multiple regression analysis revealed that temperature, pH, and DIC had significant effects on the specific growth rate of A. platensis (P < 0.05; Table 2). Temperature and pH also showed significant negative quadratic relations (A2 and B2), which imply that there are optimum values within the tested range. Since the size of coefficients in the coded model indicates the relative importance of the parameters, it can be said that temperature and pH had the most important role in determining the growth, having −0.062 and −0.092 for A and A2, and −0.043 and −0.170 for B and B2, respectively (Table 2). On the other hand, NaCl had little effect on growth within the tested range, as relatively small coefficients were found for any variables related to NaCl.
Response surfaces were graphed using the prediction model (Fig. 1). The growth rate was predicted to be high at culture temperatures of around 32 to 36 °C (Fig. 1a), having the maximum point at 34 °C. Optimum range of pH was 9.5–10.0, and the maximum point was 9.8. Arthrospira platensis grew well in the DIC range of 0.16–0.3 mol L−1, having maximum at 0.23 mol L−1. NaCl had little effects on the growth rate within the tested range (0.017–0.17 mol L−1), and the contour showed a saddleback shape, in which no optimum value is determined within the factorial design. Within the design space, the highest growth rate was observed with a NaCl concentration of 0.17 mol L−1.
Dunaliella salina NIES-2257
Dunaliella salina grew relatively more slowly than other two species (max. 0.17 day−1). In the ANOVA analysis of the multiple regression parameters, it was found that NaCl concentration had the strongest effects on the growth (Table 2). The coefficient for NaCl (D) was positive (0.056), indicating that higher concentration of NaCl was more favorable to the growth of this alga. Furthermore, there was a significant interrelationship between DIC and NaCl, indicating that sodium concentration was key to the growth of D. salina. The lowest sodium concentration (low DIC and low NaCl) was predicted to output the lowest growth rate within the tested region. The pH also had a significant effect on the growth, with higher pH improving the growth rate.
Dunaliella salina exhibited its fastest growth at a lower temperature (20 °C), especially when pH was high (pH 8–9; Fig. 1). The highest growth was observed with NaCl concentration of 2 mol L−1 and pH of 9.0. Although the effect of DIC was not significant, the best growth was observed at high DIC concentration, which could be suitable for CO2 absorption. However, the highest growth rate only went up to 0.17 day−1, which was less than half of that of A. platensis.
Euhalothece sp. Z-M001
The model parameter analysis revealed that temperature and pH were the most influential factors for Euhalothece sp. The growth was high at high temperatures. The previous study reported that Euhalothece sp. Z-M001 grows faster at 40 °C than at 35 °C, but the growth at 40 °C was susceptible to change in conditions and instability of culture (Chi et al. 2014). Since all the quadratic coefficients of ANOVA analysis revealed negative values, it is suggested that there are optimized points within tested space. Euhalothece sp. was able to grow well at the highest pH (10) and DIC concentration (1.1 mol L−1) among others.
Comparison of the optimum conditions
This study revealed the optimum temperature, pH, DIC, and NaCl concentration of three alkalihalophilic microalgae (Table 3). The two cyanobacteria, A. platensis and Euhalothece sp., exhibited high specific growth rates and presented a similar optimum temperature and pH. However, Euhalothece sp. was more resistant to high DIC and NaCl concentration. The total ion concentration of optimized M medium (Euhalothece sp.) was approximately 1.7 mol L−1, while that of SOT medium (A. platensis) was 0.28 mol L−1. Since some soda lakes have highly concentrated salts during dry season evaporation (Mikhodiuk et al. 2008), Euhalothece sp. could have been adapted to such concentrated environment.
Growth characteristics of D. salina differed from the other two species. The maximum specific growth rate of D. salina was less than half of the other two species. The optimum ion concentration of D. salina in this study was as high as approximately 2.5 mol L−1. The DIC to NaCl ratio of the optimum medium was 0.25, while those of A. platensis and Euhalothece sp. were 1.35 and 2.34. This lower ratio of optimum DIC was probably because this strain was originally collected from a saline lake with a high concentration of salt, while the other two were collected from soda lakes with high alkalinity.
Compared with other commonly cultivated algal strains, the optimized growth conditions of the tested strains were higher in DIC and pH (Fig. 2). For example, the growth condition of Chlorella vulgaris ESP-31 was reported to be about DIC 0.014 mol L−1 and pH 6.2 (Yeh et al. 2010). On the other hand, the optimum DIC concentration was 0.23, 0.5, and 1.1 mol L−1, and the optimum pH was 9.8, 9, and 10 for A. platensis, D. salina, and Euhalothece sp., respectively. The high DIC concentration is beneficial to CO2 absorption process, since it prevents pH reduction during CO2 absorption, ending up increasing the total amount of CO2 a liter of the medium can absorb.
Optimum pH and dissolved inorganic carbon (DIC) of previous and these studies. White circles represent previously reported data, and black rectangles represent this study. a Synechococcus sp. (Silva et al. 2016). b Thermosynechococcus sp. (Su et al. 2012). c Chlorella protothecoides (Gris et al. 2014). d Chlorella vulgaris (Yeh et al. 2010). e Scenedesmus sp. (Pancha et al. 2015). f Scenedesmus sp. (Tripathi et al. 2015). g Haematococcus pluvialis (Kang et al. 2005). h Nannochloropsis salina (White et al. 2013)
To evaluate the pH buffer function during CO2 absorption, the change in pH with CO2 absorption/desorption was modeled using the equation below (Fig. 3):
where K 1 and K 2 are the dissociation constants of bicarbonate and carbonate ion, K w is the water dissociation constant, c 0 is the NaHCO3 concentration, c b is the NaOH concentration for the adjustment of pH, and ΔDIC is the change in DIC from the initial value due to absorption/desorption of CO2. Although A. platensis (SOT medium) had pH buffer much stronger than non-buffered medium, much higher pH stability was observed in Euhalothece sp. (M medium) owing to its extremely high alkalinity (Fig. 3). While the pH of optimized A. platensis medium (SOT medium) reduces to less than 9 with 0.08 mol L−1 of CO2, that of Euhalothece sp. (M medium) remains above 9 until 0.4 mol L−1 of CO2 is absorbed (Fig. 3). The high pH buffer capacity enables culture stability during CO2 absorption process.
Based on the response surface analysis, the maximum specific growth rates were predicted to be 0.48, 0.17, and 0.43 day−1 for A. platensis, D. salina, and Euhalothece sp., respectively. The growth of A. platensis was among the highest, but the highest optimum DIC concentration was found with Euhalothece sp., which may be beneficial for CO2 absorption process. In order to compare the CO2 fixation properties, biomass productivity (g L−1 day−1) under optimum conditions was evaluated.
Carbon fixation properties of three microalgae under optimized conditions
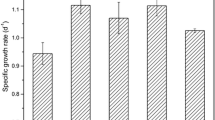
Batch cultures of the three microalgal species were conducted to test their carbon fixation properties at the light-limiting condition. All cultures clearly exhibited log-growth phase, linear growth phase, and stationary phase (Fig. 4a). During the exponential growth phase, the maximum specific growth rates of A. platensis, D. salina, and Euhalothece sp. were 1.12, 0.63, and 1.67 day−1, respectively (Fig. 4b). All the specific growth rates were faster than the respective values during the optimization experiment, probably owing to the improved agitation in the current experiment, since other conditions such as light and aeration remained the same.
During the stationary phase, the average biomass productivity of D. salina was significantly lower than those of A. platensis and Euhalothece sp. (P < 0.001); the difference was approximately three times (Table 4). There was no significant difference between the average biomass productivities of A. platensis and Euhalothece sp. (P > 0.05). According to the calculation, A. platensis and Euhalothece sp. were able to assimilate 0.391 and 0.422 gC L−1 day−1 of inorganic carbon, which is equal to fixation of 0.73 and 0.79 NL-CO2 L−1 day−1, respectively. The biomass productivity (g L−1 day−1) becomes higher when maintenance energy (i.e. respiration) is low and light utilization efficiency is high (Lee et al. 2013). The efficiency can be modeled with the equation:
where μ max is the maximum specific growth rate, I is the photon flux density, and K I is the light saturation constant, that is the photon flux density required to achieve half of the maximum specific growth rate (Lee et al. 2013). It is reported that K I of green algae is high compared to cyanobacteria, diatoms, and dinoflagellates, and green algae have higher respiration rate than others (Richardson et al. 1983). With high K I value, stronger incident light is necessary to achieve the same growth rate. It is inferred that light utilization efficiencies of Euhalothece sp. and A. platensis were higher than that of D. salina under the tested light condition. The low CO2 fixation rate of D. salina NIES-2257 suggests that this strain may not be a suitable strain for a CO2 recovery process.
As mentioned above, Euhalothece sp. exhibited a strong buffer function (Fig. 3). However, the requirement of high bicarbonate/carbonate in the medium may result in a great quantity of chemical additives to raise medium alkalinity. The CO2 absorption capacity per medium DIC was more than four times higher in A. platensis than in Euhalothece sp. (Table 4). While the source of inorganic carbon can be supplied from waste gases (e.g. flue gases), alkaline chemicals such as sodium hydroxide (NaOH) are necessary to raise the pH and alkalinity to the optimum values. For example, theoretically, 1.8 mol L−1 of NaOH is required to prepare optimized M medium, while less, 0.34 mol L−1, is needed for the SOT medium. In addition, unnecessary DIC in the medium may increase the amount of untreated DIC into the wastewater stream, which may eventually enter the natural environment. If a larger amount of DIC than the amount recovered from the CO2 recovery process is discharged, the net CO2 balance can be negative.
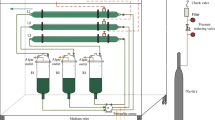
Circulation of cultured medium back to the CO2 absorption column would alleviate the chemical cost and CO2 balance, as suggested previously (Chi et al. 2011). The suggested circulating CO2 recovery process is shown in Fig. 5. The medium in a CO2 absorption column semi-continuously flows into an algal photobioreactor, at which DIC is photosynthetically fixed into algal biomass. Harvested biomass is the fixed carbon output from the system. The medium is circulated back to the absorption column (Fig. 5). For the nutrient supply and the medium sterilization, a certain amount of medium needs to be replaced periodically, at a dilution rate D (day−1). The addition of DIC into the system (DIC in ; mmol-C L−1 day−1) is
where DIC m is the optimized DIC concentration of SOT medium (230 mmol-C L−1) and M medium (1100 mmol-C L−1). In order to at least balance the medium DIC input and CO2 recovery, DIC in needs to be lower than the daily volumetric carbon fixation rate, which was found to be 33 and 35 mmol-C L−1 day−1 for A. platensis and Euhalothece sp., respectively, in this study. According to the above calculation, the dilution rate (D) of A. platensis and Euhalothece sp. needs to be lower than 0.14 and 0.03 day−1, respectively. These dilution rates correspond to HRT of 7 and 33 days. From a carbon footprint viewpoint, A. platensis is nearly five times more efficient in CO2 fixation than Euhalothece sp. based on the DIC requirement in the medium.
In this study, the two cyanobacteria, A. platensis and Euhalothece sp., were found to efficiently fix CO2 in different levels of alkaline-carbonate conditions, compared to a lower fixation rate of D. salina. Euhalothece sp. was found to tolerate extremely high alkalinity, which enables pH stability of the system and a much greater CO2 absorption capacity. This high alkalinity system may be beneficial for a process that requires large CO2 holding capacities, such as those which require transportation of absorbent between a CO2 source (e.g. a power plant) and algal bioreactors, since it reduces the required liquid volume of transportation. On the other hand, A. platensis was found to fix CO2 efficiently with less requirement of medium DIC. The comparably lower pH buffer capacity than that of Euhalothece sp. requires frequent circulation between an absorption column and the algal bioreactor for pH stabilization (Meier et al. 2015; Toledo-Cervantes et al. 2016). The high CO2 fixation efficiency per added DIC of A. platensis enables relatively rapid replacement of culture medium. Therefore, A. platensis may be suitable for a relatively small facility that produces high-value products that require greater sterility.
Conclusions
This study compared the carbon fixation characteristics of three alkalihalophilic algae, A. platensis, D. salina, and Euhalothece sp., under optimized culture conditions. Euhalothece sp. was tolerant to the highest alkalinity, enabling a stable medium pH during CO2 absorption. On the other hand, the CO2 recovery efficiency per medium DIC was superior in A. platensis. The findings suggest that, if little variation in CO2 supply rate or algal growth rate is expected, too high alkalinity may not be always desirable.
References
Ben-Amotz A, Polle JEW, Subba Rao DV (eds) (2009) The alga Dunaliella: biodiversity, physiology, genomics and biotechnology. Science Publishers
Borowitzka MA (1999) Commercial production of microalgae: ponds, tanks, tubes and fermenters. J Biotechnol 70:313–321
Chi Z, Elloy F, Xie Y, Hu Y, Chen S (2014) Selection of microalgae and cyanobacteria strains for bicarbonate-based integrated carbon capture and algae production system. Appl Biochem Biotechnol 172:447–457
Chi Z, O’Fallon JV, Chen S (2011) Bicarbonate produced from carbon capture for algae culture. Trends Biotechnol 29:537–541
Chi Z, Xie Y, Elloy F, Zheng Y, Hu Y, Chen S (2013) Bicarbonate-based integrated carbon capture and algae production system with alkalihalophilic cyanobacterium. Bioresour Technol 133:513–521
Gerasimenko LM, Mikhodyuk OS (2009) Halophilic algal-bacterial and cyanobacterial communities and their role in carbonate precipitation. Paleontol J 43:940–957
González-López CV, Acién Fernández FG, Fernández-Sevilla JM, Sánchez Fernández JF, Molina Grima E (2012) Development of a process for efficient use of CO2 from flue gases in the production of photosynthetic microorganisms. Biotechnol Bioeng 109:1637–1650
Gris B, Sforza E, Vecchiato L, Bertucco A (2014) Development of a process for an efficient exploitation of CO2 captured from flue gases as liquid carbonates for Chlorella protothecoides cultivation. Ind Eng Chem Res 53:16678–16688
Kang CD, Lee JS, Park TH, Sim SJ (2005) Comparison of heterotrophic and photoautotrophic induction on astaxanthin production by Haematococcus pluvialis. Appl Microbiol Biotechnol 68:237–241
Lee Y-K (1997) Commercial production of microalgae in the Asia-Pacific rim. J Appl Phycol 9:403–411
Lee Y-K, Chen W, Shen H, Han D, Li Y, Jones HDT, Timlin JA, Hu Q (2013) Basic culturing and analytical measurement techniques. In: Richmond A, Hu Q (eds) Handbook of microalgal culture: applied phycology and biotechnology, 2nd edn. John Wiley & Sons, Ltd., pp 37–68
Meier L, Pérez R, Azócar L, Rivas M, Jeison D (2015) Photosynthetic CO2 uptake by microalgae: an attractive tool for biogas upgrading. Biomass Bioenergy 73:102–109
Mikhodiuk OS, Gerasimenko LM, Akimov VN, Ivanovskiĭ RN, Zavarzin GA (2008) Ecophysiology and polymorphism of the unicellular extremely natronophilic cyanobacterium Euhalothece sp. Z-M001 from Lake Magadi. Mikrobiologiia 77:805–813
NIST/SEMATECH (2015) e-Handbook of Statistical Methods. In: E-handb. Stat. Methods. http://www.itl.nist.gov/div898/handbook/
Ogawa T, Terui G (1970) Studies on the growth of Spirulina platensis: (I) on the pure culture of Spirulina platensis. J Ferment Technol 48:361–367
Pancha I, Chokshi K, Ghosh T, Paliwal C, Maurya R, Mishra S (2015) Bicarbonate supplementation enhanced biofuel production potential as well as nutritional stress mitigation in the microalgae Scenedesmus sp. CCNM 1077. Bioresour Technol 193:315–323
Radmer RJ, Parker BC (1994) Commercial applications of algae: opportunities and constraints. J Appl Phycol 6:93–98
Richardson K, Beardall J, Raven JA (1983) Adaption of unicellular algae to irradiance: an analysis of strategies. New Phytol 93:157–191
Rodrigues MS, Ferreira LS, Converti A, Sato S, Carvalho JCM (2010) Fed-batch cultivation of Arthrospira (Spirulina) platensis: potassium nitrate and ammonium chloride as simultaneous nitrogen sources. Bioresour Technol 101:4491–8. Doi:
Rosenberg JN, Mathias A, Korth K, Betenbaugh MJ, Oyler GA (2011) Microalgal biomass production and carbon dioxide sequestration from an integrated ethanol biorefinery in Iowa: a technical appraisal and economic feasibility evaluation. Biomass Bioenergy 35:3865–3876
Rosenberg JN, Oyler GA, Wilkinson L, Betenbaugh MJ (2008) A green light for engineered algae: redirecting metabolism to fuel a biotechnology revolution. Curr Opin Biotechnol 19:430–436
Sathasivam R, Juntawong N, Program B (2013) Modified medium for enhanced growth of Dunaliella strains. Int J Curr Sci 5:67–73
Silva CEDF, Gris B, Sforza E, Rocca NL, Bertucco A (2016) Effects of sodium bicarbonate on biomass and carbohydrate production in Synechococcus PCC 7002. Appl Biochem Biotechnol 49:241–246
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101:87–96
Su CM, Hsueh HT, Chen HH, Chu H (2012) Effects of dissolved inorganic carbon and nutrient levels on carbon fixation and properties of Thermosynechococcus sp. in a continuous system. Chemosphere 88:706–711
Toledo-Cervantes A, Serejo ML, Blanco S, Pérez R, Lebrero R, Muñoz R (2016) Photosynthetic biogas upgrading to bio-methane: boosting nutrient recovery via biomass productivity control. Algal Res 17:46–52
Tripathi R, Singh J, Thakur IS (2015) Characterization of microalga Scenedesmus sp. ISTGA1 for potential CO2 sequestration and biodiesel production. Renew Energy 74:774–781
Vonshak A (ed) (1997) Spirulina platensis (Arthrospira): physiology, cell-biology and biotechnology. Taylor & Francis, London
Walsh BJ, Rydzak F, Palazzo A, Kraxner F, Herrero M, Schenk PM, Ciais P, Janssens IA, Peñuelas J, Niederl-Schmidinger A, Obersteiner M (2015) New feed sources key to ambitious climate targets. Carbon Balance Manag 10:26. doi:10.1186/s13021-015-0040-7
White DA, Pagarette A, Rooks P, Ali ST (2013) The effect of sodium bicarbonate supplementation on growth and biochemical composition of marine microalgae cultures. J Appl Phycol 25:153–165
Yeh KL, Chang JS, Chen WM (2010) Effect of light supply and carbon source on cell growth and cellular composition of a newly isolated microalga Chlorella vulgaris ESP-31. Eng Life Sci 10:201–208
Acknowledgements
This study was partially supported by Japan Science and Technology Agency (JST)/Japan International Cooperation Agency (JICA), Science and Technology Research Partnership for Sustainable Development (SATREPS). The authors would like to offer deep appreciation to Ms. Maki Kobayashi, Mr. Kenta Nagatsuka, Ms. Yumi Hosokawa, Ms. Hidemi Onouchi, Ms. Mako Tagawa, and Ms. Midori Goto for their support in sampling and analyzing the data.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 23 kb)
Rights and permissions
About this article
Cite this article
Kishi, M., Toda, T. Carbon fixation properties of three alkalihalophilic microalgal strains under high alkalinity. J Appl Phycol 30, 401–410 (2018). https://doi.org/10.1007/s10811-017-1226-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1226-z