Abstract
Kappaphycus alvarezii was cultured in vitro under salinities ranging from 15 to 55 psu for 35 days to determine the differential effect on growth rate, carrageenan yield, and cellular structure. Plants kept in 15 psu died after 3 days, while plants cultured in 55 psu presented low growth rates during the entire experimental period (0.28% day−1). Plants cultured in 25, 35, and 45 psu showed growth rates normally associated with this species (between 3% and 4% day−1) and similar cellular morphology. Carrageenan yield was significantly higher in plants cultured in 25 psu in relation to the other treatments. As observed by light microscopy, plants cultured in 15 psu showed cellular turgidity and increased cell wall thickness, both consequences of hyposalinity. Chloroplasts and other membranous organelles underwent rupture and considerable disorganization in ultrastructure. Although branches from the 55 psu samples showed plasmolysis, cells were able to maintain chloroplast integrity, despite their rudimentary features. In high salinities, great concentrations of floridean starch grains were observed in subcortical cells, indicating their probable participation in osmoregulation. Based on these results, we defined the range of 25 to 45 psu as the limits of saline tolerance for K. alvarezii. While new field studies are required to confirm these results, it can be concluded that new sites, such as inactive or abandoned shrimp tanks with salinities up to 25 psu, could be considered for commercial farming.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Kappaphycus alvarezii (Doty) Doty ex P. Silva is an important commercial seaweed species because it is the main source of kappa carrageenan, a phycocolloid widely used in the food, pharmaceutical and cosmetic industries, among others, as a thickener and stabilizing agent. The production of this species is totally based on commercial farms, and cultivation is normally made in off-bottom systems or in floating rafts kept at depths ranging from 0.5 to 1.0 m. However, these cultivation methods can be influenced by tides, exposing the seedlings to desiccation or rapid decreases in salinity during tropical downpours (Ask and Azanza 2002). Studies reporting on the physiology, cultivation, and commercial importance of K. alvarezii can easily be found (Li et al. 1990; Areces 1995; Largo et al. 1995; Paula et al. 2001; Ask and Azanza 2002; Hayashi et al. 2007a, b, 2008a, b), but works related to cultivation under different salinities are rare. Similarly, while several authors (Yokoya and Oliveira 1992; Katz et al. 2000; Conitz et al. 2001; Kakinuma et al. 2006) have reported on the salinity tolerance of several species, studies showing the effects of salinity on cellular structure are uncommon.
It has been suggested that seaweeds can regulate their cellular volume by modifying the internal water potential in response to the change of salinity (Yu and Pedersén 1990; Ekman et al. 1991; Goulard et al. 2001; Eggert et al. 2007; Bondu et al. 2009). According to Kirst (1989), osmotic acclimation is a fundamental mechanism of salinity tolerance, preserving intracellular stability (homeostasis), which is essential to the maintenance and efficient function of the cells. Generally, under hypersaline conditions, the accumulation of osmotically active substances by caption and biosynthesis can be observed. In contrast, excretion and degradation are observed under hyposaline conditions (Kirst 1989). Kakinuma et al. (2006) suggest that the maintenance of constant cell turgor with the change of osmotic potential, which is created by alterations in the concentrations of inorganic ions and organic osmolytes, is a mechanism typical of seaweeds.
In this work, K. alvarezii was cultured in vitro under salinities ranging from 15 to 55 psu for 35 days to determine the differential effect on growth rate, carrageenan yield and cellular structure, with the aim of establishing the limits of saline tolerance to provide a basis for selecting the most suitable locations to establish commercial farming.
Material and methods
Branches of green tetrasporophyte of K. alvarezii having a total weight of 1 g were maintained in unialgal culture at the Laboratório de Algas Marinhas (LAMAR) of the Universidade Federal de Santa Catarina and were cultured in 500 mL sterilized seawater enriched with 50% von Stosch solution (according to McLachlan 1973). Cultures were grown in a 12 h photoperiod with temperature at 25 ± 1°C, photon flux density of 200 ± 10 μmol photons m−2 s−1 and constant aeration.
Five treatments were tested: (a) 15, (b) 25, (c) 35 (considered as control), (d) 45, and (e) 55 psu. Low salinities were obtained with the addition of distilled water, while high salinities were obtained through gradual freezing and thawing of seawater until the final concentration was reached. For each treatment, three replicates were made (n = 3). Plants were cultured for 35 days, and culture media were renewed weekly, at which time seaweeds were weighed for growth rate analyses (GR), according to Lignell and Pedersén (1989): \( {\hbox{GR}}\left( {\% {\hbox{ da}}{{\hbox{y}}^{ - {1}}}} \right) = \left[ {{{\left( {{W_t}/{W_{\rm{i}}}} \right)}^{{1}/n}}--{1}} \right] \times {1}00 \), where W i = initial wet weight and W t = wet weight after t days. After the experimental period, each treatment produced fragments from which 1 cm of the main thallus under the first ramification were removed and fixed to light and electron microscopy analyses. The remains were oven-dried for carrageenan extraction.
Carrageenan extraction
Samples of each treatment (n = 3) were digested in distilled water for 4 h at 60°C, according to Hayashi et al. (2007b), following the proportion 1:100 (w/v). The digestion product was filtered under low pressure and precipitated in a solution of isopropyl alcohol (85%) and KCl (0.2%). The carrageenan fibers were recovered and oven-dried at 60°C for approximately 12 h.
Light microscopy
Fragments of all treatments were fixed in 2.5% paraformaldehyde in 0.1 M sodium phosphate buffer overnight. Afterwards, samples were dehydrated in increasing concentrations series of ethanol aqueous solutions, according to Bouzon et al. (2000). Pre-infiltration and infiltration of samples were made in glycol methacrylate Historesin (modified by Arnold et al. 1975). Sections of 4–5 μm were stained according to the following histochemical techniques: toluidine blue (AT-O) to identify acidic polysaccharides by metachromatic reaction (Gordon and McCandless 1973) and periodic acid–Schiff (PAS) to identify the presence of neutral polysaccharides (Gahan 1984). All stained material was analyzed using a Leica DM 500 with image capture. Since plants cultured, in 25, 35, and 45 psu showed similar patterns, we present only the results of 15, 35, and 55 psu treatments.
Transmission electron microscopy
Fragments cultured in extreme salinities (15 and 55 psu) and control treatment (35 psu) were fixed in a solution composed by 2.5% glutaraldehyde, 2% sucrose, 0.05% calcium chloride, and 2% paraformaldehyde in 0.1 M sodium cacodylate buffer, for 12 h at 4°C. Afterwards, washes were performed in 0.1 M sodium cacodylate buffer with reduced concentrations series of sucrose. The material was post-fixed in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer for 12 h and dehydrated in increasing concentrations of acetone. Infiltration was performed in increasing concentrations series of Spurr Resin (modified from Pueschel 1979), and ultrathin sections were obtained. Sections were post-stained with aqueous uranyl acetate followed by lead citrate. Sections were observed using a JEM 2100 transmission electron microscope at the Central Laboratory of Microscopy (Laboratório Central de Microscopia) of the Universidade Federal de Santa Catarina.
Statistical analyses
Statistical analyses were determined using unifactorial ANOVA and Fisher’s a posteriori test. All statistical analyses were performed in Statistica™ (Release 6.0) at the p < 0.05 level.
Results
Growth rates and carrageenan yield
Branches cultured in 15 psu died after 3 days. During the first culture day, thalli presented loss of pigmentation and the first symptoms of “ice-ice” disease (Fig. 1a). In control (35 psu), 25 and 45 psu treatments, the species presented similar growth patterns; however, those from the 45 psu treatment showed more ramifications (Fig. 1b–d). Plants cultured in high salinities (55 psu) presented no growth during the experimental period (Fig. 1e).
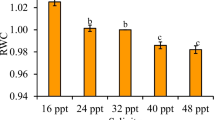
Higher growth rates were observed in branches cultured in control and 45 psu treatment (4.48% day−1 and 4.16% day−1, respectively). Significant differences were observed in plants cultured in 55 psu, when compared with the other treatments (Fig. 2a).
K. alvarezii green tetrasporophyte cultured for 35 days in different salinities. a Growth rates (% day−1); b native carrageenan yield (%). Values presented as average (n = 3). Vertical bars represent confidence intervals, and the letters represent significant differences among the treatments, according to Fisher’s a posteriori test, considering p < 0.05
Native carrageenan yield varied from 25.4% (45 psu) to 35.3% (25 psu). Significant differences were only observed in the carrageenan yield of the 25 psu treatment when compared with the others (Fig. 2b). It was not possible to extract carrageenan from plants cultured in 15 psu.
Light and transmission electron microscopy
Cross-sections of K. alvarezii cultured in normal conditions (35 psu) showed two distinct regions: (1) a cortical region composed of one outermost layer of small cells slightly elongated and two or more layers of cells less elongated and (2) a subcortical region composed of vacuolated cells with gradually increasing size toward the medullar region (Fig. 3a–c). Using transmission electron microscopy (TEM), it was possible confirm the elongated format of the cortical small cells (Fig. 4a) and observe the cytoplasm of subcortical cells with variously sized vacuoles, elongated chloroplasts and floridean starch grains well distributed. Still, pit connections were noted between two subcortical cells (Fig. 4b).
Light microscopy of the cross-sections of K. alvarezii cultured for 35 days in salinities of 35 psu (a–c), 15 psu (d–f), and 55 psu (g–j). a Section stained with PAS. PAS-positive floridean starch grains in cortical and subcortical cells and cellulose in the cell wall. b Section stained with TB-O. Metachromatic reaction of the cell wall, indicating the presence of carrageenan. c Detail of cortical cells showing cell wall thickening at the outer surface of the thallus (arrows) and a thin layer of mucilage (arrowhead). d Section stained with PAS. PAS-positive floridean starch grains uniformly distributed in cortical and first layers of subcortical cells; in the lower layers of subcortical cells, floridean starch grains were dislocated to the periphery by turgid vacuole. Presence of cellulose in cell wall. e Section stained with TB-O. Metachromatic reaction of the cell wall, indicating the presence of carrageenan and small cortical cells with round form. f Detail of the cortical cells showing dome-shaped cell wall thickening at the outer surface of the thallus (arrows). g Section stained with PAS. PAS-positive floridean starch grains in the subcortical cells and presence of pit connections (arrowheads). h Section stained with TB-O. Metachromatic reaction of the cell wall, indicating the presence of carrageenan and dissociation process of subcortical cells (arrow). i Detail of cortical cells showing dome-shaped cell wall thickening at the outer surface of the thallus (arrow). j Detail of two subcortical cells showing plasmolysis process. CC cortical cells, SC subcortical cells
Transmission electron microscopy (TEM) micrograph of cortical and subcortical cells of K. alvarezii cultured in 35 psu a–b, 15 psu c–d and 55 psu e–f for 35 days. a Detail of cortical cells with elongated form. b Detail of subcortical cells with large vacuoles, rich in floridean starch grains and chloroplasts, and pit connection (arrow). S starch, C chloroplast. c Detail of cortical cells showing total cellular disorganization, with remaining thylakoids (arrows). d Detail of subcortical cells with remaining chloroplasts (arrowheads) and numerous plastoglobuli (arrows). e Detail of isodiametric cortical cell with reduced vacuole, chloroplast rudiments and large number of plastoglobuli (arrows). CW cell wall. f Detail of subcortical cells in plasmolysis process with floridean starch grains scattered in the cytoplasm (arrow)
Samples stained with PAS exhibited strong reaction mainly in the cytoplasm of cortical and subcortical cells (35 psu), showing the presence of floridean starch grains. The reaction also occurred in the cell wall, suggesting the presence of cellulosic compounds (Fig. 3a). Toluidine blue (TB-O) staining revealed a metachromatic reaction in the cell wall, indicating the presence of carrageenan (Fig 3b). Cortical cells showed wall thickening at the outer surface of the thallus, with a thin layer of mucilage weakly positive to AT-O (Fig. 3c). As observed by TEM, the cell wall was composed of concentric cellulose microfibrils with different electron-densities. These cellulose microfibrils were embedded in an amorphous matrix consisting mainly of carrageenan, as observed by light microscopy (Fig 4b).
Cross-sections of branches cultured in 15 psu for 3 days presented cortical cells from the outermost layer less elongated than those observed in the 35 psu treatment, showing a turgidity process (Fig. 3d–f). These cells also showed a deposition of dome-shaped concentric layers in the cell wall in the outer surface of the thallus; however, the thin layer of mucilage observed in plants from the control (35 psu) was not observed in this treatment (Fig. 3f). When stained with PAS, cortical and subcortical cells presented a large amount of floridean starch grains uniformly distributed in the cytoplasm. In the lower layers of subcortical cells, the vacuole acquired large proportions because of the turgidity, dislocating the floridean starch grains to the periphery. Cellulose in the cell walls was also observed (Fig. 3d). The presence of carrageenan was revealed as a metachromatic reaction to TB-O (Fig. 3e). Profound changes caused by osmotic shock could be observed in the ultrastructural organization of cortical and subcortical cells, resulting in a full cellular disorganization (Fig. 4c–d). Chloroplasts and other organelles underwent membrane disruption, losing their characteristic conformation. Remaining chloroplasts were observed in the form of thylakoid traces; however, floridean starch grains and plastoglobuli kept the typical organization of red seaweeds (Fig. 4c–d).
Cross-sections of branches cultured in 55 psu stained with PAS showed high amounts of floridean starch grains, mainly in subcortical cells, when compared to other treatments. The distribution pattern of these grains in the cytoplasm of subcortical cells indicated vacuole retraction as a consequence of plasmolysis (Fig. 3g), which was also evident when observed with TB-O staining (Fig. 3j). Both cell wall and mucilage layer in the outer surface of the thallus presented weak reaction to PAS (Fig 3g). TB-O staining confirmed the presence of carrageenan in this treatment and a slight dissociation process of subcortical cells (Fig. 3h). Cortical cells presented a dome-shaped cell wall thickening at the outer surface of the thallus, similar to samples receiving 15 psu treatment (Fig. 3i). Changes in the ultrastructural organization of cortical and subcortical cells were also observed as a consequence of hypersalinity (Fig. 4e–f). Cortical cells became isodiametric, losing their elongated shape, similar to samples receiving 35 psu treatment. Reduced vacuoles and chloroplast rudiments with a large number of plastoglobuli were observed (Fig. 4e). As a result of plasmolysis, subcortical cells presented considerable vacuole reduction (Fig. 4f).
Discussion
Branches cultured in salinities from 25 to 45 psu presented growth rates (between 3 and 4% day−1) as expected for this species cultured in vitro (Paula et al. 1999; Paula et al. 2001; Bulboa and Paula 2005; Bulboa et al. 2007). These results agree with those from Yokoya and Oliveira (1992) in Gracilaria verrucosa (Hudson) Papenfuss, Gracilaria sp., Pterocladiella capillacea (Gmelin) Santelices and Hommersand, Hypnea musciformis (Wulfen) Lamouroux, Meristiella echinocarpum (J.E. Areschoug) Cheney and Gabrielson and Hypnea cornuta (Kützing) J. Agardh, which presented maximum growth in species cultured between 25 and 40 psu.
Low tolerance to 15 psu salinity could be related to the environment where this species normally occurs or is cultivated. Plants under this treatment showed bleaching and died after 3 days, similar to the observations of Dawes et al. (1999) in Gracilaria cornea J. Agardh. Yokoya and Oliveira (1992) also observed that species they studied became white and died after a few days under hyposaline conditions (below 15 psu), while Kumar et al. (2010) observed that Gracilaria corticata (J. Agardh) J. Agardh grown at 15 psu showed the loss of thallus rigidity and pigmentation after 9 days of exposure. On the other hand, Gelidium coulteri Harvey cultured in 15 psu or above presented gradual increase of growth rates over 5 weeks toward control values (35 psu), but salinities of 10 psu or below were lethal (Macler 1988). This last author observed that chlorophyll and phycobiliprotein levels decreased with decreasing salinity for plants at salinities below 30 psu, with values approaching zero at salinities of 10 psu and below. In the present work, the bleaching could also be explain by total disorganization in the cell ultrastructure and the chloroplast disruptions of turgid cells, even though floridean starch grains maintained their integrity, as observed in TEM. These results agree with those observed by Tropin et al. (2003) in cells of Fucus and Ascophyllum cultured in 10 psu. According to them, in this salinity, degradation of chloroplasts and mitochondria, as well as a decreased number of contacts between the organelles, indicated inhibition of the energy exchange process.
Carrageenan yield of K. alvarezii treated in 35 psu was similar to that normally obtained for the species (Azanza-Corrales and Sa-a 1990; Ohno et al. 1996; Hayashi et al. 2007a, b). Branches cultured in 25 psu presented significantly higher yield when compared to the other treatments. These results agree with those of Hurtado-Ponce and Pondevida (1997), who studied agar extracted from Gracilariopsis bailiniae (Zhang and Xia) Zhang and Xia cultured in different salinities, and those of Daugherty and Bird (1988) in G. verrucosa. According to these last authors, under long-term low salinity, the deposition of phycocolloid in the cell wall should provide additional structural support for turgid cells, increasing the yield.
Kappaphycus alvarezii treated in 55 psu presented low growth rates during the entire experimental period, as did most of the species studied by Yokoya and Oliveira (1992) cultured in higher salinities (between 45 and 60 psu). According to Kirst (1989), growth can be sacrificed near the salinity limits of tolerance in order to maintain osmotic adjustment, which can guarantee survival for short periods. The growth reduction can also be a consequence of the cumulative effects of enzymes and reduced turgor pressure that inhibits cell division (Lobban and Harrison 1994). Plasmolysis was well evidenced in this treatment (55 psu) as verified under light microscopy and TEM. A higher quantity of floridean starch grains was observed in subcortical cells, suggesting a possible transference from cortical to subcortical cells, which have larger size and, consequently, a larger surface area of water loss. According to Reed (1990), starch grains are the visible portion of reserve carbohydrate and can be the source of osmotically active low molecular weight solute production, which, together with ions, can be either accumulated or degraded in response to changes in salinity, thus contributing to osmoregulation. In rhodophyceans the main solutes identified were florideoside and digeneaside (Kirst 1989). Despite being a source for florideoside synthesis, the amount of floridean starch did not seem to decrease with the likely increased production of florideosides at high salinity, as observed here in cross-sections of branches cultured in 55 psu and stained with PAS. In fact, Goulard et al. (2001) observed in Solieria chordalis (C. Agardh) J. Agardh cultured in hypersaline condition that the content of floridean starch grains, in contrast to the florideoside level, was not affected. These authors observed that florideoside is normally located in the cytoplasm, suggesting that this component could have an osmolytic effect, protecting enzyme activity. In the present work, this hypothesis can be confirmed, mainly by the integrity of these grains observed in branches cultured in 55 psu and analyzed by TEM. The cell wall thickening of these samples could also be a protecting reaction of hypersaline conditions. Different from that observed in 15 psu, it seems that at high salinity, the thickening could mean extra protection to minimize water loss, since the carrageenans are potentially hydrophilic colloids (Stanley 1987) and, as such, would assist in water retention.
The present results showed that the tolerance limits to different salinities of K. alvarezii range between 25 and 45 psu when cultured for 35 days. In salinities lower than 15 psu or higher than 55 psu, there are profound cellular alterations, causing death of the species in the first case, or the damage of the cellular organization, affecting the growth, as observed in the second case. Floridean starch grains can also have a fundamental role in osmoregulation of the species, possibly as a carbon source for osmolyte biosynthesis, such as florideosides. While new field studies are required to confirm these results, it can be concluded that new sites, such as inactive or abandoned shrimp tanks with salinities up to 25 psu, could be considered for commercial farming.
References
Areces AJ (1995) Cultivo comercial de carragenófitas del genero Kappaphycus Doty. In: Alveal K, Ferrario ME, Oliveira EC, Sar E (eds) Manual de Metodos Ficológicos. Universidad de Concepción, Concepción, pp 529–549
Arnold W, Mitrenga D, Mayersbach H (1975) Gefriertrocknung und einbsettung in glycolmethacrylat (GMA)—ergehnisse histochemischer reaktion. Acta Histochem 14:271–277
Ask E, Azanza RV (2002) Advances in cultivation technology of commercial eucheumatoid species: a review with suggestions for future research. Aquaculture 206:257–277
Azanza-Corrales R, Sa-a P (1990) The farmed Eucheuma species (Gigartinales, Rhodophyta) in Danajon Reef, Philippines: carrageenan properties. Hydrobiologia 204/205:521–525
Bondu S, Cerantola S, Kervarec N, Deslandes E (2009) Impact of the salt stress on the photosynthetic carbon flux and 13C-label distribution within floridoside and digeneaside in Solieria chordalis. Phytochem 70:173–184
Bouzon ZL, Miguens F, Oliveira EC (2000) Male gametogenesis in the red algae Gracilaria and Gracilariopsis (Rhodophyta, Gracilariales). Crypt Algol 21:33–47
Bulboa CR, Paula EJ (2005) Introduction of non-native species of Kappaphycus alvarezii (Rhodophyta, Gigartinales) in subtropical waters: comparative analysis of growth rates of Kappaphycus alvarezii and Kappaphycus striatum in vitro and in the sea in south-eastern Brazil. Phycol Res 53:183–188
Bulboa CR, Paula EJ, Chow F (2007) Laboratory germination and sea out-planting of tetraspore progeny from Kappaphycus striatum (Rhodophyta) in subtropical waters of Brazil. J Appl Phycol 19:357–363
Conitz JM, Fagen R, Lindstrom SC, Plumley FG, Stekoll MS (2001) Growth and pigmentation of juvenile Porphyra torta (Rhodophyta) gametophytes in response to nitrate, salinity and inorganic carbon. J Appl Phycol 13:423–431
Daugherty BK, Bird KT (1988) Salinity and temperature effects on agar production from Gracilaria verrucosa strain G-16. Aquaculture 75:105–113
Dawes CJ, Orduña-Rojas J, Robledo D (1999) Response of the tropical seaweed Gracilaria cornea to temperature, salinity and irradiance. J Appl Phycol 10:419–425
Eggert A, Nitschke U, West JA, Michalik D, Karsten U (2007) Acclimatation of the intertidal red alga Bangiopsis subsimplex (Stylonematophyceae) to salinity changes. J Exp Mar Biol Ecol 343:176–186
Ekman P, Yu S, Pedersen M (1991) Effects of altered salinity, darkness and algal nutrient status on floridoside and starch content, α-galactosidase activity and agar yield of cultured Gracilaria sordida. Br Phycol J 26:123–131
Gahan PB (1984) Plant histochemistry and cytochemistry: an introduction. Academic, London
Gordon EM, Mccandless EL (1973) Ultrastructure and histochemistry of Chondrus crispus Stack. Proc Nova Scotia Inst Sci 27:111–133
Goulard F, Diouris M, Quere G, Deslandes E, Floćh J-Y (2001) Salinity effects on NDP-sugars, floridoside, starch, and carrageenan yield, and UDP-glucose-pyrophophorylase and -epimerase activities of cultured Solieria chordalis. J Plant Physiol 158:1387–1394
Hayashi L, Paula EJ, Chow F (2007a) Growth rate and carrageenan analyses in four strains of Kappaphycus alvarezii (Rhodophyta, Gigartinales) farmed in the subtropical waters of São Paulo State, Brazil. J Appl Phycol 19:393–399
Hayashi L, Oliveira EC, Bleicher-Lhonneur G, Boulenguer P, Pereira RTL, von Seckendorff R, Shimoda VT, Leflamand A, Vallée P, Critchley AT (2007b) The effects of selected cultivation conditions on the carrageenan characteristics of Kappaphycus alvarezii (Rhodophyta, Solieriaceae) in Ubatuba Bay, São Paulo, Brazil. J Appl Phycol 19:505–511
Hayashi L, Yokoya NS, Kikuchi DM, Oliveira EC (2008a) Callus induction and micropropagation improved by colchicines and phytoregulators in Kappaphycus alvarezii (Rhodophyta, Solieriaceae). J Appl Phycol 20:653–659
Hayashi L, Yokoya NS, Ostini S, Pereira RTL, Braga ES, Oliveira EC (2008b) Nutrients removed by Kappaphycus alvarezii (Rhodophyta, Solieriaceae) in integrated cultivation with fishes in re-circulating water. Aquaculture 277:185–191
Hurtado-Ponce AQ, Pondevida HB (1997) The interactive effect of some environmental factors on the growth, agar yield and quality of Gracilariopsis bailinae (Zhang et Xia) cultured in tanks. Bot Mar 40:217–223
Kakinuma M, Coury DA, Kuno Y, Itoh S, Kozawa Y, Inagaki E, Yoshiura Y, Amano H (2006) Physiological and biochemical responses to thermal and salinity stresses in a sterile mutant of Ulva pertusa (Ulvales, Chlorophyta). Mar Biol 149:97–106
Katz S, Kizner Z, Dubinsky Z, Friedlander M (2000) Responses of Porphyra linearis (Rhodophyta) to environmental factors under controlled culture conditions. J Appl Phycol 12:535–542
Kirst GO (1989) Salinity tolerance of eukaryotic marine algae. Annu Rev Plant Physiol Plant Mol Biol 40:21–53
Kumar M, Kumari P, Gupta V, Reddy CRK, Jha B (2010) Biochemical responses of red alga Gracilaria corticata (Gracilariales, Rhodophyta) to salinity induced oxidative stress. J Exp Mar Biol Ecol. doi:10.1016/j.jembe.2010.06.001
Largo DB, Fukami F, Nishijima T, Ohno M (1995) Laboratory-induced development of ice-ice disease of the farmed red algae Kappaphycus alvarezii and Eucheuma dnticulatum (Solieriaceae, Gigartinales, Rhodophyta). J Appl Phycol 7:539–543
Li R, Li J, Wu CY (1990) Effect of ammonium on growth and carrageenan content in Kappaphycus alvarezii (Gigartinales, Rhodophyta). Hydrobiologia 204/205:499–503
Lignell A, Pedersén M (1989) Ágar composition as a function of morphology and growth rate. Studies on some morphological strains of Gracilaria secundata and Gracilaria verrucosa (Rhodophyta). Bot Mar 32:219–227
Lobban CS, Harrison PJ (1994) Seaweed ecology and physiology. Cambridge University Press, Cambridge, p 366
Macler BA (1988) Salinity effects on photosynthesis, carbon allocation, and nitrogen assimilation in the red alga Gelidium couteri. Plant Physiol 88:690–694
McLachlan J (1973) Growth media-marine. In: Stein JR (ed) Handbook of phycological methods. Culture methods and growth measurements. Cambridge University Press, Cambridge, pp 25–51
Ohno M, Nang HQ, Hirase S (1996) Cultivation and carrageenan yield and quality of Kappaphycus alvarezii in the waters of Vietnam. J Appl Phycol 8:431–437
Paula EJ, Pereira RTL, Ohno M (1999) Strain selection in Kappaphycus alvarezii var. alvarezii (Solieriaceae, Rhodophyta) using tetraspore progeny. J Appl Phycol 11:111–121
Paula EJ, Erbert C, Pereira RTL (2001) Growth rate of the carrageenophyte Kappaphycus alvarezii (Rhodophyta, Gigartinales) in vitro. Phycol Res 49:155–161
Pueschel CM (1979) Ultrastructure of tetrasporogenesis in Palmaria palmata (Rhodophyta). J Phycol 15:409–424
Reed RH (1990) Solute accumulation and osmotic adjustment. In: Cole KM, Sheath RG (eds) Biology of red algae. Cambridge University Press, Cambridge, p 517
Stanley N (1987) Production, properties and uses of carrageenan. FAO Fish Tech Pap 288:116–146
Tropin IV, Radzinskaya NV, Voskoboinikov GM (2003) The influence of salinity on the rate of dark respiration and structure of the cells of brown algae thalli from the Barents Sea littoral. Biol Bull 30:40–47
Yokoya NS, Oliveira EC (1992) Effects of salinity on the growth rate, morphology and water content of some Brazilian red algae of economic importance. Cienc Mar 18:49–64
Yu S, Pedersén M (1990) The effect of salinity changes on the activity of α-galactosidase of the red algae Gracilaria sordida and Gracilaria tenuistipitata. Bot Mar 33:385–391
Acknowledgments
This study received financial support from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), the Coordenação de Aperfeiçoamento de Nível Superior (CAPES) and the Brazilian Ministry of Fisheries and Aquaculture. We thank Dr. Eurico Cabral de Oliveira for the critical analysis of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hayashi, L., Faria, G.S.M., Nunes, B.G. et al. Effects of salinity on the growth rate, carrageenan yield, and cellular structure of Kappaphycus alvarezii (Rhodophyta, Gigartinales) cultured in vitro. J Appl Phycol 23, 439–447 (2011). https://doi.org/10.1007/s10811-010-9595-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-010-9595-6