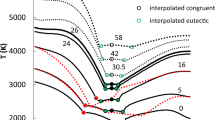
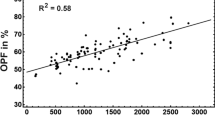
Abstract
The forsterite-melt partition coefficients K are determined experimentally for a large number of mono-, di-, tri-, and tetravalent impurities. The energies of native defects and impurities (E d) and the solution energies (E s) of impurities in forsterite are evaluated using computer simulation. The defect energy is shown to vary linearly with the difference in ionic radius between the host and substituent atoms (Δr) and with the impurity cation charge, while the partition coefficient and solution energy of impurities are quadratic functions of these parameters. The plots of lnK versus (Δr)2 and E s versus (Δr)2 for isovalent substitutions and Me xMg and Me xSi ) pass close to the origin, in contrast to the plots for heterovalent substitutions (Me ′Mg and Me ⋅Si ). The significant y intercept of the latter plots is interpreted as evidence for the formation of extra defects maintaining electroneutrality. The y intercept of the plot of E s versus (Δr)2 is 2 eV, which is about half the formation energy of Frenkel defects in forsterite. The best fit equations representing the correlation between the partition coefficients and solution energies of impurities demonstrate that heterovalent substitutions increase the entropy contribution to the free energy of solution of impurities.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Nisel’son, L.A. and Yaroshevskii, A.G., Mezhfazovye koeffitsienty raspredeleniya. Ravnovesiya kristall-zhidkost’ i zhidkost’-par (Partition Coefficients for Solid-Liquid and Liquid-Vapor Equilibria), Moscow: Nauka, 1992.
Urusov, V.S., Tauson, V.L., and Akimov, V.V., Geokhimiya tverdogo tela (Geochemistry of Solids), Moscow: GEOS, 1997.
Allan, N.L., Blundy, J.D., Purton, J.A., et al., Trace Element Incorporation in Minerals and Melts, EMU Notes Mineral., 2001, vol. 3, pp. 251–302.
Purton, J.A., Blundy, J.D., and Allan, N.L., Computer Simulation of High-Temperature Forsterite-Melt Partitioning, Am. Mineral., 2000, vol. 85, pp. 1087–1091.
Kristallizatsiya iz rasplavov: Spravochnik (Crystal Growth from the Melt: A Handbook), Bartel, I., et al., Eds., Moscow: Metallurgiya, 1987.
Onuma, N., Higuchi, H., Wakita, H., and Nagasawa, H., Trace Element Partition between Two Pyroxenes and the Host Lava, Earth Planet. Sci. Lett., 1968, vol. 5, pp. 47–51.
Jensen, B.B., Patterns of Trace Element Partitioning, Geochim. Cosmochim. Acta, 1973, vol. 37, pp. 2227–2242.
Philpotts, J.A., The Law of Constant Rejection, Geochim. Cosmochim. Acta, 1978, vol. 42, pp. 909–920.
Beattie, P., Systematics and Energetics of Trace-Element Partitioning between Olivine and Silicate Melts: Implications for the Nature of Mineral/Melt Partitioning, Chem. Geol., 1994, vol. 117, pp. 57–71.
Dudnikova, V.B., Zharikov, E.V., Urusov, V.S., et al., Effects of the Ionic Radius and Charge State of Impurities on Their Forsterite-Melt Partition Coefficient, Mater. Elektron. Tekh., 2000, no. 2, pp. 11–14.
Urusov, V.S., Teoriya izomorfnoii smesimosti (Theory of Isomorphous Miscibility), Moscow: Nauka, 1977.
Blundy, J.D. and Wood, B.J., Prediction of Crystal-Melt Partition Coefficients from Elastic Moduli, Nature (London), 1994, vol. 372, pp. 452–454.
Wood, B.J. and Blundy, J.D., A Predictive Model for Rare Earth Element Partitioning between Clinopyroxene and Anhydrous Silicate Melt, Contrib. Mineral. Petrol., 1997, vol. 129, pp. 166–181.
Blundy, J.D., Robinson, J.A.C., and Wood, B.J., Heavy REE Are Compatible in Clinopyroxene on the Spinel Iherzolite Solidus, Earth Planet. Sci. Lett., 1998, vol. 160, pp. 493–504.
Blundy, J.D. and Dalton, J.A., An Experimental Comparison of Clinopyroxene-Melt Partitioning in Silicate and Carbonate Systems and Implications for Mantle Metasomatism, Contrib. Mineral. Petrol., 2000, vol. 139, pp. 356–371.
Brenan, J.M., Shaw, H.F., Ryerson, F.J., and Phinney, D.L., Experimental Determination of Trace Element Partitioning between Pargasitic Amphibole and Synthetic Hydrous Melt, Earth Planet. Sci. Lett., 1995, vol. 135, pp. 1–12.
La Tourette, T., Hervig, R.L., and Holloway, J.R., Trace Element Partitioning between Amphibole, Phlogopite, and Basanite Melt, Earth Planet. Sci. Lett., 1995, vol. 135, pp. 13–30.
Moller, P., The Dependence of Partition Coefficients on Differences of Ionic Volumes in Crystal-Melt Systems, Contrib. Mineral. Petrol., 1988, vol. 99, pp. 62–69.
Dudnikova, V.B., Urusov, V.S., Bykov, A.B., and Kolesov, G.M., Forsterite-Melt Partitioning of Trivalent Trace Elements, Geokhimiya, 1992, no. 3, pp. 444–447.
Shannon, R.D., Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, vol. 32, no.5, pp. 751–767.
Dudnikova, V.B., Zharikov, E.V., Eremin, N.N., et al., Forsterite-Melt Partitioning of Vanadium and the Structural and Valence States of Vanadium, Geokhimiya, 2001, no. 7, pp. 734–743.
Dudnikova, V.B., Gaister, A.V., Zharikov, E.V., et al., Forsterite-Melt Partitioning of Cr at Different Redox Conditions and Cr Concentrations in the Melt, Geokhimiya, 2005, no. 5.
Shenjun, L., Lin, L., Zulun, W., et al., Growth and Characteristics of Mg2SiO4:Ti Crystal, J. Cryst. Growth, 1994, vol. 139, pp. 327–331.
Kobayashi, T. and Takei, H., Distribution of Some Trivalent Ions between Melt and Single Crystals Mg2SiO4 Grown by Czochralski Method, Earth Planet. Sci. Lett., 1977, vol. 36, pp. 231–236.
Wood, B.J. and Blundy, J.D., The Effect of Cation Charge on Crystal-Melt Partitioning of Trace Element, Earth Planet. Sci. Lett., 2001, vol. 188, pp. 59–71.
Birle, I.D., Gibbs, G.V., Moore, P.B., and Smith, J.V., Crystal Structure of Natural Olivines, Am. Mineral., 1968, vol. 53, pp. 807–825.
Gale, J.D., GULP: A Computer Program for the Symmetry Adopted Simulation of Solids, J. Chem. Soc., Faraday Trans., 1997, vol. 93, pp. 629–637.
Freeman, C.M. and Catlow, C.R.A., A Computer Modeling Study of Defect and Dopant States in SnO2, J. Solid State Chem., 1990, vol. 85, pp. 65–75.
Lewis, G.V. and Catlow, C.R.A., Potential Models for Ionic Oxides, J. Phys. C: Solid State Phys., 1985, vol. 18, pp. 1149–1161.
Sanders, M.J., Leslie, M.J., and Catlow, C.R.A., Inter-atomic Potentials for SiO2, J. Chem., Soc. Chem. Commun., 1984, vol. 18, pp. 1271–1273.
Hazen, R.M., Effects of Temperature and Pressure on the Crystal Structure of Forsterite, Am. Mineral., 1976, vol. 61, pp. 1280–1293.
Smyth, D.M. and Stocker, R.L., Point Defects and Non-Stoichiometry in Forsterite, Phys. Earth Planet. Inter., 1975, vol. 10, pp. 183–192.
Andersson, K., Borchart, G., Scherrer, S., and Weber, S., Self Diffusion in Mg2SiO4 (Forsterite) at High Temperature: A Model Case Study for SIMS Analyses on Ceramic Surfaces, Fresenius’ J. Anal. Chem., 1989, vol. 333, pp. 383–385.
Jaoul, O., Bertran-Alvarez, Y., Liebermann, R.C., and Price, G.D., Fe-Mg Interdiffusion in Olivine up to 9 GPa at T = 600–900°C; Experimental Data and Comparison with Defect Calculations, Phys. Earth Planet. Inter., 1995, vol. 89, pp. 199–218.
Purton, J.A., Allan, N.L., Blundy, J.D., and Wasserman, E.A., Isovalent Trace Element Partitioning between Minerals and Melts: A Computer Simulation Study, Geochim. Cosmochim. Acta, 1996, vol. 60, pp. 4977–4987.
Purton, J.A., Allan, N.L., and Blundy, J.D., Calculation Solution Energies of Heterovalent Cations in Forsterite and Diopside: Implication for Trace Element Partitioning, Geochim. Cosmochim. Acta, 1997, vol. 61, pp. 3927–3936.
Rajamani, V., Brown, G.E., and Prewitt, C.T., Cation Ordering in Ni-Mg Olivine, Am. Mineral., 1975, vol. 60, pp. 292–299.
McCormick, T.C., Smyth, J.R., and Lofgren, G.E., Site Occupancies of Minor Elements in Synthetic Olivines as Determined by Channeling-Enhanced X-ray Emission, Phys. Chem. Miner., 1987, vol. 14, pp. 368–372.
Gaite, J.M., Pseudo-Symmetries of Crystallographic Coordination Polyhedra. Application to Forsterite and Comparison with Some EPR Results, Phys. Chem. Miner., 1980, vol. 6, pp. 9–17.
Mass, J.L., Burlitch, J.M., Markgraf, S.A., et al., Oxygen Activity Dependence of Chromium(IV) Population in Chromium-Doped Forsterite Crystals Grown by Floating Zone Technique, J. Cryst. Growth, 1996, vol. 165, pp. 250–257.
Rager, H., Electron Spin Resonance of Trivalent Chromium in Forsterite, Mg2SiO4, Phys. Chem. Miner., 1977, vol. 1, pp. 371–378.
Rager, H., Electron-Nuclear Hyperfine Interaction of 53Cr3+ in Mg2SiO4 (Forsterite), Z. Naturforsch., A, 1980, vol. 3, pp. 1296–1303.
Budil, D.E., Park, D.G., Freed, J.H., et al., 9.6 GHz and 34 GHz Electron Paramagnetic Resonance Studies of Chromium-Doped Forsterite, J. Chem. Phys., 1994, vol. 101, pp. 3538–3548.
Urusov, V.S. and Dudnikova, V.B., Energetics of Heterovalent Microisomorphism with Vacancy Formation in Ionic Crystals, Geokhimiya, 1987, no. 9, pp. 1219–1230.
Dudnikova, V.B., Gaister, A.V., Zharikov, E.V., et al., Effect of Compensation Doping on Chromium Solubility in Forsterite, Neorg. Mater., 2003, vol. 39, no.8, pp. 985–990 [Inorg. Mater. (Engl. Transl.), vol. 39, no. 8, pp. 845–850].
Author information
Authors and Affiliations
Additional information
__________
Translated from Neorganicheskie Materialy, Vol. 41, No. 6, 2005, pp. 720–732.
Original Russian Text Copyright © 2005 by Dudnikova, Urusov, Zharikov.
Rights and permissions
About this article
Cite this article
Dudnikova, V.B., Urusov, V.S. & Zharikov, E.V. Crystal-Melt Partition Coefficients of Impurities in Forsterite, Mg2SiO4: Experimental Determination, Crystal-Chemical Analysis, and Thermodynamic Evaluation. Inorg Mater 41, 627–638 (2005). https://doi.org/10.1007/s10789-005-0181-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10789-005-0181-2