Abstract
Although some environmental risks and resources are known to affect the evolution of primate social groups, we know little about the effect of major natural disturbances on primate populations. Hurricane Iris hit the Monkey River watershed in southern Belize in October 2001, presenting a unique opportunity to document the effects of a natural disaster under circumstances wherein some pre-hurricane data were available. We measured the characteristics of the population of black howlers in the affected forest 3.5 years after the storm and compared the population data with pre-hurricane data from a 52-ha study area, that may represent the larger continuous riverine forest and from which all monkeys were known. From February to May 2004, we sampled 28.77 km2 of the 96-km2 forest fragment via five transects walked 12 times each. From these data we estimate that the population in the watershed has dropped from 9784 to 1181 monkeys, a reduction of 88%, reflected by both a 79% drop in the number of social groups and a 38% reduction in group size. Before the storm, 75% of the social groups were multimale; after the storm, 74% of the groups were unimale. While the ratio of adult females to males improved slightly, the ratio of adults to immatures, and adult females to immatures more than doubled, indicating a much lower potential for growth. These data provide a quantitative assessment of how a major natural disturbance can affect a primate population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Alouatta pigra occurs in southeastern Mexico, Belize, and northern Guatemala (Crockett and Eisenberg 1987; Ostro et al. 2001; Estrada et al. 2002; Rylands et al. 2006) in a variety of habitats (Crockett 1998; Silver et al. 1998; Estrada et al. 2004), including riverine and seasonally flooded areas (Horwich and Johnson 1986; Silver et al. 1998). The habitat of A. pigra is undergoing fragmentation through its rapid conversion to pasture lands and agricultural fields (Estrada et al. 2002; 2004), and the fragmentation may lead to important shifts in demographic variables, such as mean population density (Van Belle and Estrada 2006). However, tropical forests and their animal populations are affected not only by anthropogenic activities but also by natural occurrences, such as droughts and hurricanes (Gould et al. 1999; Wright 1999) which are equally important for shaping specific characteristics. Hurricanes in particular are a major agent of catastrophic disturbance in the Caribbean and along the Atlantic coast of Mesoamerica (Scatena and Larsen 1991). Although studies of the effect of hurricanes are rare, largely due to the absence of pre-hurricane comparative data (Tanner and Kapos 1991), there is a small number of studies on the aftermath of hurricanes in tropical and subtropical forests (Dittus 1985a; Reilly 1991; Ferreira and Laurance 1997; Burslem and Whitmore 1999; Alexander et al. 2006) and animal populations (invertebrates: Willig and Camilo 1991; Schowalter 1994; Bloch and Willig 2006; birds: Askins and Ewert 1991; Will 1991; Waide 1991; bats: Gannon and Willig 1994; frogs: Marsh and Pearman 1997). Dittus (1985b) reported on the effect of leaf monkeys on their feeding trees (as browsers) after a cyclone in Sri Lanka. Klinger (2006) examined the effects of natural disturbances, including hurricanes, on mammal communities in the Bladen Nature Reserve of south western Belize.
On October 8, 2001, a riverine forest fragment in southern Belize along with its resident black howler (A. pigra) population was severely damaged by Hurricane Iris, a category four storm that sustained winds of 145 miles per hour. The 96-km2 fragment that was affected includes the forested area of the Monkey River watershed, bordered on the north and south by coastal plain savanna, on the east by the Caribbean Sea, and on the west by the Southern Highway and human settlements including agriculture. Local extinction of the howler population in Monkey River was predicted from the first assessment of the area after the storm (Meerman 2001) based on the severity of damage to the forest. Geographic Information System (GIS) analyses of satellite data later showed that Hurricane Iris increased the amount of edge habitat, decreased patch size, and increased the number and isolation of patches within the Monkey River watershed (Alexander et al. 2006).
Although a full survey of the watershed was not conducted before the storm, Hurricane Iris, by striking an area where some pre-hurricane data were available, created a unique opportunity to document the effect of a natural disturbance on an endangered species. We report on a survey of the Monkey River watershed forest fragment aimed at assessing the black howler population 3.5 yr after Hurricane Iris and compare the results to pre-hurricane true count data from a small area that might representative the larger forest fragment. We compare population and group density, total population size and number of groups, size and composition of social groups, and age and sex ratios (as indicators of potential for population growth) pre- and post-hurricane.
Methods
We derived pre-hurricane population estimates from a study of the behavioral ecology of the black howlers in the Monkey River watershed which took place between May 1999 and October 2001. All monkeys in a 52-ha area, located north of the Monkey River on the eastern edge of the larger 96-km2 watershed fragment, were monitored daily by trained observers from May to August 1999, January to June 2000 and 2001, and in October of 2001. Immediately before the hurricane on October 6 and 7, 2001, researchers were able to locate, count, and obtain age-sex compositions of most of the study groups before evacuating on the morning of October 8. The group compositions were unchanged from June. One week after the storm researchers were able to regain limited access to the site to monitor the study groups. Since then, the study area has been continuously monitored by a minimum of two researchers conducting daily demographic surveys, vegetation samples, and dawn-to-dusk follows of individual groups.
We obtained pre-hurricane values from the intensive study of the 52-ha study area. We used them as the basis for the comparison with post-hurricane data based on the assumption that the 52-ha area, a small proportion of the total watershed forest, represents the larger forest; there is no pre-hurricane survey of the larger forest. Visits to other areas of the watershed by members of the research team and multiple accounts by local forest guides strengthened our confidence that the monkey population in the study area was typical of the area. Also, the measurement of 96 km2 for the watershed fragment was based on an infrared spectrograph (IRS) rectified image of Monkey River and the surrounding area (321 fusion) (Alexander et al. 2006) and included only those areas that were continuous and of the same vegetation type: semi-evergreen lowland broadleaf riparian forest. Additionally, post-hurricane data from the study site (Pavelka 2003; Pavelka et al. 2003; Pavelka and Chapman 2006) and from the census show the study site to be representative of the larger forest. All pre-hurricane data presented and to which we refer derive from the study area, and all post-hurricane data derive from the survey described below.
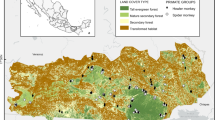
To assess the population after the storm, we augmented the continued observations of the study groups with line transect surveys of the 96-km2 watershed forest between January and June 2004. We sampled five transects (Fig. 1, Table I) 12 times each for a total area sampled of 28.77 km2. Because there has been no previous survey of the area, we chose transect routes that utilized existing roads and trails or major waterways. Although the ecological conditions within the area sampled are relatively homogeneous, we selected transects to represent some of the available variation. Transects one and two sampled from the riverside, three and four sampled areas not directly on the river, and transect five sampled closer to the forest edge.
We conducted surveys on alternate weeks, either on foot or by canoe between 06:00 h/15:00 h. At the start of each transect we recorded the time, the location (UTM coordinates using a Garmin GPS), and the weather. At the end of each transect we recorded the time, location, total kilometers traveled, moving time (km/h), time spent stopped (min), and average moving speed (km/h). Each time was rented a group or solitary monkey, we recorded the time, location, perpendicular distance to the center of the group (in meters via a range finder), compass bearing of the group, group size, group spread, and group composition.
We determined transect length based on UTM coordinates taken on the ground via a handheld global positioning system (GPS). We then entered the coordinates into ArcView software and joined them to form a semistraight line from which we measured total distance. Via this method, we sought to avoid overestimating the area sampled, which the mean trip odometer value would have done, particularly along the winding river. We calculated transect width using the sightings data and the effective distance method of Whitesides et al. (1988), which is widely regarded as the most accurate technique to estimate population densities (Fashing and Cords 2000) and uses effective distance and species typical mean group spread to calculate transect width. We calculated transect area via the formula Area = Lt × W, wherein Lt = total length of all samples (i.e., length of transect × number of transect walks) and W = width. We calculated the area sampled for each transect, and summed them to get the total area sampled. We calculated the population and group densities for each transect by dividing the total number of individuals and groups sighted by the area sampled post-hurricane and by 52-ha pre-hurricane. We obtained estimates of total population size and number of groups in the watershed pre- and post-hurricane by multiplying population and group densities (per km2) by the total available habitat (96 km2).
Our calculations of group size, group type, number of solitary individuals, and age and sex ratio are based on unique groups or individuals identified in the sightings data or both. Plotting all monkey sightings on a satellite image of the watershed allowed us to identify unique groups and solitaries. We considered groups and individuals that were sighted in the same area and which consisted of the same or a very similar group composition to be the same group. If we were unsure about whether groups or solitaries were the same, we treated them as different groups. Males reach full adult size at ca. 5 yr and have a large throat with full beard and often a heart-shaped head. Females reach full adult size by 4 yr and are identifiable by their size and/or the presence of an infant. Males and females are easily distinguishable by the size and shape differences and the presence or absence of prominent testicles. Researchers were experienced in identifying howlers of known age and sex from many months in the study area, thus ensuring reliability of identification in the survey. We calculated sex ratio as the total number of adult males to the total number of adult females and age ratios as the total number of all adults and then just adult females to the total number of immatures.
Results
Before the storm, 53 monkeys in eight social groups (and two solitary individuals that maintained site fidelity) inhabited the 52-ha study site (Pavelka 2003). Within the 28.77 km2 of the watershed surveyed during 12 samples of five transects in the post-hurricane study, we observed 354 individuals in 114 total sightings – 94 in groups plus 20 solitary individuals. The data (Table II) yield a pre-hurricane population density of 102 ind/km2 vs. a post-hurricane density of 12 ind/km2, an 88% reduction; the estimated population area 10 000 individuals had been reduced to just over 1100 monkeys.
The remaining rows of Table II are based on comparisons of the known population in the study area before the hurricane with the unique individuals (n = 187) and groups (n = 46) identified in the post-hurricane survey data (i.e., not from the total sightings data). Before the hurricane, eight social groups, each ranging from two to ten individuals, lived in the study area. One group each contained two, three, six, and ten individuals; the other four groups contained eight individuals, yielding a mean group size of 6.6 and a mode of eight. Survey data from after the storm revealed that the social groups had dropped in size, ranging from two to six individuals, though groups of six individuals were rare (only 1/46). Mean group size dropped 39% after the storm to four, and modal group size dropped 38% to five. Solitary individuals (not included in the calculations of group size) became 84% more prevalent in the population, though their density dropped in direct accordance with the drop in population density in general. The proportion of unimale versus multimale groups was reversed in the pre- and post-hurricane data: before the storm 75% of groups were multimale, and after the storm 74% of groups were unimale.
In addition to the changes in population and group size, the age and sex structure was altered by the hurricane. The sex ratio in the study area before the storm was 1:0.81 (M:F), with slightly more males than females (including those males living in the area but not in social groups); it dropped to 1:1.08 after the storm, with slightly more females than males. Before the storm, the ratio of adults to immatures was 1:0.39, and it more than doubled to 1:0.18 during the survey. The adult female to immature ratio was 1:0.88 before the storm, and it increased by almost 150%, with a ratio of 1:0.35 adult females to immatures, after the storm.
Discussion
Dramatic reductions in the black howler population in the Monkey River watershed forest fragment are evident 3.5 yr after Hurricane Iris, with an 88% reduction in population size and density from an estimated 9784 monkeys in the 96-km2 fragment pre-hurricane to only 1181 in the spring of 2004. Previous publications have described the immediate effect of the hurricane on the population (Pavelka et al. 2003), food supply (Pavelka and Behie 2005), diet, and activity (Behie and Pavelka 2005), and on the population (Pavelka and Chapman 2006) in the 52 ha study area over the first 3 yr. The new survey data confirm what we suspected to be true of the larger population based on our continued daily monitoring of the study site since the storm. The hurricane resulted in an initial 42% loss of monkeys (Pavelka et al. 2003), and the population at the study site shows the same pattern of a drop in group size and number of groups (Pavelka and Chapman 2006) that we found in the current survey of the larger watershed area.
Our data show a relationship between population density, group size, and multimale groups for A. pigra. Groups of ≤ 10 individuals are the norm for A. pigra (Horwich and Gebard 1983; Gonzalez-Kirchner 1996; Horwich et al. 2001; Pavelka 2003). Before the storm we had a dense population, and most social groups were large (for A. pigra) and multimale. Post-hurricane, Monkey River is characterized by a low population density and small unimale groups. Positive correlations among population density, group size, and proportion of multimale groups have been reported for other populations of A. pigra by Horwich et al. (2001) Ostro et al. (2001) and Estrada et al. (2002) and for A. seniculus by Rumiz (1990) and Rudran and Fernandez-Duque (2003) and A. palliata by Chapman and Balcomb (1998).
Estrada et al. (2002) suggested that small group size may be an adaptation for A. pigra living in fragmented environments, and Van Belle and Estrada (2006) found that in fragmented forests mean group sizes were smaller and there were fewer males in comparison to groups living in an extensive forest. Group size increased when a fragment increased in surface area and quality (Rodriguez-Toledo et al. 2003), and small group size can indicate reduced resources (Clarke et al. 2002) and persistent feeding stress (Ratsimbazafy et al. 2002). Feeding stress may be evidenced by the dietary shift observed in Monkey River. Rivera and Calme (2006) found that howlers living in fragmented forests were forced to consume alternate plant species due to a lower availability of Ficus spp. We also found that A. pigra in Monkey River switched to a primarily folivorous diet due to a lack of fruiting trees (Behie and Pavelka 2005).
The reason for the consistently small group sizes in A. pigra, one of the main factors that lead to the recognition of it as a separate species from its geographic neighbor and close relative A. palliata (Smith 1970), is unknown (Chapman and Pavelka 2005; Knopff and Pavelka 2006). Wright (1999) suggested a connection between cyclones in the north of Madagascar and the consistently small lemur social groups, and in southeastern Madagascar, Ratsimbazafy (2002a) examined a population of black and white ruffed lemurs before and after a large cyclone and found a reduction in group size from four to eight individuals before the cyclone to a maximum group size of four afterwards. The connection between a hurricane and a reduction in group size in A. pigra again raises the possibility that natural disasters may play an important role in determining, or constraining, primate group size, or at least that smaller group sizes in A. pigra may reflect more frequent hurricanes. A cursory investigation of hurricane tracks in and around Central America over the past 150 years show a concentration of hurricane paths crossing onto land (National Oceanic & Atmospheric Administration website, 2007) in precisely the range of A pigra, with a few of them crossing into the range of A. palliata. We are currently investigating this link between consistent hurricane strikes and the current and historic distribution of A. pigra, however it should also be noted that some South American howler species also typically show small group size (A. seniculus: Crockett 1998; Iwanaga and Ferrari 2002; Rudran & Fernandez-Duque 2003; A. caraya: Zunino et al. 2001).
Age structure and sex ratio can be important indicators of the overall health of a population (Fedigan and Jack 2001) and provide researchers with insights into whether a population is growing or declining (Crockett 1998). The small remaining black howler population at Monkey River showed no sign of strong growth potential at the time of our study. In A. palliata, Heltne et al. (1975) argued that for a population to remain stable, a female-to-immature ratio ≥ 1:1.5 was necessary, and Monkey River was far below this value. Populations of A. seniculus have a larger proportion of infants when they are growing than when they are declining (Rudran and Fernandez-Duque 2003), and populations of A. caraya likewise show higher adult-to-immature and adult female-to-juvenile and infant ratios when the population is declining (Rumiz 1990). The slight increase in the proportion of adult females in the post-hurricane survey may be positive for population growth, but the high ratio of adult females to immatures is not. The survey data do not permit us to determine if production or survival of infants is the problem post-hurricane, but it is clear that the number of immature individuals in the population is low, much lower than before the storm. From the data collected at the long-term study site (Pavelka and Chapman 2006), we know that infant production was halted for > 1 year, and then survivorship was low for the next 2–3 years. Infant survivorship in the study area is beginning to increase, and the rate of population decline may be leveling off (Pavelka, unpublished data).
Our results indicate that natural disasters, such as hurricanes, can have major impacts on primate population dynamics, causing dramatic changes to population and group size and density to the age and sex ratios. A clearer understanding of these processes may allow us to better understand the role of natural disturbances on the evolution of primate social groups. In the case of Monkey River, we will continue to monitor an expanded study area and repeat the watershed survey in order to determine if a recovery has begun.
References
Alexander, S. M., Pavelka, M. S. M., & Bywater, N. H. (2006). Quantifying fragmentation of black howler (Alouatta pigra) habitat after Hurricane Iris (2001), Southern Belize. In A. Estrada, P. A. Garber, M. S. M. Pavelka, & L. Luecke (Eds.), New perspectives in the study of Mesoamerican primates: Distribution, ecology, behavior, and conservation (pp. 539–561). New York: Springer.
Askins, R. A., & Ewert, D. N. (1991). Impact of hurricane Hugo on bird populations on St. John, U.S. Virgin islands. Biotropica, 23, 481–487.
Behie, A. M., & Pavelka, M. S. M. (2005). The short-term effects of a hurricane on the diet and activity of black howlers (Alouatta pigra) in Monkey River, Belize. Folia Primatologica, 76, 1–9.
Bloch, C. P., & Willig, M. R. (2006). Context-dependence of long-term responses of terrestrial gastropod populations to large-scale disturbance. Journal of Tropical Ecology, 22, 111–122.
Burslem, D. F. R. P., & Whitmore, T. C. (1999). Species diversity, susceptibility to disturbance and tree population dynamics in tropical rain forest. Journal of Vegetation Science, 10, 767–776.
Chapman, C. A., & Balcomb, S. R. (1998). Population characteristics of howlers: ecological conditions or group history. International Journal of Primatology, 19, 385–403.
Chapman, C. A., & Pavelka, M. S. M. (2005). Group size in folivorous primates: ecological constraints and the influence of social factors. Primates, 46(1), 1–9.
Clarke, M. R., Collins, D. A., & Zucker, E. L. (2002). Responses to deforestation in a group of mantled howlers (Alouatta palliata) in Costa Rica. International Journal of Primatology, 23, 365–381.
Crockett, C. M. (1998). Conservation biology of the genus Alouatta. International Journal of Primatology, 19, 549–578.
Crockett, C. M., & Eisenberg, J. F. (1987). Howlers: Variations in group size and demography. In B. B. Smuts, D. L. Cheney, R. M. Seyfarth, R. W. Wrangham, & T. T. Struhsaker (Eds.), Primate societies (pp. 54–68). Chicago and London: The University of Chicago Press.
Dittus, W. (1985a). The influence of leaf-monkeys on their feeding trees in a cyclone disturbed environment. Biotropica, 17, 100–106.
Dittus, W. (1985b). The influence of cyclones on the dry evergreen forest of Sri Lanka. Biotropica, 17, 1–14.
Estrada, A., Luecke, L., Van Belle, S., Barrueta, E., & Meda, M. R. (2004). Survey of black howler (Alouatta pigra) and spider (Ateles geoffroyi) monkeys in the Mayan sites of Calakmul and Yaxchilán, Mexico and Tikal, Guatemala. Primates, 45, 33–39.
Estrada, A., Mendoza, A., Castellanos, L., Pacheco, R., Van Belle, S., Garcia, Y., et al. (2002). Population of the black howler monkey (Alouatta pigra) in a fragmented landscape in Palenque, Chiapas, Mexico. American Journal of Primatology, 58, 45–55.
Fashing, P. J., & Cords, M. (2000). Diurnal primate densities and biomass in the Kakamega forest: An evaluation of census methods and a comparison with other forests. American Journal of Primatology, 50, 139–152.
Fedigan, L. M., & Jack, K. (2001). Neotropical primates in a regenerating Costa Rican dry forest: a comparison of howler and capuchin population patterns. International Journal of Primatology, 22, 689–713.
Ferreira, L. V., & Laurance, W. F. (1997). Effects of forest fragmentation on mortality and damage of selected trees in Central Amazonia. Conservation Biology, 11, 797–801.
Gannon, M. R., & Willig, M. R. (1994). The effects of Hurricane Hugo on bats of the luquillo experimental forest of Puerto Rico. Biotropica, 23, 320–331.
Gonzalez-Kirchner, J. P. (1996). Group size and population density of the black howler monkey (Alouatta pigra) in Muchukux forest, Quintana Roo, Mexico. Folia Primatologica, 69, 260–265.
Gould, L., Sussman, R. W., & Sauther, M. L. (1999). Natural disasters and primate populations: The effects of a 2-year drought on a naturally occurring population of ring-tailed lemurs (Lemur catta) in southwestern Madagascar. International Journal of Primatology, 20, 69–84.
Heltne P. G., Turner D. C., & Scott, N. J., Jr. (1975). Comparison of census data on Alouatta palliata from Costa Rica and Panama. In R. W. Thorington Jr., & P. G. Heltne (Eds.). Neotropical primates: field studies and conservation (pp. 10–19). Washington D.C.: National Academy of Sciences.
Horwich, R. H., Brockett, R. C., James, R. A., & Jones, C. B. (2001). Population growth in the Belizean black howling monkey (Alouatta pigra). Neotropical Primates, 9, 1–7.
Horwich, R. H., & Gebard K. (1983). Roaring rhythms in black howler monkeys (Alouatta pigra) of Belize. Primates, 24, 290–296.
Horwich, R. H., & Johnson, E. D. (1986). Geographical distribution of the black howler (Alouatta pigra) in Central America. Primates, 27, 53–62.
Iwanaga, S., & Ferrari, S. F. (2002). Geographic distribution of red howlers (Alouatta seniculus) in southwestern Brazilian Amazonia, with notes on Alouatta caraya. International Journal of Primatology, 23, 1245–1256.
Klinger, R. (2006).The interaction of disturbances and small mammal community dynamics in a lowland forest in Belize. Journal of Animal Ecology, 75, 1227–1238.
Knopff, K. H., & Pavelka, M. S. M. (2006). Determinants of group size in the Central American black howler monkey (Alouatta pigra): Evaluating the importance of ecological constraints. International Journal of Primatology, 27, 1059–1078.
Marsh, D. M., & Pearman, P. B. (1997). Effects of habitat fragmentation on the abundance of two species of Leptodactylid frogs in an Andean montane forest. Conservation Biology, 11, 1323–1328.
Meerman, J. (2001). A first assessment of damage to terrestrial ecosystems in southern Belize as caused by hurricane Iris of October 8, 2001. Belize Environmental Consultancies Limited, Belize District, Belize.
National Oceanic & Atmospheric Administration. (2007). http://www.noaa.gov/.
Ostro, L. E. T., Silver, S. C., Koontz, F. W., Horwich, R. H., & Brockett, R. (2001). Shifts in social structure of black howler (Alouatta pigra) groups associated with natural and experimental variation in population density. International Journal of Primatology, 22, 733–748.
Pavelka, M. S. M. (2003). Group, range, and population size of Alouatta pigra at Monkey River, Belize. Neotropical Primates, 11, 187–191.
Pavelka, M. S. M., & Behie, A. M. (2005). The effect of hurricane Iris on the food supply of black howlers (Alouatta pigra) in southern Belize. Biotropica, 37, 102–108.
Pavelka, M. S. M., Brusselers, O. T., Nowak, D., & Behie, A. (2003). Population reduction and social disorganization in Alouatta pigra following a hurricane. International Journal of Primatology, 24, 1037–1055.
Pavelka, M. S. M., & Chapman, C. A. (2006). Population structure of black howlers (Alouatta pigra) in southern Belize and responses to hurricane Iris. In A. Estrada, P.A. Garber, M. S. M. Pavelka, & L. Luecke (Eds.), New perspectives in the study of mesoamerican primates: Distribution, ecology, behavior, and conservation (pp.143–163). New York: Springer.
Ratsimbazafy, J. H. (2002a). On the brink of extinction and the process of recovery: Responses of black-and-white ruffed lemurs (Varecia variegata variegata) to disturbance in Manombo Forest, Madagascar. Dissertation Abstracts International.
Ratsimbazafy, J. H., Ramarosandratana, H. V., & Zaonarivelo, R. J. (2002). How do black-and-white ruffed lemurs still survive in a highly disturbed habitat? Lemur News, 7, 7–10.
Reilly, A. E. (1991). The effects of hurricane Hugo in three tropical forests in the U.S. Virgin Islands. Biotropica, 23, 414–419.
Rivera, A., & Calme, S. (2006). Forest fragmentation and its effects on the feeding ecology of black howlers (Alouatta pigra) from the Calakmul area in Mexico. In A. Estrada, P. A. Garber, M. S. M. Pavelka, & L. Luecke (Eds.), New perspectives in the study of Mesoamerican primates: Distribution, ecology, behavior, and conservation (pp. 189–215). New York: Springer.
Rodriguez-Toledo, E. M., Mandujano, S., & Garcia-Orduna, F. (2003). Relationships between forest fragments and howler monkeys (Alouatta palliata mexicana) in southern Veracruz, Mexico. In L. K. Marsh, (Ed.), Primates in fragments: Ecology and conservation (pp. 79–96). New York, Boston, Dordrecht, London, Moscow: Kluwer Academic/Plenum Publishers.
Rudran, R., & Fernandez-Duque, E. (2003). Demographic changes over thirty years in a red howler population in Venezuela. International Journal of Primatology, 24, 925–947.
Rumiz, D. I. (1990). Alouatta caraya: population density and demography in northern Argentina. American Journal of Primatology, 21, 279–294.
Rylands, A. B., Groves, C. P., Mittermeier, R. A., Cortes-Ortiz, L., & Hines, J. J. H. (2006). Taxonomy and distributions of Mesoamerican primates. In A. Estrada, P. A. Garber, M. S. M. Pavelka, & L. Luecke (Eds.), New perspectives in the study of Mesoamerican primates: Distribution, ecology, behavior, and conservation (pp. 29–81). New York: Springer.
Scatena, F. N., & Larsen M. C. (1991). Physical aspects of hurricane Hugo in Puerto Rico. Biotropica, 23, 317–323.
Schowalter, T. D. (1994). Invertebrate community structure and herbiovry in a tropical rain forest canopy in Puerto Rico following hurricane Hugo. Biotropica, 26, 312–319.
Silver, S. C., Ostro, L. E. T., Yeager, C. P., & Horwich, J. (1998). Feeding ecology of black howler monkey (Alouatta pigra) in northern Belize. American Journal of Primatology, 45, 263–279.
Smith, J. D. (1970). The systematic status of the black howler monkey, Alouatta pigra Lawrence. Journal of Mammalogy, 51, 358–369.
Tanner, E. V. J., & Kapos, V. (1991). Hurricane effects on forest ecosystems in the Caribbean. Biotropica, 23, 513–521.
Van Belle, S., & Estrada, A. (2006). Demographic features of Alouatta pigra populations in extensive and fragmented forests. In A. Estrada, P. A. Garber, M. S. M. Pavelka, & L. Luecke (Eds.), New perspectives in the study of Mesoamerican primates: distribution, ecology, behavior, and conservation (pp. 121–143). New York: Springer.
Waide, R. B. (1991). Summary of the response of animal populations to hurricanes in the Caribbean. Biotropica, 23, 508–512.
Whitesides, G. H., Oates, J. F., Green, S. M., & Kluberdanz, R. P. (1988). Estimating primate densities from transects in a west African rain forest: A comparison of techniques. Journal of Animal Ecology, 57, 345–367.
Will, T. (1991). Birds of a severely hurricane damaged Atlantic coastal rain forest in Nicaragua. Biotropica, 23, 497–507.
Willig, M. R., & Camilo, G. R. (1991). The effect of hurricane Hugo on six invertebrate species in the Luquillo experimental forest of Puerto Rico. Biotropica, 23, 455–461.
Wright, P. C. (1999). Lemur traits and Madagascar ecology: coping with an island environment. Yearbook of Physical Anthropology, 42, 31–72.
Zunino, G. E., Gonzalez, V., Kowalewski, M. M., & Bravo, S. P. (2001). Alouatta caraya. Relations among habitat, density and social organization. Primate, Report, 61, 37–46.
Acknowledgments
We would like to thank the people of Monkey River for their help throughout this project and especially to our forest guides, as well as Tracy Wyman and Aaron Osicki for maps and Shelley Alexander for satellite images and for their assistance with all the mapping aspects of this project. We are grateful to all of the researchers who helped with the data collection, including Allison Maclean, Greg Bridgett, and Alison Behie, and to Linda Fedigan and Colin Chapman for helpful discussions regarding the framework of the paper. We would like to acknowledge our funding sources, including the Department of Anthropology, Faculty of Social Sciences, and University Research Grants Committee at the University of Calgary, Province of Alberta Graduate Scholarship, National Geographic, and Natural Sciences and Engineering Research Council of Canada Post Graduate Scholarship and Discovery Grant Programs. Finally, the manuscript was greatly improved by helpful comments by two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pavelka, M.S.M., McGoogan, K.C. & Steffens, T.S. Population Size and Characteristics of Alouatta pigra Before and After a Major Hurricane. Int J Primatol 28, 919–929 (2007). https://doi.org/10.1007/s10764-007-9136-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10764-007-9136-6