Abstract
Surveys of populations of spider and howler monkeys were conducted at the Mayan sites of Calakmul and Yaxchilán, Mexico and Tikal, Guatemala. The forests in which these sites are found are part of the largest landmass of tropical rain forests present in Mesoamerica, encompassing about 4 million ha. Triangulation of monkey vocalization combined with ground surveys was used to determine the presence of howler and spider monkey groups. Howler monkey mean troop size at these sites varied from 6.6±2.1 individuals in Yaxchilán to 7.5±1.9 in Calakmul to 8.7±2.2 in Tikal. Density estimates varied from 12.8 individuals/km2 in Yaxchilán to 15.2 individuals/km2 in Calakmul to 17.8 individuals/km2 in Tikal. Mean spider monkey subgroup size varied from 4.7±2.6 individuals in Tikal to 5.6±3.0 individuals in Yaxchilán to 7.7±3.8 individuals in Calakmul. Spider monkey density varied from 17.0 individuals/km2 in Yaxchilán to 17.2 individuals/km2 in Calakmul to 56.4 individuals/km2 in Tikal. All sightings of both howler and spider monkeys at the three sites were in undisturbed rain forest vegetation and spider monkeys in general were more frequently sighted at higher tree heights than howlers. We discuss the value of further acquiring data on howler and spider monkey populations existing in extensive forest tracts and on the conservation value for both primate species of the forests surrounding the Mayan ruins found in this area of Mesoamerica.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The black howler monkey, Alouatta pigra, is a primate species endemic to an area of the Mesoamerican region shared by Mexico, Guatemala, and Belize, and it is the only Alouatta species present in the Yucatán peninsula (Horwich and Johnson 1986; Watts and Rico-Gray 1987; Rylands et al. 1995). Population studies of A. pigra are available only for two sites in Belize (Horwich and Johnson 1986; Ostro et al. 1999), the Mayan site of Tikal in Guatemala (Coelho et al. 1976; Schlichte 1978), and more recently Muchukux and Palenque in Mexico (Gonzales-Kirchener 1998; Estrada et al. 2002a, 2002b). Such paucity of information and the rapid fragmentation and conversion of A. pigra‘s natural habitat to pasture lands and agricultural fields outside of protected forested areas makes the task of conservation particularly difficult for populations of this regionally endemic primate species.
Two subspecies of spider monkeys are present in Mexico, Ateles geoffroyi vallerosus and A. g. yucatanensis. The former subspecies is present in most of southern Mexico, while the latter is restricted to the Yucatán peninsula (Watts and Rico-Gray 1987). Population data are particularly scanty for both subspecies in the area of Mesoamerica shared by Mexico, Guatemala, and Belize and are only available for the site of Tikal, Guatemala (Coelho et al. 1976) and the Muchukux forest and the Punta Lagunas reserve in the Yucatan peninsula, Mexico (Gonzáles-Kirchner 1999; Ramos-Fernández and Ayala-Orozco 2003). Because their habitats are destroyed and fragmented, and because they are among the most frequently hunted monkeys for their meat and for the trafficking of infants as pets, spider monkeys are among the most endangered primates in Mesoamerica (Kinzey 1997; Rylands et al. 1997). Data on subgroup size, age, and sex composition and density for populations of A. geoffroyi are essential to document the existence of those areas where populations of spider monkeys are preserved and those areas where representatives of the species may be at risk.
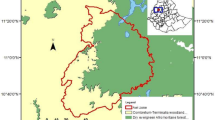
The largest protected landmass of tropical rain forest existing in Mesoamerica is represented by the Calakmul biosphere reserve in the southern portion of the Yucatan peninsula in the state of Campeche, Mexico, the adjacent Maya Reserve and Tikal National Park in Guatemala, and the Montes Azules biosphere reserve in Chiapas, Mexico. As a group, they constitute about 4 million ha of tropical rain forest (Fig. 1). Populations of A. geoffroyi and Alouatta pigra occur within this forested landmass, but information on where these populations are found and data on their size and state of conservation is almost inexistent (but see Coelho et al. 1976).
Location of study sites (black dots with smaller capital letters). Larger capital letters refer to the protected reserves. Calakmul is located within the Calakmul biosphere reserve (C; ca. 700,000 ha). Tikal is in Tikal National Park (T; ca. 6,000 ha) attached to the 3 million ha Maya reserve (M). Yaxchilán Natural Monument (Y; 2,700 ha) is located at the edge of the Usumacinta river that demarcates the international boundary between Guatemala and Mexico. This reserve is connected to the Community Reserve “La Cojolita” (35,000 ha), which in turn connects to the Biosphere Reserve of Montes Azules (MA; 300,000 ha). Vegetation type in Calakmul and Tikal is semideciduous tropical rain forest while in Yaxchilán high evergreen rain forest is the dominant vegetation type
In this article we present the results of initial surveys of populations of A. pigra and of Ateles geoffroyi in three sites within the above landmass of tropical rain forest. Two of these are located in Mexico, namely, the Mayan site of Calakmul, in the biosphere reserve of Calakmul, Campeche and the Mayan site of Yaxchilán in Chiapas. The third site was the Mayan site of Tikal in Tikal National Park, Guatemala. Our surveys in Tikal were aimed at gathering comparative data for the same primate species at a location outside of Mexico, but within the protected landmass of tropical rain forest. The forest of Tikal harbors a population of Alouatta pigra and of Ateles g. vellerosus that were surveyed about 30 years ago (Coelho et al. 1976).
Methods
Study sites
The Mayan site of Calakmul located in Campeche is found in the center of the southern portion of the 700,000-ha Calakmul biosphere reserve (18°06′N, 89°48′W; elevation ranges from 50 to 300 m above sea level) and it covers an area of 30 km2. The vegetation present in the area encompassed by the archeological site is semideciduous rain forest with trees reaching heights of 15–20 m with many species losing their leaves in the dry season (November to April; Rojas 2000). The climate is hot and humid and average annual precipitation is 820 mm, with a dry season between November and April (average monthly rainfall 21.4±25.4 mm) and a wetter period between May and January (average monthly rainfall 123.3±43.2 mm). Mean annual temperature is 25±2.2°C (range 21–28°C).
The Mayan site of Yaxchilán in the state of Chiapas is found within the protected forest of Natural Monument Yaxchilan (NMY; 16°53′N, 90°57′W; elevation 250 m above sea level) by the Usumacinta river bordering with Guatemala. The NMY encompasses about 2,700 ha, of which 1,130 ha are contained within an omega-shaped area by the river with the rest extending inland. This forest is connected to 35,000 ha of protected rain forest that form the Community Reserve “La Cojolita,” which in turn is connected to the Montes Azules biosphere reserve (Fig. 1). Tall evergreen rain forest (tree heights between 15 and 45 m) is the dominant vegetation at the study site (Valle 2000). The climate is hot and humid and average annual precipitation is 1,951 mm, with a dry season between December and April (average monthly rainfall 42.4±12.7) and a wetter period between May and November (average monthly rainfall 256.0±100.1 mm). Mean annual temperature is 25.5±2.2°C (range 21–28°C).
The archeological site of Tikal is located in Tikal National Park (TNP; 17°15′N, 89°35′W) in northern Guatemala. TNP encompasses an area about 576 km2 attached to the Maya biosphere reserve (Fig. 1). While the ancient Mayan city occupied about 16 km2, the major structures are distributed in an area 5 km2 in size surrounded or covered by rain forest vegetation. The vegetation present at TNP is tropical semideciduous forest (Lundel 1937). The weather is hot and humid and average annual precipitation is 1,762 mm, but with a distinct dry season between December and April, when average monthly rainfall is 60 mm. Average daily temperature is 25.0±8.5°C (Coelho et al. 1976).
Primate surveys
In Calakmul, surveys of black howler (Alouatta pigra) and spider monkeys (Ateles geoffroyi yucatanensis) were conducted in January, June, and November 2001 for a total of 30 days and in March 2002 for an additional 8-day period. Surveys were concentrated in a 4.0-km2 area around the major archeological buildings. In Yaxchilán, surveys of primates (Alouatta pigra and Ateles g. yucatanensis) were conducted in February, July, and November 2001 for a total of 30 days and in March 2002 for 8 extra days. These surveys were concentrated in a 1.0-km2 area to the north, south, and west of the major buildings. The eastern side of the site is formed by the Usumacinta river. Two additional surveys of howler and spider monkeys were conducted from a boat by navigating 13.7 km along the Usumacinta river following the contour of the 1,100-ha omega-shaped area where the site of Yaxchilán is located. These surveys started at 0500 hours and were conducted down river (S–N direction) with the outboard motor off, allowing for a gentle and silent flow of the boat down river. With a global positioning system (GPS; Garmin GPS III, Kansas, USA) we recorded the location of the boat when howler and/or spider monkey vocalizations were heard or when monkeys were sighted at the river’s edge. In Tikal, Guatemala, primate surveys (Alouatta pigra and Ateles g. vellerosus) were conducted in March 2002 for 8 days and were concentrated in the 5.0-km2 area in which howler and spider monkeys were studied 30 years earlier by Coelho et al.(1976), and in which the major archeological ruins are found.
During each field session in each site we triangulated during 2–3 consecutive mornings from 0500 to 0700 hours howler monkey howling from the top of the tallest (30–64 m) Mayan structures. Teams of 2–3 people were assigned to different Mayan structures to provide coverage of all parts of the forest surrounding the Mayan site. Participants recorded the time of the howling and estimated the approximate distance of the howling troop from the observers and its compass direction. When heard, spider monkey vocalizations were triangulated as well, but these were very occasional. Resulting information was placed on daily maps. Overlapping points provided an estimate of the number of howler troops in the area. All team member had ample experience in censusing howler (Alouatta pallliata and A. pigra) and spider monkeys at various sites in southern Mexico (e.g. Los Tuxtlas, Palenque).
For the ground surveys, our group was divided into teams of 2–3 people. Each team was assigned a different area of the study site for exploration, but these areas were not shared to avoid duplicity of monkey counts. In each area a team slowly walked (1.0 km/h) on existing trails (the system of trails at each site varied from 8 km in Calakmul to 7.5 km in Yaxchilán to 11 km in Tikal) or through the forest in search of the troops heard howling from the top of the Mayan buildings. Because of the deciduous nature of the vegetation at the time of our surveys, maximum visibility of monkeys on each side of the trails in Calakmul was approximately 40 m, in Yaxchilán about 20 m, and in Tikal about 30 m. When a troop of howler monkeys or a subgroup of spider monkeys was sighted, we noted its location on detailed topographic maps (1:6250) of the study sites. Major archeological buildings were depicted on the maps and these served as important reference points to map location of troop contacts. A GPS was also used to obtain precise georeference points for howler troop and/or spider monkey subgroup sightings and movements, which were plotted on the daily maps. The maps from several days were overlapped to determine the consistency in the spatial location of howler troops or of subgroups of spider monkeys.
Contacted howler and spider monkey groups were followed for as long as possible and repeatedly counted to confirm number of individuals as well as age and sex composition. Individual howler and spider monkeys were classified as adults, juveniles, and infants by their size and appearance. In the adult class we did not differentiate between subadults and adults and in the infant class we considered both clinging infants and infants that could move away from the mother when stationary. Howler monkey troops contacted in the first 2–3 days were encountered and followed in subsequent days, further confirming their identities and general location. Spider monkey subgroups for which we had accurate counts and that were identified, by examining the consistency in size, age, and sex composition and spatial location, as not being repeated counts of the same subgroup were considered as independent social units. When monkeys were sighted we estimated the height at which they were found.
We expressed density (individuals per square kilometer) figures in terms of the area sampled rather than density of the species’ home range (Chapman and Balcomb 1998). Thus, in Calakmul we sampled 4.0 km2 and in Tikal 5.0 km2. In these two sites we were certain of having identified all howler troops existing within the sampled area. In Yaxchilán we sampled an area 1.0 km2 around the major Mayan building, and the river survey around the omega-shaped site allowed us to sample an additional 10.3 km2. In this site we only obtained accurate counts of troop size and age composition for those howler monkey troops present in the 1.0-km2 area around the Mayan ruins. The triangulation of troop howling in the river survey yielded information on additional troops existing in the omega-shaped area. In this survey only two of the troops detected were present at the edge of the river; the rest of the troops were inland as indicated by the estimated distances (100–500 m) from the boat. Mean group size obtained from the counts of howler troops censused in the 1.0-km2 area was multiplied by the total number of troops recorded for the entire omega-shaped area (11.3 km2) to give a gross estimate of population density.
Spider monkeys live in small temporary subgroups of unstable composition, which are part of larger groups or communities (van Roosmalen and Klein 1988; Kinzey 1997). Because of the fusion–fission nature of their social organization, it is rare to see all members of the community in the same location, suggesting that it is not easy to make generalizations on density and/or subgroup size for this primate species (Coelho et al. 1976; Klein and Klein 1977). Thus for the calculation of spider monkey density we took into account confirmed subgroup counts within each study area (4 km2 in Calakmul, 1 km2 in Yaxchilán, and 5 km2 in Tikal).
Results
Alouatta pigra
In Calakmul auditory survey indicated the presence of eight troops that were located on the ground. In these troops we counted a total of 60 howler monkeys and detected one solitary male (Table 1). Thirty-three percent of the individuals counted in the troops were adult males, 30% were adult females, 14% were juvenile males, 10% were juvenile females, and 13% were infants. Six troops had three adult males and two troops had one adult male. Adult sex ratio was 1:0.90 and in juveniles it was 1:0.75. Adult female to immature ratio was 1:1.22.
Auditory survey in Yaxchilán resulted in the detection of 11 troops of howler monkeys. Eight of these were contacted on the ground surveys and the other 3 could not be found. In the 8 troops we counted 53 howler monkeys. One solitary male was located in the area. Forty-one percent of the individuals counted in the troops were adult males, 30% were adult females, 8% were juvenile males, 6% were juvenile females, and 15% were infants (Table 2). Adult sex ratio was 1:0.73, in juveniles it was 1:0.75, and adult female to immature ratio was 1:0.97. The survey in the Usumacinta river along the contour of the 11.3-km2 omega-shaped area where Yaxchilán is located yielded the auditory identification of 11 additional troops of howler monkeys.
In Tikal auditory surveys detected ten troops that were also contacted on the ground surveys. In total we counted 88 howler monkeys in these troops and discovered two solitary males. Twenty-five percent of individuals in the troops were adult males, 33% were adult females, 7% were juvenile males, 14% were juvenile females, and 21% were infants. Ninety percent of troops had more than one adult male (Table 3). Adult sex ratio was 1:1.32 and in juveniles it was 1:2.0. Adult female to immature ratio was 1:1.23.
Mean troop size at the three sites varied from 6.6±2.1 individuals in Yaxchilán to 7.5±1.9 in Calakmul to 8.7±2.2 in Tikal. Density estimates varied from 12.8 individuals/km2 in Yaxchilán to 15.2 individuals/km2 in Calakmul to 17.8 individuals/km2 in Tikal.
Ateles geoffroyi
Nine spider monkey subgroups were clearly identified in Calakmul, yielding a total count of 69 individuals. Twenty-two percent of individuals counted were adult males, 45% were adult females, 14% were juvenile males, 7% were juvenile females, and 12% were infants (Table 1). Adult sex ratio was 1:2.07 and in juveniles it was 1:0.53. Adult female to immature ratio was 1:0.74. In Yaxchilán three subgroups of spider monkeys were counted in the 100-ha study area with a total of 17 individuals. These subgroups were observed several times in different locations and sometimes in the same trees as howler monkeys. Adult males accounted for 35.3% of individuals counted, adult females for 29.4%, juvenile males for 5.9%, juvenile females for 11.8%, and infants for 17.6% (Table 2). Adult sex ratio was 1:0.83 and in juveniles it was 1:2.0; adult female to immature ratio was 1:1.20.
In Tikal we recorded 145 sightings of Ateles and these were distributed in practically all sectors of the study area. Careful examination of the consistency in location, date, time of the day, and age and sex composition of the spider monkey subgroup indicated that the 145 sightings corresponded to 99 distinct subgroups of spider monkeys in the study area, accounting for about 282 spider monkeys (Table 3). Adult sex ratio was 1:1.61; among juveniles this ratio was 1:1.53. Adult female to immature ratio was 1:0.71.
Mean spider monkey subgroup size varied from 4.7±2.6 individuals in Tikal to 5.6±3.0 in Yaxchilán to 7.7±3.8 in Calakmul. Spider monkey density varied from 17.0 individuals/km2 in Yaxchilán to 17.2 individuals/km2 in Calakmul to56.4 individuals/km2 in Tikal.
All sightings of both howler and spider monkeys at the three sites were in undisturbed rain forest vegetation. No statistical differences were found among sites in the heights at which individuals were sighted for each species (Kruskal–Wallis test H=5.0, df=5, P>0.40 in both cases) and samples were pooled for each species. On average spider monkeys were detected at higher tree heights and occupied a broader spectrum of heights than howler monkeys (Fig. 2).
Distribution of heights at which Ateles geoffroyi and Alouatta pigra were sighted in Calakmul, Yaxchilán, Mexico, and in Tikal, Guatemala. No statistical differences (Kruskal–Wallis test) were found among sites and thus samples were pooled for each species. Spider monkeys (dark bars) were sighted, on average, at higher heights than howler monkeys and they also moved along a broader spectrum of heights than howler monkeys. Number of pooled records for spider monkeys was 230 and for howlers 120
Discussion
Our surveys showed the presence of populations of howler and spider monkeys at the sites investigated. While varying in some demographic parameters, such populations seem well preserved in the extensive forest tracts they inhabit. Density values we report for howlers in the sites surveyed are similar to those found for the species in other sites in Mexico, namely Muchukux, Quintana Roo (15.1 individuals/km2; Gonzales-Kirchener 1998) and Palenque (23.0 individuals/km2; Estrada et al. 2002a). High densities of Alouatta pigra in Belize (up to 178 individuals/km2) have been suggested to be the result of overcrowding in fragmented strips of riparian vegetation and small forest patches (Silver et al. 1998; Ostro et al. 1999, 2000; Horwich et al. 2001), a situation similar that of a population of the same species living in fragmented landscapes in Palenque, Mexico (Estrada et al. 2002b). Our data suggest that adult female to immature ratios tend to be, on average, higher in A. pigra existing in extensive forest tracts (mean for the three sites 1:1.28±0.27) than in the fragmented habitats of Belize where the species has been studied (mean 1:0.92±0.23). Our survey in Tikal suggests an increase from four to ten troops in the howler monkey population since the earlier survey 30 years ago in the same study area (Coelho et al. 1976).
The black howler monkey has been reported to occupy a variety of habitats ranging from undisturbed tropical rain forest to disturbed and riverine forests (Horwich and Johnson 1986; Horwich and Lyon 1987; Watts and Rico-Gray 1987). The species is generally found in riparian forests at elevations below 400 m; an exception to this is the population in Tikal, Guatemala (Ostro et al. 2000). Data from Calakmul and Palenque (Estrada et al. 2002a) indicate that A. pigra prefers the undisturbed nonriparian forest habitats that dominate these sites and in Palenque black howler monkeys may also be found living in forests at 500 m above sea level (Estrada et al. 2002a). In Yaxchilán our survey showed that only 9% of the troops were detected at the edge of the Usumacinta river; the rest were found inland.
Bearing in mind the problem of adequately estimating spider monkey densities at the three sites, these values ranged from 17.2 to 56.4 individuals/km2. At other sites in the Yucatan peninsula spider monkey density varies from 6.3 to 89.5 individuals/km2 (Gonzales-Kirchener 1999; Ramos-Fernandez and Ayala-Orozco 2003). The density estimate we report for Ateles geoffroyi in Tikal(56.4 individuals/km2)is similar to that of 45 individuals/km2 reported 30 years earlier (Coelho et al. 1976), but Cant (1978) in a subsequent study reported densities of 26 individuals/km2 for the species in the same site. We estimated the presence of 282 spider monkeys in the study area, a figure not too different from the estimated 225 spider monkeys reported about 30 years earlier for the same study area (Coelho et al. 1976).
Spider monkeys in the three sites consistently displayed a preference for the canopy of tall trees. They also displayed much vertical mobility when traveling and opportunistically foraged on low trees bearing ripe fruit, as has been reported for this primate at other localities (van Roosmalen and Klein 1988; Yoneda 1990). In contrast, howler monkeys in the same forest preferred lower heights than spider monkeys.
Habitat destruction and fragmentation outside of protected areas is a major cause of decline of primate populations in the Neotropics (Estrada and Coates-Estrada 1996; Rylands et al. 1997; Cuarón 2000). Hunting and trafficking of infants as pets are important additional pressures that contribute to declines of populations of primates in Mesoamerica and these are exacerbated under conditions of habitat fragmentation (Watts and Rico-Gray 1987; Ráez-Luna 1995; Estrada and Coates-Estrada 1996). Habitat isolation may have a range of effects that can result in local extinction due to genetic and/or demographic stochasticity and in alteration of demographic structures and reductions in population growth rates (Crockett 1988; Laurance et al. 2002). In this context, the study and monitoring of primate populations in national parks and biosphere reserves is fundamental to providing baseline and long-term information on variability of demographic traits. Such information is essential for improving our understanding of species’ tolerance to anthropogenic disturbance and to assess whether these protected forests are a functioning network and an effective conservation tool (Sánchez-Azofeifa et al. 2003).
The protection and connectedness of the forests constituted by the Calakmul biosphere reserve, the Maya Reserve, Tikal National Park, Yaxchilan Natural Monument, and the Montes Azules biosphere reserve seem to be ensuring the existence of populations of Alouatta pigra and Ateles geoffroyi, but more information is needed about the location of these populations as these reserves encompass a very large land mass and a variety of habitat types. The concentration of howler and spider monkey groups in the core area of the Mayan ruins at each of the sites investigated is noteworthy. Such concentration could be the result of the abundance in these areas of tree species of the genus Brosimum, Ficus, and Poulsenia (Moraceae), Manilkara, Pouteria (Sapotaceae), Spondias (Anacardiaceae), and Bursera (Burseraceae; Lundel 1937; Rojas 2000; Valle 2000), among others, known to be important in the diet of howler and spider monkeys in the region (Coelho et al. 1976; Cant 1990; Silver et al. 1998). More importantly, the conservation of primate populations in these sites may be the result of the continuous protection of these cultural and touristic areas by the governments of Mexico and Guatemala. For now, our surveys in Calakmul, Yaxchilán, and Tikal, as well as those in Palenque (Estrada et al. 2002a), show that the protection of the forests surrounding several Mayan sites has resulted in important foci of conservation of populations of Alouatta pigra and Ateles geoffroyi in this area of Mesoamerica.
References
Cant JGH (1978) Population survey of the spider monkey (Ateles geoffroyi) at Tikal, Guatemala. Primates 19:525–535
Cant JGH (1990) Feeding ecology of spider monkeys, Ateles geoffroyi at Tikal, Guatemala. Hum Evol 5:269–281
Chapman CA, Balcomb SR (1998) Population characteristics of howlers: ecological conditions of group history. Int J Primatol 19:385–403
Coelho AM Jr, Coelho L, Bramblett C, Bramblett S, Quick LB (1976) Ecology, population characteristics, and sympatric associations in primates: a sociobioenergetic analysis of howler and spider monkeys in Tikal, Guatemala. Yearb Phys Anthropol 20:96–135
Crockett C (1998) Conservation biology of the genus Alouatta. Int J Primatol 19:549–578
Cuarón AD (2000) Effects of land-cover changes on mammals in a Neotropical region: a modeling approach. Conserv Biol 14:1676–1692
Estrada A, Coates-Estrada R (1996) Tropical rain forest fragmentation and wild populations of primates at Los Tuxtlas. Int J Primatol 5:759–783
Estrada A, Castellanos L, García Y, Franco B, Muñoz D, Ibarra A, Rivera A, Fuentes E, Jimenez C (2002a) Survey of the black howler monkey, Alouatta pigra, population at the Mayan site of Palenque, Chiapas, Mexico. Primates 44:51–58
Estrada A, Mendoza A, Castellanos L, Pacheco R, Van Belle S, García Y, Muñoz Z (2002b) Population of the black howler monkey (Alouatta pigra) in a fragmented landscape in Palenque, Chiapas, Mexico. Am J Primatol 58:45–55
Gonzales-Kirchener JP (1998) Group size and population density of the black howler monkey (Alouatta pigra) in Muchukux forest, Quintana Roo, Mexico. Folia Primatol 69:260–265
Gonzales-Kirchener JP (1999) Habitat use, population density and subgrouping pattern of the Yucatan spider monkey (Ateles geoffroyi yucatanensis) in Quintana Roo, Mexico. Folia Primatol 70:55–60
Horwich RH, Johnson EW (1986) Geographic distribution of the black howler monkey (Alouatta pigra) in Central America. Primates 27:53–62
Horwich RH, Lyon J (1987) An experimental technique for the conservation of private lands. J Med Primatol 17:169–176
Horwich RH, Brockett RC, James RA, Jones C (2001) Population growth in the Belizean black howling monkey (Alouatta pigra). Neotrop Primates 9:1–7
Kinzey WG (1997) Ateles. In: Kinzey WG (ed) New world primates: ecology, evolution and behavior. Aldine de Gruyter, New York, pp 192–199
Klein LL, Klein DJ (1977) Neotropical primates: aspects of habitat usage, population density and regional distribution in La Macarena, Colombia. In: Thorington RW Jr, Heltne PG (eds) Neotropical primates: field studies and conservation. National Academy of Sciences, Washington, D.C, pp 70–99
Laurance WF, Lovejoy TE, Vasconcelos HL, Bruna EM, Didhem RK, Stouffer PC, Gascon C, Bierregaard RO, Laurance SG, Sampelo E (2002) Ecosystem decay of Amazonian forest fragments: a 22 year investigation. Conserv Biol 16:605–619
Lundel CL (1937) The vegetation of Peten. Carnegie Institute Publ 478
Ostro LET, Silver SC, Koontz FW, Young TP, Horwich RH (1999) Ranging behavior of translocated and established groups of black howler monkeys Alouatta pigra in Belize, Central America. Biol Conserv 87:181–190
Ostro LET, Silver SC, Koontz FW, Young TP (2000) Habitat selection by translocated black howler monkeys in Belize. Anim Conserv 3:175–181
Ráez-Luna EF (1995) Hunting large primates and conservation of Neotropical rain forests. Oryx 29:43–48
Ramos-Fernández G, Ayala-Orozco B (2003) Population size and habitat use of spider monkeys in Punta Laguna, Mexico. In: Marsh LK (ed) Primates in fragments: ecology and conservation. Kluwer/Plenum Press, New York, in press
Rojas S (2000) La reserva de la biosfera de Calakmul. Arqueol Mex 7:46–51
Roosmalen MGM van, Klein LL (1988) The spider monkey, genus Ateles. In: Mittermeier RA, Rylands AB, Coimbra-Filho AF, Fonseca BAB (eds) Ecology and behavior of Neotropical primates. Academia Brasileira de Ciencias, Rio de Janeiro, pp 455–537
Rylands A, Mittermeier RA, Rodríguez-Luna E (1995) A species list for the New World primates (Platyrrhini): distribution by country, endemism, and conservation status according to the Mace-Land system. Netrop Primates 3 (Suppl):114–164
Rylands AB, Mittermeir RA, Rodriguez-Luna E (1997) Conservation of neotropical primates: threatened species and an analysis of primate diversity by country and by region. Folia Primatol 8:134–160
Sánchez-Azofeifa AG, Daily GC, Pfaff ASP, Busch C (2003) Integrity and isolation of Costa Rica’s national parks and biological reserves: examining the dynamics of land-cover change. Biol Conserv 109:123–135
Schlichte H (1978) A preliminary report on the habitat utilization of a group of howler monkeys (Alouatta villosa pigra) in the national Park of Tikal, Guatemala. In: Montgomery GG (ed) The ecology of arboreal folivores. Smithsonian Institution Press, Washington, D.C., pp 551–561
Silver SC, Ostro LET, Yeager CP, Horwich R (1998) Feeding ecology of the black howler monkey (Alouatta pigra) in northern Belize. Am J Primatol 45:263–279
Valle JA (2000) Analisis estructural de una hectarea de selva alta perennifolia en El Monumento Natural Yaxchilán (Chiapas), México. Bachelor’s thesis, University of Mexico (UNAM)
Watts E, Rico-Gray V (1987) Los primates de la peninsula de Yucatán, México: estudio preliminar sobre su distribución actual y estado de conservación. Biótica 12:57–66
Yoneda M (1990) The difference of tree size used by five Cebid monkeys in Macarena, Colombia. Field Studies of New World Monkeys, La Macarena, Colombia 3:13–18
Acknowledgements
We are grateful to the Cleveland Zoo Scott Neotropical Fund for support to conduct this study. We thank the Mexican Institute of Anthropology (INAH) for permission to work in Calakmul and Yaxchilán and thank the authorities of Tikal National Park for permission to conduct the primate surveys and for providing invaluable logistical support. LL acknowledges support from the School of Liberal Arts at University of Texas. SVB acknowledges the support of Secretaria de Relaciones Exteriores de México and of Universidad Nacional Autónoma de México. MM thanks Universidad Nacional Autónoma de México and Universidad de San Carlos, Guatemala, for support. We also thank three anonymous reviewers and M. Andrews for valuable suggestions to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Estrada, A., Luecke, L., Van Belle, S. et al. Survey of black howler (Alouatta pigra) and spider (Ateles geoffroyi) monkeys in the Mayan sites of Calakmul and Yaxchilán, Mexico and Tikal, Guatemala. Primates 45, 33–39 (2004). https://doi.org/10.1007/s10329-003-0062-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-003-0062-8