Abstract
Cepharanthine, a biscolaurine alkaloid isolated from the plant Stephania cephalantha Hayata, has been reported to have potent anti-inflammatory properties. Here, we investigated the effects of cepharanthine on the expression of CXCL10 (a CXC chemokine induced by interferon-gamma [IFN-γ] that has been observed in a wide variety of chronic inflammatory disorders and autoimmune conditions) in IFN-γ-treated human salivary gland cell lines. We observed that IFN-γ-induced CXCL10 production in NS-SV-DC cells (a human salivary gland ductal cell line), but not in NS-SV-AC cells (a human salivary gland acinar cell line). Cepharanthine inhibited the IFN-γ-induced CXCL10 production in NS-SV-DC cells. A Western blot analysis showed that cepharanthine prevented the phosphorylation of JAK2 and STAT1, but did not interfere with the NF-κB pathway. Moreover, cepharanthine inhibited the IFN-γ-mediated chemotaxis of Jurkat T cells. These results suggest that cepharanthine suppresses IFN-γ-induced CXCL10 production via the inhibition of the JAK2/STAT1 signaling pathway in human salivary gland ductal cells. Our findings also indicate that cepharanthine could inhibit the chemotaxis of Jurkat T cells by reducing CXCL10 production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Primary Sjögren’s syndrome (pSS), one of the most common autoimmune diseases [1], is characterized by the eventual total replacement of the acinar structure by marked lymphocytic infiltrates in the salivary and lacrimal glands [2]. The pathogenesis of this selective and progressive destruction of the acinar structure in the salivary glands is not yet fully understood. However, accumulated evidence indicates a close relationship between cytokine expression in salivary gland tissue and the development and progression of this disease [3, 4]. Among several cytokines, especially interferons (IFNs) [5, 6] and tumor necrosis factor-alpha (TNF-α) [7], have been suggested to play an important role in the pathogenesis of pSS.
CXCL10, a CXC chemokine induced by IFN-γ, is produced by diverse cell types, including peripheral blood mononuclear cells (PBMC), fibroblasts, and endothelial cells during Th1-type immune responses [8]. CXCL10 binds its receptor to CXCR3. CXCR3 is widely expressed on multiple types of cells of the innate immune system, including dendritic cells, natural killer (NK) cells, NKT cells, neutrophils, and macrophages [9,10,11,12]. Thus, these entirely different innate immune cells are the potential targets for CXCL10-mediated chemotaxis.
The expression of CXCL10 has been observed in pSS. Ogawa et al. [13, 14] reported that Th1 chemokines such as CXCL9, CXCL10, and CXCL11 were involved in the accumulation of T cell infiltrates in the salivary glands of pSS patients. Moreover, the inoculation of a CXCL10 antagonist into MRL/lpr mice during the early stage of sialadenitis significantly reduced the mononuclear cell infiltration and parenchymal destruction [15].
Cepharanthine (Kaken Pharmaceutical Co. Ltd., Tokyo), a biscoclaurine alkaloid extracted from the plant Stephania cephalantha Hayata, has been used widely for the treatment of a number of acute and chronic diseases, for leukopenia during radiation therapy, and as an anticancer treatment [16]. Although the exact mechanism has not been elucidated, cepharanthine exerts immunomodulatory effects by enhancing the cytotoxic effect of NK cells and macrophages [17, 18], suggesting that cepharanthine may play a role in the regulation of signaling pathways of cytokines. It has been demonstrated that cepharanthine effectively suppressed TNF-α-induced matrix metalloproteinase (MMP)-9 production, leading to the restoration of normal acinar structures in both an in vitro culture system and an in vivo murine SS model [19, 20]. Additionally, a single-center open-label study showed efficacy of cepharanthine on the increase of salivary flow in pSS patients [21]. However, an analysis to identify the influence of cepharanthine on CXCL10 expression in salivary gland cells has not been performed.
We therefore examined the effect of cepharanthine on the regulation of IFN-γ-induced CXCL10 expression by using immortalized human salivary gland cell clones in an in vitro experiment. We also investigated whether cepharanthine regulates the CXCL10-mediated chemotaxis of human T cell leukemia cells.
MATERIALS AND METHODS
Cell Culture
The characteristics of the cell lines NS-SV-DC (immortalized human salivary gland ductal cells) and NS-SV-AC (immortalized human salivary gland acinar cells) are described in detail elsewhere [22]. These cell clones were cultured at 37 °C in serum-free keratinocyte medium (Gibco Laboratories, Gaithersburg, MD) in an incubator with an atmosphere containing 5% CO2. Jurkat human leukemic T cells (Riken Cell Bank, Ibaraki, Japan) were maintained in RPMI-1640 medium (Gibco Laboratories), which was supplemented with 10% fetal bovine serum (FBS, Gibco) in a 5% CO2-humidified incubator at 37 °C.
Reagents
Recombinant human IFN-γ was purchased from R&D Systems (Minneapolis, MN). Cepharanthine was purchased from Kaken Shoyaku Co. (Tokyo).
Cell Growth Assay
NS-SV-DC cells (1 × 104 cells/well) and NS-SV-AC cells (1 × 104 cells/well) were seeded in 96-well plates (Falcon, Oxnard, CA) in serum-free keratinocyte medium. Twenty-four hours later, the cells were treated with cepharanthine (0.1–50 μg/ml). After appropriate incubation periods, an MTT reagent (Roche, Basel, Switzerland) was added to each well and incubation was continued for 4 h. The cells were dissolved in solubilization solution (Roche) and read at 570 nm in a microtiter plate reader.
Enzyme-Linked Immunosorbent Assay
NS-SV-DC and NS-SC-AC cells were plated onto 96-well plates (1 × 105 cells/well) and treated with various concentrations of IFN-γ for 6, 12, or 24 h. Each concentration of CXCL10 was determined by an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems) according to the manufacturer’s instructions. Optimal absorbance was read at 450 nm in a microtiter plate reader.
Quantitative Real-Time Reverse Transcription-Polymerase Chain Reaction
NS-SV-DC cells were treated with IFN-γ (10 ng/ml) for 6, 12, or 24 h in the presence or absence of various concentrations of cepharanthine. Total cellular RNA was isolated with TRIzol Reagent (Life Technologies, Carlsbad, CA). The cDNA was synthesized from total RNA with the use of an Advantage cDNA PCR Kit (Clontech, Palo Alto, CA). We quantitatively analyzed the expression levels of mRNAs for CXCL10 and GAPDH using an ABI Prism 7000 Sequence Detection System (Applied Biosystems Japan, Tokyo), TaqMan® Universal PCR Master Mix (Applied Biosystems), and Assays-on-Demand™ Gene Expression Products (Applied Biosystems) according to the manufacturer’s recommendations.
The thermal cycler protocol was 95 °C for 10 min, followed by 40 cycles of 95 °C for 5 s and 60 °C for 30 s. We performed an analysis of the relative gene expression data using the 2−ΔΔCT method on Sequence Detection System Software (Applied Biosystems). We calculated the fold change in the studied gene expression, normalized to an endogenous control, using the formula RQ = 2−ΔΔCT. The relative expression levels of CXCL10 mRNAs are expressed as a fold increase in the GAPDH mRNA expression.
Protein Isolation and Western Blot Analysis
The cells were treated with 10 ng/ml IFN-γ for 5, 10, 30, 60, 120, or 240 min in the presence or absence of cepharanthine (10 μg/ml). Whole cell lysates were prepared using M-PER lysis solution (Thermo Fisher Scientific, Waltham, MA) supplemented with a protease/phosphatase inhibitor mixture (Thermo Fisher Scientific). Cytosolic extracts (30 μg) were subjected to electrophoresis on 10% sodium dodecyl sulfate (SDS)-polyacrylamide gels (Bio-Rad, Hercules, CA), and then transferred onto nitrocellulose membranes. The membranes were blocked with 3% bovine serum albumin (BSA) and incubated with each of the following antibodies (all from Cell Signaling Technology, Beverly, MA, diluted at 1:1000): anti-Janus kinase 1 (JAK1), anti-phospho-JAK1, anti-JAK2, anti-phospho-JAK2, anti-STAT1, anti-phospho-STAT1, and anti-β-actin. After intervening rinses with Tris-buffered saline with Tween 20 (TBS-T), the IgG secondary antibodies (Cell Signaling Technology, diluted at 1:1000) were used for the respective primary antibodies. The immune complexes were visualized by enhanced chemiluminescence (ECL) Western Blotting Detection Reagent (GE Healthcare, Buckinghamshire, UK). The density of the visualized immune complexes was digitized using an Amersham Imager 600 (GE Healthcare).
Cell Migration Assay
We analyzed Jurkat cells’ directional migration, which was induced by conditioned medium (CM) derived from IFN-γ-treated NS-SV-DC cells, using a CytoSelect™ 96-Well Cell Migration Assay (5 μm, Fluorometric Format, Cell Biolabs, San Diego, CA) according to the manufacturer’s instructions. In brief, NS-SV-DC cells were treated with 10 ng/ml IFN-γ in the presence or absence of cepharanthine (1 or 10 μg/ml) for 24 h, and then 150 μl of CM was placed in the wells of the bottom (feeder tray). Next, 100 μl of serum-free medium containing 5 × 105 Jurkat cells was placed in the migration chamber. The chemotaxis plate was cultured at 37 °C for 12 h. After incubation, the cells that had migrated to the lower chambers were incubated for 20 min with 50 μl of Lysis Buffer/Dye Solution (Cell Biolabs). Fluorescence was read at 480/520 nm. Values were expressed as relative fluorescence units (RFU). The experiments were performed in triplicate.
Statistical Analysis
The statistical analysis was performed by Mann-Whitney U test, and p values < 0.05 were considered significant.
RESULTS
Cell Viability Assay of Cepharanthine-Treated NS-SV-DC and NS-SV-AC Cells
We used an MTT assay to determine the growth kinetics of NS-SV-DC and NS-SV-AC cells treated with various concentrations of cepharanthine for up to 3 days. The cell growth of these two cell lines decreased at concentrations of 20–50 μg/ml of cepharanthine (Fig. 1). We thus used cepharanthine at the concentrations up to 10 μg/ml in the subsequent experiments.
Growth inhibitory effect of cepharanthine on NS-SV-DC and NS-SV-AC cells. NS-SV-DC cells (a) and NS-SV-AC cells (b) (1 × 104 cells/well) were seeded into 96-well plates. After 24 h, cells were treated with cepharanthine (0.1–50 μg/ml). At different time intervals, cell growth was evaluated by MTT assay. Bar: mean ± SD. *p < 0.05. **p < 0.01.
IFN-γ-Induced CXCL10 Production in NS-SV-DC Cells
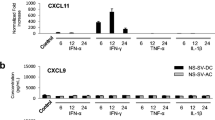
We conducted the ELISA assay to examine the production of CXCL10 after treatment with 10 ng/ml of IFN-γ for 24 h in NS-SV-DC and NS-SV-AC cells. As shown in Fig. 2a, a significant increase in the production of CXCL10 protein was detected in NS-SV-DC cells, but not in NS-SV-AC cells. We also examined the production of CXCL10 in NS-SV-DC cells by ELISA assay after treatment with 1, 10, 50, and 100 ng/ml of IFN-γ for 24 h. Figure 2b shows that the CXCL10 production was increased by treatment with IFN-γ in a dose-dependent manner.
CXCL10 production following IFN-γ stimulation. a NS-SV-DC and NS-SC-AC cells were treated with IFN-γ (10 ng/ml) for 6, 12, or 24 h. An ELISA was performed to measure CXCL10 protein in the conditioned medium. Bar: mean ± SD. *p < 0.05. b NS-SV-DC cells were treated with 1, 10, 50, or 100 ng/ml of IFN-γ for 24 h. An ELISA was performed to measure CXCL10 protein in the conditioned medium. Bar: mean ± SD. *p < 0.05.
Cepharanthine Inhibited the IFN-γ-Induced CXCL10 Production in NS-SV-DC Cells
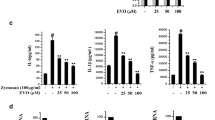
To identify the effect of cepharanthine on the IFN-γ-induced CXCL10 expression at the mRNA level, we treated NS-SV-DC cells with 0.1, 1, and 10 μg/ml of cepharanthine under stimulation of 10 ng/ml of IFN-γ for 12 h. As shown in Fig. 3a, cepharanthine (0.1–10 μg/ml) significantly suppressed the level of IFN-γ-induced CXCL10 mRNA in NS-SV-DC cells.
Cepharanthine inhibited the IFN-γ-induced CXCL10 production in NS-SV-DC cells. a NS-SV-DC cells were treated with IFN-γ (10 ng/ml) in the presence or absence of cepharanthine (0.1–10 μg/ml) for 12 h. Total RNAs were prepared, and the quantification of mRNA levels was evaluated by qRT-PCR. Bar: mean ± SD. *p < 0.05. b NS-SV-DC cells were treated with IFN-γ (10 ng/ml) in the presence or absence of cepharanthine (0.1–10 μg/ml) for 24 h. An ELISA was performed to measure the CXCL10 protein in the conditioned medium. Bar: mean ± SD. *p < 0.05.
We then examined CXCL10 protein expression using an ELISA assay. NS-SV-DC cells were treated with 10 ng/ml of IFN-γ in the presence of 0.1, 1, or 10 μg/ml of cepharanthine for 24 h. As a consequence, the IFN-γ-induced CXCL10 protein was significantly suppressed by cepharanthine (Fig. 3b).
Effects of Cepharanthine on the JAK2/STAT1 and NF-κB Signaling Pathways in IFN-γ-Stimulated NS-SV-DC Cells
IFN-γ is known to activate the JAK/STAT signaling kinases to regulate gene expression [23]. We thus evaluated the effects of cepharanthine on IFN-γ-induced JAK1, JAK2 and STAT1 activation in NS-SV-DC cells. The cells were treated with 10 ng/ml of IFN-γ in the presence or absence of cepharanthine (10 μg/ml). The Western blot analysis demonstrated that phospho-JAK2 and phospho-STAT1 were significantly increased after IFN-γ stimulation in NS-SV-DC cells (Fig. 4a), whereas the expression of JAK1 was not observed in NS-SV-DC cells (data not shown). In contrast, cepharanthine treatment led to a reduction in IFN-γ-induced JAK2 and STAT1 phosphorylation in NS-SV-DC cells (Fig. 4a, b).
Effects of cepharanthine on the JAK2/STAT1 pathway in IFN-γ-stimulated NS-SV-DC cells. a The NS-SV-DC cells were treated with 10 ng/ml of IFN-γ in the presence or absence of cepharanthine (10 μg/ml). A Western blot analysis showed that the phosphorylation of JAK2 and STAT1 was increased after IFN-γ stimulation in NS-SV-DC cells. b The bar graphs of phospho-JAK2 or phospho-STAT1 expression were normalized to β-actin.
Since CXCL10 gene contains NF-κB and STAT1-responsible elements (ISRE; interferon-stimulated response element) in the promoter regions [24], we next attempted to examine the effect of cepharanthine on NF-κB signaling in NS-SV-DC cells. However, we could not identify NF-κB activation by IFN-γ stimulation in this cell line, i.e., the degradation of IκB-α and the translocation of p65 to the nucleus were not detected in IFN-γ-stimulated NS-SV-DC cells (data not shown). This result was consistent with our previous observation that NF-κB activation was not detected in NS-SV-DC cells due to the lack of IκB-α protein and mRNA [25].
Effects of Cepharanthine on the Chemotaxis of Jurkat T Cells
Since CXCL10 has been reported to accumulate CXCR3+ T cells in lip salivary glands (LSGs) of SS patients [13], we analyzed the ability of cepharanthine to recruit Jurkat T cells in IFN-γ-treated NS-SV-DC cells. The migration assay results demonstrated that the chemotaxis of Jurkat cells was significantly increased in IFN-γ-treated NS-SV-DC cells compared to IFN-γ-untreated NS-SV-DC cells (Fig. 5). In addition, cepharanthine significantly inhibited the IFN-γ-mediated chemotaxis of Jurkat cells (Fig. 5). These results indicated that cepharanthine has the potential to inhibit the chemotaxis of CXCR3+ T cells through the inhibition of CXCL10.
Effects of cepharanthine on the chemotaxis of Jurkat T cells. NS-SV-DC cells were treated with 10 ng/ml IFN-γ in the presence or absence of cepharanthine (10 μg/ml) for 24 h. Serum-free Jurkat cells were placed in the migration chamber. The chemotaxis plate was incubated at 37 °C for 12 h. Fluorescence was read at 480/520 nm. Values were expressed as relative fluorescence units (RFU). Bar: mean ± SD. *p < 0.05.
DISCUSSION
We focused on the effects of cepharanthine on the expression of CXCL10, an IFN-γ-inducible chemokine, and the transcriptional activation of which is dependent mainly on the JAK/STAT1 signaling pathway. Our findings demonstrated, for the first time to our knowledge, the anti-inflammatory effects of cepharanthine on IFN-γ-treated human salivary gland cells.
IFN-γ is one of the key cytokines involved in many inflammatory responses, including rheumatic diseases [26]. It is established that IFN-γ treatment can result in CXCL10 overexpression in some cells, which leads to the migration of CXCR3+ T cells [27, 28]. An immunohistochemical study also indicated that IFN-γ stimulated the production of CXCL10 in salivary ductal glands of SS patients [13]. Here, we investigated the precise molecular mechanisms involved in the expression of IFN-γ-induced CXCL10 by using immortalized human salivary gland cell clones in an in vitro experiment. Our results demonstrated a significant increase in the expression of CXCL10 after treatment with IFN-γ only in human salivary gland ductal (NS-SV-DC) cells, not in human salivary gland acinar (NS-SV-AC) cells.
We recently reported that our single-center open-label study showed efficacy of cepharanthine on the increase of salivary flow in pSS patients [21]. The histology of LSGs from SS patients demonstrated that the levels of infiltrated lymphocytes were decreased after the oral administration of cepharanthine (6 mg/day for 12 months). Although the precise molecular mechanism was not investigated in that study, we speculated that cepharanthine may have an effect on CXCL10, which was produced from salivary gland ductal cells by IFN-γ.
In the present study, we observed that the IFN-γ-induced CXCL10 production in NS-SV-DC cells was inhibited by cepharanthine. IFN-γ induced the rapid phosphorylation of both JAK2 and STAT1 in NS-SV-DC cells, whereas the phosphorylation of both JAK2 and STAT1 was inhibited by cepharanthine treatment. In NS-SV-DC cells, JAK1 was not detected by Western blotting. These results suggested that cepharanthine inhibits the IFN-γ-induced CXCL10 expression in NS-SV-DC cells through the suppression of the activation of the JAK2/STAT1 pathway.
It was reported that CXCL10 gene contains NF-κB and STAT1-responsible elements (ISRE) in the promoter regions [24]. In addition, IFN-γ was reported to potentiate TNF-α-induced CXCL10 production in human monocytes by increasing the activation of STAT1 and NF-κB through JAK1 and JAK2 pathways [29]. We also attempted to study the effect of cepharanthine on NF-κB signaling, but we did not observe NF-κB activation by IFN-γ in NS-SV-DC cells. In the Western blot analysis, IκB-α degradation and the translocation of p65 to the nucleus were not detected in IFN-γ-stimulated NS-SV-DC cells. These results are consistent with our previous report [25].
Taken together, these observations indicate that IFN-γ potentiates CXCL10 production in human salivary ductal cells by the activation of the JAK2/STAT1 signaling pathway, not by the NF-κB pathway, and that cepharanthine suppresses the IFN-γ-induced CXCL10 production via an inhibition of the JAK2/STAT1 signaling.
Chemokines are a superfamily of cytokines that regulate immune cell migration under both inflammatory and normal physiological conditions [30]. The interactions between chemokines and their receptors play an important role in the induction of a selective local infiltration of specific cells in various diseases [31]. It was reported that CXCL10 was expressed in the salivary ductal glands of SS patients and that CXCL10 was accumulated in CXCR3+ T cells in LSGs of SS patients [13]. In the present study, the migration of Jurkat cells, a human acute T cell leukemia cell line, was stimulated in response to the conditioned medium obtained from IFN-γ-treated NS-SV-DC cells. This finding is consistent with the histological findings of SS salivary glands, i.e., a periductal infiltration of T cells was evident in the salivary glands of SS patients. Thus, cepharanthine significantly inhibited the IFN-γ-mediated chemotaxis of Jurkat T cells. These results suggest that cepharanthine could contribute to the inhibition of T cells’ chemotaxis through the downregulation of CXCL10, which is secreted from salivary gland ductal cells following IFN-γ stimulation.
In conclusion, the results of this study demonstrated that cepharanthine suppresses IFN-γ-induced CXCL10 expression via an inhibition of the JAK2/STAT1 signaling pathway in human salivary gland ductal cells. Our findings also indicate that cepharanthine inhibits the chemotaxis of Jurkat T cells by reducing the CXCL10 production from ductal cells. These results suggest that cepharanthine could be a potential therapeutic drug for pSS patients.
References
Alspaugh, M.A., and K. Whaley. 1981. In Sjögren’s syndrome, textbook of Rheumatology, ed. W.N. Kelley, E.D. Harris, S. Ruddy, and C.B. Sledge, 971–999. Philadelphia: W.B. Saunders.
Daniels, T.E. 1984. Labial salivary gland biopsy in Sjögren's syndrome: Assessment as a diagnostic criterion in 362 suspected cases. Arthritis and Rheumatism 27: 147–156.
Hamano, H., I. Saito, N. Haneji, Y. Mitsuhashi, and Hayashi Y. MiyasakaN. 1993. Expressions of cytokine genes during development of autoimmune sialadenitis in MRL/lpr mice. European Journal of Immunology 23: 2387–2391.
Fox, R.I., H.I. Kang, D. Ando, J. Abrams, and E. Pisa. 1994. Cytokine mRNA expression in salivary gland biopsies of Sjögren's syndrome. Journal of Immunology 152: 5532–5529.
Li, H., J.A. Ice, C.J. Lessard, and K.L. Sivils. 2013. Interferons in Sjögren’s syndrome: genes, mechanisms, and effects. Frontiers in Immunology 20 (4): 290. https://doi.org/10.3389/fimmu.2013.00290.
Hall, J.C., A.N. Baer, A.A. Shah, L.A. Criswell, C.H. Shiboski, A. Rosen, and L. Casciola-Rosen. 2015. Molecular subsetting of interferon pathways in Sjögren's syndrome. Arthritis & Rhematology 67: 2437–2446.
Nocturne, G., and X. Mariette. 2013. Advances in understanding the pathogenesis of primary Sjögren’s syndrome. Nature Reviews. Rheumatology 9: 544–556.
Luster, A.D., J.C. Unkeless, and J.V. Ravetch. 1985. Gamma-interferon transcriptionally regulates an early-response gene containing homology to platelet proteins. Nature 315: 672–676.
García-López, M.A., F. Sánchez-Madrid, J.M. Rodríguez-Frade, M. Mellado, A. Acevedo, M.I. García, J.P. Albar, C. Martínez, and M. Marazuela. 2001. CXCR3 chemokine receptor distribution in normal and inflamed tissues: Expression on activated lymphocytes, endothelial cells, and dendritic cells. Laboratory Investigation 81: 409–418.
Groom, J.R., and A.D. Luster. 2011. CXCR3 ligands: redundant, collaborative and antagonistic functions. Immunology and Cell Biology 89: 207–215.
Hartl, D., S. Krauss-Etschmann, B. Koller, P.L. Hordijk, T.W. Kuijpers, F. Hoffmann, A. Hector, E. Eber, V. Marcos, I. Bittmann, O. Eickelberg, M. Griese, and D. Roos. 2008. Infiltrated neutrophils acquire novel chemokine receptor expression and chemokine responsiveness in chronic inflammatory lung diseases. Journal of Immunology 181: 8053–8067.
Tomita, K., B.L. Freeman, S.F. Bronk, N.K. LeBrasseur, T.A. White, P. Hirsova, and S.H. Ibrahim. 2016. CXCL10-mediates macrophage, but not other innate immune cells-associated inflammation in murine nonalcoholic steatohepatitis. Scientific Reports 6: 28786.
Ogawa, N., L. Ping, L. Zhenjun, Y. Takada, and S. Sugai. 2002. Involvement of the interferon-γ-induced T cell-attracting chemokines, interferon-γ-inducible 10-kD protein (CXCL10) and monokine induced by interferon-γ (CXCL9), in the salivary gland lesions of patients with Sjögren's syndrome. Arthritis and Rheumatism 46: 2730–2741.
Ogawa, N., T. Kawanami, K. Shimoyama, L. Ping, and S. Sugai. 2004. Expression of interferon-inducible T cell alpha chemoattractant (CXCL11) in the salivary glands of patients with Sjögren’s syndrome. Clinical Immunology 112: 235–238.
Hasegawa, H., A. Inoue, M. Kohno, M. Muraoka, T. Miyazaki, M. Terada, T. Nakayama, O. Yoshie, M. Nose, and M. Yasukawa. 2006. Antagonist of interferon-inducible protein 10/CXCL10 ameliorates the progression of autoimmune sialadenitis in MRL/lpr mice. Arthritis and Rheumatism 54: 1174–1183.
Gutterman, J.U., S. Fine, J. Quesada, S.J. Horning, J.F. Levine, R. Alexanian, L. Bernhardt, M. Kramer, H. Spiegel, W. Colburn, P. Trown, T. Merigan, and Z. Dziewanowski. 1982. Recombinant leukocyte A interferon: Pharmacokinetics, single-dose tolerance, and biologic effects in cancer patients. Annals of Internal Medicine 96: 549–556.
Ono, M. 1987. Antitumor effect of cepharanthine: activation of regional lymph node lymphocytes by intratumoral administration. Clinical Immunology 19: 1061–1064.
Morioka, S., M. Ono, N. Tanaka, and K. Orita. 1985. Synergistic activation of rat alveolar macrophages by cepharanthine and OK-432. Gan to Kagaku Ryoho 12: 1470–1475.
Azuma, M., K. Aota, T. Tamatani, K. Motegi, T. Yamashita, Y. Ashida, Y. Hayashi, and M. Sato. 2002. Suppression of tumor necrosis factor α-induced matrix metalloproteinase 9 production in human salivary gland acinar cells by cepharanthine occurs via down-regulation of nuclear factor κB: A possible therapeutic agent for preventing the destruction of the acinar structure in the salivary glands of Sjögren's syndrome patients. Arthritis and Rheumatism 46: 1585–1594.
Azuma, M., Y. Ashida, T. Tamatani, K. Motegi, N. Takamaru, N. Ishimaru, Y. Hayashi, and M. Sato. 2006. Cepharanthin, a biscoclaurine alkaloid, prevents destruction of acinar tissues in murine Sjögren’s syndrome. The Journal of Rheumatology 33: 912–920.
Yamanoi, T., K. Aota, Y. Momota, and M. Azuma. 2017. Treatment with the biscoclaurine alkaloid cepharanthin for improved salivary secretion in primary Sjögren's syndrome patients. Journal of Oral Health Biosciences 29: 39–48.
Azuma, M., T. Tamatani, Y. Kasai, and M. Sato. 1993. Immortalization of normal human salivary gland cells with duct-, myoepithelial-, acinar-, or squamous phenotype by transfection of SV40 ori-mutant DNA. Laboratory Investigation 69: 24–42.
Hogan, J.C., and J.M. Stephens. 2001. The identification and characterization of a STAT 1 binding site in the PPARγ2 promoter. Biochemical and Biophysical Research Communications 287: 484–492.
Majumder, S., L.Z. Zhou, P. Chaturvedi, G. Babcock, S. Aras, and R.M. Ransohoff. 1998. p48/STAT-1alpha-containing complexes play a predominant role in induction of IFN-gamma-inducible protein, 10 kDa (IP-10) by IFN-gamma alone or in synergy with TNF-alpha. Journal of Immunology 161: 4736–4744.
Azuma, M., K. Motegi, K. Aota, Y. Hayashi, and M. Sato. 1997. Role of cytokines in the destruction of acinar structure in Sjögren’s syndrome salivary glands. Laboratory Investigation 77: 269–280.
Hall, J.C., L. Casciola-Rosen, A.E. Berger, E.K. Kapsogeorgou, C. Cheadle, A.G. Tzioufas, A.N. Baer, and A. Rosen. 2012. Precise probes of type II interferon activity define the origin of interferon signatures in target tissues in rheumatic diseases. Proceedings of the National Academy of Sciences of the United States of America 109: 17609–17614.
Lacotte, S., S. Brun, S. Muller, and H. Dumortier. 2009. CXCR3, inflammation, and autoimmune diseases. Annals of the New York Academy of Sciences 1173: 310–317.
Gangur, V., F.E. Simons, and K.T. Hayglass. 1998. Human IP-10 selectively promotes dominance of polyclonally activated and environmental antigen-driven IFN-gamma over IL-4 responses. The FASEB Journal 12: 705–713.
Qi, X.F., D.H. Kim, Y.S. Yoon, D. Jin, X.Z. Huang, J.H. Li, Y.K. Deung, and K.J. Lee. 2009. Essential involvement of cross-talk between IFN-γ and TNF-α in CXCL10 production in human THP-1 monocytes. Journal of Cellular Physiology 220: 690–697.
Kelner, G.S., J. Kennedy, K.B. Bacon, S. Kleyensteuber, D.A. Largaespada, N.A. Jenkins, N.G. Copeland, J.F. Bazan, K.W. Moore, T.J. Schall, et al. 1994. Lymphotactin: A cytokine that represents a new class of chemokine. Science 266: 1395–1399.
Murdoch, C., and A. Finn. 2000. Chemokine receptors and their role in inflammation and infectious diseases. Blood 95: 3032–3043.
Financial Support
This work was supported by Grant-in-Aid for Scientific Research from the Japan Ministry of Education, Culture, Sports, Science and Technology (No. 15K11254).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Aota, K., Yamanoi, T., Kani, K. et al. Cepharanthine Inhibits IFN-γ-Induced CXCL10 by Suppressing the JAK2/STAT1 Signal Pathway in Human Salivary Gland Ductal Cells. Inflammation 41, 50–58 (2018). https://doi.org/10.1007/s10753-017-0662-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0662-x