Abstract
With growing demand for food production in Africa, protecting wetlands and combining increased agricultural production with conservation of the ecological integrity of wetlands is urgent. The role of aboveground biomass of papyrus (Cyperus papyrus) in the storage and retention of nitrogen (N) and phosphorus (P) was studied in two wetland sites in East Africa under seasonally and permanently flooded conditions. Nyando wetland (Kenya) was under anthropogenic disturbance from agriculture and vegetation harvesting, whereas Mara wetland (Tanzania) was less disturbed. Maximum papyrus culm growth was described well by a logistic model (regressions for culm length with R2 from 0.70 to 0.99), with culms growing faster but not taller in Nyando than in Mara. Maximum culm length was greater in permanently than in seasonally flooded zones. Total aboveground biomass was higher in Mara than in Nyando. The amounts of N and P stored were higher in Mara than in Nyando. In disturbed sites, papyrus plants show characteristics of r-selected species leading to faster growth but lower biomass and nutrient storage. These findings help to optimize management of nutrient retention in natural and constructed wetlands.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
By 2050, it is expected that the African population will grow to 2.5 billion (UN, 2015). This will increase the demand for food, and the need to increase agricultural production. As the productivity of African agricultural systems is low compared with other world regions, increased production is often achieved by areal extension (van Asselen et al., 2013; Schoumans et al., 2015; OECD/FAO, 2016) at the expense of natural ecosystems like forests, natural grasslands and wetlands (UNCCD, 2017; Ramsar Convention on Wetlands, 2018; FAO, 2019). This has an impact on biodiversity, and soil and water quality (Butchart et al., 2010; UNCCD, 2017). It also poses risks for human well-being, as food production and a good living environment depend on the regulating and provisioning services of healthy, well-functioning ecosystems (MEA, 2005; IPBES, 2019). In Africa, wetlands contribute to water quality regulation and flood protection (Silvius et al., 2000; Schuyt, 2005; Verhoeven & Setter, 2010), and high wetland carbon storage may help with climate change mitigation (IPCC, 2014). Moreover, wetlands can increase the resilience of poor rural communities against climate change effects like floods and droughts and have relatively high economic value and biodiversity (Russi et al., 2013; Darwall et al., 2018; Tickner et al., 2020). There is thus a trade-off between agricultural production and the loss of ecosystem services. As agricultural development in Africa progresses, protecting wetlands and combining increased agricultural production with conservation of the ecological integrity and function of wetlands is an urgent need (Jayne et al., 2019).
Utilisation of wetlands to meet human needs has implications on ecological functions. A key ecological function affected when wetlands are converted or degraded is nutrient and sediment retention (Johnston, 1991). Wetlands often serve as buffer zones in the landscape, and their degradation, in combination with fertilizer application and soil erosion in the catchment, can increase nutrient and sediment runoff into rivers and lakes (Hecky et al., 2010). In the Lake Victoria region in East Africa, harvesting of papyrus (Cyperus papyrus L.) vegetation and seasonal agriculture in papyrus wetlands are widespread (Kipkemboi & van Dam, 2018). This has led to increased sediment and nutrient loads, contributing to ecological degradation of Lake Victoria with algal blooms and excessive growth of the water hyacinth [Eichhornia crassipes (Mart.) Solms] with serious economic consequences (Kiwango & Wolanski, 2008; Hecky et al., 2010; Olokotum et al., 2020).
Nutrient and sediment retention by papyrus wetlands involves a number of biological and physical processes. Nitrogen (N) and phosphorus (P) removal from surface water include temporary storage in above- and belowground biomass and adsorption to soil particles, and longer term storage in deeper peat layers (Gaudet, 1977; Kansiime et al., 2007; Hes & van Dam, 2019). Permanent removal of N by the wetland ecosystem occurs only through denitrification (Galloway et al., 2004; Pina-Ocha & Álvarez-Cobelas, 2006). Both natural and anthropogenic disturbance can interfere with these processes, and change the nutrient retention characteristics of the wetlands. For example, natural disturbances include flooding and drying cycles, and grazing by herbivores. Anthropogenic disturbances include livelihood activities like seasonal or permanent conversion to agriculture and vegetation harvesting. To illustrate this, drainage will lower the moisture content of the soil, which has consequences for adsorption properties and denitrification. It will also affect the growth of the papyrus plants, which changes the flow and storage of nutrients in above- and belowground biomass. Different patterns of harvesting and removal of aboveground biomass have an impact on nutrient retention (Terer et al., 2012; Hes & van Dam, 2019). Some areas are cleared of papyrus vegetation entirely for seasonal agriculture in the dry season, after which the vegetation grows back when the wetland floods again in the rainy season (van Dam et al., 2013). In other areas harvesting is more selective because different uses (e.g. fibre for making chairs, roof thatch, mat making and other household crafts) require different grades of papyrus culms. When the belowground biomass of the plants is left intact, the fast regrowth of papyrus can restore the aboveground biomass in about 6–9 months (Terer et al., 2012), demonstrating a high resilience of this system. As pressure from harvesting and agriculture increases, it is important to understand the recovery of the aboveground biomass and associated N and P accumulation under different utilization regimes.
Research quantifying the role of aboveground biomass in nutrient balances is limited to modelling (van Dam et al., 2007; Hes & van Dam, 2019) and measurements on constructed wetlands (Kengne et al., 2008). To understand how much N and P are stored aboveground, the biomass density (in g dry weight [DW] m−2) and the N and P content of biomass (in % DW) need to be quantified. Reported productivity of aboveground papyrus biomass is 5–37 g DW m−2 day−1, aboveground biomass 1384–6045 g DW m−2, and N and P content 0.65–1.75% DW and 0.024–0.13% DW, respectively (reviewed in Hes & van Dam, 2019). Immature papyrus shoots have a higher N and P content than mature plants (Gaudet, 1977; Rongoei & Kariuki, 2019), and this influences the amounts of N and P present in aboveground biomass. How these characteristics change over time depends on how the growth rates of shoots respond to the prevailing environmental conditions (e.g., flooding levels, water quality, harvesting, grazing). Papyrus culms can grow very fast (up to 4–5 m within about 6 months) and typically exhibit six vegetative growth stages (Fig. 1). While the impact of environmental conditions on biomass and density of papyrus has been studied (Gaudet, 1975; Muraza et al., 2013; Opio et al., 2017; Geremew et al., 2018), there is no data on the effect of disturbances on N and P content of culms of the different growth stages.
Papyrus with development stages: (I) closed umbel; (II) opening umbel; (III) fully opened umbel; (IV) flowering; (V) mature; (VI) senescing and points for measurements: (A) base of umbel; (B) top of scale leaves; (C) base of culm. Figure adapted by permission from the Licensor: Springer Nature, Economic Botany (Muthuri & Kinyamario, 1989)
Growth of papyrus biomass (culm and umbel) was described and simulated by a logistic model, with estimated values for maximum biomass and instantaneous growth rates based on literature values (van Dam et al., 2007; Hes et al., 2014; Opio et al. 2014; Hes & van Dam, 2019). The amounts of N and P stored in aboveground biomass can be estimated by combining the logistic growth model with the N and P content at different growth stages. The overall aim of this study was to quantify the relationship between aboveground papyrus biomass and retention of N and P to better manage the balance between use and conservation of natural systems and improve performance of constructed wetlands for wastewater treatment. The three specific objectives were to: (1) describe growth of aboveground biomass with a logistic model and estimate the model parameters in sites with different disturbance conditions (flooding, intensity of human use); (2) compare standing biomass, growth of aboveground biomass and uptake of N and P under different disturbance conditions; and (3) assess the role of living aboveground biomass in retention of N and P under different conditions and identify implications for sustainable use. Our hypothesis is that papyrus culm growth follows a logistic model, N and P content decreases while the culms develop from young to mature, and that culm development and density is influenced by natural conditions and anthropogenic pressures. Based on two growth strategy theories (r/K selection and CSR triangle) we expect disturbed and drier areas will have faster growth, lower biomass and less fully matured plants compared with undisturbed wet areas (Pianka, 1970; Grime, 1977). As a result, undisturbed areas would have higher biomass, but lower N and P content. Therefore, there may be more N and P stored in mildly disturbed areas with lower biomass and higher N and P content. To explore this, we conducted a field experiment in natural papyrus wetlands in Kenya and Tanzania.
Materials and methods
Description of the field sites
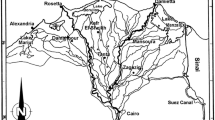
The field experiments were carried out during the period November 2010–January 2011 in Nyando wetland in Kenya and Mara wetland in Tanzania (Fig. 2). Nyando wetland (0°11′-0°19′ S to 34°47′-34°57′ E) borders Lake Victoria at Winam Gulf at an altitude of 1140 m and is adjacent to Kenya’s third biggest city, Kisumu within the district Kisumu East, with a population of 475,000 at the time of this study (KNBS, 2010). The total wetland area was estimated at 30–50 km2 (Khisa et al., 2013) and the intact part is largely dominated by papyrus. Mara wetland (1° 27′-1° 37′ S to 33° 55′-34° 28′ E) borders Lake Victoria at the mouth of the Mara River at an altitude of 1134 m and is a papyrus-dominated wetland of more than 350 km2 (Bregoli et al., 2019). The population in and around the wetland was 56,000, divided over 20 villages (The United Republic of Tanzania, 2013). Just upstream of the wetland is a mining area (North Mara Gold Mine) and an associated town, Nyamongo. Both Nyando and Mara are similar papyrus dominated floodplain wetlands and surrounded by predominantly agricultural land used for crops and cattle grazing. While the overall anthropogenic pressure in Nyando leads to a reduction of wetland area (Khisa et al., 2013; Rongoei et al., 2013), in the Mara the wetland is expanding (Bregoli et al., 2019). Comparing the state of the two field sites we observed that papyrus growth in general is better in Mara with less anthropogenic disturbance (harvesting and agriculture) and larger undisturbed permanently flooded areas.
Locations of field sites and transects for sampling with: A Africa; B Lake Victoria; C Nyando Wetland; D Mara wetland; E Ogenya sampling site; F Singida sampling site; G Mara sampling site. In E, F and G: white dashed line = transect; solid line = transition between zones; SF-zone seasonally flooded zone; PF-zone permanently flooded zone (satellite images
In both sites, two experiments were carried out. The first experiment monitored the growth of aboveground biomass (culms). In the second experiment, biomass, culm density and nitrogen and phosphorus content of the plants were measured. Both experiments were done in permanently and in seasonally flooded zones dominated by papyrus. The permanently flooded zone was defined as saturated soils with standing water all year around and the seasonally flooded zone was not fully saturated all year round. Because of the different conditions in the two locations, there were slight differences in experimental set-up in both sites that are described below.
Monitoring of aboveground biomass growth
In Nyando wetland, two transects were cut through the papyrus dominated vegetation stands: one in Singida at Bwaja River bordering the agricultural zone, and another in Ogenya at the shore of Winam Gulf (Fig. 2C, E, F). In Mara wetland, three transects were cut perpendicular to the main channel of Mara River (Fig. 2D, G). Because of time and resource limitations, we could only monitor the first and the third transect (Mara 1 and Mara 3) for this experiment. All transects were made from the seasonally flooded zone to the permanently flooded zone, perpendicular to the open water (Nyando) or the river (Mara). In both zones of each transect, a 10 m path was cut perpendicular to the transect (Fig. 2).
The development stages of the papyrus plants were assessed using the description by Muthuri & Kinyamario (1989). Along the transect paths, four (Nyando) or five (Mara) culms of each development stage were selected randomly and marked with a waterproof marker (Fig. 2). The development of these individual culms was followed for 8 weeks. At the start, and then every two weeks, the following measurements were taken: culm length (cl, in cm); culm length from the top of the scale leaf (clsl, in cm); culm width at the top of the scale leaf (width, in cm); culm girth at the top of the scale leaf (girth, in cm). Culm lengths were measured with a wooden stick and tape measure, culm width with a vernier calliper, and culm girth with a length of rope and a tape measure.
To describe general conditions, the following water quality measurements were taken at the start of the experiment, after 4 weeks and after 8 weeks: electrical conductivity (EC, in µS/cm), temperature (in °C), pH, dissolved oxygen concentration (DO, in mg/L) (all with a model 3210 SET multimeter; Wissenschaftlich-Technische Werkstätten, Hamburg, Germany), ammonium-nitrogen (NH4-N in mg/L), nitrate-nitrogen (NO3-N in mg/L), soluble reactive phosphorus (PO4-P in µg/L), total nitrogen (TN in mg/L) and total phosphorus (TP in mg/L). All N and P analysis followed standard methods (APHA, 1992). Nyando analysis were carried out in Njoro, Kenya at Egerton University. In the Mara site, only pH, DO, NH4-N, NO3-N and TN were measured, due to limitations in the Musoma, Tanzania laboratory.
Biomass and culm density
In Nyando, two quadrats of 1 × 5 m were selected in each zone (eight in total) along the same transects (Fig. 2). In Mara, three 1 × 5 m quadrats were selected in each zone of transects 1 and 3, and two quadrats in both zones of transect 2 (16 in total). In all quadrats, the entire aboveground biomass was harvested by cutting the culms just above the rhizome. The culms were counted and classified according to their development stage. Culms were initially sun-dried, and then oven-dried at 80 °C to determine their dry weight.
Nitrogen, phosphorus and carbon content in plant organs
In each of the Nyando 1 × 5 m quadrats, two 1 × 1 m quadrats (Fig. 2) were harvested for both above- and belowground biomass. All culms were cut just above the rhizome and divided into development classes (Fig. 1), counted and measured (lengths, width and girth as described above). After sun and oven drying, the combined dry weight of all culms of the same development stage was measured. Then, the plant material was cut in pieces of approximately 2 cm, mixed and one sample for each development stage for each zone was taken to determine nitrogen (N), phosphorus (P) and carbon (C) content. The rhizomes and roots were sun- and oven-dried to constant weight and dry weight was determined. After cutting into 2 cm pieces and mixing, a sub-sample was taken to determine N, P and C content. For Mara wetland the same procedure was followed with two 1 × 1 m quadrats in the seasonally and permanently flooded zones of transects 2 and 3 (Fig. 2).
All samples were analysed at the laboratory of the Chemical Biological Soil Laboratory of Wageningen University, the Netherlands. After grinding the samples to 1 mm, N and P (mg/g DW) were determined by digestion of the samples with H2SO4/Se/salicyclic acid and H2O2 and segmented flow analysis (SFA).
Determining logistic growth models
The growth pattern of the papyrus can be described using the logistic growth equation:
With L = culm length (cm), t = time (day), r = instantaneous growth rate (day−1) and K = the maximum culm length (cm). Dividing the equation (Eq. 1) by L gives:
a linear relationship with the relative growth in length (dL/dt /L) as dependent variable, length (L) as the independent variable, intercept r and slope r/K. For each 2-week growth period of the culms, individual relative culm growth (L2 − L1)/days and mean culm length ((L1 + L2)/2) were calculated. For the culms showing maximum growth, model II linear regression lines were estimated between mean culm length (X) and relative growth rate (Y) (Fig. 3), using the lmodel2 package in R. The parameters r and K of the logistic curve were calculated for each transect from the intercept and slope (Rongoei & Outa, 2016). The same method for determining the logistic growth models was used for width and girth.
Relative growth rate (day−1) in relation to culm length of papyrus. Data points represent 2-week growth intervals for individual culms among three sites and two zones per site with OgP = Ogenya permanent flooded zone (n = 77); OgS = Ogenya seasonally flooded zone (n = 54); SiP = Singida permanently flooded zone (n = 70); SiS = Singida seasonally flooded zone (n = 68); MaP = Mara permanently flooded zone (n = 187) and MaS = Mara seasonally flooded zone (n = 119). Solid circles = maximum relative growth rate and open circles = relative growth rate. Model II regression lines were calculated for the points with maximum growth. For details see text
Data analysis
Confidence intervals (95%) for the parameters of the logistic growth curves (r, K) were estimated based on the standard error estimates for the regression coefficients (Sheather, 2009). Differences in mean culm length, biomass, culm density, and N and P content among growth stages were compared using one-way Type III analysis of variance (ANOVA). Within growth stages, differences in the same variables among transect locations (Ogenya, Singida, Mara) and inundation zones (seasonal or permanent) were compared using two-way Type III ANOVA. All variables were checked for normal distribution and log10-transformed when necessary (biomass, culm density). All statistical tests were performed using R version 4.0.2 (R Core Team, 2020).
Results
All regression models were significant, but more significant for both culm length and culm length from top of scale leaf than for width and girth. R2 values were highest for culm length between 0.89 and 0.99 and one of 0.70 (Table 1 and Fig. 3), especially compared with culm length from top of scale leaf. The regression models based on culm length were therefore used to estimate the carrying capacity (K, maximum length) and the instantaneous growth rate (r) (Fig. 3).
The results of the regression models based on culm length showed the highest instantaneous growth rate (0.097 day−1) in Ogenya seasonal flooded zone. Growth rates were all higher in Nyando (between 0.081 and 0.097 day−1) than in Mara (0.071 day−1 in Mara permanently flooded zone and 0.072 day−1 in Mara seasonally flooded zone). In both Nyando sites, the growth rates were higher in the seasonally flooded zones (0.091 day−1 in Ogenya seasonally flooded zone and 0.081 day−1 in Ogenya permanently flooded zone; 0.094 day−1 in Singida seasonally flooded zone and 0.082 day−1 in Singida permanently flooded zone) and in Mara there was no difference between the zones (Fig. 4). The maximum culm length (K) was higher in the permanently flooded zones for each site, most notably in Ogenya (413 and 287 cm respectively). In Mara (484 and 464 cm respectively), the maximum culm length was higher than in Nyando (between 287 and 413 cm). Based on the confidence intervals, the growth difference between permanent and seasonal flooding was only significant (P < 0.05) in Ogenya (Fig. 4).
Estimated growth parameters of the logistic curve for papyrus in six transects. A Instantaneous growth rate r; B maximum length K; and c culm length over time among three sites and two zones per site with OgP = Ogenya permanently flooded zone, OgS = Ogenya seasonally flooded zone, SiP = Singida permanently flooded zone, SiS = Singida seasonally flooded zone, MaP = Mara permanently flooded zone and MaS = Mara seasonally flooded zone. Error bars in A and B indicate 95% confidence intervals
Culm length increased significantly from growth stage I to growth stage IV (Fig. 5). In both Singida and Ogenya, the average culm length was higher in the permanently flooded zone than in the seasonally flooded zone. In the most mature phase (V), the culms in Mara were significantly longer than in both Nyando sites (Fig. 5a). In all locations, there was no significant difference between the amount of biomass classified as stage I, II, III or IV per m2. However, the highest amount of biomass was in growth stage V, especially for Mara and within Mara in the permanently flooded zone (Fig. 5b). Density for most stages was below 5 culms per m2, with no difference between seasonally—or permanently flooded zones. The stage V (mature culms) counts were higher, especially in Mara (≈ 15 culms m−2, while stage IV (flowering) was low or absent in Mara and always present in both Nyando sites (Fig. 5c). The content of N and P in the culms were highest during stage I and II and decreased with every consecutive stage, a pattern seen in all the sites or zones (Fig. 6a, b).
Field measurements of A culm length; B biomass and C culm density per growth stage among three sites and two zones per site with OgP = Ogenya permanently flooded zone, OgS = Ogenya seasonally flooded zone, SiP = Singida permanently flooded zone, SiS = Singida seasonally flooded zone, MaP = Mara permanently flooded zone and MaS = Mara seasonally flooded zone. Note: in B the two data points in the gray rectangle (MaP & MaS) were plotted on a different scale, see second y-axis. Error bars indicate standard error. Growth stages sharing the same capital letter were not significantly different (1-way Type III ANOVA, P < 0.05). Within growth stages, sites sharing the same lower case letter were not significantly different (2-way Type III ANOVA, P < 0.05)

taken from vegetation quadrats. Error bars indicate standard error. Growth stages sharing the same capital letter were not significantly different (1-way Type III ANOVA, P < 0.05). Within growth stages, sites sharing the same lower case letter were not significantly different (2-way Type III ANOVA, P < 0.05)
Nitrogen (A) and phosphorus (B) content of culm per growth stage among three sites and two zones per site with OgP = Ogenya permanently flooded zone, OgS = Ogenya seasonally flooded zone, SiP = Singida permanently flooded zone, SiS = Singida seasonally flooded zone, MaP = Mara permanently flooded zone and MaS = Mara seasonally flooded zone. Data points are means of composite culm samples (2 for Ogenya and Singida, 4 for Mara)
The culm density, amount of biomass and average length of all culms of growth stage I to V were highest in the permanently flooded zone of Mara wetland, followed by Mara seasonally flooded zone. In both Singida and Ogenya the culm density was higher in the seasonally flooded zone than the permanently flooded zone, while the average length was higher in the permanently flooded zone than the seasonally flooded zone (Fig. 7).
Comparison of average culm density, culm biomass and culm length among three sites and two zones per site with OgP = Ogenya permanently flooded zone, OgS = Ogenya seasonally flooded zone, SiP = Singida permanently flooded zone, SiS = Singida seasonally flooded zone, MaP = Mara permanently flooded zone and MaS = Mara seasonally flooded zone. Error bars indicate standard error, except for MaP and MaS in A as length for all culm classes together in Mara was not measured, but calculated based on culm density per class and average length per class
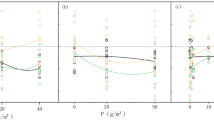
As a result of the higher culm density and biomass, the amount of N and P per m2 was also higher in Mara compared with the Nyando sites (Table 2). In Mara around two thirds of the N and P was stored in mature culms (growth stage V), while in Nyando this was more evenly spread over the growth stages II-V (Table 2).
The belowground (root and rhizome) and detritus biomass were highest in the permanently flooded zone in Mara and then Mara’s seasonally flooded zone. Values for Ogenya and Singida were considerably lower in both zones and similar with each other (Table 3). Content of N and P in the belowground biomass were similar to the values in the aboveground biomass (Fig. 6) and Table 3), with no significant differences among the sites or zones. Nitrogen values in the detritus were similar to content in living biomass, while the values for phosphorus were lower (Table 3).
Oxygen concentrations in Ogenya and Singida seasonally flooded zones were around 2 mg/L, which was higher than in the permanently flooded zones and in both zones in Mara (all below 1 mg/L). The pH was around 6 in all sites and zones, except for Singida permanently flooded zone (7.5 ± 2.1). The water temperature was around 21 °C in both Mara sites, around 22 °C in the Nyando permanently flooded sites and around 24.5 °C in the Nyando seasonally flooded sites (Table 4). Ammonium concentration was highest in Singida (0.31 ± 0.09 mg/L) and nitrate was higher in Mara than in both Nyando sites. Total N was around 1 mg/L in all sites.
Discussion
Papyrus culms grew faster, but not as tall, in the Nyando sites (Singida and Ogenya), compared with the Mara site. The maximum culm length (K) in all sites was greater in the permanently flooded zones than in the seasonally flooded zones. The growth rate (r) in the seasonally flooded zones in the Nyando sites were higher compared with the permanently flooded zones in Nyando, and with both zones in Mara. Biomass was higher in Mara compared with Nyando, but culm density was not significantly different. The regression models used to estimate r and K were based on the relation between maximum growth and length, and describe a growth pattern for each site. Culms growing at a slower rate were not used for estimating the maximum growth, as these are likely limited by nutrient availability or light availability due to self-shading (Jones, 1988; Saunders et al., 2014). The models were highly significant, with high coefficients of determination (R2 > 0.89), confirming that the growth pattern of papyrus culms can be adequately described using a logistic growth model. The only exception was the seasonally flooded zone in Singida (R2 = 0.70), which had fewer measurements of longer culms which may have reduced the coefficient of determination. The logistic growth pattern is well known from other studies on papyrus (Kansiime et al., 2003; Opio et al., 2014), but also for other emergent macrophytes such as Phragmites australis (Cav.) Trin. ex Steud. (Zemlin et al., 2000; Clevering et al., 2001) and Typha domingensis Pers. (Lorenzen et al., 2001; Lagerwall et al., 2012).
Culm growth was measured with non-destructive methods as proxies for biomass increase. A standard method, like the diameter at breast height (DBH) for trees, is not available for emergent macrophytes like papyrus. Therefore different methods were tried. Culm length from the scale leaf, width at the scale leaf, and girth at scale leaf were measured, however culm length measured from the base of the culm at the rhizome (Fig. 1) gave the best results. Possibly the growth rate of the scale leaf is different from that of the whole culm as competitive plants are known to change allometry under stress (Grime, 1977), which could influence the consistency of the three other methods. Besides, for girth and width small measuring mistakes result in larger errors, which could be reduced by the use of high quality digital Vernier calipers. Another improvement in growth parameter (r and K) estimates could be realized by more frequent (weekly, instead of bi-weekly) measurements (Deegan et al., 2007), and by including more culms of growth stages I and II in the experiment to have more data points at lower culm lengths. There may be seasonal differences in growth rates or resource allocation (Pianka, 1970; Grime, 1977). The experiments ran in between the so called short and long rainy seasons, although because of variability in the rainfall patterns in Nyando and Mara the seasons are not always very distinct (Gabrielsson et al., 2013). According to Opio et al. (2017), productivity of papyrus was not affected much by seasonal variations.
The results of the growth analysis indicate that zones with different disturbance levels show differences in papyrus growth strategy. With higher pressure from livelihood activities such as harvesting, grazing and cropping (Nyando) and with seasonal flooding, culms grow faster but shorter. Where disturbance is less (Mara) and with permanent flooding, culms grow slower but taller. This is consistent with theories that characterize species based on their growth strategies, and growth and recovery rates (Soissons et al., 2019). One of those theories is r/K selection, where ‘K’ refers to carrying capacity and ‘r’ to the intrinsic rate of natural increase. Rapid development, high growth rates, early reproduction, and small body size are traits of r-selected species. K-selected species are characterised by slower development, great competitiveness, delayed reproduction, and larger size (Pianka, 1970). A second theory is the CSR triangle, where species are classified as Competitive (C), Stress Tolerant (S), Ruderal (R), or as a combination. S is the equivalent of K-selected and R of r-selected, and C species combine traits of the others (Grime, 1974, 1977). The vegetation response in Nyando (growing faster, but less tall) is characteristic of C-R species which under favourable conditions grow fast (clonal growth), dominate large areas and make large and rapid changes in their allometry and allocation in response to stress (Weiner, 2004) for example by increased seed production. When harvested, burned or grazed (in the seasonally flooded zones, and more so in Nyando than in Mara) they re-grow quickly, but shorter, following the classic strategy of an r-selected species. Under light competition and when close to carrying capacity (the permanently flooded zones, especially in Mara) the papyrus showed more traits of K-selected or R species: slow, tall, and strong (Pianka, 1970).
When looking at the culm densities per growth stage, a similar growth strategy emerges. Under higher pressure from livelihood activities (Nyando), there is more seed production (stage IV), characteristic of R type plants. However, clonal reproduction was observed in both Nyando and Mara, and allowed for fast spreading and dominance, characteristics of a competitive (C) strategy. The relatively high density of mature culms (stage V) in Mara and the lower number of flowering plants (stage IV) indicate a combined C-R strategy. This was confirmed by findings of experiments under longer periods of stress, e.g. droughts and high sediment loads, where sexual reproduction and seedling recruitment become a more prevalent growth strategy for papyrus plants (Terer et al., 2014; Geremew et al., 2018).
Mara wetland was a more productive environment than Nyando, as evidenced by the higher biomass per m2. The total biomass in Mara (4.4–7.5 kg m−2) was in the upper range, and in Nyando (1.4–2.5 kg m−2) in the lower range of values for papyrus (1.4–8.7 kg m−2; summarized in Hes and van Dam, 2019). This confirms that conditions were less affected by livelihood activities in Mara and that papyrus follows a C strategy under good conditions, outcompeting other species with fast clonal growth (Geremew et al., 2018). Nyando, however had a higher aboveground biomass production rate compared with Mara. The biomass was far below the carrying capacity, with growth, therefore, closer to the exponential part of the logistic growth curve than Mara. The observed differences in total aerial biomass between the more affected Nyando site and the more pristine conditions in Mara were in line with the findings in Terer et al. (2012) indicating that frequent harvesting impacts re-growth of papyrus and leads to a lower overall biomass.
Nitrogen and phosphorus content of the shoots at all sites was highest in stages I and II and decreased with every growth stage (III to V), with no significant differences among the sites or zones. The pattern of higher N and P content in earlier development of the shoots confirms earlier findings (Gaudet, 1977; Muturi & Jones, 1997), and can be explained by active translocation from the rhizome to support fast culm development (Rejmánaková, 2005; Asaeda et al., 2008). The amount of N found in aboveground biomass was at the lower end, and P at the higher end of the range reported in the literature (0.65–1.75%DW for N; 0.024–0.13%DW for P; summarized in Hes & van Dam, 2019). Lake Victoria has received increasing P loads since the 1960s (Hecky et al., 2010), which may have increased P uptake and storage of papyrus in Lake Victoria's riparian zones. The N and P content in the belowground biomass was comparable with the values in the aboveground biomass, with no significant differences among the sites or zones. In detritus, the P content was lower than in the living biomass, but N content was similar to that in living biomass. P release from dead culms was reported to be faster than N release, both in papyrus (Gaudet, 1977) and in other macrophytes (Chimney & Pietro, 2006). This is likely from faster leaching of P, especially in the tropics under humid conditions, while N decomposition depends more on microbial processes (Manzoni et al., 2010).
As a result of the higher aboveground biomass, the N and P amounts per m2 were higher in Mara than in Nyando. Most of the N and P in Mara was located in the mature (stage V) culms. In the Nyando sites there was a more equal distribution over the different growth stages. The amount of N stored in both Nyando sites was low (7–12 g N m−2). Even the values found in Mara (22–36 g N m−2) were lower than reported from other studies (48–104 g N m−2; Gaudet, 1977; Boar et al., 1999; Boar, 2006). For Nyando, this can be explained by the low biomass. For Mara, the exclusion of senescing culms (Stage VI) from our analysis may explain the lower N values. The lower amount of P in the Nyando sites (1.8–2.5 g P m−2) compared with reported values (5.4–5.7 g P m−2; Gaudet, 1977; Boar, 2006) are likely because of relatively low biomass. The relatively high values in Mara P (4.9–8.3 g P m−2) may be a result of increased P loading to the lake, as mentioned above. The study by Boar (2006) was carried out at Lake Naivasha, and the experiments described by Gaudet (1977) were carried out in the early 1970s, when the P loading to Lake Victoria was lower.
Natural and anthropogenic disturbance influenced N and P retention by aboveground biomass in our field sites. Based on different conditions, papyrus adopted different growth strategies and this influenced nutrient retention. Retention was higher in permanently inundated zones than seasonally inundated zones, and higher in sites with low disturbance (Mara) than in sites with livelihood activities (Nyando). Under ideal conditions, a K-selected or Competitive growth strategy leads to large mature culms, leaving less space for young culms and storing nutrients in the aboveground biomass. Under stressful conditions there is a shift to more r-selected growth, with young shoots growing fast, but attaining a lower height (Pianka, 1970), retranslocation of N and P to the rhizome for storage, and morphological flexibility with smaller shoots and denser rhizomes (Geremew et al., 2018). Under severe or long term stress, seed production can increase, ensuring survival after e.g. a long absence of water (Terer et al., 2014). The flexibility to switch between growth strategies makes papyrus wetlands resilient to environmental and anthropogenic pressure, and amenable to management measures for conservation and restoration (Morrison et al., 2012).
The results provide input for a wider application of modelling N and P retention (Hes & van Dam, 2019). Incorporating the effects of disturbance and the variation of N and P content of the culms can improve the estimation of regulating services like nutrient retention and water purification, and improve our understanding of trade-offs related to provisioning services such as food production (agriculture and fisheries). Quantitative models can contribute to the land-sharing/land-sparing debate (Kremen, 2015). Currently, agricultural development in eastern Africa follows a 'sharing' pattern, in which papyrus wetlands are used for extensive dry-season agriculture, grazing and harvesting of culms from which they recover in the wet season when access is limited. A 'sparing' approach would conserve papyrus areas with no or very limited anthropogenic pressures, but intensify crop production in designated agricultural zones. Sparing could have a positive impact on other ecosystems services including food production, biodiversity and general ecosystem integrity (Kiwango & Wolanski, 2008; Pacini et al., 2018), whereas sharing, with frequent harvesting, may be beneficial for optimizing or balancing a limited number of ecosystem services such as wetland based agriculture and N and P retention (Hes & van Dam, 2019). Modelling can support this debate by quantifying N and P impacts on water quality in different land-use scenarios, specifically through quantifying the impact of harvesting and other livelihood activities on retention and long term effects on regeneration of aboveground biomass (Terer et al., 2012).
Another application for these results can be found in constructed wetlands for wastewater treatment (Kengne et al., 2008; Perbangkhem & Polprasert 2010). Our results can improve management of removal efficiency. A simulation of N and P in culms (Fig. 8) showed that the maximum amount of N and P per culm was reached before the culm reached maximum length and after approximately 90 days. This was a result of increasing biomass (growth) and decreasing N and P concentrations (Fig. 6), probably caused by active retranslocation of N and P even before maximum length was reached (Snyder & Rejmánková, 2015). If harvesting is done when culms reach the maximum amount of N and P stored (and not when they are fully grown), N and P removal through harvesting can increase by almost 50%. Environmental conditions, higher nutrient loading, controlled flow and residence time are different in constructed wetlands and likely lead to differences in growth and N and P content (Chale, 1987; Kengne et al., 2008; Perbangkhem & Polprasert, 2010). The results of this study can help optimize N and P removal for a constructed wetland system with known growth rates and N and P content per growth stage, by increased frequency of harvesting and higher amount of N and P removed per harvest.
Nutrient content and length of one shoot over time: a nitrogen; and b phosphorus. Length was plotted with a logistic equation with average r (0.081 day−1) and K (414.2 cm) of all sites. N and P were plotted by multiplying length with an estimated 2nd polynomial equation describing the relationship between average length of one shoot and N or P per shoot per growth stage of all sites, with N and P in one shoot as the dependent variable. Equation for N: y = − 7*10–5 x2 + 0.0318x in which y = N (mg N/cm) and x = culm length (cm), R2 = 0.88; and for P: y = − 2*10–5 x2 + 0.0089 × in which y = P (mg P/cm) and x = culm length (cm), R2 = 0.67. Graphs were drawn with Stella Professional Version 1.7.1, ISEE systems and calculations made with MS Excel Professional Plus 2013
In conclusion, our findings can help to understand and quantify the impacts of livelihood activities and inundation on retention of N and P in natural wetlands, as well as optimize management for removal of N and P from wastewater by harvesting aboveground papyrus biomass in constructed wetlands. We recommend further research and model development on the role of biomass and accumulation of organic matter in N and P retention, and to incorporate our findings in local contexts to support management of both natural and constructed wetlands.
Data availability
Data available from author upon request.
References
APHA, 1992. Standard methods for examination of water and wastewater. American Public Health Association, Washington DC, USA.
Asaeda, T., L. Rajapakse & T. Fujino, 2008. Applications of organ-specific growth models; modelling of resource translocation and the role of emergent aquatic plants in element cycles. Ecological. Modelling 215: 170–179.
Boar, R. R., 2006. Responses of a fringing Cyperus papyrus L. swamp to changes in water level. Aquatic Botany 84: 85–92.
Boar, R. R., D. M. Harper & C. S. Adams, 1999. Biomass allocation in Cyperus papyrus in a tropical wetland, Lake Naivasha, Kenya. Biotropica 31: 411–421.
Bregoli, F., A. Crosato, P. Paron & M. E. McClain, 2019. Humans reshape wetlands: Unveiling the last 100 years of morphological changes of the Mara Wetland, Tanzania. Science of the Total Environment 691: 896–907.
Butchart, S. H. M., M. Walpole, B. Collen, A. van Strien, J. P. W. Scharlemann, E. A. Almond et al., 2010 Global Biodiversity: Indicators of Recent Declines. Science 328: 1164–1168
Chale, F. M. M., 1987. Plant biomass and nutrient levels of a tropical macrophyte (Cyperus papyrus L.) receiving domestic wastewater. Hydrobiological Bulletin 21: 167-170.
Chimney, M. J., & K. C. Pietro, 2006. Decomposition of macrophyte litter in a subtropical constructed wetland in south Florida (USA). Ecological Engineering 27: 301-321.
Clevering O. A., H. Brix & J. Lukavská, 2001. Geographic variation in growth responses in Phragmites australis. Aquatic Botany 69: 89-108.
Darwall W. R. T., V. Bremerich, A. De Wever, A. I. Dell, J. Freyhof, M. O. Gessner, H-P. Grossart, I. Harrison, K. Irvine, S. C. Jähnig, J. M. Jeschke, J. J. Lee, C. Lu, A. M. Lewandowska, M. T. Monaghan, J. C. Nejstgaard, H. Patricio, A. Schmidt-Kloiber, S. N. Stuart, M. Thieme, K. Tockner, E. Turak & O. Weyl, 2018. The Alliance for Freshwater Life: A global call to unite efforts for freshwater biodiversity science and conservation. Aquatic Conservation 28: 1015-1022.
Deegan, B. M., S. D. White & G. G. Ganf, 2007. The influence of water level fluctuations on the growth of four emergent macrophyte species. Aquatic Botany 86: 309-315.
FAO, 2019. The State of the World’s Biodiversity for Food and Agriculture. In J. Bélanger & D. Pilling (eds), FAO Commission on Genetic Resources for Food and Agriculture Assessments, Rome.
Gabrielsson, S., S. Brogaard & A. Jerneck, 2013. Living without buffers—illustrating climate vulnerability in the Lake Victoria basin. Sustainability Science 8: 143-157.
Galloway, J. N., F. J. Dentener, D. G. Capone, E. W. Boyer, R. W. Howarth, S. P. Seitzinger, G. P. Asner, C. C. Cleveland, P. A. Green, E. A. Holland, D. M. Karl, A. F. Michaels, J. H. Porter, A. R. Townsend, C. J. Vorosmarty, 2004. Nitrogen cycles: past, present, and future. Biogeochemistry 70: 153-226.
Gaudet, J. J., 1975. Mineral Concentrations in Papyrus in Various African Swamps. Journal of Ecology 63: 483-491.
Gaudet, J. J., 1977. Uptake, Accumulation, and Loss of Nutrients by Papyrus in Tropical Swamps. Ecology 58: 415-422.
Geremew, A., I. Stiers, T. Sierens, A. Kefalew & L. Triest, 2018. Clonal growth strategy, diversity and structure: A spatiotemporal response to sedimentation in tropical Cyperus papyrus swamps. PLoS ONE 13: e0190810.
Grime, J. P., 1974. Vegetation classification by reference to strategies. Nature 250: 26-31.
Grime, J. P., 1977. Evidence for the Existence of Three Primary Strategies in Plants and Its Relevance to Ecological and Evolutionary Theory. The American Naturalist 111: 1169-1194.
Hecky, R. E., R. Mugidde, P. S. Ramlal, M. R. Talbot & G. W. Kling, 2010. Multiple stressors cause rapid ecosystem change in Lake Victoria. Freshwater Biology 55: 19-42.
Hes, E. M. A. & A. A. van Dam, 2019. Modelling nitrogen and phosphorus cycling and retention in Cyperus papyrus dominated natural wetlands. Environmental Modelling & Software 122: 104531.
Hes, E. M. A., R. Niu & A. A. van Dam, 2014. A simulation model for nitrogen cycling in natural rooted papyrus wetlands in East Africa. Wetlands Ecology and Management 22: 157-176.
IPBES, 2019. Global assessment report on biodiversity and ecosystem services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. E. S. Brondizio, J. Settele, S. Díaz & H. T. Ngo (eds). IPBES secretariat, Bonn.
IPCC, 2014. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. R. K. Pachauri & L. A. Meyer (eds). IPCC, Geneva.
Jayne, T. S., S. Snapp, F. Place & N. Sitko, 2019. Sustainable agricultural intensification in an era of rural transformation in Africa. Global Food Security 20: 105-113.
Johnston, C. A., 1991. Sediment and nutrient retention by freshwater wetlands: Effects on surface water quality. Critical Reviews in Environmental Control 21: 491-565.
Jones, M. B., 1988. Photosynthetic responses of C3 and C4 wetland species in a tropical swamp. Journal of Ecology 76: 253-262.
Kansiime, F., E. Kateyo, H. Oryem-Origa & P. Mucunguzi, 2007. Nutrient status and retention in pristine and disturbed wetlands in Uganda: Management implications. Wetlands Ecology and Management 15: 453-467.
Kansiime, F., M. Nalubega, J. J. van Bruggen & P. Denny, 2003. The effect of wastewater discharge on biomass production and nutrient content of Cyperus papyrus and Miscanthidium violaceum in the Nakivubo wetland, Kampala, Uganda. Water Science and Technology 48: 233-40.
Kengne, I. M., A. Akoa, E. K. Soh, V. Tsama, M. M. Ngoutane, P. H. Dodane & D. Koné, 2008. Effects of faecal sludge application on growth characteristics and chemical composition of Echinochloa pyramidalis (Lam.) Hitch. and Chase and Cyperus papyrus L. Ecological Engineering 34: 233-242.
KNBS, 2010. The 2009 Kenya Population and Housing Census. Kenya National Bureau of Statistics, Nairobi.
Khisa, P. S., S. Uhlenbrook, A. A. van Dam, J. Wenninger, A. van Griensven & M. Abira, 2013. Ecohydrological characterization of the Nyando wetland, Lake Victoria, Kenya: A State of System (SoS) analysis. African Journal of Environmental Science and Technology 7: 417-434.
Kipkemboi, J. & A. A. van Dam, 2018. Papyrus Wetlands. In: The Wetland Book II: Distribution, Description, and Conservation. C. M. Finlayson, G. R. Milton, R. C. Prentice, N. C. Davidson (eds). Springer Netherlands, Dordrecht: 183–197.
Kiwango, Y. A. & E. Wolanski, 2008. Papyrus wetlands, nutrients balance, fisheries collapse, food security, and Lake Victoria level decline in 2000–2006. Wetland Ecology and Management 16: 89-96.
Kremen, C., 2015. Reframing the land-sparing/land-sharing debate for biodiversity conservation. Annals of the New York Academy of Sciences 1355: 52-76.
Lagerwall, G., G. Kiker, R. Muñoz-Carpena, M. Convertino, A. James & N. Wang, 2012. A spatially distributed, deterministic approach to modeling Typha domingensis (cattail) in an Everglades wetland. Ecological Processes 1: 10
Lorenzen, B., H. Brix, I. A. Mendelssohn, K. L. McKee & S. L. Miao, 2001. Growth, biomass allocation and nutrient use efficiency in Cladium jamaicense and Typha domingensis as affected phosphorus and oxygen availability. Aquatic Botany 70: 117-133.
Manzoni, S., J. A. Trofymow, R. B. Jackson & A. Porporato, 2010. Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecological Monographs 80: 89-106.
MEA, 2005. Ecosystems and Human Well-being: Wetlands and Water Synthesis. World Resources Institute, Washington DC.
Morrison E. H. J., C. Upton, K. Odhiambo-K'oyooh & D. M. Harper, 2012. Managing the natural capital of papyrus within riparian zones of Lake Victoria, Kenya. Hydrobiologia 692: 5-17.
Muraza, M., A. W. Mayo, J. Norbert, 2013. Wetland Plant Dominance, Density and Biomass in Mara River Basin Wetland Upstream of Lake Victoria in Tanzania. International Journal of Scientific & Technology Research 2: 348-359.
Muthuri, F. M. & M. B. Jones, 1997. Nutrient distribution in a papyrus swamp: Lake Naivasha, Kenya. Aquatic Botany 56: 35-50.
Muthuri, F. M., J. I. Kinyamario, 1989. Nutritive Value of Papyrus (Cyperus papyrus, Cyperaceae), a Tropical Emergent Macrophyte. Economic Botany 43: 23-30.
Namaalwa, S., A. A. van Dam, G. M. Gettel, R. Kaggwa, I. Zsuffa & K. Irvine, 2019. The impact of wastewater discharge and agriculture on water quality and nutrient retention of Namatala wetland, Eastern Uganda. Frontiers in Environmental Science 8: https://doi.org/10.3389/fenvs.2020.00148.
OECD/FAO, 2016. “Agriculture in Sub-Saharan Africa: Prospects and challenges for the next decade”. In OECD-FAO Agricultural Outlook 2016-2025, OECD Publishing, Paris.
Olokotum, M., V. Mitroi, M. Troussellier, R. Semyalo, C. Bernard, B. Montuelle, W. Okello, C. Quiblier & J. F. Humbert, 2020. A review of the socioecological causes and consequences of cyanobacterial blooms in Lake Victoria. Harmful Algae 96:101829.
Opio, A., M. B. Jones, F. Kansiime & T. Otiti, 2014. Growth and Development of Cyperus papyrus in a Tropical Wetland. Open Journal of Ecology 4: 113-123.
Opio, A., M. B. Jones, F. Kansiime & T. Otiti, 2017. Response of Cyperus papyrus productivity to changes in relative humidity, temperature and photosynthetically active radiation. African Journal of Plant Science 11: 133-141.
Pacini, N., P. Hesslerová, J. Pokorný, T. Mwinami, E. H. J. Morrison, A. A. Cook, S. Zhang & D. M. Harper, 2018. Papyrus as an ecohydrological tool for restoring ecosystem services in Afrotropical wetlands. Ecohydrology & Hydrobiology 18: 142-154.
Perbangkhem, T., & C. Polprasert, 2010. Biomass production of papyrus (Cyperus papyrus) in constructed wetland treating low-strength domestic wastewater. Bioresource Technology 101: 833–835.
Pianka, E. R., 1970. On r- and K-Selection. The American Naturalist 104: 592-597.
Pina-Ochoa, E., & M. Álvarez-Cobelas, 2006. Denitrification in aquatic environments: a cross-system analysis. Biogeochemistry 81: 111-130.
R Core Team, 2020. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/.
Ramsar Convention on Wetlands, 2018. Global Wetland Outlook: State of the World’s Wetlands and their Services to People. Ramsar Convention Secretariat, Gland.
Rejmánková, E., 2005. Nutrient resorption in wetland macrophytes: comparison across several regions of different nutrient status. New Phytologist 167: 471–482.
Rongoei, P. J. K., J. Kipkemboi, J. B. Okeyo-Owuor & A. A. van Dam, 2013. Ecosystem services and drivers of change in Nyando floodplain wetland, Kenya. 7: 274-291.
Rongoei, P. J. K. & N. O. Outa, 2016. Cyperus papyrus L. Growth Rate and Mortality in Relation to Water Quantity, Quality and Soil Characteristics in Nyando Floodplain Wetland, Kenya. Open Journal of Ecology 6: 714-735.
Rongoei, P. J. K. & Kariuki, S, 2019. Implications of Papyrus (Cyperus papyrus L.) Biomass Harvesting on Nutrient Regulation in Nyando Floodplain Wetland, Lake Victoria, Kenya. Open Journal of Ecology 9: 443-457.
Russi, D., P. ten Brink, A. Farmer, T. Badura, D. Coates, J. Förster, R. Kumar & N. Davidson, 2013. The Economics of Ecosystems and Biodiversity for Water and Wetlands. IEEP, London and Brussels; Ramsar Secretariat, Gland.
Saunders, M. J., F. Kansiime & M. B. Jones, 2014. Reviewing the carbon cycle dynamics and carbon sequestration potential of Cyperus papyrus L. wetlands in tropical Africa. Wetlands Ecology and Management 22: 413-155.
Schoumans, O. F., F. Bouraoui, C. Kabbe, O. Oenema & K. C. van Dijk, 2015. Phosphorus management in Europe in a changing world. Ambio 44: 180–192.
Schuyt, K. D., 2005. Economic consequences of wetland degradation for local populations in. Africa. Ecological Economics 53: 177–190.
Sheather, S. J., 2009. A modern approach to regression with R. Springer Science + Business Media, New York.
Silvius, M. J., M. Oneka & A. Verhagen, 2000. Wetlands: lifeline for people at the edge. Physics and Chemistry of the Earth, Part B 25: 645–652.
Snyder, J.M. & E. Rejmánková, 2015. Macrophyte root and rhizome decay: the impact of nutrient enrichment and the use of live versus dead tissue in decomposition studies. Biogeochemistry 124(1): 45-59.
Soissons, L. M., M. M. van Katwijk, B. Li, Q. Han, T. Ysebaert, P. M. J. Herman & T. J. Bouma, 2019. Ecosystem engineering creates a new path to resilience in plants with contrasting growth strategies. Oecologia 191: 105-1024.
Terer, T., L. Triest & A. M. Muasya, 2012. Effects of harvesting Cyperus papyrus in undisturbed wetland, Lake Naivasha, Kenya. Hydrobiologia 680: 135-148.
Terer, T., A. M. Muasya, S. Higgins, J. J. Gaudet, L. Triest, 2014. Importance of seedling recruitment for regeneration and maintaining genetic diversity of Cyperus papyrus during drawdown in Lake Naivasha, Kenya. Aquatic Botany 116: 93-102.
The United Republic of Tanzania, 2013. 2012 Population and housing census, population Distribution by Administrative Areas. National Bureau of Statistics, Ministry of Finance, Dar es Salaam & Office of Chief Government Statistician President’s Office, Finance, Economy and Development Planning, Zanzibar.
Tickner, D., J. J. Opperman, R. Abell, M. Acreman, A. H. Arthington, S. E. Bunn, S. J. Cooke, J. Dalton, W. Darwall, G. Edwards, I. A. N. Harrison, K. Hughes, T. I. M. Jones, D. Leclère, A. J. Lynch, P. Leonard, M. E. Mcclain, D. Muruven, J. D. Olden, S. J. Ormerod, J. Robinson, R. E. Tharme, M. Thieme, K. Tockner, M. Wright & L. Young, 2020. Bending the Curve of Global Freshwater Biodiversity Loss: An Emergency Recovery Plan Forum. BioScience 70: 330–342.
UN, 2015. World Population Prospects: The 2015 Revision, Key Findings and Advance Tables. Working Paper No. ESA/P/WP.241. UN Department of Economic and Social Affairs, Population Division, New York.
UNCCD, 2017. The Global Land Outlook, first edition. Secretariat of the United Nations Convention to Combat Desertification, Bonn.
van Asselen, S., P. H. Verburg, J. E. Vermaat & J. H. Janse, 2013. Drivers of wetland conversion: a global meta-analysis. PLoS ONE 8: e81292.
Van Dam, A. A., A. Dardona, P. Kelderman & F. Kansiime, 2007. A simulation model for nitrogen retention in a papyrus wetland near Lake Victoria, Uganda (East Africa). Wetlands Ecology and Management 15: 469-480.
Van Dam, A. A., J. Kipkemboi, M. M. Rahman & G. M. Gettel, 2013. Linking hydrology, ecosystem function, and livelihood outcomes in African papyrus wetlands using a Bayesian network model. Wetlands 33: 381-397.
Verhoeven, J. T. A., T. L. Setter, 2010. Agricultural use of wetlands: opportunities and limitations. Annals of Botany 105: 155 –163.
Weiner, J., 2004. Allocation, plasticity and allometry in plants. Perspectives in Plant Ecology, Evolution and Systematics 6: 207-215
Zemlin, R., H. Kühl, J.-G. Kohl, 2000. Effects of seasonal temperature on shoot growth dynamics and shoot morphology of common reed (Phragmites australis). Wetlands Ecology and Management 8: 447-457.
Acknowledgements
The authors would like to thank: IHE Delft Institute for Water Education for time allocated to this research; the Netherlands Fellowship Programme (NFP) for funding part of the field work; and Do Phuong Hien, Hawa Nakitende and Kuenzang Tshering for their invaluable contributions to the field experiments.
Funding
The Netherlands Fellowship Programme (NFP) funded part of the field work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Handling editor: Eric R. Larson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hes, E.M.A., Yatoi, R., Laisser, S.L. et al. The effect of seasonal flooding and livelihood activities on retention of nitrogen and phosphorus in Cyperus papyrus wetlands, the role of aboveground biomass. Hydrobiologia 848, 4135–4152 (2021). https://doi.org/10.1007/s10750-021-04629-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-021-04629-3