Abstract
Mangrove forest encroachment into coastal marsh habitats has been described in subtropical regions worldwide in recent decades. To better understand how soil processes may influence vegetation change, we studied soil surface elevation change, accretion rates, and soil subsurface change across a coastal salinity gradient in Florida, USA, an area with documented mangrove encroachment into saline marshes. Our aim was to identify if variations in the soil variables studied exist and to document any associated vegetation shifts. We used surface elevation tables and marker horizons to document the soil variables over 5 years in a mangrove-to-marsh transition zone or ecotone. Study sites were located in three marsh types (brackish, salt, and transition) and in riverine mangrove forests. Mangrove forest sites had significantly higher accretion rates than marsh sites and were the only locations where elevation gain occurred. Significant loss in surface elevation occurred at transition and salt marsh sites. Transition marshes, which had a significantly higher rate of shallow subsidence compared to other wetland types, appear to be most vulnerable to submergence or to a shift to mangrove forest. Submergence can result in herbaceous vegetation mortality and conversion to open water, with severe implications to the quantity and quality of wetland services provided.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ecosystem regime shifts have become increasingly common as a consequence of human activities that reduce natural system resilience, such as resource exploitation, pollution, altered disturbance regimes, and land-use and climate change (Folke et al., 2004). Coastal wetlands have been described as some of the most vulnerable yet economically important ecosystems on earth (Kirwan & Megonigal, 2013), and sea-level rise (SLR) is expected to have a major influence on the function and resiliency of these wetlands (Morris et al., 2002; Kirwan & Temmerman, 2009; McKee et al., 2012; Crosby et al., 2016). On subtropical coasts, the transition zone, or ecotone, between mangrove forests and herbaceous marshes is vulnerable to shifting boundaries from climate change-driven alterations in temperature and precipitation as well as SLR (Osland et al., 2016; Gabler et al., 2017). Several studies have documented vegetation shifts within the mangrove–marsh ecotone (hereafter called ecotone); these shifts were often identified as mangrove forest encroachment into brackish and salt marshes (Perry & Mendelssohn, 2009; Krauss et al., 2011; Saintilan et al., 2014; Armitage et al., 2015; Howard et al., 2017). In addition, freshwater marsh displacement by mangroves has occurred in southeast Florida, USA (Ross et al., 2000; Meeder et al., 2017). Mangroves are cold-intolerant, and range expansion is facilitated by an increased interval between winter freeze events (Sherrod et al., 1986; Stevens et al., 2006; Cavanaugh et al., 2015). Chronic warming between freeze events, however, can stimulate growth of wetland grasses, leading to increased levels of interspecific competition that may slow the progression of mangrove dominance (Coldren et al., 2016). Human-caused hydrologic alteration (e.g., canal construction) has also been identified as a factor that can enhance mangrove encroachment into marshes (Krauss et al., 2011). Such vegetation shifts within the ecotone have implications in terms of ecosystem services provided (reviewed in Kelleway et al., 2017). Notably, increased aboveground biomass and carbon storage have resulted when marsh vegetation was replaced by mangrove forest (Doughty et al., 2016; Yando et al., 2016; Simpson et al., 2017). In addition to species shifts, plant community structural changes (e.g., stem density, stem height, belowground biomass) in response to SLR can also affect services provided.
The ability of wetlands to track rising sea levels vertically by increasing soil surface elevation involves feedbacks between ecogeomorphic processes, including plant growth, organic matter accumulation, mineral sedimentation, and subsidence, and physical factors that are associated with the position of the wetland platform, including tidal inundation duration and exposure to erosional forces. Global SLR was estimated at 1.1 ± 0.3 mm year−1 in the 20th century prior to 1990, with an increase to 3.1 ± 1.4 mm year−1 between 1993 and 2012 (Dangendorf et al., 2017). Trends of accelerated local or regional SLR compared to global levels have been identified along the Atlantic (Sallenger et al., 2012; Valle-Levinson et al., 2017) and Gulf of Mexico (Donoghue, 2011; Wahl et al., 2014) coasts of the USA; wetlands of the latter region are also characterized by high subsidence rates (Törnqvist et al., 2008; Yuill et al., 2009; Day et al., 2011). As noted by Reed (1999), when increased flooding resulting from subsidence is accompanied by enhanced sediment supply, marshes may be able to maintain elevation under higher sea levels. In addition to sedimentation, wetland surface elevation is affected by vegetation through subsurface processes such as root growth and sediment expansion (Nyman et al., 2006; McKee, 2011; Krauss et al., 2014). When surface elevation change does not equal or exceed relative SLR through enhanced sedimentation and/or increased primary production, species composition may shift to more flood-tolerant species.
Interspecific and intraspecific variations in tolerance to flooding stressors have been found in many herbaceous wetland species (Fraser & Karnezis, 2005; Spalding & Hester, 2007; Visser & Sandy, 2009; Howard et al., 2016). If shifts in plant species composition do not occur in response to increased flooding, open water areas can form. Ponding is the conversion of interior vegetated coastal wetlands to open water (DeLaune et al., 1994). The drivers that can lead to pond formation are varied and complex (Day et al., 2000) and, in addition to the inability of some species to tolerate flooding stress (Mendelssohn & McKee, 1988; Nyman et al., 1993), include mortality induced by salt stress (DeLaune et al., 1994), intense herbivory (Stevenson et al., 1985), and enhanced subsidence resulting from subsurface fluid withdrawal for hydrocarbon production (Morton et al., 2006). The ability of coastal wetlands to tolerate and/or recover from stressors can be influenced by plant interactions (e.g., competition, facilitation) that alter species growth and survival, and the resulting structural and composition changes in vegetation communities may affect soil process through impacts on plant productivity. Changes in plant productivity can affect elevation change and therefore the ability of a marsh to offset the effects of rising sea levels (Baustian et al., 2012).
Wetland sedimentation often has both organic and mineral components (Nyman et al., 1993; Reed, 1995; Anisfeld et al., 2016), and the relative importance of these components in influencing surface elevation varies across the coastal landscape. Surface elevation influences coastal wetland hydrology, affecting tidal inundation frequency, flooding duration, and flooding depth. These hydrologic conditions affect soil oxidation state, erosion, and accretion (i.e., surface accretion or deposition of surface material by sedimentation), which, in turn, influence plant growth. A strong relationship between the rate of salt marsh surface elevation change and the rate of mangrove encroachment was identified in southeast Australia; salt marsh sites with elevation deficits exhibited high rates of mangrove encroachment (Rogers et al., 2006). Patterns in soil surface elevation and accretion in ecotones during the shift from herbaceous to mangrove species were examined in two studies. In New South Wales, Australia, surface elevation increase was lower at sites where mangrove encroachment occurred compared to sites in a mature mangrove forest and in a salt marsh; accretion in the encroachment zone, however, was intermediate between the two habitats, with highest accretion occurring in the forest (Rogers et al., 2013). In contrast, no difference was found in elevation change or accretion among salt marsh, young mangrove (less than 30 years old), and mixed marsh/mangrove sites in Louisiana, USA (McKee & Vervaeke, 2018). The discrepancy between these studies indicates that variability can exist in the relationships between mangrove encroachment and soil elevation change and accretion. Our study aims to provide additional information on these relationships.
In this study, we examined soil vertical accretion and relative soil surface elevation change across a salinity gradient in coastal southwest Florida, a region that is experiencing upslope expansion of mangrove forests into saline marshes. We addressed the following questions: (1) Do patterns of surface elevation change and surface accretion rate vary across the environmental gradient? (2) Are vegetation shifts, either among herbaceous species, from herbaceous species to mangroves, or to open water, associated with surface elevation change? and (3) Are soil process changes and/or vegetation shifts corroborated by data derived from aerial imagery on mangrove forest expansion and pond formation?
Materials and methods
Study area
The study area at Ten Thousand Islands National Wildlife Refuge (TTINWR), located in southwest Florida (25° 57′ N, 81° 32′ W), includes a gradient from brackish marsh to mangrove fringe forest. The refuge was established in 1996 and is comprised of about 14,165 ha of mangrove forests, emergent marshes, open ponds, and small coastal hammocks. The subtropical climate is characterized by mean annual rainfall between 127.0 and 152.4 cm; seasons alter between wet (May–October) and dry (November–April) (Lodge, 2010). Winter daytime temperatures in south Florida are often above 25 °C, with an average daily minimum temperature of 10 °C; however, temperatures can drop below freezing during the passage of cold fronts, which can occur frequently in some years (Duever et al., 1994). Soils are predominately histosols, consisting of peat and muck deposits of varying thickness over sand, marl, and limestone (Brown et al., 1990). Historically, freshwater flowed from north to south seasonally as sheetflow across TTINWR into the Gulf of Mexico (Gulf); however, road, canal, and housing infrastructure development that began in the 1920s altered regional hydrologic patterns so that water became unevenly distributed through a network of canals (Booth et al., 2014). Based on position in the landscape and historic flow patterns, the more northern brackish marsh sites likely receive higher surface and groundwater freshwater inflow compared to salt and transition marshes (Booth et al., 2014), while the mangrove forest sites receive riverine and marine sediment subsidies. Variation in salinity has been documented among habitat types on TTINWR. Salinity gradually decreased with distance inland from mangrove forests to brackish marshes, ranging from 35.3 to 17.9 psu, and interstitial water salinity tended to increase with depth beneath the soil surface (Krauss et al., 2011). A study of vegetation communities on the refuge recorded interstitial water salinity values at 15 cm below the marsh soil surface between 4 and 30 psu during the fall (Howard et al., 2017). Among the marsh types, interstitial salinity was higher in transition marshes compared to salt and brackish marshes over time, but salt and brackish marshes often had similar salinity levels (Howard et al., 2017). At the southern boundary of TTINWR, Pumpkin Bay has a mean tidal range of 0.88 m, with a diurnal tidal range (difference in height between mean higher high water and mean lower low water) of 1.15 m (NOAA, 2018a).
Three species of mangroves—Avicennia germinans (L.) L. (black mangrove), Laguncularia racemosa (L.) C.F. Gaertn. (white mangrove), and Rhizophora mangle L. (red mangrove)—are found on TTINWR and form a mixed-species forest that is contiguous with the Gulf on the southern portion of the refuge. Inland of the mangrove forests is salt marsh dominated by Distichlis spicata (L.) Greene (saltgrass) and Juncus roemerianus Scheele (needle rush), with brackish marshes dominated by Spartina bakeri Merr. (sand cordgrass) and Typha domingensis Pers. (southern cattail) at the northern refuge boundary. Open water channels and ponds throughout TTINWR are often bordered by mangrove forest. Between 1927 and 2005, a 35% increase in mangrove coverage on TTINWR was identified (Krauss et al., 2011); this increase was attributed to sea-level rise and hydrological changes resulting from canals constructed to drain wetlands to provide sites for housing and commercial development. Hydrologic alterations in the region surrounding the refuge were also linked to vegetation loss on the refuge and an increase in ponded areas (Andres et al., 2019). A recent model analysis predicted that, under a sea-level rise of 0.5 m and macroclimate change (i.e., temperature and evapotranspiration increases, altered precipitation), mangrove forests in this region of Florida will migrate up to 15 km inland by 2060 (Flower et al., 2017). Vegetation communities on TTINWR will also be affected by a major hydrologic restoration project that is part of the Comprehensive Everglades Restoration Plan (https://www.nps.gov/ever/learn/nature/cerp.htm); the Picayune Strand Restoration Project, located directly north of the refuge, has the goal of increasing freshwater sheet flow from north to south. The project includes construction of three pumping stations, with the final station completed in the summer of 2019 (US Army Corps of Engineers, 2019).
Surface accretion and elevation
We used the rod surface elevation table-marker horizon (RSET-MH) method (Cahoon et al., 2002; Cahoon, 2015) to document changes in relative soil surface elevation and vertical accretion. While the surface elevation table technique has been deployed worldwide since its development in the 1990s (Webb et al., 2013), relatively few stations exist in the mangrove–marsh ecotone along the northern Gulf of Mexico coast (Osland et al., 2017). When tied to a survey datum, RSETs establish absolute measures of vertical land motion (VLMw, or elevation) of the wetland soil surface relative to the base of the rod, which can be related to tide gauge data (Cahoon, 2015). The RSET is a portable device that is attached to a series of connected steel rods driven into the substrate to refusal (i.e., a benchmark), and elevation measurements are obtained relative to the base of the benchmark (see Cahoon, 2015 for details). Marker horizons, consisting of a layer of powdered, light-colored feldspar (clay) material deposited on the soil surface (Cahoon & Turner, 1989), are placed in the vicinity of RSETs for simultaneous measurement of sediment vertical accretion. Shallow subsidence (in the form of soil compaction) is indicated when vertical accretion exceeds VLMw (Cahoon et al., 1995). Root zone expansion is indicated when VLMw exceeds vertical accretion (Cahoon et al., 2000; McKee et al., 2007a); root zone expansion is defined as the upward movement of the soil surface from root volume expansion occurring below the soil surface.
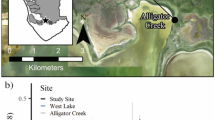
We installed RSET stations in June and December 2011 at eight sites across the gradient from brackish marsh at the northern boundary of TTINWR to riverine mangrove forest near the southern refuge boundary along the Gulf. There were two sites in each of four wetland types: brackish marsh, salt marsh, transition marsh (defined as saline marsh adjacent to a mangrove forest contiguous to the Gulf), and mangrove forest (Fig. 1). The RSETs in the three marsh types were located within 10 m of permanent transects established in 2007 to record vegetation composition (Howard et al., 2017). The transects extended perpendicularly from near the edge of the mangrove forest (transition marsh) or the edge of a mangrove stand along a waterway (brackish, salt marshes) into marsh that did not support mature mangrove trees (Howard et al., 2017). Two RSET stations were located at each site, resulting in four RSETs in each habitat type and 16 total stations. The length of the steel rods that were driven to refusal at each station was recorded.
Map of Ten Thousand Islands National Wildlife Refuge, Naples, Florida, showing locations of eight sites where rod sediment elevation (RSET) stations were installed. There were two RSET stations at each site. Asterisks indicate sites where data collected were related to information on long-term pond formation as reported by Andres et al. (2019)
Initial elevation data were collected in June 2012. Three marker horizons were established at this time within 4.5 × 4.5 m (20.25 m2) plots established at each RSET installation; the benchmark was located at the plot center. No mangrove seedlings were present in the marsh plots when initial data were collected. VLMw was measured by attaching the RSET arm to the benchmark at four compass positions. At each position, nine individual elevation measurements were made by positioning fiberglass pins to touch the soil surface and then recording pin length (to nearest mm) extending above the arm. Any obstructions (e.g., woody debris) or soil surface damages (e.g., invertebrate burrow) were noted for each pin location. With an RSET arm length of 62 cm, elevation measurements were obtained within a circular area of approximately 1.2 m2. During the subsequent VLMw measurement in October 2012, soil cores were extracted from each of the three marker horizon stations in each RSET plot using a small (2.5 cm diameter × 18 cm length) modified Russian peat corer, also known as a Macaulay corer (Jowsey, 1966). Accretion, measured using calipers as the depth (mm) of accumulated soil material above the feldspar layer, was recorded for three positions on each core (the middle and both sides of the D-shaped core). Elevation and accretion measurements were obtained using these techniques four additional times over 5 years, in June 2013, 2014, 2015, and 2017.
To relate elevation change to marsh conversion to open water, we used information from a previous study on historical pond formation on TTINWR. The previous study examined wetland geomorphic and vegetation change in southwest Florida and included sites on TTINWR (Andres et al., 2019). As part of that study, 1 km2 (100 ha) marsh sections were delineated and evaluated for the creation, expansion, and merger of ponds, also referred to as “pocks” based on their appearance, over time using aerial imagery from 1953 and 2009. Two of our RSET-MH sites—the western salt and western transition marsh sites—were located in one of the marsh sections delineated by Andres et al. (2019). Between 1953 and 2009 pond area in the 1 km2 marsh section that overlapped with our four RSET-MH plots increased by 54,562 m2 (5.5 ha) (Andres, 2016). We examined soil variables at the four plots to determine if our findings complement the historically documented increase in pond formation by indicating decreased soil surface elevation, which can result in vegetation mortality and conversion to open water.
Vegetation
Herbaceous vegetation cover and mangrove seedlings
Cover values of herbaceous plant species present within the 20.25 m2 RSET plots in marsh habitats (brackish, salt, and transition) were visually estimated and recorded to the nearest 5%; species with less than 2.5% estimated cover were designated as 1% cover. Data were obtained each year during elevation and accretion measurements beginning in June 2012. With the exception of the second measurement in October 2012, subsequent measurements were made mid-growing season (i.e., June 2013–2015, 2017) for a total of six measurement times. The number of mangrove seedlings in the marsh plots was recorded by species in 2015 and 2017. The mangrove RSET plots were located in mature forests where herbaceous cover was absent or very sparse and few mangrove seedlings were present. Because of this low occurrence and the study focus on documenting marsh species shifts and vegetation loss, we did not estimate cover or record seedling numbers in forest RSET plots.
Mangrove forest extent
The mangrove boundary of TTINWR was previously mapped over a 78-year period using a combination of T-sheets (1927), black and white aerial photographs (1940), and true-color aerial imagery (2005), documenting the addition of approximately 1878 ha of new mangrove area at the expense of marsh habitat on TTINWR (Krauss et al., 2011). Since then, mangrove encroachment has visibly slowed following freeze events in January and December 2010, when temperatures fell below − 2° C for several hours (Howard et al., 2017), and a controlled burn in March 2012 (A. From, personal observation). We extended the mapping period an additional 9 years to overlap RSET-MH measurements and report mangrove habitat change through 2014, using sub-meter resolution true-color imagery taken in December 2014 (Collier Country Property Appraiser’s Office, Naples, Florida, USA). The National Standard for Spatial Data Accuracy (NSSDA) for this imagery at 95% was 0.516 m. The North American Datum of 1983 (Florida East) was the spatial reference used for mapping. The images were analyzed using ArcGIS (Version 10.4 ESRI, Inc., Redlands, California, USA), and were rectified to a common geoid and projection to facilitate comparisons with the map sets from 2005. Classification was simple, with polygons being assigned as either mangrove or non-mangrove; mangrove species identity was not differentiated. There was no dedicated field ground truthing for analysis of the 2014 imagery; however, stratified random sampling along transects and spot checks were conducted in 2009 for the Krauss et al.'s (2011) study on mangrove forest change.
Data analysis
For VLMw analyses, invalid measurements (e.g., pin touched a root or other unmovable debris) were not included. The mean of pin measurements for each RSET arm position was determined, and elevation for each RSET station was calculated as the mean of the four arm measurements. Mean VLMw data of the four RSET replicates in each wetland type were obtained for each measurement time; these data were then expressed as cumulative change from the baseline (i.e., initial) measurement. Data were analyzed using simple linear regression analysis, which was applied to each wetland type separately. The regression slopes were examined for significant variation from zero (i.e., baseline condition). The mean of the three accretion measures in each soil core was determined, and the mean for the RSET plot was calculated using cores from the three feldspar plots. Accretion data were analyzed in the same manner as VLMw data. Subsurface change was calculated by subtracting VLMw from vertical accretion, with positive values indicating shallow subsidence (Cahoon et al., 1995; Cahoon, 2015). Linear rates of change among wetland types for VLMw, accretion, and subsurface change were compared using regression analysis.
A separate analysis of VLMw, accretion, and subsurface change was conducted using the methods described above for the four RSET stations (western salt and western transition marsh plots) located within a study site delineated by Andres et al. (2019). Statistical analysis of the relationship between the soil variables and pond formation was not possible because of the different scales of the data sets (i.e., elevation was measured within an area of approximately 1.2 m2, ponding was measured within 1-km2 sections) and different times of data collection. Therefore, we examined our results to determine if they complement the historic increase in pond formation identified by Andres et al. (2019) by indicating elevation loss.
The relationship of total herbaceous vegetation cover in the marsh RSET plots and VLMw, accretion, and subsurface change was investigated using a Pearson correlation analysis; the marsh types were examined separately. For these analyses, five measurement times were included; baseline values were deleted because the soil variables were set at zero by definition. Variation in vegetation cover among the marsh types over time was examined using repeated-measures analysis of variance (ANOVA). We believe that the repeated-measures framework was required based on the probable lack of independence between measurement times (i.e., influence of seed rain and vegetative expansion through rhizomes). Marsh type was the independent variable in these analyses, with total herbaceous vegetation cover and cover of the most common species within the plots as dependent response variables.
To examine differences in woody encroachment among marsh types, one-way ANOVA was applied with marsh type as the independent variable and final mangrove seedling number as the response variable. The relationships between remotely sensed mangrove forest cover information and the site-specific soil variables were not examined using statistical methods because of the different scales of the data sets and different times of data collection. Rather, we examined mangrove forest cover change interpreted from aerial imagery to identify patterns that may corroborate trends identified for the small-scale measurements of the soil variables.
Regression (PROC REG), correlation (PROC CORR), ANOVA (PROC GLM), and repeated-measures ANOVA (PROC MIXED) analyses were conducted using SAS software (ver. 9.3; SAS Institute, Inc., Cary, North Carolina). Response variables that did not meet required assumptions for normality and homogeneity were transformed, and the significance level for analyses was α = 0.05. Main effect comparisons were examined using Tukey’s test, and interaction effects were examined using the Bonferroni test adjusted for the number of comparisons.
Results
Surface accretion and elevation
Regression analyses by wetland type comparing change from baseline conditions identified significant accretion in all types and significant VLMw change in salt marshes, transition marshes, and mangrove forests that ranged from − 6.45 to 4.36 mm year−1 (Table 1). Significant shallow subsidence was found in all types, but was accentuated in the transition marsh (9.74 mm year−1) where elevation loss was the greatest (Table 1). Comparisons of rates of change among wetland types indicated significant differences (P < 0.0001, all models) in all soil variables (accretion, VLMw, subsurface change) among the types. Accretion rate was significantly higher in mangroves (6.15 mm year−1) compared to the three marsh types (mean = 3.03 mm year−1), while it was equivalent among the marsh types. The rate of VLMw change at transition marshes (− 6.45 mm year−1) was significantly lower than that at the other three wetland types (mean = 0.90 mm year−1). Among those three types, VLMw was significantly lower in salt marshes compared to mangroves, but it was equivalent in brackish marshes compared to both salt marshes and mangroves. Subsurface change in transition marshes was significantly greater compared to the other wetland types, while it did not differ between brackish marsh, salt marsh, and mangrove forest plots (Table 1). The rod depth to refusal for the 16 RSET benchmarks ranged from 3.3 to 13.9 m, with an overall mean of 9.0 ± 0.78 m (data presented as mean ± 1 SE). Based on refusal depth from most shallow to deepest, the wetland types were ranked as follows: transition marsh (5.5 ± 1.49 m), mangrove forest (8.4 ± 1.08 m), salt marsh (10.4 ± 0.78 m), and brackish marsh (11.5 ± 1.15 m).
Analyses of the subset of RSET-MH installations within the marsh section described by Andres et al. (2019) showed significant accretion (P < 0.0001), VLMw change (P = 0.0011) and subsurface change (P < 0.0001). Results were as follows: accretion, 2.12 ± 0.40 mm year−1; VLMw change, − 2.67 ± 0.72 mm year−1; and subsurface change, 4.79 ± 0.77 mm year−1.
Vegetation
Herbaceous vegetation cover and mangrove seedlings
A total of 13 species was recorded in the marsh RSET plots during the study (Table S1). The most common species in brackish and salt marsh plots were S. bakeri and D. spicata; S. bakeri was absent from plots in transition marshes, where the dominant species was D. spicata. Sesuvium portulacastrum (L.) L. was the only species in addition to D. spicata that was present in all marsh types. Total herbaceous species cover was not correlated with VLMw in any marsh type, and in transition marshes, there was also a lack of correlation of cover with accretion and subsurface change (Table 2). The direction of correlation between cover and accretion and cover and subsurface change varied among brackish and salt marshes. Cover in brackish marshes was positively correlated with both variables, while in salt marshes, it was negatively correlated with both (Table 2).
Total cover and S. portulacastrum cover responded to the interaction between marsh type and time (Table 3). Total cover in salt marsh plots in June 2017 was lower than that at the previous five measurement times (Fig. 2); total cover in brackish and transition marsh plots did not vary significantly over time. Marsh type differences in total cover identified were lower initial (June 2012) cover in brackish compared to salt marsh plots and lower final cover (June 2017) in salt compared to transition marsh plots (Fig. 2). Cover of D. spicata differed among all three marsh types regardless of measurement time and was lowest in brackish marsh plots (Table 4A). A significant difference in S. portulacastrum cover was identified for June 2017, when it was lower in salt and transition compared to brackish plots (Table 4B). Spartina bakeri cover responded to both time and marsh type; it was lower in brackish compared to salt plots and in June 2017 compared to all other measurement times (Table 4C).
There were no mangrove seedlings present in the marsh RSET plots at installation. During baseline measurements in June 2012, however, one seedling of R. mangle was noted in a brackish marsh plot. In June 2013, there were R. mangle seedlings present in 5 of the 12 plots. The presence of L. racemosa was first noted in June 2014, when it was found in two plots. In June 2017, A. germinans was noted for the first time as six seedlings in a single transition marsh plot. There was a total of 82 mangrove seedlings comprised of the three species distributed across 11 of the 12 marsh plots in June 2017; L. racemosa was most abundant with 39 seedlings occurring in salt and transition marsh plots. Mangrove seedling number in June 2017 differed by marsh type (P = 0.0309); it was lower in brackish marshes (1.75 ± 0.85) compared to transition marshes (10.25 ± 4.11), while seedling number in salt marshes (8.50 ± 1.55) was equivalent to both other marsh types.
Mangrove forest extent
Mangrove forest coverage showed areas of both contraction and expansion in 2014 imagery compared to imagery from 2005. While northward latitudinal movement (Fig. 3) of about 550 m was documented, there was a negligible (10 ha) decrease in mangrove coverage, from 7274 ha in 2005 (Krauss et al., 2011) to 7264 ha in 2014 (our study). Patches of mangrove mortality ranging in size from about 0.1 to 1.6 ha were noted in the imagery (Fig. 3).
Recent mangrove cover change on Ten Thousand Islands National Wildlife Refuge, Naples, Florida. Mangrove stands identified in imagery from both years appear in gray. Purple indicates healthy mangrove strands in 2005 that were dead or had transitioned to other habitat types in 2014, while blue indicates non-mangrove habitats in 2005 that were dominated by mangroves in 2014
Discussion
Our study identified differences in elevation and accretion dynamics across the coastal gradient at TTINWR. Mangrove forest sites, located closest to the Gulf, had significantly higher accretion rates compared to marsh sites and were the only locations where elevation gain was documented. Although the mangrove sites in our study experienced significant shallow subsidence, the rate of subsidence was offset by a higher rate of vertical accretion. Higher sediment accretion in mangrove compared to salt marsh habitats was also found in Western Port Bay in southeast Australia, an area where mangrove landward expansion is occurring (Rogers et al., 2005). The surface elevation increase documented at the mangrove sites (4.36 ± 0.31 mm year−1) exceeds the recent trend (1965–2017) of relative SLR (2.8 ± 0.45 mm year−1) recorded at the nearest tide gauge, in Naples, FL (NOAA, 2018b). These results indicate that the mangrove forests we studied are resilient to SLR over the time frame studied and, in the absence of an acceleration in SLR rate, will maintain, or gain, elevation over time. This is consistent with a study at a mangrove forest near Rookery Bay, also in southwest Florida, where fringe mangrove forests located along a protected shoreline were found to be generally tracking SLR over a 2.5-year study (Cahoon & Lynch, 1997). The rate of shallow subsidence documented at our riverine mangrove sites, at 1.65 ± 0.71 mm year−1, was lower than that found in Rookery Bay fringe forests, at 3 – 4 mm year−1 (Cahoon & Lynch, 1997). However, a different study in Rookery Bay fringe forests documented a subsidence rate of about 5 mm year−1 with a surface elevation change of only 0.61 ± 1.84 mm year−1, which was lower than relative sea-level rise (McKee, 2011). The discrepancies between the Rookery Bay studies indicate that local variability in processes affecting elevation can be important; large differences in surface elevation dynamics can occur over very short distances in coastal wetlands.
In contrast to our mangrove forest sites, significant loss in soil surface elevation occurred at transition and salt marsh sites in TTINWR. This implies that these sites are vulnerable to flooding stressors that can reduce plant growth and survival, such as the development of an anaerobic soil environment and sulfide toxicity (Li et al., 2009; Lamers et al., 2013). Transition marshes, with a significantly higher rate of subsurface change compared to the other three wetland types, therefore appear to be the most vulnerable to submergence and transition to open water. The significant positive subsurface change (i.e., subsidence) found at the marsh sites is consistent with the study by Rogers et al. (2013), where high subsidence was documented in a mangrove encroachment zone in southeast Australia. It is possible that low allochthonous sediment input to the more interior salt and transition marsh sites on TTINWR contributed to the relatively low accretion rates at these sites, and that the low accretion cannot compensate for the higher subsidence rates as compared to subsidence rates in brackish marsh and mangrove sites. It is interesting to note that the rod depth to refusal during RSET installation was lowest at the transition marsh sites, where the rate of subsidence was highest. Depth to refusal cannot be considered a definitive indicator of soil depth above the bedrock because the rods may be deflected from the perpendicular or enter porous rock. However, because the same installation technique was used at all sites, variations in depth to refusal among the sites may reflect relative variation in soil depth above the bedrock. The comparatively shallow depth to refusal at the transition marsh sites may therefore be related to low sediment input.
A significant loss in soil surface elevation was identified for the four stations (two in salt and two in transition marshes) located within a 100-ha section identified by Andres (2016) as displaying an increase in pond area of 5.5 ha between 1953 and 2009, equivalent to a 5% loss of vegetation. Although that study did not cover the time frame of our data collection, the long-term change (i.e., over 56 years) in pond coverage documented may be relevant to our findings of negative VLMw at the four salt and transition marsh plots. The decreasing soil surface elevation we identified may be driving mortality of emergent marsh species and conversion to open water. Increasing open water on TTINWR is consistent with studies conducted along the Atlantic coast. Interior ponding was identified as the major cause of wetland loss on the eastern shore of Chesapeake Bay in Maryland (Kearny et al., 1988; Schepers et al., 2017), and extensive interior ponding was documented in Maine coastal wetlands (Wilson et al., 2010). A higher rate of increase in pond density on TTINWR was documented close to Faka Union Canal, located on the eastern border of the refuge, compared to areas farther away; this pattern was attributed to hydrologic alterations associated with the canal (Andres et al., 2019). The Faka Union Canal was constructed in the early 1970s and cut off the Faka Union River, disrupting water flow patterns that supported wetland development and persistence. The increased tidal signature in the northeastern portion of TTINWR attributed to these alterations (Krauss et al., 2011) may be a driving factor for marsh loss.
Total vegetation cover was not correlated with VLMw in the RSET plots, and conflicting results were obtained for comparisons between cover and accretion and between cover and subsurface change, depending on marsh type (i.e., positive, negative, or absent). For example, there was a significant decrease in surface elevation in transition marsh plots, where there was a lack of correlation between cover and subsurface change (Table 2). However, in salt marsh plots where significant elevation loss also occurred, but to a lesser degree compared to transition plots, a negative correlation between the variables indicated that subsurface change increased (i.e., subsidence increased) as cover decreased. This relationship would be expected if vegetation is contributing to elevation increase through root zone expansion or organic matter contribution. In a confusing contrast to results in the subsiding marsh plots, however, there was a positive correlation of cover and subsurface change in brackish marsh plots where elevation was maintained, indicating that subsurface change increased (subsidence increased) as cover increased. These analyses, therefore, did not lead to consistent conclusions regarding the relationship of vegetation cover to the soil processes studied at TTINWR.
Differences in vegetation cover and species assemblage among the marsh zones were identified. Lower total vegetation cover in salt marsh plots and lower S. bakeri cover in both salt and brackish marsh plots at the end of the study indicate a vulnerability to submergence in these marshes. Total cover at brackish marsh plots tended to increase, but the increase was insignificant; this result was likely due to large variation at the final measurement time. Decreasing S. bakeri cover was visually striking at some sites (Fig. 4), and loss of this species has been noted for the refuge overall (Howard et al., 2017; Andres et al., 2019). Counterintuitively, we did not find a decrease in either total cover or D. spicata (the dominant species) cover at transition marsh sites, where the highest rate of shallow subsidence was documented. It is possible that the elevation of the marsh platform at the transition sites had not yet reached a threshold point below which mortality of herbaceous species would occur. Both S. bakeri and D. spicata occur in intertidal high-marsh habitats (Maricle et al., 2007). In general, D. spicata is considered to be relatively intolerant of flooding stress (Lonard et al., 2013). The position of D. spicata on the marsh platform has been described as being about 10–15 cm above mean high water to about 10 cm below mean high water (Lefor et al., 1987), and in marshes with average water levels of 5 cm above to 15 cm below the soil surface (Shiflet, 1963). Aboveground biomass and tiller density of D. spicata were reduced in a greenhouse study under high inundation (semi-diurnal tide with high tide at 5 cm above the soil surface) compared to low inundation (high tide at 15 cm below soil surface), but only under high nutrient conditions (Watson et al., 2015). In another greenhouse study, continuous flooding to a depth of 10 cm reduced D. spicata stem number regardless of salinity levels (0, 10, and 25 psu; Howard & Rafferty, 2006). Little information exists on the flooding tolerance of S. bakeri. However, there is some evidence of variation in the tolerance of S. bakeri and D. spicata to environmental stressors. In a greenhouse study that simulated a 20-cm semi-diurnal tidal cycle, D. spicata biomass was significantly reduced under tidal (water 15 cm above the soil surface at high tide) compared to saturated (continuous flooding depth of 1 cm) conditions, whereas S. bakeri biomass was equivalent under both hydrologic conditions (Howard et al., 2016).
Photographic views along two vegetation transects located about 10 m from rod surface elevation table stations on Ten Thousand Islands National Wildlife Refuge, Naples, Florida. The views are orientated north to south along the 50-m long transects and arrows indicate the position of the south marker post. The left photographs are of a western salt marsh transect taken in a September 2008 and b September 2016. The dominant vegetation in 2008 was Spartina bakeri; the mangroves present in 2016 were Laguncularia racemosa and Rhizophora mangle. The right photographs are of a central (eastern location on Fig. 1) brackish marsh transect taken in c September 2009 and d September 2016. Dominant species in 2009 were S. bakeri and Distichlis spicata, whereas in 2016 the transect was primarily open water with a few seedlings of L. racemosa and R. mangle among sparse cover of D. spicata and Typha domingensis
Mangrove encroachment was documented at all marsh sites, and the number of colonizing mangrove seedlings was significantly greater in transition compared to brackish marsh plots. If seedling survival is high, transition marsh sites may convert to forested sites rather than to open water habitat despite high subsidence rates. Because transition marsh sites were adjacent to mangrove forests that are contiguous to the Gulf of Mexico to the south, it is possible that greater propagule availability contributed to relatively high seedling recruitment at these marshes. Mangrove establishment success in marshes can vary depending on marsh species composition. Facilitation is indicated by the enhancement of mangrove propagule entrapment by some marsh plants (Lewis, 2005; McKee et al., 2007b; Donnelly & Walters, 2014); however, entrapment success varies with the growth form of the herbaceous species (Peterson & Bell, 2012) and mangrove propagule size (McKee et al., 2007b). In contrast, competitive suppression of mangrove seedling growth by herbaceous plants has been documented in field studies (Patterson et al., 1993; Zhang et al., 2012; Guo et al., 2013) and in greenhouse experiments (McKee & Rooth, 2008; Howard et al., 2015). The dominant species in transition marshes in our study, D. spicata, did not affect establishment of A. germinans or L. racemosa, but it did suppress mangrove seedling growth, in a greenhouse study (Howard et al., 2015). Long-term studies are required to document if degrading marshes that shift from herbaceous species to mangrove dominance change from a system characterized by elevation loss to one characterized by elevation gain as the forest matures.
In contrast to our results indicating sedimentation and elevation differences among vegetation assemblages along a coastal gradient, studies comparing adjacent Spartina alterniflora and A. germinans stands in Louisiana found similar accretion rates (Perry & Mendelssohn, 2009), and both similar accretion rates and elevation trajectories (McKee & Vervaeke, 2018), between the vegetation types. The Louisiana studies were conducted near the latitudinal limit for A. germinans, where this species is characterized by relatively short stature (less than 5 m) with multiple stems (Osland et al., 2014). The similarity in elevation dynamics between marsh species and A. germinans was attributed to similar structural densities (marsh culm and A. germinans pneumatophore numbers) and root matter accumulation rates (McKee & Vervaeke, 2018). In comparison, riverine mangrove forests in south Florida, including our plots on TTINWR, include additional species that do not form pneumatophores and are often composed of trees that exceed 20 m in height (Odum & McIvor, 1990).
Mangrove forest cover change analysis on TTINWR from 2005 to 2014 indicated a continuation of the trend identified by Krauss et al. (2011) for the previous 78 years; that is, the northern boundary of the mangrove forest moved inland. However, an increase in overall mangrove cover on the refuge resulting from this expansion was not found. Rather, areas of mangrove mortality offset the gains, resulting in a 10-ha reduction in overall cover. Large-scale tree mortality in mangrove forests has been attributed to various factors depending on geographic location, including drought, climate variability, sediment infilling, conversion to aquaculture, and hydrologic modifications that restrict tidal exchange (Blasco et al., 1996; Lewis et al., 2016; Richard & Friess, 2016; Duke et al., 2017; Lovelock et al., 2017; Krauss et al., 2018). Regeneration following tree mortality can be adversely affected by biotic factors, including propagule herbivory by crabs and insects (Clarke & Kerrigan, 2002; Cannicci et al., 2008), and abiotic factors such as hydrology (Lewis, 2005). Although we do not have supporting data, the patchy mangrove loss we identified within established forest stands at TTINWR was likely related to hydrologic changes. Factors that can cause gap mortality within mangrove forests include lightning strikes, impoundment, and intense winds associated with tropical storms (Smith et al., 1994; Doyle et al., 1995; McCoy et al., 1996; Sherman et al., 2000; Cahoon et al., 2003). An intense hurricane that affected TTINWR during the period covered by our mapping effort was Wilma, which made landfall as a Category 3 in October 2005 near Cape Romano, Florida. Following tree mortality, the absence of mangrove root growth often leads to peat collapse and, consequently, increased standing water levels that may prevent mangrove regeneration (Lugo, 1997; Sherman et al., 2000; Cahoon et al., 2003). Mud flats within the mangrove forest on TTINWR and degraded forest stands have been noted in recent years (A. From, personal observation). Higher water levels under the current conditions of increasing rates of SLR may have synergistic effects that can lead to pond formation within mangrove forests on TTINWR, contributing to an overall increase in open water habitats on the refuge. In contrast to stress related to increasing water levels, mangrove encroachment into salt marshes in southeast Australia was linked to drought conditions that led to soil compaction and elevation loss in salt marshes (Rogers et al., 2006).
Woody species encroachment into grasslands leads to increased aboveground primary production in sub-humid regions (Bargar et al., 2011) and higher aboveground carbon storage (Scharenbroch et al., 2010; Pellegrini et al., 2014; Doughty et al., 2016). As noted previously, vegetation shifts such as those documented at TTINWR (Krauss et al., 2011; Howard et al., 2017) also can affect soil characteristics. Global data analyses comparing soils of herbaceous and mangrove-dominated wetlands indicated that soil carbon sequestration rates were similar in salt marshes and mangrove forests, leading to the conclusion that regional or local factors (e.g., precipitation, temperature, hydroperiod) are dominant controls on soil carbon sequestration rates (Chmura et al., 2003; Feher et al., 2017). This conclusion is supported by a study conducted along the northern Gulf of Mexico coast, which indicated that carbon storage in marshes and mangrove forests varied with environmental setting (Yando et al., 2016). At high-precipitation sites in Louisiana, there was little difference in soil properties between the assemblages, including organic matter content (Perry & Mendelssohn, 2009; Henry & Twilley, 2013). At semi-arid, low precipitation sites in Texas, however, soil carbon storage was increased in mangrove forests compared to marshes at some sites (Bianchi et al., 2013; Yando et al., 2016; Guo et al., 2017), but was reduced at others (Comeaux et al., 2012). A recent study in Florida documented an increase in total soil carbon in an area of mangrove encroachment into salt marshes over just three years (Simpson et al., 2019). As noted by Kelleway et al. (2016), studies at the poleward limit of mangroves (e.g., Louisiana, Florida) may reflect the episodic nature of mangrove expansion, where climate-mediated factors can set back expansion at 30-year or shorter intervals. In a region in Australia with a 70-year record of continuous mangrove encroachment belowground carbon storage was increased as mangroves replaced salt marshes (Kelleway et al., 2016).
Conversion of marsh and mangrove habitats to open water clearly leads to a loss in aboveground carbon storage, but effects on belowground carbon sequestration are not so obvious. When vegetation cover and root mass are removed, stored carbon may be eroded and oxidized to CO2 (Mcleod et al., 2011). In contrast, anoxic conditions resulting from permanent flooding may prevent decomposition and thereby maintain long-term carbon storage in soils, and an increase in salinity resulting from sea-level rise in tidal areas may slow the decomposition and carbon mineralization (Mcleod et al., 2011; Luo et al., 2019). Vegetation loss due to ponding will also reduce capacity for coastal storm protection (reviewed in Duarte et al., 2013) and reduce habitat availability for marsh-dependent wildlife (Kelleway et al., 2017). Considering the vegetation shifts occurring on TTINWR, the capacity of the system for aboveground carbon storage will be substantially decreased if marsh loss continues without replacement by mangroves, resulting in conversion to open water.
Conclusions
The differences we identified in VLMw, soil accretion, and subsurface change along the coastal gradient at TTINWR suggest a trajectory of future habitat change on the refuge. Significant shallow subsidence and loss of elevation in interior regions of the refuge (i.e., salt and transition marsh), likely the result of stress from rising sea levels augmented by low allochthonous mineral sediment input, will drive the loss of herbaceous marsh species. Conversion of marsh to mangrove forest habitat, a trend on the refuge over the last several decades, will continue if conditions for mangrove recruitment (e.g., water depth, salinity, propagule predation pressure) remain favorable. Alternately, low mangrove recruitment combined with local hydrologic alterations may drive an increase in pond formation and an overall increase in open water, with a consequent decrease in wetland services provided. It is expected that increased freshwater input to TTINWR following full implementation of the large-scale hydrologic restoration project (Picayune Strand Restoration Project) will enhance the productivity of marsh species, thereby increasing both autochthonous and allochthonous sources for sedimentation and increasing elevation capital. Healthy marsh habitats are expected to reduce the rate of inland encroachment of mangrove forests and the extent of pond formation. Future studies will be required to document if these expectations are realized.
References
Andres, K. D., 2016. Coastal wetland geomorphic and vegetation change: effects of sea-level rise and water management on brackish marshes. M.S. Thesis: Fort Myers, FL, Florida Gulf Coast University: 191 pp.
Andres, K. D., M. Savarese, B. Bovard & M. Parsons, 2019. Coastal wetland geomorphic and vegetation change: effects of sea-level rise and water management on brackish marshes. Estuaries and Coasts 42: 1308–1327.
Anisfeld, S. C., T. D. Hill & D. R. Cahoon, 2016. Elevation dynamics in a restored versus a submerging salt marsh in Long Island Sound. Estuarine, Coastal and Shelf Science 170: 145–154.
Armitage, A. R., W. E. Highfield, S. D. Brody & P. Louchouarn, 2015. The contribution of mangrove expansion to salt marsh loss on the Texas Gulf Coast. PLoS ONE 10: e0125404.
Bargar, N. N., S. R. Archer, J. L. Campbell, C. Huang, J. A. Morton & A. K. Knapp, 2011. Woody plant proliferation in North American drylands: a synthesis of impacts on ecosystem carbon balance. Journal of Geophysical Research 116: G00K07.
Baustian, J. J., I. A. Mendelssohn & M. A. Hester, 2012. Vegetation’s importance in regulating surface elevation in a coastal salt marsh facing elevated rates of sea level rise. Global Change Biology 18: 3377–3382.
Bianchi, T. S., M. A. Allison, J. Zhao, R. S. Comeaux, R. A. Feagin & R. W. Kulawardhana, 2013. Historical reconstruction of mangrove expansion in the Gulf of Mexico: linking climate change with carbon sequestration in coastal wetlands. Estuarine, Coastal and Shelf Science 119: 7–16.
Blasco, F., P. Saenger & E. Janodet, 1996. Mangroves as indicators of coastal change. Catena 27: 167–178.
Booth, A. C., L. E. Soderqvist & M. C. Berry, 2014. Flow monitoring along the western Tamiami trail between County Road 92 and State Road 29 in support of the comprehensive Everglades Restoration Plan, 2007–2010. U.S. Geological Survey Data Series 831, U.S. Geological Survey, Reston, Virginia.
Brown, R. B., E. L. Stone & V. W. Carlisle, 1990. Soils. In Meyers, R. L. & J. J. Ewel (eds), Ecosystems of Florida. University of Central Florida Press, Orlando: 35–69.
Cahoon, D. R., 2015. Estimating relative sea-level rise and submergence potential at a coastal wetland. Estuaries and Coasts 38: 1077–1084.
Cahoon, D. R. & R. E. Turner, 1989. Accretion and canal impacts in a rapidly subsiding wetland II: feldspar marker horizon technique. Estuaries 12: 260–268.
Cahoon, D. R. & J. C. Lynch, 1997. Vertical accretion and shallow subsidence in a mangrove forest of southwestern Florida, USA. Mangroves and Salt Marshes 1: 173–186.
Cahoon, D. R., D. J. Reed & J. W. Day Jr., 1995. Estimating shallow subsidence in microtidal salt marshes of the southeastern United States: Kaye and Barghoorn revisited. Marine Geology 128: 1–9.
Cahoon, D. R., J. R. French, T. Spencer, D. Reed & I. Möhher, 2000. Vertical accretion versus elevational adjustments in UK saltmarshes: an evaluation of alternative methodologies. In Pye, K. & J. R. L. Allen (eds), Coastal and estuarine environments: sedimentology, geomorphology and geoarchaeology. Special Publication 175. The Geographical Society of London, London: 223–238.
Cahoon, D. R., P. Hensel, J. Rybczyk, K. L. McKee, C. E. Proffitt & B. C. Perez, 2003. Mass tree mortality leads to mangrove peat collapse at Bay Islands, Honduras after Hurricane Mitch. Journal of Ecology 91: 1093–1105.
Cahoon, D. R., J. C. Lynch, B. C. Perez, B. Segura, R. D. Holland, C. Stelly, G. Stephenson & P. Hensel, 2002. High-precision measurements of wetland sediment elevation: II. The rod surface elevation table. Journal of Sedimentary Research 72: 734–739.
Cannicci, S., D. Burrows, S. Fratini, T. J. Smith III, J. Offenberg & F. Dahdouh-Guebas, 2008. Faunal impact on vegetation structure and ecosystem function in mangrove forests: a review. Aquatic Botany 89: 186–200.
Cavanaugh, K. C., J. D. Parker, S. C. Cook-Patton, I. C. Feller, A. P. Williams & J. R. Kellner, 2015. Integrating physiological threshold experiments with climate modeling to project mangrove species’ range expansion. Global Change Biology 21: 1928–1938.
Chmura, G. L., S. C. Anisfeld, D. R. Cahoon & J. C. Lynch, 2003. Global carbon sequestration in tidal, saline wetland soils. Global Biogeochemical Cycles 17: 1–12.
Clarke, P. J. & R. A. Kerrigan, 2002. The effects of seed predators on the recruitment of mangroves. Journal of Ecology 90: 728–736.
Coldren, G. A., C. R. Barreto, D. D. Wykoff, E. M. Morrissey, J. A. Langley, I. C. Feller & S. K. Chapman, 2016. Chronic warming stimulates growth of marsh grasses more than mangroves in a coastal wetland ecotone. Ecology 97: 3167–3175.
Comeaux, R. S., M. A. Allison & T. S. Bianchi, 2012. Mangrove expansion in the Gulf of Mexico with climate change: implications for wetland health and resistance to rising sea levels. Estuarine, Coastal and Shelf Science 96: 81–95.
Crosby, S. C., D. F. Sax, M. E. Palmer, H. S. Booth, L. A. Deegan, M. D. Bertness & H. M. Leslie, 2016. Salt marsh persistence is threatened by predicted sea-level rise. Estuarine, Coastal and Shelf Science 181: 93–99.
Dangendorf, S., M. Marcos, G. Wöppelmann, C. P. Conrad, T. Frederikse & R. Riva, 2017. Reassessment of 20th century global mean sea level rise. Proceedings of the National Academy of Sciences 114: 5941–5946.
Day Jr., J. W., L. D. Britsch, S. R. Hawes, G. P. Shaffer, D. J. Reed & D. Cahoon, 2000. Pattern and process of land loss in the Mississippi Delta: a spatial and temporal analysis of wetland habitat change. Estuaries 23: 425–438.
Day, J. W., G. P. Kemp, D. J. Reed, D. R. Cahoon, R. M. Boumans, J. J. Suhayda & R. Gambrell, 2011. Vegetation death and rapid loss of surface elevation in two contrasting Mississippi delta salt marshes: the role of sedimentation, autocompaction and sea-level rise. Ecological Engineering 37: 229–240.
DeLaune, R. D., J. A. Nyman & W. H. Patrick Jr., 1994. Peat collapse, ponding, and wetland loss in a rapidly submerging coastal marsh. Journal of Coastal Research 10: 1021–1030.
Donnelly, M. & L. Walters, 2014. Trapping of Rhizophora mangle propagules by coexisting early successional species. Estuaries and Coasts 37: 1562–1571.
Donoghue, J. F., 2011. Sea level history of the northern Gulf of Mexico coast and sea level rise scenarios for the near future. Climatic Change 107: 17–33.
Doughty, C. L., J. A. Langley, W. S. Walker, I. C. Feller, R. Schaub & S. K. Chapman, 2016. Mangrove range expansion rapidly increases coastal carbon storage. Estuaries and Coasts 39: 385–396.
Doyle, T. W., T. J. Smith III & M. B. Robblee, 1995. Wind damage effects of Hurricane Andrew on mangrove communities along the southwest coast of Florida, USA. Journal of Coastal Research SI 21: 159–169.
Duarte, C. M., I. J. Losada, I. E. Hendriks, I. Mazarrasa & N. Marba, 2013. The role of coastal plant communities for climate change mitigation and adaptation. Nature Climate Change 3: 961–968.
Duever, M. J., J. F. Meeder, L. C. Meeder & J. M. McCollom, 1994. The climate of south Florida and its role in shaping the Everglades ecosystem. In Davis, S. M. & J. C. Ogden (eds), Everglades, the ecosystem and its restoration. St. Lucie Press, Delray Beach: 225–248.
Duke, N. C., J. M. Kovacs, A. D. Griffiths, L. Preece, D. J. E. Hill, P. van Oosterzee, J. Mackenzie, H. S. Morning & D. Burrows, 2017. Large-scale dieback of mangroves in Australia’s Gulf of Carpentaria: a severe ecosystem response, coincidental with an unusually extreme weather event. Marine and Freshwater Research 68: 1816–1829.
Feher, L. C., M. J. Osland, K. T. Griffith, J. B. Grace, R. J. Howard, C. L. Stagg, N. M. Enwright, K. W. Krauss, C. A. Gabler, R. H. Day & K. Rogers, 2017. Linear and nonlinear effects of temperature and precipitation on ecosystem properties in tidal saline wetlands. Ecosphere 8: e01956.
Flower, H., M. Rains & C. Fits, 2017. Visioning the future: scenarios modeling of the Florida coastal Everglades. Environmental Management 60: 989–1009.
Folke, C., S. Carpenter, B. Walker, M. Scheffer, T. Elmqvist, L. Gunderson & C. S. Holling, 2004. Regime shifts, resilience, and biodiversity in ecosystem management. Annual Review of Ecology and Systematics 35: 557–581.
Fraser, L. H. & J. P. Karnezis, 2005. A comparative assessment of seedling survival and biomass accumulation for fourteen wetland plant species grown under minor water depth differences. Wetlands 25: 520–530.
Gabler, C. A., M. J. Osland, J. B. Grace, C. L. Stagg, R. H. Day, S. B. Hartley, N. M. Enwright, A. S. From, M. L. McCoy & J. L. McLeod, 2017. Macroclimate change expected to transform coastal wetland ecosystems this century. Nature Climate Change Letters 7: 142–147.
Guo, H., C. Weaver, S. P. Charles, A. Whitt, S. Dastidar, P. D’Odorico, J. D. Fuentes, J. S. Kominoski, A. R. Armitage & S. C. Pennings, 2017. Coastal regime shifts: rapid response of coastal wetlands to changes in mangrove cover. Ecology 98: 762–772.
Guo, H., Y. Zhang, L. Zhenjiang & S. C. Pennings, 2013. Biotic interactions mediate the expansion of black mangrove (Avicennia germinans) into salt marshes under climate change. Global Change Biology 19: 2765–2774.
Henry, K. M. & R. R. Twilley, 2013. Soil development in a coastal Louisiana wetland during a climate-induced vegetation shift from salt marsh to mangrove. Journal of Coastal Research 29: 1273–1283.
Howard, R. J., K. W. Krauss, N. Cormier, R. H. Day, J. Biagas & L. Allain, 2015. Plant-plant interactions in a subtropical mangrove-to-marsh transition zone: effects of environmental drivers. Journal of Vegetation Science 26: 1198–1211.
Howard, R. J., J. Biagas & L. Allain, 2016. Growth of common brackish marsh macrophytes under altered hydrologic and salinity regimes. Wetlands 36: 11–20.
Howard, R. J., R. H. Day, K. W. Krauss, A. S. From, L. Allain & N. Cormier, 2017. Hydrologic restoration in a dynamic subtropical mangrove-to-marsh ecotone. Restoration Ecology 25: 471–482.
Howard, R. J., A. S. From & L. Allain, 2019. Soil surface elevation dynamics in a mangrove-to-marsh ecotone characterized by vegetation shifts. U.S. Geological Survey data release. https://doi.org/10.5066/P9XZYJ2X.
Howard, R. J. & P. S. Rafferty, 2006. Clonal variation in response to salinity and flooding stress in four marsh macrophytes of the northern Gulf of Mexico, USA. Environmental and Experimental Botany 56: 301–313.
Jowsey, P. C., 1966. An improved peat sampler. New Phytologist 65: 245–248.
Kearny, M. S., R. E. Grace & J. C. Stevenson, 1988. Marsh loss in Nanticoke Estuary, Chesapeake Bay. Geographical Review 78: 205–220.
Kelleway, J. J., K. Cavanaugh, K. Rogers, I. C. Feller, E. Ens, C. Doughty & N. Saintilan, 2017. Review of the ecosystem service implications of mangrove encroachment into salt marshes. Global Change Biology 23: 3967–3983.
Kelleway, J. J., N. Saintilan, P. I. MacReadie, C. G. Skilbeck, A. Zawadzki & P. J. Ralph, 2016. Seventy years of continuous encroachment substantially increases ‘blue carbon’ capacity as mangroves replace intertidal salt marshes. Global Change Biology 22: 1097–1109.
Kirwan, M. L. & J. P. Megonigal, 2013. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504: 53–60.
Kirwan, M. & S. Temmerman, 2009. Coastal marsh response to historical and future sea-level acceleration. Quaternary Science Reviews 28: 1801–1808.
Krauss, K. W., A. S. From, T. W. Doyle, T. J. Doyle & M. J. Barry, 2011. Sea-level rise and landscape change influence mangrove encroachment onto salt marsh in the Ten Thousand Islands region of Florida, USA. Journal of Coastal Conservation 15: 629–638.
Krauss, K. W., A. W. J. Demopoulos, N. Cormier, A. S. From, J. P. McClain-Counts & R. R. Lewis III, 2018. Ghost forests of Marco Island: mangrove mortality driven by belowground soil structural shifts during tidal hydrologic alteration. Coastal, Estuarine and Shelf Science 212: 51–62.
Krauss, K. W., K. L. McKee, C. E. Lovelock, D. R. Cahoon, N. Saintilan, R. Reef & L. Chen, 2014. How mangrove forests adjust to rising sea level. The New Phytologist 202: 19–34.
Lefor, M. W., W. C. Kennard & D. L. Civco, 1987. Relationship of salt-marsh plant distributions to tidal levels in Connecticut, USA. Environmental Management 11: 61–68.
Lamers, L. P. M., L. L. Govers, I. C. J. M. Janssen, J. J. M. Geurts, M. E. W. Van der Welle, M. M. Van Katwijk, T. Van der Heide, J. G. M. Roelofs & A. J. P. Smolders, 2013. Sulfide as a soil phytotoxin – a review. Frontiers in Plant Science. https://doi.org/10.3389/fpls.2013.00268.
Lewis III, R. R., 2005. Ecological engineering for successful management and restoration of mangrove forests. Ecological Engineering 24: 403–418.
Lewis III, R. R., E. C. Milbrandt, B. Brown, K. W. Krauss, A. S. Rovai, J. W. Beever III & L. L. Flynn, 2016. Stress in mangrove forests: early detection and preemptive rehabilitation are essential for future successful worldwide mangrove forests management. Marine Pollution Bulletin 109: 764–771.
Li, S., I. A. Mendelssohn, H. Chen & W. H. Orem, 2009. Does sulphate enrichment promote the expansion of Typha domingensis (cattail) in the Florida Everglades? Freshwater Biology 54: 1909–1923.
Lodge, T. E., 2010. The Everglades Handbook: Understanding the Ecosystem, 3rd ed. CRC Press, Boca Raton.
Lonard, R. I., F. W. Judd & R. Stalter, 2013. The biological flora of coastal dunes and wetlands: Distichlis spicata (C. Linnaeus) E. Greene. Journal of Coastal Research 29: 106–117.
Lovelock, C. E., I. C. Feller, R. Reef, S. Hickey & M. C. Ball, 2017. Mangrove dieback during fluctuating sea levels. Scientific Reports 7: 1680.
Lugo, A. E., 1997. Old-growth mangrove forests in the United States. Conservation Biology 11: 11–20.
Luo, M., J. Huang, W. Zhu & C. Tong, 2019. Impacts of increasing salinity and inundation on rates and pathways of organic carbon mineralization in tidal wetlands: a review. Hydrobiologia 827: 31–49.
Maricle, B. R., D. R. Cobos & C. S. Campbell, 2007. Biophysical and morphological leaf adaptations to drought and salinity in salt marsh grasses. Environmental and Experimental Botany 60: 458–467.
McCoy, E. D., H. R. Mushinsky, D. Johnson & W. E. Meshaka Jr., 1996. Mangrove damage caused by Hurricane Andrew on the southwestern coast of Florida. Bulletin of Marine Science 59: 1–8.
Mcleod, E., G. L. Chmura, S. Bouillion, R. Salm, M. Björk, C. M. Duarte, C. E. Lovelock, W. H. Schlesinger & B. R. Silliman, 2011. A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Frontiers in Ecology and the Environment 9: 552–560.
McKee, K. L., 2011. Biophysical controls on accretion and elevation change in Caribbean mangrove ecosystems. Estuarine, Coastal and Shelf Science 91: 475–483.
McKee, K. L. & J. E. Rooth, 2008. Where temperate meets tropical: multifactorial effects of elevated CO2, nitrogen enrichment, and competition on a mangrove-salt marsh community. Global Change Biology 14: 1–14.
McKee, K. L. & W. C. Vervaeke, 2018. Will fluctuations in salt marsh-mangrove dominance alter vulnerability of a subtropical wetland to sea-level rise? Global Change Biology 24: 1224–1238.
McKee, K. L., D. R. Cahoon & I. C. Feller, 2007a. Caribbean mangroves adjust to rising sea level through biotic controls on change in soil elevation. Global Ecology and Biogeography 16: 545–556.
McKee, K. L., J. E. Rooth & I. C. Feller, 2007b. Mangrove recruitment after forest disturbance is facilitated by herbaceous species in the Caribbean. Ecological Applications 17: 1678–1693.
McKee, K. L., K. Rogers & N. Saintilan, 2012. Response of salt marsh and mangrove wetlands to changes in atmospheric CO2, climate, and sea level. In Middleton, B. A. (ed.), Global Change and the Function and Distribution of Wetlands. Springer, Dordrecht: 63–96.
Meeder, J. F., R. W. Parkinson, P. L. Ruiz & M. S. Ross, 2017. Saltwater encroachment and prediction of future ecosystem response to the Anthropocene Marine Transgression, southeast saline Everglades, Florida. Hydrobiologia 803: 29–48.
Mendelssohn, I. A. & K. L. McKee, 1988. Spartina alterniflora dieback in Louisiana: time-course investigation of soil waterlogging effects. Journal of Ecology 76: 509–521.
Morris, J. T., P. V. Sundareshwar, C. T. Nietch, B. Kjerfve & D. R. Cahoon, 2002. Response of coastal wetlands to rising sea levels. Ecology 83: 2869–2877.
Morton, R. A., J. C. Bernier & J. A. Barras, 2006. Evidence of regional subsidence and associated interior wetland loss induced by hydrocarbon production, Gulf Coast region, USA. Environmental Geology 50: 261–274.
NOAA, 2018a. Tides and Currents, Station Information. https://tidesandcurrents.noaa.gov/stationhome.html?id=8724963. Accessed 27 Sept 2018.
NOAA, 2018b. Tides and Currents, Sea Level Trends. https://tidesandcurrents.noaa.gov/sltrends/sltrends.shtml. Accessed 20 Mar 2018.
Nyman, J. A., R. D. DeLaune, H. H. Roberts & W. H. Patrick Jr., 1993. Relationship between vegetation and soil formation in a rapidly submerging coastal marsh. Marine Ecology Progress Series 96: 269–279.
Nyman, J. A., R. J. Walters, R. D. DeLaune & W. H. Patrick Jr., 2006. Marsh vertical accretion via vegetative growth. Estuarine, Coastal and Shelf Science 69: 370–380.
Odum, W. E. & C. C. McIvor, 1990. Mangroves. In Meyers, R. L. & J. J. Ewel (eds), Ecosystems of Florida. University of Central Florida Press, Gainesville: 517–548.
Osland, M. J., R. H. Day, J. C. Larriviere & A. S. From, 2014. Aboveground allometric models for freeze-affected black mangroves (Avicennia germinans): equations for a climate sensitive mangrove-marsh ecotone. PLoS ONE 9: e99604.
Osland, M. J., K. T. Griffith, J. C. Larriviere, L. C. Feher, D. R. Cahoon, et al., 2017. Assessing coastal wetland vulnerability to sea-level rise along the northern Gulf of Mexico coast: gaps and opportunities for developing a coordinated regional sampling network. PloS ONE 12: e0183431.
Osland, M. J., N. M. Enwright, R. H. Day, C. A. Gabler, C. L. Stagg & J. B. Grace, 2016. Beyond just sea-level rise: considering macroclimate drivers within costal wetland vulnerability assessments to climate change. Global Change Biology 22: 1–11.
Patterson, C. S., I. A. Mendelssohn & E. M. Swenson, 1993. Growth and survival of Avicennia germinans seedlings in a mangle/salt marsh community in Louisiana, USA. Journal of Coastal Research 9: 801–810.
Pellegrini, A. F. A., W. A. Hoffman & A. C. Franco, 2014. Carbon accumulation and nitrogen pool recovery during transitions from savanna to forest in central Brazil. Ecology 95: 342–352.
Perry, C. L. & I. A. Mendelssohn, 2009. Ecosystem effects of expanding populations of Avicennia germinans in a Louisiana salt marsh. Wetlands 29: 396–406.
Peterson, J. M. & S. S. Bell, 2012. Tidal events and salt-marsh structure influence black mangrove (Avicennia germinans) recruitment across and ecotone. Ecology 93: 1648–1658.
Reed, D. J., 1995. The response of coastal marshes to sea-level rise: survival or submergence? Earth Surface Processes and Landforms 20: 39–48.
Reed, D. J., 1999. Response of mineral and organic components of coastal marsh accretion to global climate change. Current Topics in Wetland Biogeochemistry 3: 90–99.
Richard, D. R. & D. A. Friess, 2016. Rates and drivers of mangrove deforestation in Southeast Asia, 2000–2012. Proceedings of the National Academy of Sciences 113: 344–349.
Rogers, K., N. Saintilan & H. Heijnis, 2005. Mangrove encroachment of salt marsh in Western Port Bay, Victoria: the role of sedimentation, subsidence, and sea level rise. Estuaries 28: 551–559.
Rogers, K., K. M. Wilton & N. Saintilan, 2006. Vegetation change and surface elevation dynamics in estuarine wetlands of southeast Australia. Estuarine, Coastal and Shelf Science 66: 559–569.
Rogers, K., N. Saintilan, A. J. Howe & J. F. Rodríguez, 2013. Sedimentation, elevation, and marsh evolution in a southwestern Australian estuary during changing climatic conditions. Estuarine, Coastal and Shelf Science 133: 172–181.
Ross, M. E., J. F. Meeder, J. P. Sah, P. L. Ruiz & G. J. Telesnicki, 2000. The southeast saline Everglades revisited: 50 years of coastal vegetation change. Journal of Vegetation Science 11: 101–112.
Saintilan, N., N. C. Wilson, K. Rogers, A. Rajkaran & K. W. Krauss, 2014. Mangrove expansion and salt marsh decline at mangrove poleward limits. Global Change Biology 20: 147–157.
Sallenger Jr., A. H., K. S. Doran & P. A. Howd, 2012. Hotspots of accelerated sea-level rise on the Atlantic coast of North America. Nature Climate Change 2: 884–888.
Scharenbroch, B. C., M. L. Flores-Mangual, B. Lepore, J. G. Bockheim & B. Lowery, 2010. Tree encroachment impacts carbon dynamics in a sand prairie in Wisconsin. Soil Science Society of America Journal 74(956–96): 8.
Schepers, L., M. Kirwan, G. Guntenspergen & S. Temmerman, 2017. Spatio-temporal development of vegetation die-off in a submerging coastal marsh. Limnology and Oceanography 62: 137–150.
Sherman, R. E., T. J. Fahey & J. J. Battles, 2000. Small-scale disturbance and regeneration dynamics in a neotropical mangrove forest. Journal of Ecology 88: 165–178.
Sherrod, C. L., D. L. Hockaday & C. McMillan, 1986. Survival of red mangrove, Rhizophora mangle, on the Gulf of Mexico coast of Texas. Contributions in Marine Science 29: 27–36.
Shiflet, T. N., 1963. Major ecological factors controlling plant communities in Louisiana marshes. Journal of Range Management 16: 231–235.
Simpson, L. T., T. Z. Osborne, L. J. Duckett & I. C. Feller, 2017. Carbon storage along a climate induced coastal wetland gradient. Wetlands 37: 1023–1035.
Simpson, L. T., C. M. Stein, T. Z. Osborne & I. C. Feller, 2019. Mangroves dramatically increase carbon storage after 3 years of encroachment. Hydrobiologia 834: 13–26.
Smith III, T. J., M. B. Robblee, H. R. Wanless & T. W. Doyle, 1994. Mangroves, hurricanes, and lightning strikes. BioScience 44: 256–262.
Spalding, E. A. & M. W. Hester, 2007. Interactive effects of hydrology and salinity on oligohaline plant species productivity: implications of relative sea-level rise. Estuaries and Coasts 30: 214–225.
Stevens, P. W., S. L. Fox & C. L. Montague, 2006. The interplay between mangroves and saltmarshes at the transition between temperate and subtropical climate in Florida. Wetlands Ecology and Management 14: 435–444.
Stevenson, J. C., M. S. Kearney & E. C. Pendleton, 1985. Sedimentation and erosion in a Chesapeake Bay brackish marsh system. Marine Geology 67: 213–235.
Törnqvist, T. E., D. J. Wallace, J. E. A. Storms, J. Wallinga, R. L. Van Dam, M. Blaauw, M. S. Derksen, C. J. W. Klerks, C. Meijneken & E. M. A. Snijders, 2008. Mississippi Delta subsidence primarily caused by compaction of Holocene strata. Nature Geoscience 1: 173–176.
U.S. Army Corps of Engineers, 2019. Picayune Strand Restoration Project facts and information. https://usace.contentdm.oclc.org/utils/getfile/collection/p16021coll11/id/3143. Accessed 1 Sept 2019.
Yando, E. S., M. J. Osland, J. M. Willis, R. H. Day, K. W. Krauss & M. W. Hester, 2016. Salt marsh-mangrove ecotones: using structural gradients to investigate the effects of woody plant encroachment on plant-soil interactions and ecosystem carbon pools. Journal of Ecology 104: 1020–1031.
Yuill, B., D. Lavoie & D. J. Reed, 2009. Understanding subsidence processes in coastal Louisiana. Journal of Coastal Research SI54: 23–36.
Valle-Levinson, A., A. Dutton & J. B. Martin, 2017. Spatial and temporal variability of sea level rise hot spots over the eastern United States. Geophysical Research Letters. https://doi.org/10.1002/2017GL073926.
Visser, J. M. & E. R. Sandy, 2009. The effects of flooding on four common Louisiana marsh plants. Gulf of Mexico Science 1: 21–29.
Wahl, T., F. M. Calafat & M. E. Luther, 2014. Rapid changes in the seasonal sea level cycle along the US Gulf coast from the late 20th century. Geophysical Research Letters. https://doi.org/10.1002/2013GL058777.
Watson, E. B., H. M. Andrews, A. Fischer, M. Cencer, L. Coiro, S. Kelley & C. Wigand, 2015. Growth and photosynthesis responses of two co-occurring marsh grasses to inundation and varied nutrients. Botany 93: 671–683.
Webb, E. L., D. A. Friess, K. W. Krauss, D. R. Cahoon, G. Guntenspergen & J. Phelps, 2013. A global standard for monitoring coastal wetland vulnerability to accelerated sea-level rise. Nature Climate Change 3: 458–465.
Wilson, K. R., J. T. Kelley, B. R. Tanner & D. F. Belknap, 2010. Probing the origins and stratigraphic signature of salt ponds from north-temperate marshes in Maine, U.S.A. Journal of Coastal Research 26: 1007–1026.
Zhang, Y., G. Huang, W. Wang, L. Chen & G. Lin, 2012. Interactions between mangroves and exotic Spartina in an anthropogenically disturbed estuary in southern China. Ecology 9: 588–597.
Acknowledgements
Funding for this study was provided by the U.S. Fish and Wildlife Service (Intragovernmental Agreements 4500035235, 4500081468) and the U.S. Geological Survey Ecosystems Mission Area. We thank Kevin Godsea, Wade Gurley, and Mark Danaher, U.S. Fish and Wildlife Service, for logistical and technical support. Darren Johnson, Cherokee Nation Technologies, Wetland and Aquatic Research Center, provided data analyses. Comments provided by Donald Cahoon and anonymous reviewers helped to improve the manuscript. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. The data are available at https://doi.org/10.5066/P9XZYJ2X (Howard et al., 2019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Iacopo Bertocci
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Howard, R.J., From, A.S., Krauss, K.W. et al. Soil surface elevation dynamics in a mangrove-to-marsh ecotone characterized by vegetation shifts. Hydrobiologia 847, 1087–1106 (2020). https://doi.org/10.1007/s10750-019-04170-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-04170-4