Abstract
Temporary pools in semiarid regions are influenced by climatic seasonality and the extremes of moisture/desiccation effects influence mechanisms for plant survival, which should be analyzed based on competitor (C), ruderal (R), or stress tolerator (S) strategies. The aim of the present study was to evaluate how aridity exerts an influence on CSR functional groups of aquatic plants in temporary pools. Aquatic plants were sampled from 27 temporary pools in areas with a climatic gradient of evaporation, precipitation, and temperature. The distribution of functional groups along the environmental gradient was analyzed through redundancy analysis and patterns of CSR strategies were analyzed using linear correlation. We found that assemblages of macrophyte ponds were composed mainly of intermediate R/CR strategies. Evaporation exerted a positive influence on the occurrence of species with larger specific leaf area and a negative influence on the occurrence of taller species. Moreover, ponds in regions with greater evaporation had greater species richness (r2 = 0.2, P < 0.01). The same was found for competitors (r2 = 0.2, P < 0.01) but not for ruderal or tolerant classes. In conclusion, climatic conditions influence CSR classes and exert a specific influence on the specific functional traits of species in temporary pools of semiarid regions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The morfo-physiological components of organisms are associated with environmental variations and the performance of individuals and these integrative processes reverberate on higher organizational levels, such as plant communities (Violle et al., 2007). The intrinsic characteristics or “functional traits” of organisms have been used in studies on the ecology of restricted ecosystems (Wiens et al., 2010) as well as studies addressing higher scales, such as plant geography (Swenson & Weiser, 2010), where variations within and between different functional groups could be used for the estimation of parameter values in vegetation models.
The most commonly analyzed functional groups of plant species were classified by Grime (1977, 2001), who considered selection processes of adaptive characteristics based on competitor (C), ruderal (R), and stress tolerator (S) strategies. With this classification, competitive species tend to have a long lifecycle, high vegetative biomass and low investment in reproduction, predominating in environments with low degrees of disturbance and stress; ruderal species have a short lifecycle and high investment in reproduction and are related to environments with high degrees of disturbance and stress; and species tolerant to stress are characterized by a long lifecycle and low vegetative biomass, occurring more often in environments with a high degree of stress, but low disturbance (Grime & Pierce, 2012).
The CSR model applied to lentic systems under the influence of a temperate climate has revealed a relationship between the limiting factors of these systems (depth and productivity) and the selection of specific functional groups, as such environments are also conditioned to variables influenced by the period of greater precipitation, which leads to an increase in the water column in spring (Leira & Cantonati, 2008). As a predictive model of functional groups in assemblages in permanent lentic environments located in temperate regions, the CSR concept demonstrates the predominance of species with submersion tolerance, which are typically S strategists (Brock et al., 1987; Riis & Hawes, 2002). In shallow lakes, where the depth gradient is not a limiting factor, functional groups are influenced by the trophic pattern of the system, as species with intensive vegetative reproduction and competitive traits predominate in environments with high productivity and disturbance due to anthropogenic desiccation, whereas species with less biomass and greater investment in sexual reproduction, which are characteristics of ruderal species, predominate in systems with limited nutrients and low productivity (Arthaud et al., 2012). However, other systems have different hydrodynamic patterns with accentuated seasonal changes in the water column, such as temporary pools in the semiarid region of northeast Brazil, where assemblages of aquatic plants are structured over time and space by the simultaneous action of environmental filters and interspecific competition (Ferreira et al., 2015) and where the dominant aquatic plant species have complementary life growth forms to enable their co-existence (Tabosa et al., 2012).
Climatic variables play a fundamental role in the dynamics of the assemblages of temporary aquatic environments (Pedro et al., 2006) due to the direct relationship between precipitation and the depth of the water column as well as between temperature and evaporation, which determine the length of the existence of temporary pools (Murray-Hudson et al., 2014). Moreover, the magnitude of the response of vegetation to rainfall patterns increases with the increase in environmental aridity (Djebou et al., 2015), such that seasonal climatic conditions tend to influence the intensity of stress and disturbance in these temporary pools.
As the intensity of stress and disturbance in these temporary pools may be related to the aridity gradient of the semiarid region, the selection of functional groups occurs in plant assemblages in such a way as to favor fitness and, consequently, frequencies of occurrence in pools with increasing environmental severity and a longer drought period. Thus, in the present study, we test the hypothesis that environmental severity (represented by climatic conditions in a semiarid region) influences CSR functional groups in assemblages of aquatic plants in temporary pools. Several conditions (e.g., high temperature and evaporation) tend to select species with ruderal strategies due to the desiccation of the habitat and the consequent loss of biomass during the dry season. Specifically, a greater proportion of species with ruderal-stress tolerator (RS) strategy traits is expected in environments with more severe climatic variables due to the shorter length of time in which the aquatic habitat remains in existence.
Materials and methods
Study area
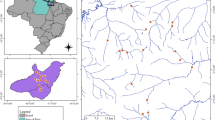
The temporary pools analyzed were located in the State of Ceará in northeast Brazil between latitudes 3°27′ and 3°45′S and longitudes 39°17′ and 40°22′W (Fig. 1). The region has a warm semiarid climate (BSh in the Köppen-Geiger classification system) (Peel et al., 2007), characterized by a mean temperature of 24 to 28°C and mean precipitation of 916.5 mm (Fig. S1, Electronic Supplementary Material 1), with irregular rainfall between January and June (Alvares et al., 2014). The soil in the region is superficial and has low permeability, which leads to the appearance of temporary pools in depressions in the relief due to the accumulation of rainfall in the wet season, followed by complete desiccation in the dry season.
The total area covered approximately 14,000 km2, spanning from coastal formations to the semiarid region in the interior of the State of Ceará, Brazil. The temporary pools located in the coastal region are surrounded by a complex of arboreal-bush vegetation that developed on terrains of the Tertiary (Barreiras Formation) and border the semiarid Caatinga Phytogeographical Domain (Ab’Sáber, 2003). The temporary pools located in the interior of the state occur among the ‘Caatinga’ vegetation, which refers to a seasonally dry tropical forest (Silva et al., 2017). The Caatinga vegetation develops on a crystalline complex of Precambrian origin, Proterozoic eon, which includes intracratonic sedimentary basins with deposits of Cretacean sediments, constituting a predominantly flat relief (altitude: 300 to 500 m) (Andrade, 1968). These landscapes are influenced by the orographic effects of residual reliefs at an altitude of approximately 900 m that exert an influence on the differentiated distribution of precipitation and evaporation (Souza & Oliveira, 2006).
Sampling design
Assemblages of aquatic plants were sampled in 27 temporary pools (Fig. 1). The sampling units were chosen based on water surface (two to four hectares) and depth (one to two meters). Climate data for each sampling point were obtained from the WorldClim databank (Hijmans et al., 2005) using DIVA-GIS (Hijmans, 2001) with a precision of 2.5 min (~ 5 km), which we used 19 climatic variables (Annual Mean Temperature, Mean Monthly Temperature Range, Isothermality, Temperature Seasonality, Max Temperature of Warmest Month, Min Temperature of Coldest Month, Temperature Annual Range, Mean Temperature of Wettest Quarter, Mean Temperature of Driest Quarter, Mean Temperature of Warmest Quarter, Mean Temperature of Coldest Quarter, Annual Precipitation, Precipitation of Wettest Month, Precipitation of Driest Month, Precipitation Seasonality, Precipitation of Wettest Quarter, Precipitation of Driest Quarter, Precipitation of Warmest Quarter, Precipitation of Coldest Quarter). To this set of variables, we added data on Evaporation, which were extracted from the climate databank of the government of the State of Ceará (Funceme, 2017). Due to collinearity, we selected only Evaporation, Annual Precipitation, and Annual Mean Temperature (Table S1, Electronic Supplementary Material 1).
Sampling was performed only once per site during the wet season between May and July 2017, which was a year with less than average rainfall (Fig. S1, Electronic Supplementary Material 1). We used the line-intercept method (Canfield, 1941) adapted to sampling only the organisms intercepted by the lines. A total of ten parallel lines perpendicular to margin were determined in each temporary pool. The lines covered the entire extension of the water surface and separated by a distance of 2 m. We used the concept put forth by Sculthorpe (1967), who defines hydrophytes as vascular herbaceous plants with growth in aquatic environments or inundated soils, and we characterized them by the following growth forms: (a) free-floating hydrophytes and (b) hydrophytes rooted to the substrate, which are categorized as emergent, submerged or plants only with floating leaves.
The samples were processed using traditional conservation methods described by Bridson & Forman (1998), identified and deposited at the EAC Herbarium. The taxonomic identification was performed using the specialized literature (Cook, 1996). The taxonomic terminology and abbreviations of authors’ names are in accordance with the International Plant Names Index (IPNI, 2018).
Traits were selected due to their relationship to life history, which is evidenced by the establishment capacity of individuals in aquatic plant assemblages (Pierce et al., 2013; Fu et al., 2015), and the relationship to the action of stress and disturbance factors in the habitat or CSR strategy (Grime, 1977) (Table 1). Specimens with signs of herbivory, parasitism or partial deterioration were not included in this analysis. A total of 313 individuals were intercepted by the lines in the 27 temporary pools sampled. Thus, the measures of functional traits were obtained from this quantity of organisms. The specimens were placed in plastic bags with water and transported to the laboratory under refrigeration at approximately 4°C. In the laboratory, the samples were submitted to the following: (a) determination of plant height, calculated by the distance from the base of the stem or petiole (in rhizomatous plants) to the top of the photosynthetic tissues using a metric tape; (b) determination of plant diameter, measured at the base of the stem or petioles (maximum of 2 cm above rhizome) using calipers; and (c) count of the number of leaves and flowers on the individual (Fu et al., 2012). Leaf analysis was performed based on Pérez-Harguindeguy et al. (2013): (Electronic Supplementary Material 2): leaf fresh weight, photosynthesizing leaf area, specific leaf area, leaf dry weight, leaf dry matter content, and total biomass of the individual.
Statistical analysis
The climate variables and functional traits were submitted to the variance inflation factor (VIF) test for the exclusion of multicollinear variables (Zuur et al., 2009), which were performed with the aid of the R program analyse VIF in the Regclass package (R Core Team, 2019); and variables with values > 3 were not considered in subsequent analyses. The results of the VIF test for functional traits indicated that plant height was collinear with biomass, leaf area, leaf fresh weight, leaf dry weight and plant diameter (Table S2) and all of these traits were related to the characteristics of competitor species. The non-collinear climate variables were: Annual Precipitation (BIO12), Evaporation (Evap) and Annual Mean Temperature (BIO1) (Table S3). These variables were submitted to non-metric multidimensional scaling (NMDS) analysis to determine whether the distribution pattern of the points was related to the climate gradient. As NMDS orders can only be interpreted when the stress values are lower than 0.2 (Quinn & Keough, 2002). The non-collinear functional traits were plant height, number of leaves, number of flowers, specific leaf area and leaf dry matter content (Table S2, Electronic Supplementary Material 1).
To determine the proportion of species according to characteristics and CSR strategies, principal component analysis was performed for the relationship between the functional traits selected and the species using the values of the functional traits standardized as mean zero and one standard deviation (z-score). Relationships between functional traits and climate variables were investigated using canonical redundancy analysis (RDA). This procedure is commonly used to reveal changes in the mean expression of characteristics in communities as a function of climate factors (Kleyer et al., 2012). A data matrix was created from the community-weighted mean (CWM) of the functional trait values of the species per sampling point (Garnier et al., 2007). We used the CWM restricted by environmental variables in the RDA and forward selection to discriminate the climate variables that exerted the greatest influence on the distribution of the functional traits. These analyses were performed with the aid of the R software in the vegan package (R Core Team, 2019).
The analysis of the relative proportion of species per CSR strategy was performed using a StrateFy matrix provided by Pierce et al. (2017). For this analysis, we used leaf area, leaf dry matter content and specific leaf area, despite the collinearity among these variables. The same test was used in the analysis of the relative proportion of the assemblages for each sampling point to determine the existence of variation in the percentage of CSR strategies as a function of climatic factors. This test furnishes the percentage of each CSR strategy and classifies the strategy based on predominance. We then performed linear regression analyses between each adaptive CSR and the number of species on Axis 1 of the RDA, which summarizes linear relationships between components of response climate variables that are explained by a set of explanatory trait variables. Thus, the regression analyses demonstrated the influence of climate factors on patterns of adaptive strategies in the assemblages.
Results
Overall, 17 species were collected from the 27 temporary pools, all of which belonged to variations of ruderal strategies (Table 2), with a predominance of typical characteristics of the ruderal (R, lines 1–9, column 5) and ruderal/competitor-ruderal strategies (R/CR, lines 10–14, column 5). However, species belonging to the same strategy had different growth forms (Table 2). In the R strategy class, there was a predominance of emergent species together with species with floating leaves and free-floating species (Table 2, lines 1–9, column 4). In the R/CR strategy, there was a predominance of emergent species with floating leaves and only one emergent species (Table 2, lines 10–14, column 4). The other categories were represented by only one growth form (Table 2, lines 15–17, column 4).
Considering the percentages of species based on patterns of CSR strategies, the majority of assemblages in the semiarid region had a predominance of the R/CR strategy over the R and ruderal/competitor-ruderal-stress tolerator (R/CSR) strategies, with no occurrences of assemblages in the ruderal/ruderal-stress tolerator class (Table 3). Considering the individual percentages of the CSR strategies, there was a predominance of R and C strategies in all assemblages, whereas only four assemblages (sampling points 21, 9, 15, and 16) had the stress tolerator strategy (S) in their composition (Table 3).
The NMDS analysis resulted in a one-dimensional solution for Axis 1 (Fig. 2), the stress of which was 0.1399 and R2 was 0.946. This analysis revealed the organization of sampling points on an increasing climatic gradient, indicating the occurrence of a gradient ranging from points with less rainfall and greater evaporation (sampling points 1–9, Table S1) to points with greater rainfall and less evaporation (sampling points 20–27, Table S1) (Fig. 2).
The PCA explained 59.9% of the variation in the functional traits of the species. Axis 1 (explained 33.6%) was positively related with specific leaf area and number of flowers and a negatively related with leaf dry matter content. On the other hand, Axis 2 was positively related with plant height and number of leaves (Fig. 3). Plant height was related to competitive interaction traits (Table 1) and the PCA related larger values of this variable species of the genus Nymphaea (Fig. 3). The species Hydrocleys martii Seub. and Nymphoides humboldtiana (Kunth) Kuntze had the highest specific leaf area values, which were related to ruderal traits, and species of the genera Eleocharis and Neptunia had the highest leaf dry matter content, which represents a stress tolerator trait (Fig. 3).
Principal component analysis (PCA) between functional traits and species data. HP height plant, LN number of leaves, FN number of flowers, SLA specific leaf area, LDMC leaf dry matter contents, Eacut Eleocharis acutangula (Roxb.) Schult., Ediv Eichhornia diversifolia (Vahl) Urb., Ehet Eichhornia heterosperma Alexander, Eint Eleocharis interstincta (Vahl) Roem. & Schult., Emut Eleocharis mutata (L.) Roem. & Schult., Epan Eichhornia paniculata (Spreng.) Solms., Esub Echinodorus subalatus (Mart.) Griseb., Hmar Hydrocleys martii Seub., Lhel Ludwigia helminthorrhiza (Mart.) H.Hara., Nama Nymphaea amazonum Mart. & Zucc., Nhum Nymphoides humboldtiana (Kunth) Kuntze, Nlasi Nymphaea lasiophylla Mart. & Zucc., Nlin Nymphaea lingulata Wiersema, Nole Neptunia oleracea Lour., Npul Nymphaea pulchella DC., Pcor Pontederia cordata var. ovalis (Mart.) Solms, Pstr Pistia stratiotes L
The RDA revealed the influence of climate variables (evaporation, rainfall and temperature) on the distribution of the weighted functional traits per sampling point (CWM) (F = 2.19, P = 0.01) (Fig. 4). The first axis of the RDA explained 81.5% of the variation in the data, with the influence of rainfall on plant height and number of leaves as well as the influence of evaporation and temperature on specific leaf area and leaf dry matter content. The second axis of the RDA explained 17.4% of the variation in the data, with the influence of rainfall on number of leaves and plant height (Fig. 4). The result of the forward selection process among the climate variables revealed that evaporation exerted the greatest influence on the structuring of the functional traits in the assemblages (R2adj = 0.14; F = 5.57; P = 0.001).
Redundancy analysis plot based on relationship between percentages of functional traits of each assembly located at sampling points and climate variables. EVAP evaporation, PREC precipitation, TEMP average annual temperature, HP plant height, LN number of leaves, FN number of flowers, SLA specific leaf area, LDMC leaf dry matter content
We found a negative association between the number of species and Axis 1 of the RDA (Fig. 5a), indicating the occurrence of a greater number of species in environments with less rainfall and greater evaporation. Moreover, we found a positive association between the percentage of competitive strategies and Axis 1 of the RDA, revealing that competitive strategies tend to occur at sites with greater rainfall and less evaporation (Fig. 5b). No association was found between Axis 1 of the RDA and stress tolerator (Fig. 5c) or ruderal (Fig. 5d) strategies.
Discussion
The main purpose of the present study was to investigate the importance of climatic conditions to explaining the functional structure of the macrophyte assemblage in a semiarid region. We found that evaporation rates greatly structured CSR strategies and the composition of functional traits. Evaporation constitutes a considerable disturbance in the region, as affects the amount of water in temporary pools (Sánchez-Carrillo et al., 2004). The importance of this type of disturbance was demonstrated by the clear homogeneity of species belonging to R/CR classes, indicating that intermittence in pools tends to lead to convergent functional strategies in the semiarid region. Indeed, climatic severity favors assemblages composed of species with high adaptation to regional conditions (Rodrigues-Filho et al., 2018a). This results in distribution of functional traits with strongly specific responses along the disturbance intensity gradient. Locations with high climate severity tend to have species with considerable similarity in terms of functional attributes, which somewhat intensifies agonistic interactions (e.g., competition) (Larned et al., 2010). This suggests that climate severity may also influence the local functional structuring of macrophyte assemblages. For example, the climatic gradient favored a stratified phytofacies as a form of spatial occupation of the small water body in such a way as to avoid the effects of overlap and competition in local assemblages (Tabosa et al., 2012). Competitor species (C-strategy) were more representative in lakes with lower evaporation rates and higher precipitation. Thus, our findings within and between assemblages suggest that macrophyte communities in semiarid regions are structured by regional disturbances (direct effect; predominance of ruderal strategies), which, in turn, exerts an influence on local competition for resources (indirect effect; changes in evaporation act mainly by selecting functional traits related to strong competitive ability). The present results are in agreement with data described in previous studies reporting that regional factors are important to understanding the structure of biological assemblages (Whittaker et al., 2001; Ricklefs, 2006; Ernst et al., 2013; Alahuhta, 2015; Alahuhta et al., 2016; Rodrigues-Filho et al., 2018b).
Despite the predominance of species of the R/CR classes, the present results indicate that changes in climatic conditions are capable of generating a functional continuum, with the substitution of species with competitor strategies. This reveals a fine adaptation of macrophyte species to climate variation, which promotes strong environmental selection in ponds in semiarid environments (Ferreira et al., 2015). For example, ponds in regions with greater evaporation rates have a shorter permanence time, which hinders the investment of species in costly structures, such as the number of leaves (Camacho et al., 2016). In contrast, ponds in regions with greater precipitation have a longer permanence time and less selection from the loss of habitat due to evaporation, enabling species to increase their investment in structures that confer greater competitive capacity (e.g., greater size and number of leaves). Thus, while ruderality is a general adaptation to the semiarid climate, changes in the balance between evaporation and precipitation mainly contribute to the selection of functional attributes related to competitor species (C). Indeed, we found that the increase in precipitation favors assemblages with greater species richness, corresponding to a greater frequency of attributes related to competition but not ruderality (Fig. 5). This intricate relation between the regional influence (favoring ruderality) and the local influence (selecting competitor attributes) of climate conditions is explained further in the next section.
How do climatic conditions shape the functional structure of local assemblages of pond macrophytes?
The consequence disturbance operating at the broadest scale in local assemblage favors the colonization and permanence of species with taller plants with a larger number of flowers and leaves, which permit the colonization and permanence in environments for a longer period and reflects the R/CR pattern found in the assemblages analyzed. Plant height is related to an investment in vertical growth, favoring competition for light (Fu et al., 2015) and the stratified occupation of the water column, which is a formation pattern of aquatic plant communities in the semiarid (Tabosa et al., 2012). Temporary pools in locations with less precipitation and greater evaporation had assemblages with ruderal strategy, with a greater specific leaf area and smaller number of leaves per specimen. This reveals less investment in the number of leaves without compromising the efficiency in capturing light, which characterizes plants with a short lifecycle in environments with a shorter existence (Pierce et al., 2013; Camacho et al., 2016). This can be observed more specifically for species of the family Pontederiaceae. These species exhibited smaller internodes; characteristic of ruderal strategies in semiarid regions due to the little, water availability and shorter existence of these environments. Another result related to the environmental influence occurred in groups of Eudicotiledons in a desiccation experiment that demonstrated phenotypic adjustments in comparison to a control group, with an increase in dry matter content (Wilde et al., 2014). Thus, the present findings on the influence of evaporation and the investment in specific leaf area lend support to the hypothesis of the adaptation of herbaceous species in arid environments related to investment in growth cycles and rapid reproduction along the aridity gradient (Ottaviani & Keppel, 2017).
The increase in the number of species was not related to predominance of stress tolerant and ruderal strategies, as no direct relationship was found between these separate strategies and the environmental gradient. Despite being counterintuitive, this pattern has been reported in other studies (e.g., Chappuis et al., 2014) and may derive from two possible explanations: (1) high local richness values or (2) low local richness values accompanied by high turnover in species composition. The second explanation sounds more parsimonious. In addition to the homogeneity of ruderal strategies, we may infer that groups of species with such strategies exhibit a large variety of functional combinations in semiarid regions (Nicolet et al., 2004). Moreover, this differentiation of strategies may favor lower competition rates in local assemblages. An example of populations with the profile of intermediate characteristics between ruderal and competitive strategies was found in species of the genus Nymphaea (Table 2), which, unlike data reported in the literature, seemed to be related to the competitor strategy due to the exclusive investment in leaves, which enables the occupation of the water surface, with higher cover indices during the wet season (Tabosa et al., 2012). Likewise, species of the genera Eleocharis and Neptunia (Table 2) are recognized by their characteristics of resistance to the desiccation of the environment (Fu et al., 2012; Deng et al., 2017), but these populations demonstrated characteristics of intermediate strategies between stress tolerator and ruderal in the semiarid region. Thus, although each species invests in adaptive strategies that are intrinsic to its fitness, an influence of the environment on functional traits was found, implying the selection of different survival strategies for the intermittent pools of the semiarid region. Furthermore, there was variation in the growth forms of these classes (Table 2). These assemblages are structured by species with complementary growth forms that compose a stratified phytofacies as a form of spatial occupation of the small water body in such a way as to avoid the effects of overlap and competition, as reported by Tabosa et al. (2012). Thus, the variation in growth forms within the same strategy class may be related to this tendency toward the structuring of assemblages due to spatial competition.
The competitor traits in temporary pools with less disturbance reveals the influence of the inter-population interaction and dominance relationships during the occupation of the aquatic environment. This explains the occurrence of intermediate strategies (R/CR) in the assemblages. Rosado & de Mattos (2017) report a similar finding on sandy coastal plains, in which plant assemblages exhibited a predominance of intermediate stress tolerant/competitor-stress tolerant (S/CS) strategies, indicating that survival characteristics and the inter-population relationship are common among species that occur in disturbed systems. Indeed, these findings suggest that good competitors were generally dominant in pools with less disturbance, thereby supporting the original theory put forth by Grime et al., (1997). Another finding that underscores the importance of intermediate strategies in these environments was the occurrence of species with a smaller number of competitive characteristics in environments with a short period of water column establishment. Analyzing the influence of the proportion of competitive characteristics along the gradient, sites with greater precipitation had assemblages with characteristics related to competition. This occurred due to the fact that the water column in these environments had a longer period of existence, which enabled more time for establishment and investment in vegetative growth for the spatial occupation phenomenon commonly found in permanent shallow lakes in temperate or tropical regions (Fu et al., 2014; Ecke et al., 2016) and competition (Michalet et al., 2015). Thus, the structuring of the local assemblage in wet temporary environments may be related to the biotic process of competition in the Brazilian semiarid region.
What can we learn from assemblages of pond macrophytes in a semiarid region?
Interestingly, the analysis of the direct effect explained more than 80% of the functional structure of the macrophyte assemblages. This percentage differs from data reported in numerous studies conducted on the same taxonomic group, but in tropical and temperate regions (Bini et al., 1999; Capers et al., 2010; Chappuis et al., 2014; Alahuhta, 2015). In general, these studies explain approximately 20% of the structure of assemblages, even when considering local processes (e.g., local environmental conditions). This suggests that assemblages in semiarid regions may be easier to predict based on climatic conditions that generate homogeneity with regards to functional strategies (e.g., ruderal). Taking such predictability into consideration together with the accentuated climate change expected for the upcoming years, assemblages in semiarid regions may be good models for predicting how future climate change will affect the functional structure of local assemblages. It is noteworthy that studies conducted in regions with disturbances of low intensity predict similar patterns to those found in the present investigation (Rosset et al., 2010; Alahuhta et al., 2011; Netten et al., 2011). Thus, projecting how future climate change will affect assemblages in semiarid regions may help us project changes on more distant temporal scales.
Conclusion
In conclusion, high levels of evaporation and high temperatures promote the predominance of species with ruderal strategies and competition potentially generates high trait variability in temporary pools. These responses were strongly predicted for semiarid regions, suggesting that assemblages located in regions with climatic severity tend to present high functional redundancy of CSR strategies, but considerable morphological variation to avoid competition. We also conclude that the method of analysis of leaf traits enabled the determination of strategies along environmental gradients, lending support to the application of CSR theory to the study of aquatic plant assemblages.
References
Ab’Sáber, A. N., 2003. Os Domínios de Natureza no Brasil: Potencialidades Paisagísticas. Ateliê Editorial, São Paulo.
Alahuhta, J., 2015. Geographic patterns of lake macrophyte communities and species richness at regional scale. Journal of Vegetation Science 26: 564–575.
Alahuhta, J., J. Heino & M. Luoto, 2011. Climate change and the future distributions of aquatic macrophytes across boreal catchments. Journal of Biogeography 38: 383–393.
Alahuhta, J., S. Hellsten, M. Kuoppala & J. Riihimäki, 2016. Regional and local determinants of macrophyte community compositions in high-latitude lakes of Finland. Hydrobiologia 812: 99–114.
Alvares, C. A., J. L. Stape, P. C. Sentelhas, J. L. M. Gonçalves & G. Sparovek, 2014. Koppen’s climate classification map for Brazil. Meteorologische Zeitschrift 22: 711–728.
Andrade, G. O., 1968. Gênese do relevo nordestino: estado atual dos conhecimentos. Estudos Universitários 3: 1.
Arthaud, F., D. Vallod, J. Robin & G. Bornette, 2012. Eutrophication and drought disturbance shape functional diversity and life-history traits of aquatic plants in shallow lakes. Aquatic Science 74: 471–481.
Bini, L. M., S. M. Thomaz, K. J. Murphy & A. F. M. Camargo, 1999. Aquatic macrophytedistribution in relation to water and sediment conditions in the Itaipu Reservoir, Brazil. Hydrobiologia 415: 147–154.
Bridson, D. & L. Forman, 1998. The Herbarium Handbook, 3rd ed. Whitstable Litho Printers, Great Brain.
Brock, T. C. M., G. Van der Velde & H. M. Van de Steeg, 1987. The effects of extreme water level fluctuations on the wetland vegetation of a Nymphaeid-dominated oxbow lake in the Netherlands. Archiv fur Hydrobiologie 27: 57–73.
Camacho, A., N. Murueta, E. Blasco, A. C. Santamans & A. Picazo, 2016. Hydrology-driven macrophyte dynamics determines the ecological functioning of a model Mediterranean temporary lake. Hydrobiologia 774: 93–107.
Canfield, R., 1941. Application of the line interception method in sample range vegetation. Journal of Forestry 39: 322–331.
Capers, R. S., R. Selsky & G. J. Bugbee, 2010. The relative importance of local conditions and regional processes in structuring aquatic plant communities. Freshwater Biology 55: 952–966.
Chappuis, E., E. Gacia & E. Ballesteros, 2014. Environmental factors explaining the distribution and diversity of vascular aquatic macrophytes in a highly heterogeneous mediterranean region. Aquatic Botany 113: 72–82.
Cook, C. D. K., 1996. Aquatic Plant Book. SPB Academic Publishers, Amsterdam.
Deng, Z., Y. Xie, X. Chen, F. Li, C. Hu & N. Liu, 2017. Effect of clone size on submergence tolerance and post-submergence growth recovery in Carex brevicuspis (Cyperaceae). Journal of Limnology 76: 424–430.
Djebou, D. C. S., D. C. Singh & O. W. Frauenfeld, 2015. Vegetation response to precipitation across the aridity gradient of the southwestern United States. Journal Arid Environments 115: 35–43.
Ecke, F., S. Hellsten, J. Köhler, A. W. Lorenz, J. Rääpysjärvi, S. Scheunig, J. Segersten & A. Baattrup-Pedersen, 2016. The response of hydrophyte growth forms and plant strategies to river restoration. Hydrobiologia 769: 41–54.
Ernst, R. E., E. Pereira, M. A. Hamilton, S. A. Pisarevsky, J. Rodrigues, C. C. G. Tassinari, W. Teixeira & V. Van-Dunem, 2013. Mesoproterozoic intraplate magmatic ‘barcode’ record of the Angola portion of the Congo Craton: newly dated magmatic events at 1505 and 1110 Ma and implication for Nuna (Columbia) supercontinent reconstructions. Precambrian Research 230: 103–118.
Ferreira, F. S., A. B. Tabosa, R. B. Gomes, F. R. Martins & L. Q. Matias, 2015. Spatiotemporal ecological drivers of an aquatic plant community in a temporary tropical pool. Journal of Arid Environments 115: 66–72.
Fu, P. L., Y. J. Jiang, A. Y. Wang, T. J. Brodribb, J. L. Zhang, S. D. Zhu & K. F. Cao, 2012. Stem hydraulic traits and leaf water-stress tolerance are co-ordinated with the leaf phenology of angiosperm trees in an Asian tropical dry karst forest. Annals of Botany 110: 189–199.
Fu, H., J. Zhong, G. L. Yuan, P. Xie & T. Cao, 2014. Functional traits composition predict macrophytes community productivity along a water depth gradient in a freshwater lake. Ecology and Evolution 4: 1516–1523.
Fu, H., J. Zhong, G. Yuan, C. Guo & Q. Lou, 2015. Predicting changes in macrophyte community structure from functional traits in a freshwater lake: a test of maximum entropy model. PLoS ONE 10: e0131630.
FUNCEME—Fundação Cearense de Meteorologia e Recursos Hídricos, 2017. Índice de aridez para o Ceará, Fortaleza, Brazil. http://www.funceme.br/index.php/areas/17-mapas-tematicos/542-indice-de-aridez-para-o-ceara.
Garnier, E., S. Lavorel, P. Ansquer, H. Castro, P. Cruz & J. Dolezal, 2007. Assessing the effects of land-use change on plant traits, communities and ecosystem functioning in grasslands: a standardized methodology and lessons from an application to 11 European sites. Annals of Botany 99: 967–985.
Grime, J. P., 1977. Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. The American Naturalist 111: 1169–1194.
Grime, J. P., 2001. Plant Strategies, Vegetation Processes and Ecosystem Properties, 2nd ed. Wiley, Chichester.
Grime, J. P. & S. Pierce, 2012. The Evolutionary Strategies that Shape Ecosystems. Wiley-Blackwell, Chichester.
Grime, J. P., K. Thompson, R. Hunt, J. G. Hodgson, J. H. C. Cornelissen, I. H. Rorison, G. A. F. Hendry, T. W. Ashendenl, A. P. Askew, S. R. Band, R. E. Booth, C. C. Bossard, B. D. Campbell, J. E. L. Cooper, A. W. Davison, P. L. Gupta, W. Hall, D. W. Hand, M. A. Hannah, S. H. Hillier, D. J. Hodkinson, A. Jalili, Z. Liu, J. M. L. Mackey, N. Matthews, M. A. Mowforth, A. M. Neal, R. J. Reader, K. Reiling, W. Ross-Fraser, R. E. Spencer, F. Sutton, D. E. Tasker, P. C. Thorpe & J. Whitehouse, 1997. Integrated screening validates primary axes of specialization in plants. Oikos 79: 259–281.
Hijmans, R. J., 2001. Computer tools for spatial analysis of plant genetic resources data: 1. DIVA-GIS. Plant Genetic Resources Newsletter 127: 15–19.
Hijmans, R. J., S. E. Cameron, J. L. Parra, P. G. Jones & A. Jarvis, 2005. Superposições climáticas interpoladas de alta resolução para áreas terrestres globais. International Journal of Climatology 25: 1965–1978.
IPNI, 2018. The International Plant Names Index. http://www.ipni.org.
Kleyer, M., S. Dray, F. de Bello, J. Leps, R. J. Pakeman & B. Strauss, 2012. Assessing species and community functional responses to environmental gradients: which multivariate methods? Journal of Vegetation Science 23: 805–821.
Larned, S. T., T. Datry, D. B. Arscott & K. Tockner, 2010. Emerging concepts in temporary-river ecology. Freshwater Biology 55: 717–738.
Leira, M. & M. Cantonati, 2008. Effects of water-level fluctuations on lakes: an annotated bibliography. Hydrobiologia 613: 171–184.
Michalet, R., J. P. Maalouf, P. Choler, B. Clément, D. Rosebery, J. M. Royer, C. Schöb & C. J. Lortie, 2015. Competition, facilitation and environmental severity shape the relationship between local and regional species richness in plant communities. Ecography 38: 335–345.
Murray-Hudson, M., P. Wolski, F. Murray-Hudson, M. T. Brown & K. Kashe, 2014. Disaggregating hydroperiod: components of the seasonal flood pulse as drivers of plant species distribution in floodplains of a tropical wetland. Wetlands 34: 927–942.
Netten, J., J. Van Zuidam, S. Kosten & E. T. H. M. Peeters, 2011. Differential response to climatic variation of free-floating and submerged macrophytes in ditches. Freshwater Biology 56: 1761–1768.
Nicolet, P., J. Biggs, G. Fox, M. J. Hodson, C. Reynolds, M. Whitfield & P. Williams, 2004. The wetland plant and macroinvertebrate assemblages of temporary ponds in England and Wales. Biology Conservation 120: 261–278.
Ottaviani, G. & G. Keppel, 2017. Contrasting intraspecific foliar trait responses to stressful conditions of two rhizomatous granite outcrop species at different scales in southwestern Australia. Austral Ecology 43: 249–256.
Pedro, F., L. Maltchick & I. Bianchini-Jr, 2006. Hydrologic cycle and dynamics of aquatic macrophytes in two intermittent rivers of the semi-arid region of Brazil. Brazilian Journal of Biology 66: 575–585.
Peel, M. C., B. L. Finlayson & T. A. McMahon, 2007. Updated world map of the Köppen-Geiger climate classification. Hydrology and Earth System Sciences 11: 1633–1644.
Pérez-Harguindeguy, N., S. Díaz, E. Garnier, S. Lavorel, H. Poorter & P. Jaureguiberry, 2013. New handbook for standardized measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167–234.
Pierce, S., G. Brusa, I. Vagge & B. E. L. Cerabolini, 2013. Allocating CSR plant functional types: the use of leaf economics and size traits to classify woody and herbaceous vascular plants. Functional Ecology 27: 1002–1010.
Pierce, S., D. Negreiros, B. E. L. Cerabolini, J. Kattge, S. Díaz, M. Kleyer, B. Shipley, S. J. Wright, N. A. Soudzilovskaia, V. G. Onipchenko, P. M. van Bodegom, C. Frenette-Dussault, E. Weiher, B. X. Pinho, J. H. C. Cornelissen, J. P. Grime, K. Thompson, R. Hunt, P. J. Wilson, G. Buffa, O. C. Nyakunga, P. B. Reich, M. Caccianiga, F. Mangili, R. M. Ceriani, A. Luzzaro, G. Brusa, A. Siefert, N. P. U. Barbosa, F. S. Chapin, W. K. Cornwell, J. Y. Fang, G. W. Fernandes, E. Garnier, S. Le Stradic, J. Peñuelas, F. P. L. Melo, A. Slaviero, M. Tabarelli & D. Tampucci, 2017. A global method for calculating plant CSR ecological strategies applied across biomes world-wide. Functional Ecology 31: 444–457.
Quinn, G. & M. Keough, 2002. Experimental Design and Data Analysis for Biologists. Cambridge University Press, Cambridge.
R Core Team, 2019. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna.
Ricklefs, R. E., 2006. Evolutionary diversification and the origin of the diversity-environment relationship. Ecology 87: 3–13.
Riis, T. & I. Hawes, 2002. Relationships between water level fluctuations and vegetation diversity in shallow water of New Zealand lakes. Aquatic Botany 74: 133–148.
Rodrigues-Filho, C. A. S., R. P. Leitão, J. Zuanon, J. I. Sánchez-Botero & F. B. Baccaro, 2018a. Historical stability promoted higher functional specialization and originality in Neotropical stream fish assemblages. Journal of Biogeography 45: 1345–1354.
Rodrigues-Filho, C. A. S., R. C. Gurgel-Lourenço, L. A. V. Valões, E. F. Oliveira, R. F. Leitão, D. S. Garcez & J. I. Sánchez-Botero, 2018b. How are local fish communities structured in Brazilian semiarid headwater streams? Hydrobiologia 819: 93–108.
Rosado, B. H. P. & E. A. de Mattos, 2017. On the relative importance of CSR ecological strategies and integrative traits to explain species dominance at local scales. Functional Ecology 31: 1969–1974.
Rosset, V., A. Lehmann & B. Oertli, 2010. Warmer and richer? Predicting the impact of climate warming on species richness in small temperate waterbodies. Global Change Biology 16: 2376–2387.
Sánchez-Carrillo, S., D. G. Angeler, R. Sánchez-Andrés, M. Alvarez-Cobelas & J. Garatuza-Payán, 2004. Evapotranspiration in semi-arid wetlands: relationships between inundation and the macrophyte-cover: open-water ratio. Advances in Water Resources 27: 643–655.
Sculthorpe, C. D., 1967. The Biology of Aquatic Vascular Plants. Edward Arnald, London.
Silva, J. M., L. C. F. Barbosa, I. R. Leal & M. Tabarelli, 2017. The Caatinga: understanding the challenges. In Silva, J. M., M. Tabarelli & I. R. Leal (eds), The Largest Tropical Dry Forest Region in South America. Springer, Gewerbestrasse.
Souza, M. J. N. & V. P. V. Oliveira, 2006. Os enclaves úmidos e subúmidos do semi-árido do nordeste brasileiro. Mercator 9: 85–102.
Swenson, N. G. & M. D. Weiser, 2010. Plant geography upon the basis of functional traits: an example from eastern North American trees. Ecology 91: 2234–2241.
Tabosa, A. B., L. Q. Matias & F. R. Martins, 2012. Live fast and die young: the aquatic macrophyte dynamics in a temporary pool in the Brazilian semiarid region. Aquatic Botany 102: 71–78.
Violle, C., M. Navas, D. Vile, E. Kazakou, C. Fortunel, I. Hummel & E. Garnier, 2007. Let the concept of trait be functional! Oikos 116: 882–892.
Whittaker, R. J., K. J. Willis & R. Field, 2001. Scale and species richness: towards a general, hierarchical theory of species diversity. Journal of Biogeography 28: 453–470.
Wiens, J. J., D. D. Ackerly, A. P. Allen, B. L. Anacker, L. B. Buckley, H. V. Cornell, E. I. Damschen, T. J. Davies, J. A. Grytnes, S. P. Harrison, B. A. Hawkins, R. D. Holt, C. M. McCain & P. R. Stephens, 2010. Niche conservatism as an emerging principle in ecology and conservation biology. Ecology Letters 13: 1310–1324.
Wilde, M., N. Sebei, S. Puijalon & G. Bornette, 2014. Responses of macrophytes to dewatering: effects of phylogeny and phenotypic plasticity on species performance. Evolutionary Ecology 28: 1155–1167.
Zuur, A. F., E. N. Ieno, N. J. Walker, A. A. Saveliev & G. M. Smith, 2009. Mixed effects Models and Extensions in Ecology with R. Springer, New York.
Acknowledgements
This study was financed in part by the Brazilian Fostering Agency Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES—Coordination for the Advancement of Higher Education Personnel)—Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: André Padial
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Albuquerque, A.C., Rodrigues-Filho, C.A.S. & Matias, L.Q. Influence of climatic variables on CSR strategies of aquatic plants in a semiarid region. Hydrobiologia 847, 61–74 (2020). https://doi.org/10.1007/s10750-019-04072-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-04072-5