Abstract
The hydrologic and physical structure of streams strongly influences the biological composition of benthic macroinvertebrate communities. Research on step–pool systems in high-gradient streams has focused primarily on physical processes rather than on ecological characteristics. This study examined both the biological and physical attributes of 27 step–pool sequences in three steep mountain streams of the Smith River Basin in northern California, USA. Multivariate analysis using non-metric multidimensional scaling (NMS) found a biological separation of step and pool sites (based on benthic macroinvertebrate data) in all three study watersheds. Step habitats had greater taxa richness, diversity, %Plecoptera, %Heptageniidae, %Nemouridae, and %clingers compared to pools. Steps also differed from pools in physical characteristics such as grain size distribution, whereby steps were dominated by boulders compared to pools characterized by gravel and cobbles. Moreover, steps had higher dissolved oxygen, greater water velocity, and shallower water depths compared to pools. NMS ordinations showed a correlation between physical factors and biological communities. These results suggest the ecological importance of step–pools streams, in that the development of step sequences creates and maintains a repetitive pattern of high-quality ecological environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The hydraulic conditions and physical habitat of streams are known to influence the biological composition of benthic macroinvertebrate communities (e.g., Statzner, 1981; Statzner & Higler, 1986; Gore et al., 2001; Lytle & Poff, 2004; Sagnes et al., 2008). Physical factors such as stream velocity and turbulence provide hydraulic constraints on benthic invertebrates (e.g., Sagnes et al., 2008) and can also affect biotic interactions such as competition and predation (Lancaster & Hildrew, 1993; Hart & Finelli, 1999). Benthic macroinvertebrate distribution often differs in low-velocity compared to high-velocity habitats within a stream (Mérigoux & Dolédec, 2004; Dolédec et al., 2007). These differences are often linked to biological traits (Bêche et al., 2006) such as stream-lined body morphology (Statzner, 1988; Weissenberger et al., 1991), holdfasts for attaching to stream substrate (Hora, 1930; Wichard et al., 2002), avoidance of current (Vogel, 1994), feeding behavior (Merritt et al., 2008), oxygen demands (Collier, 1994), and biotic interactions (Hansen et al., 1991).
The step–pool channel is a reach type in which stream velocities, turbulence, and physical habitat vary at the microhabitat scale in a recurring, interchanging fashion (Montgomery & Buffington, 1997). Step–pool systems are unique environments commonly found in high-gradient (>3%) mountain streams characterized by a preponderance of steps and pools that produce a repetitive staircase-like longitudinal profile (Chin, 1999). Step–pools provide resistance (Abrahams et al., 1995) and ensure continuous and intensive energy dissipation that otherwise would heavily erode and degrade channels in high-gradient environments (Heede, 1981; Chin, 1989). The main topographic characteristic of step–pool streams is an alternating sequence of steps and pools that contrasts sharply with adjacent physical conditions within the same stream.
The rhythmic nature of the step–pool streambed has long captured the attention of scientists (e.g., Judd, 1964), but study into the nature of this spatial rhythmicity has focused primarily on physical processes. Application of spectral analysis identified periodicities in the occurrence of step–pools (Chin, 2002), for example, thereby suggesting a fundamental adjustment in the streambed to energy expenditure, and an analogy to meandering in the vertical dimension. Despite possible external forcings such as vegetation and bedrock, a rhythmic streambed suggests that underlying internal adjustments to energy expenditure are nevertheless detectable in steep headwater streams, resulting in a fluvially organized form Chin & Phillips (2007).
Despite these and other efforts that have increased understanding of the geomorphology of step–pool systems (e.g., Zimmermann, 2013), corresponding levels of analysis on the biological character of step–pool streams are not as numerous. Headwater mountain streams have long been known as important sources of nutrients, organic matter, and colonization for downstream reaches (Hynes, 1970; Gomi et al., 2002; Hall et al., 2002; Karlsson et al., 2005). Step–pool streams are also recognized as critical habitat for salmonids (Montgomery et al., 1999; Buffington et al., 2004) and tailed frogs (Welsh & Ollivier, 1998; Dupuis et al., 2000). In 1999, Scheuerlein set the stage for future research by theorizing that the contrasting abiotic features of step–pool environments would enhance their ecological value (Scheuerlein, 1999). Subsequently, Wang et al. (2009) found higher density and diversity of benthic macroinvertebrates in step–pool systems compared to nearby streams that lacked step–pools.
Benthic macroinvertebrates offer a distinctive way to characterize the biological communities in step–pools. They are ubiquitous in stream systems and have biological traits that indicate adaptation to specific habitat types (e.g., Poff, 1997; Usseglio-Polatera et al., 2000; Bêche et al., 2006; Bonada et al., 2007; Statzner et al., 2008; Tullos et al., 2009). Certain taxa in this diverse and ecologically important group are sensitive to physical and chemical perturbations (Carter et al., 2007); thus they can be used to assess the ecological health of streams (Barbour et al., 1999). Correlating fundamental data on biological characteristics of benthic macroinvertebrates with physical aspects of steps and pools therefore provides an essential basis to developing theoretical concepts for the ecological significance of step–pool mountain streams.
Ultimately, an integration of well-developed geomorphological principles with new bio-physical knowledge can contribute to greater understanding of the physical and biological interactions in step–pool streams. Moreover, because step–pools serve as important habitat for threatened aquatic species (e.g., Montgomery et al., 1999), a deeper understanding of the biological community in step–pool systems is needed to guide sustainable management of biotic and water resources, as well as for restoration of steep river channels impacted by human activity.
This study explored the composition of benthic macroinvertebrate assemblages in alternating steps and pools using three steep mountain streams of the Smith River Basin (Del Norte County, California, USA). The following hypotheses were tested: (1) benthic macroinvertebrate assemblages differ between steps and pools, (2) biological characteristics relate to physical attributes of steps and pools, and (3) ecological attributes of steps and pools are similar among the three study streams.
Materials and methods
Study area
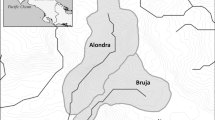
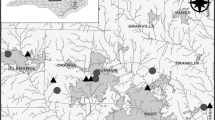
The study was conducted on Eighteenmile Creek and the West and East Forks of Patrick’s Creek; three tributary streams of the Middle Fork Smith River in Del Norte County, California (Fig. 1). The Smith River Basin is located in northern California and southern Oregon (USA) with a catchment area of 1,860 km2. One of the only large, undammed rivers in California, the Smith River has a wild and scenic status as part of the National Wild and Scenic Rivers System [designated by the Wild and Scenic Rivers Act (16 USC 1271-1287)-Public Law 90-542].
Study streams were selected based on similar physical and biological features, including watershed area, elevation, gradient, riparian vegetation, geology, soils, substrate size, and the presence of stable step–pool sequences with no signs of erosion or deposition (Table 1; for photos see Fig. 2). Historic land uses within the Eighteenmile and Patrick’s Creeks Watersheds included road and trail building, water diversion, mining, hunting, fishing, homesteading, timber harvest, and back-country recreation (Bowman, 2012). Despite these historic impacts, study sites were selected based on relative lack of disturbance in the upstream watershed and other features that might influence intact stream biological communities. All sampling sites were located within the Smith River National Recreation Area and Six Rivers National Forest (US Forest Service). Mean annual precipitation of the study area is 2540 mm with most of the rainfall occurring between October and May (United States Department of Agriculture, USDA, 2014).
Three 50-m long reaches were selected on each of the three study streams (for a total of nine reaches). Reaches were spaced at least 50 m apart. Three paired step–pools, randomly selected within each 50-m reach, provided a total of 18 samples per stream (i.e., 9 steps and 9 pools). Sampling areas were based on the size of the habitat sampled (step or pool) in order to be representative of the biotic community therein. A total of 54 samples of benthic macroinvertebrates were collected across the three study streams, along with surveys of physical characteristics. Field work was conducted on 1, 2, and 16 October 2011 with two minor flow events occurring through the period of data collection. Flow conditions were similar for all sampling events and represent baseflow conditions (0.064–0.076 m3/s).
Biological characteristics
Sampling of benthic macroinvertebrates proceeded in a downstream–upstream direction within each 50-m sampling reach. Sampling began at the most downstream pool within a study reach, followed by the adjacent upstream step, until all three randomly selected step–pools in the 50-m reach were sampled. Pools were sampled using a traditional 30-cm-wide D-frame net with 50 μm mesh. Agitation by kicking the bottom substrate for 1 min dislodged benthic macroinvertebrates in the pool substrate for collection. Then, a swooping motion with the D-frame net through each pool captured benthic macroinvertebrates suspended in the water column. Step habitats were sampled using a 30-cm-wide flexible frame D-net with 50 μm mesh, similar to one tested in a previous study (Chin et al., 2009a). The flexible frame D-net was placed at the downstream end of the step, while rubbing and agitating the upstream step for 1 min to dislodge benthic macroinvertebrates attached to the steps or within the interstitial spaces of the steps. Statistical analysis methods took into account the differing sampling techniques and sampling areas in steps and pools (see “Data analysis” section below).
Benthic macroinvertebrates were sorted from sediment and organic debris in the laboratory using a dissecting microscope (×10–30). All benthic macroinvertebrates sampled at a given site were sorted into a single 20-ml scintillation vial and labeled with the site identification code and habitat type (i.e., step or pool). An aquatic taxonomy laboratory (Aquatic Biology Associates, Inc., Corvallis, Oregon) identified the sorted benthic macroinvertebrates to the standard taxonomic effort (i.e., generally genus or species for insect taxa, and class, subclass, or family for non-insect invertebrates).
Physical characteristics
Physical characteristics of the study reaches were collected immediately following the biological sampling of steps and pools. Physical characteristics measured at each of the 54 sampling sites included grain size, wetted width, water depth, water velocity, water temperature, conductivity, total dissolved solids, pH, and dissolved oxygen. Pebble counts (adapted from Wolman, 1954) characterized grain size in each pool by measuring the intermediate axis of 50 randomly selected particles. For boulder steps, measurement of the five largest rocks within each step followed methods from Chin (1999). Dissolved oxygen was measured with an YSI 55 DO meter that simultaneously recorded dissolved oxygen and temperature. A salinity meter (YSI EcoSense EC 300) simultaneously registered conductivity, salinity, total dissolved solids, and temperature. Lastly, a pH meter (pH Testr Basic) inserted into the water column recorded pH at each sampling site.
To calculate discharge, water depth and velocity measurements were taken along one stable cross section at each tributary. A measuring rod recorded water depth, while a Marsh-McBirney meter measured velocity. Depth and velocity were measured along each transect at equal intervals across the channel, so that a minimum of five measurements were made. Discharge was calculated by using the standard equation Q = vA (where Q is discharge, v is velocity, and A is cross-sectional area).
Data analysis
Non-metric multidimensional scaling (NMS) was used to compare biological communities in steps and pools (PC-ORD, version 6.08, MjM Software, Gleneden Beach, Oregon, 2011). NMS is the preferred multivariate ordination technique to analyze these data (compared to CCA or PCA) because it is designed to look for patterns and groupings in biological data that are highly heterogeneous or have non-linear relationships among responses (McCune & Grace, 2002; Peck, 2010). Because the differing sampling methods used for steps and pools were tailored to the morphology of the substrata, sampling areas were not uniform in size. Therefore, benthic macroinvertebrate abundance or density data were not considered in the data analysis. To address the unequal sampling areas, rarefaction was used to subsample to 300 individuals per sample; samples containing less than 300 individuals retained their original count. Following rarefaction, taxa found at <5% of all sites were removed prior to running the NMS ordination (as recommended by McCune & Grace, 2002). The rarefied data were ordinated using the Sorensen (Bray–Curtis) similarity coefficient (Bray & Curtis, 1957; McCune & Grace, 2002), 200 maximum iterations, 100 real runs, and a stability criterion of 0.000001. Sites were grouped by habitat (i.e., step or pool) and stream. Data analysis was conducted for each stream separately as well as all three streams together. NMS was also used to relate ecological characteristics to physical attributes by overlaying physical attributes as vectors in the ordination plot. A multi-response permutation procedure (MRPP) was used in PC-ORD to test for differences between steps and pools.
Biological metrics were calculated using the rarefied benthic macroinvertebrate data to test study hypotheses of differences between steps and pools, biological patterns related to physical parameters, and similarities among streams. Metrics of tolerance and sensitivity (e.g., %intolerant taxa, % of the generally sensitive orders Ephemeroptera, Plecoptera and Trichoptera or %EPT) were calculated to see if steps or pools tended to support higher percentages of sensitive or tolerant groups. For example, steps might provide higher quality habitat that can support a higher percentage of sensitive/intolerant organisms compared to pools. Trophic structure metrics such as functional feeding groups (e.g., collector-filterer, scraper, predator) were explored to see if food resources differed among steps and pools. Habit metrics (e.g., clinger, sprawler, burrower) were calculated to reflect the types of habitat available in steps and pools. A non-parametric Wilcoxon matched-pair signed-rank test was used to examine differences in biological metrics among streams and habitat type (steps and pools). A two-way ANOVA using habitat and stream as fixed factors enabled examination of physical and water quality variables in steps compared to pools. Statistical analyses were performed in SPSS (version 22, IBM, 2013) with an α of 0.05.
Results
The biological samples collected in this study contained a total of 104 benthic macroinvertebrate taxa. Two of the samples did not contain any benthic macroinvertebrates; therefore, 52 of the 54 biological samples collected were analyzed. Twenty-eight rare taxa found at <5% (~2 sites) of sites were omitted from analysis, resulting in 72 taxa used for the NMS ordination.
Hypothesis #1
Benthic macroinvertebrate assemblages differ between steps and pools.
NMS of rarefied data revealed differences in benthic macroinvertebrate assemblages between steps and pools (Fig. 3). This partitioning of the step and pool samples in the NMS ordination plot indicated that the taxonomic composition of biological communities in steps and pools was overall quite different. The final stress for a three-dimensional solution was 11.62. Axis 1 described 30.9% of the variability in the data, Axis 2 described 14.8%, and Axis 3 described 35.6% (cumulative 81.3% of the variability explained). Therefore, Axes 1 and 3 were selected for the final ordination plot because these axes cumulatively explained the greatest amount of variability in the data. NMS plots with data transformed to presence/absence revealed similar patterns to the plot shown in Fig. 3.
Non-metric multidimensional scaling (NMS) plot of sites sampled. Symbols are coded by habitat type (dark triangles pools, open circles steps) and show a partitioning by habitat type. Labeled vectors indicate physical and water quality variables correlated to step and pool sites (all vectors shown in the plot have r 2 > 0.285). Length of vector indicates strength of relationship. Final stress = 11.62. Plot generated in PC-ORD version 6.08
MRPP was used in PC-ORD to directly test differences between steps and pools. MRPP is a non-parametric procedure for testing the hypothesis of no difference between two or more groups (Mielke & Berry, 2001) and was conducted in addition to the NMS to gain further confirmation of group differences. The distance measure used was Euclidean (Pythagorean). Groups were defined as step or pool, and the chance-corrected within-group agreement (A) was 0.1843. The within-group agreement is a measure of effect size and provides an indication of how similar the sample units are to one another within a group (within-group homogeneity compared to random expectation) (Peck, 2010). The highly significant result of the MRPP (P = 0.000000031) indicated that the benthic macroinvertebrate assemblages in step and pool groups were significantly different.
The presence of all step sites on the right side of the NMS ordination plot (i.e., higher Pearson correlations to Axis 1) and most pools samples on the left side of the plot (lower correlations to Axis 1) indicates that Axis 1 represents a gradient from pool to step features (Fig. 3). Moreover, the taxa most correlated with Axis 1 in the NMS ordination (Table 2) were similar to the most common and dominant taxa found in the step samples. The top correlated taxa with Axis 1 (and hence more abundant and common in step habitats) were the mayfly Baetis tricaudatus Dodds 1923 (r 2 = 0.463), the caddisfly genus Hydropsyche (r 2 = 0.408), the predatory caddisfly Rhyacophila hyalinata Banks 1905 (r 2 = 0.338), and the stonefly Doroneuria baumanni Stark and Gaufin 1974 (r 2 = 0.264; Table 2). Conversely, the taxa with the lowest Pearson correlation to Axis 1 represent taxa correlated with pool sites. These pool-related taxa included the limnephilid caddisfly Hydatophylax hesperus Banks 1914 (r 2 = −0.073), the caddisfly Heteroplectron californicum McLachlan 1871 (r 2 = −0.069), the midge fly genus Dixa (r 2 = −0.039), and the caddisfly genus Lepidostoma (r 2 = −0.01).
When coding the site symbols by stream, the NMS ordination plot revealed a grouping of Eighteenmile Creek mostly separate from that of East and West Forks of Patrick’s Creek (Fig. 4). Therefore, separate NMS ordinations were run for Eighteenmile Creek and the East and West Forks of Patrick’s Creek. The ordinations separated by stream revealed similar separation of steps and pools within each stream; therefore, further analysis of each stream separately was not extensively pursued.
Some overlap occurred among common taxa found in both steps and pools. Taxa commonly found in step samples included the mayfly B. tricaudatus (found in 96% of step samples), the perlid stoneflies Calineuria californica Banks 1905 and D. baumanni (both found in 85% of step samples), and the trichopteran genus Hydropsyche (found in 85% of step samples). Taxa most commonly found in pool samples included ephemeropterans B. tricaudatus (in 76% of pool samples) and Ephemerella dorothea infrequens McDunnough 1924 (in 44% of pool samples), and the trichopteran genus Micrasema (in 60% of pool samples). Micrasema was the dominant taxon in both steps and pools constituting 29.4% of all individuals in steps and 25.5% in pools.
Using the Wilcoxon matched-pair signed-rank test, analysis of biological metrics revealed significant differences (α = 0.05) between steps and pools in several of the metrics examined (Table 3; Fig. 5). For example, steps had significantly greater taxa richness (following rarefaction), Shannon–Weaver diversity, and percentage of the sample composed of Plecoptera, Heptageniidae (Ephemeroptera), Nemouridae (Plecoptera), and clingers (organisms that have adaptations for clinging to benthic substrata) compared to pools (Table 3; Fig. 5). Pool habitats had a higher percentage of sprawlers (organisms that sprawl on the surface of substrate) than steps (Table 3; Fig. 5). In contrast, several metrics did not differ significantly between steps and pools. For example, %intolerant taxa, %Ephemeroptera, %Trichoptera, %EPT (a metric that combines the generally pollution sensitive orders Ephemeroptera, Plecoptera, and Trichoptera), %collector-filterer, %scraper, and %burrower were not significantly different in steps compared to pools (Table 3; Fig. 5).
Comparison of mean percentage composition of biological metrics in steps (shaded bars) and pools (white bars). The data presented in these graphs is pooled from all three study streams. The graph on the left includes biological metrics related to tolerance (intol %intolerant, EPT %Ephemeroptera, Plecoptera, and Trichoptera, Ephem %Ephemeroptera, Plecop %Plecoptera, Trichop %Trichoptera, Heptag %Heptageniidae, Nemour %Nemouridae). Graph on the right includes metrics related to functional feeding group (CF %collector-filterer, SC %scraper) and habit (CL %clinger, SP %sprawler, BU %burrower). Asterisks indicate significant differences between steps and pools (α < 0.05)
Because of the differences in benthic macroinvertebrate composition in Eighteenmile Creek compared to that of East and West Forks of Patrick’s Creek (shown by the NMS), the metrics between steps and pools were also compared by separating Eighteenmile Creek from East and West Forks of Patrick’s Creek (Table 3). Results found that significant differences between steps and pools varied among streams. For example, East and West Forks of Patrick’s Creek had higher Shannon–Weaver diversity, %Plecoptera, %Heptageniidae, %Nemouridae, %collector-filterer, %clinger, and %sprawler in steps compared to pools (Table 3). However, Eighteenmile Creek alone failed to show significant differences in these metrics. Conversely, %Trichoptera was significantly higher in steps than pools in Eighteenmile Creek, but not in East and West Forks of Patrick’s Creek (Table 3). Despite some differences between study streams, the overall pattern when pooling data from the three streams was a difference in benthic macroinvertebrate communities between steps and pools.
Hypothesis #2
Biological characteristics relate to physical attributes within steps and pools.
Examination of the physical dimensions of the stream channel and flow characteristics revealed notable differences between steps and pools (Table 4). Because a two-way ANOVA using habitat and creek as fixed factors revealed several differences among creeks, data for physical characteristics and water quality were considered with the three streams combined and separately (Table 4). However, a comparison of Eighteenmile Creek separate from East and West Forks of Patrick’s Creek did not change whether a variable was significantly different between steps and pools (α = 0.05). Overall, water depths were significantly shallower in steps (0.09 ± 0.01 m) compared to pools (0.21 ± 0.01 m, P < 0.0001), wetted widths were not significantly different in steps compared to pools (P = 0.636), and water velocity was significantly faster in the steps (0.50 ± 0.04 m/s) compared to the pools (0.07 ± 0.04 m/s, P < 0.0001).
Water quality data revealed both similarities and differences in steps compared to pools (Table 4). For example, temperature (P = 1.00), conductivity (P = 0.939), total dissolved solids (P = 0.566), and pH (P = 0.294) did not differ significantly in steps versus pools across the three study streams. However, dissolved oxygen (as measured in % and mg/l) was significantly higher in steps than pools (P < 0.0001).
Sediment analysis revealed different grain size distributions in steps compared to pools across the three study streams (Fig. 6) and when examining each stream separately. The median grain size in steps (519.5 mm) was much larger than that of pools (56.0 mm). Steps on all three streams were dominated by boulders (\(\bar{x}\) = 87%), while pools were characterized by gravels (\(\bar{x}\) = 56%) and cobbles (\(\bar{x}\) = 39%; Fig. 6).
NMS ordination showed how water quality and physical variables related to the biological communities found in steps and pools. Labeled vectors shown in Fig. 3 indicate that biological communities sampled from step habitats (as represented by Axis 1) were associated with larger grain size (r 2 = 0.389), higher water velocity (r 2 = 0.238), and higher dissolved oxygen (r 2 = 0.206). Conversely, biological communities found in pools were associated with a greater water depth.
Hypothesis #3
Ecological attributes of steps and pools are similar among the three study streams.
Comparing attributes across the three study streams provided insight into the extent to which physical and ecological patterns might hold true across other streams and watersheds. The three study streams had similar dissolved oxygen, pH, total dissolved solids, and salinity, but Eighteenmile Creek had slightly higher water temperature and conductivity. Water temperature was approximately 2°C warmer in Eighteenmile Creek (14.5 ± 0.1°C) than in East Fork Patrick’s (12.6 ± 0.1°C) and West Fork Patrick’s Creeks (12.3 ± 0.1°C), but temperature fluctuated very little within each stream (average s.d. of water temperature was 0.26°C). Conductivity was higher in Eighteenmile Creek (133.3 ± 0.7 μS) compared to East Fork Patrick’s (73.3 ± 0.6 μS) and West Fork Patrick’s Creeks (127.04 ± 0.3 μS). Discharge was similar across the three streams: 0.065 m3/s in Eighteenmile Creek, 0.064 m3/s in East Fork Patrick’s Creek, and 0.076 m3/s in West Fork Patrick’s Creek.
Comparison of pebble counts among the three streams revealed similar grain size distributions. As noted above, steps on all three streams were dominated by boulders (87–100%), with small percentages of cobble on Eighteenmile Creek (11%) and East Fork Patrick’s Creek (8%; Fig. 7a). Pools were dominated by gravels (54–58%) and cobbles (37–42%), with similar proportions found in each stream (Fig. 7b). For steps, the mean intermediate axis of particles was 486.3 mm in Eighteenmile Creek, 486.9 mm at East Fork Patrick’s Creek, and significantly larger 632.38 mm at West Fork Patrick’s (P = 0.01). In pools, the mean intermediate axes measured were 75.0 mm in Eighteenmile Creek, 84.2 mm in East Fork Patrick’s Creek, and 85.6 mm in West Fork Patrick’s Creek (not significantly different, P < 0.05).
NMS plots revealed that benthic macroinvertebrate assemblages were different between steps and pools regardless of the watershed they were sampled in, as described above and shown in Fig. 4. When examining the same plot coded by habitat (Fig. 3), the separation of step and pool biological communities was still apparent despite the differences among watersheds. In other words, step samples across the three streams were more similar to each other than a step and pool sample within a single stream.
Discussion
Overall, this study found distinctive biological signatures in step compared to pool habitats. These findings reinforce the importance of hydraulic forces and physical habitat structure on biological organisms and their influence on the distribution of benthic macroinvertebrates on a small scale (e.g., Statzner, 1981; Statzner & Higler, 1986; Sagnes et al., 2008). The differences found between steps and pools are even more fascinating because these habitats draw from the same species pool within a single stream.
Examination of specific taxa more commonly found in steps or pools (or with high or low correlations to Axis 1) can help explain some of the mechanisms driving these patterns. Attributes such as morphology and behavior (life history, feeding, etc.) likely relates to physical factors in steps and pools, as shown in a few key example taxa below.
Taxa more abundant in step samples
Individuals of the caddisfly genus Hydropsyche (Trichoptera: Hydropsychidae) were common and abundant in step habitats sampled for this study and are known to inhabit erosional habitats such as step–pools and spin nets in fixed retreats (Edington, 1968; Fuller & Mackay, 1980). Fixed retreats of Hydropsyche capture particulate organic matter in the water column such as diatoms, green algae, detritus, and other invertebrates (Benke & Wallace, 1980). This feeding mode puts Hydropsyche in the collector-filterer functional feeding group (Merritt et al., 2008). Hydropsyche individuals tend to be found in higher densities in larger substrate such as cobble and gravel compared to sand or fine sand (Poepperl, 2000) and prefer habitats in high shear velocity conditions (Mérigoux & Dolédec, 2004; Sagnes et al., 2008) rather than low shear stress. These habitat preferences may explain why Hydropsyche individuals were more abundant in step habitat samples in this study where shear stress and water velocity would likely be higher and aid in the capture of food resources traveling downstream through the water column.
Rhyacophila hyalinata (Trichoptera: Rhyacophilidae) were also more abundant in step habitats and are known to be free ranging clingers (Lloyd, 1921) and predators of other invertebrates (Burton & McRae, 1972; Manuel & Folsom, 1982). Smith (1968) recorded the larval habitat of R. hyalinata as turbulent mountain streams with rocky beds. Mérigoux & Dolédec (2004) found Rhyacophila sp. abundant in the high shear stress environments of a Mediterranean-type tributary of the Rhône River in southern France; a finding that is in concordance with the findings of this study that observed R. hyalinata more commonly in step than in pools samples.
The benthic macroinvertebrate “habit” of particular note that was found in a significantly higher percentage in steps than pools was a group called the ‘clingers’ that have adaptations to cling to the substrate in high-velocity environments (Merritt et al., 2008). One group of clingers known for its ability to cling to benthic substrate is the coleopteran family Psephenidae (commonly known as water pennies). Smith & Dartnall (1980) found that the psephenid’s stream-lined form and powerful grip of its thoracic legs enable it to persist in habitats with a high velocity. The dorso-ventrally flattened individuals of Psephenidae were five times more abundant in step compared to pool samples in this study.
Taxa more abundant in pool samples
The life history and feeding traits of the caddisfly genus Hydatophylax (Trichoptera, Limnephilidae) show a preference for pools or slow-moving depositional habitats (Anderson, 1976). Zhang (1996) found that Hydatophylax larvae live chiefly among emergent water plants and submerged roots of terrestrial plants. Hydatophylax larvae form rough, cylindrical cases out of wood, bark, minerals, and sticks (Lloyd, 1921) presumably as a form of camouflage among detritus. In terms of functional feeding group, Hydatophylax are obligate shredders and detritivores (Anderson et al., 1978). Holomuzki et al. (2013) found that H. hesperus preferred small-order streams with dense canopies and high allochthonous inputs. Hydatophylax sp. egg masses are generally found on substrate in pools near the surface of the water where vegetation and other stream features shade oviposition sites (Hoffmann & Resh, 2003).
Individuals in the caddisfly genus Heteroplectron (Trichoptera: Calamoceratidae), similar to Hydatophylax, are usually found in depositional habitats such as pools (Lloyd, 1921; Anderson, 1976). Heteroplectron can be collected by searching leaf packs in pools of high-gradient streams (Patterson & Vannote, 1979). Larvae are generally detritivores that feed on decaying leaf litter and wood (Anderson et al., 1978, 1984). Heteroplectron larvae make their cases out of a hollowed out stick or piece of bark (Anderson et al., 1984). Holomuzki et al. (2013) found Heteroplectron in slow-flowing areas where fine sediment or benthic organic matter accumulate in Elder Creek and Fox Creek (Mendocino Co., California). These findings confirm that Heteroplectron individuals would more likely be collected in pool habitats than steps.
Taxa found in both step and pool samples
Palmer (1995) found that individuals of Baetis bicaudatus Dodds 1923 achieved a higher maximum body size in rapid compared to slow-moving current environments and thus might have a preference for higher velocity step–pool environments. Shallower step habitats may be more amenable to egg-laying for Baetis females that crawl beneath the water and lay rows of eggs on submerged substrate (Merritt et al., 2008). However, pools provide easier foraging habitat with less turbulence thus explaining preferences for both step and pool habitat types. Baetis are generally swimmers, but are also clingers at rest, and are collector-gatherers and facultative scrapers (Gilpin & Brusven, 1970). These plastic feeding, growth, and mobility patterns lend themselves to inhabiting both steps and pools.
Individuals of Baetis rhodani Pictet 1843 were found to inhabit areas with high hydraulic stress when small in size and lower hydraulic stress as they grew larger in a Black Forest stream (Statzner & Borchardt, 1994) indicating their ability to inhabit areas of both high and low shear stress depending on their body size. Mérigoux & Dolédec (2004) also found Baetis sp. at both high and low levels of shear stress further indicating their ability to exist in both hydraulic conditions. These shifts in the types of habitat used based on body size (Statzner & Borchardt, 1994) may indicate that certain species utilize both step and pool habitats during different parts of their life depending on their body size. The samples in this study were conducted in the autumn, but samples collected in a different time of year may yield different results for taxa that shift habitat preferences as they grow in size.
The caddisfly genus Micrasema (Trichoptera: Brachycentridae) was the dominant taxon in both steps and pools constituting 29.4% of all individuals in steps and 25.5% in pools and are commonly found in mountain streams (Anderson, 1976). Several functional feeding groups are included within Micrasema such as shredders, collectors-gatherers, and grazers (Becker, 1990; Merritt et al., 2008) indicating the ability of different species within this genus to exploit a variety of food resources in differing habitats. Several studies have explored the impact of Micrasema grazing on stream algae (e.g., Dudley & D’Antonio, 1991; Katano et al., 2005, 2007). Katano et al. (2005, 2007) found that individuals of Micrasema quadriloba Martynov in a Japanese mountain stream controlled the abundance of periphyton at the reach scale. Micrasema have also been found to be positively affected by the presence of macroalgae due to structural habitats created by algae (Dudley et al., 1986). Therefore, distribution of algae may be more of an influence on Micrasema distribution than habitat structure related to steps and pools.
Influence of physical attributes on biological assemblages within steps and pools
Scheuerlein (1999) postulated that the turbulent physical conditions of a step habitat at lower flow have the potential to exclude most macroinvertebrate taxa, but our study found the opposite to be true. A myriad of taxa are adapted to the high-velocity step environment or have developed the ability to stay out of high-velocity areas by taking refuge in the interstitial spaces within a step (Muehlbauer & Doyle, 2012). Step habitats in step–pool streams may also provide a function analogous to riffles in riffle–pool streams in which riffles contain more biodiversity and abundance of benthic macroinvertebrates compared to pools (Brown & Brussock, 1991). Bryant et al. (2007) suggested that large quantity of organic and inorganic sediment stored in log steps may make them biological hotspots in headwater streams.
Differences in physical characteristics between steps and pools were reflected in particle size, dissolved oxygen, water depth, and water velocity of each habitat type (Table 4). The physical structure of a step (sharp drop in elevation) along with the coarse particles, shallow water depths, high water velocity, and turbulence combines to increase dissolved oxygen at a localized scale. In contrast, pools exhibit lower dissolved oxygen, smaller sediment sizes (such as sands, gravels and cobbles), lower stream velocity, and deeper water (e.g., Wohl et al., 1997; Wang et al., 2009). The level of dissolved oxygen can influence the biotic integrity of a stream (Connolly et al., 2004) and may also stratify the types of microhabitats that benthic macroinvertebrates colonize. Jacobsen (2008) found a decrease in taxa richness as dissolved oxygen and temperature decreased with higher altitude.
Step–pool systems provide habitat heterogeneity for aquatic organisms, which may explain the relatively high richness of taxa found in this study (n = 104 taxa prior to rarefaction). Muehlbauer & Doyle (2012) examined microscale heterogeneity in knickpoints (physically analogous to steps) and attributed the high density of benthic macroinvertebrate assemblages to the ability of the knickpoint to provide refugia during low flow conditions. Principe et al. (2007) found the highest richness, diversity, and evenness of macrophytes and macroalgae in heterogeneous hydraulic units located in the upper watershed of the Carcarana River Basin near Cordoba, Argentina.
Similar physical characteristics of the three study streams supported their use for a comparative study of this nature and can provide insight for regional patterns. For example, similar grain size distributions were found in steps on all three streams, as was the case for pools, reflecting a similar alternating staircase-like physical habitat structure for benthic macroinvertebrates. Moreover, physical attributes such as water depth and discharge were similar among the study streams and may indicate that these patterns exist in watersheds of similar size, geology, and gradient (Vogel & Kroll, 1992). We would also expect that ecological patterns of steps and pools would be similar throughout high-gradient mountain streams in this region.
Implications for theory and management
Results from this study contribute to the development of ecological theory and management pertaining to the biological significance of step habitats. The higher taxa richness, diversity, and percentage composition of sensitive benthic macroinvertebrate groups in steps compared to pools underscores that the presence of varying habitats is important to support a wide variety of organisms. The higher diversity and richness of benthic communities in steps also emphasizes the biological importance of step–pool formations in streams.
An increased interest in the use of artificial steps in stream restoration has sparked research in the physical and biological responses to step–pool restoration projects. Comiti et al. (2009) found that artificial steps were more effective at maintaining ecological function (i.e., retention of coarse particulate organic matter and macrobenthos richness and diversity) compared with traditional check dams. Similarly, artificial steps designed to mimic natural steps were linked to improvements in the aquatic ecosystems (as measured by increases of benthic macroinvertebrate density and taxa richness) in southwest China (Yu et al., 2010). Initial investigation into biological processes in restored step–pool channels (Purcell et al., 2002) also revealed a tendency toward specialized fauna in steps compared with pools (Chin et al., 2009a) and linkages to geomorphological processes (Chin et al., 2009b, 2010).
In summary, this study illustrates the contrasting patterns of biological and physical attributes of steps and pools across three tributary streams in the Smith River Basin. Environmental and biological interactions apparently influence the abundance and diversity of benthic macroinvertebrates on a habitat scale. Future research should explore if similar patterns exist in other watersheds, bioregions, and hydroclimatic conditions in order to develop greater cumulative generalizations. Regardless, step–pools serve as important habitat for aquatic species and contribute to the development of stable and diverse freshwater landscapes, and thus should be a priority for conservation, management, and restoration efforts.
References
Abrahams, A. D., G. Li & J. F. Atkinson, 1995. Step–pool streams: adjustment to maximum flow resistance. Water Resources Research 31: 2593–2602.
Anderson, N. H., 1976. The distribution and biology of the Oregon Trichoptera. Oregon Agricultural Experiment Station Technical Bulletin 134: 1–152.
Anderson, N. H., J. R. Sedell, L. M. Roberts & F. J. Triska, 1978. The role of aquatic invertebrates in processing wood debris in coniferous forest streams. American Midland Naturalist 100: 64–82.
Anderson, N. H., R. J. Steedman & T. Dudley, 1984. Patterns of exploitation by stream invertebrates of wood debris. Verhandlungen des Internationalen Verein Limnologie 22: 1847–1852.
Barbour, M. T., J. Gerritsen, B. Snyder & S. James, 1999. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers: Periphyton, Benthic Macroinvertebrates, and Fish. U.S. Environmental Protection Agency, Office of Water, Washington, DC.
Bêche, L. A., E. P. McElravy & V. H. Resh, 2006. Long-term seasonal variation in the biological traits of benthic macroinvertebrates in two Mediterranean-climate streams in California, USA. Freshwater Biology 51: 56–75.
Becker, G., 1990. Comparison of the dietary composition of epilithic trichopteran species in a first order stream. Archiv für Hydrobiologie 120: 13–40.
Benke, A. C. & J. B. Wallace, 1980. Trophic basis of production among net-spinning caddis flies in a southern Appalachian stream. Ecology 61: 108–118.
Bonada, N., M. Rieradevall & N. Prat, 2007. Macroinvertebrate community structure and biological traits related to flow permanence in a Mediterranean river network. Hydrobiologia 589: 91–106.
Bowman, S. W., 2012. Patrick Creek Watershed 2011 Level II Stream Survey. Report submitted to Six Rivers National Forest, Smith River National Recreation Area, Gasquet Ranger District, Gasquet.
Bray, J. R. & J. T. Curtis, 1957. An ordination of the upland forest communities in southern Wisconsin. Ecological Monographs 27: 325–349.
Brown, A. V. & P. P. Brussock, 1991. Comparisons of benthic invertebrates between riffles and pools. Hydrobiologia 220: 99–108.
Bryant, M. D., T. Gomi & J. J. Piccolo, 2007. Structures linking physical and biological processes in headwater streams of the Maybeso watershed, Southeast Alaska. Forest Science 53: 371–383.
Buffington, J. M., D. R. Montgomery & H. M. Greenberg, 2004. Basin-scale availability of salmonid spawning gravel as influenced by channel type and hydraulic roughness in mountain catchments. Canadian Journal of Fisheries and Aquatic Sciences 61: 2085–2096.
Burton, G. J. & T. M. McRae, 1972. Observations on trichopteran predators on aquatic stages of Simulium damnosum and other Simulium species in Ghana. Journal of Medical Entomology 9: 289–294.
California Geological Survey Website. [available on internet at http://www.consrv.ca.gov/]. Accessed 14 October 2014.
Carter, J. L., V. H. Resh, M. J. Hannaford & M. J. Myers, 2007. Macroinvertebrates as biotic indicators of environmental quality. In Hauer, F. R. & G. A. Lamberti (eds), Methods in Stream Ecology, 2nd ed. Academic, San Diego: 805–831.
Chin, A., 1989. Step–pools in stream channels. Progress in Physical Geography 13: 391–408.
Chin, A., 1999. The morphologic structure of step–pool in mountain streams. Geomorphology 27: 191–204.
Chin, A., 2002. The periodic nature of step–pool mountain streams. American Journal of Science 302: 144–167.
Chin, A. & J. D. Phillips, 2007. The self-organization of step–pools in mountain streams. Geomorphology 83: 346–358.
Chin, A., A. H. Purcell, J. Quan & V. H. Resh, 2009a. Assessing geomorphological and ecological responses in restored step–pool systems. In James, L. A., S. L. Rathburn & G. R. Whittcar (eds), Management and Restoration of Fluvial Systems with Broad Historical Changes and Human Impacts: Geological Society of America Special Paper 451: 199–217.
Chin, A., S. Anderson, A. Collison, B. Ellis-Sugai, J. Haltiner, J. Hogervorst, G. M. Kondolf, L. S. O’Hirok, A. H. Purcell, A. L. Riley & E. Wohl, 2009b. Linking theory and practice for restoration of step–pool streams. Environmental Management 43: 645–661.
Chin, A., F. Gelwick, D. Laurencio, L. R. Laurencio, M. S. Byers & M. Scoggins, 2010. Linking geomorphological and ecological responses in restored urban pool–riffle streams. Ecological Restoration 28: 460–475.
Collier, K. J., 1994. Influence of nymphal size, sex and morphotype on microdistribution of Deleatidium (Ephemeroptera: Leptophlebiidae) in a New Zealand river. Freshwater Biology 31: 35–42.
Comiti, F., L. Mao, M. A. Lenzi & M. Siligardi, 2009. Artificial steps to stabilize mountain rivers: a post-project ecological assessment. River Research and Applications 25: 639–659.
Connolly, N. M., M. R. Crossland & R. G. Pearson, 2004. Effect of low dissolved oxygen on survival, emergence, and drift of tropical stream macroinvertebrates. Journal of the North American Benthological Society 23: 251–270.
Dolédec, S., N. Lamouroux, U. Fuchs & S. Mérigoux, 2007. Modelling the hydraulic preferences of benthic macroinvertebrates in small European streams. Freshwater Biology 52: 145–164.
Dudley, T. L. & C. M. D’Antonio, 1991. The effects of substrate texture, grazing and disturbances on macroalgae establishment in streams. Ecology 72: 297–309.
Dudley, T. L., S. D. Cooper & N. Hemphill, 1986. Effects of macroalgae on a stream invertebrate community. Journal of the North American Benthological Society 5: 93–106.
Dupuis, I. A., F. L. Bunnel & P. A. Friele, 2000. Determinants of the tailed frog’s range in British Columbia, Canada. Northwest Science 74: 109–115.
Edington, J. M., 1968. Habitat preferences in net-spinning caddis larvae with special reference to the influence of water velocity. Journal of Animal Ecology 37: 675–692.
Fuller, R. L. & R. J. Mackay, 1980. Field and laboratory studies of net-spinning activity by Hydropsyche larvae (Trichoptera: Hydropsychidae). Canadian Journal of Zoology 58: 2006–2014.
Gilpin, B. R. & M. A. Brusven, 1970. Food habits and ecology mayflies of the St. Mary’s River in Idaho. Melanderia 4: 19–40.
Gomi, T., R. C. Sidle & J. S. Richardson, 2002. Understanding processes and downstream linkages of headwater systems. Bioscience 52: 905–916.
Gore, J. A., J. B. Layzer & J. Mead, 2001. Macroinvertebrate instream flow studies after 20 years: a role in stream management and restoration. Regulated Rivers: Research and Management 17: 527–542.
Hall, R. O., E. S. Bernhardt & G. E. Likens, 2002. Relating nutrient uptake with transient storage in forested mountain streams. Limnology and Oceanography 47: 255–265.
Hansen, R. A., D. D. Hart & R. A. Merz, 1991. Flow mediates predator–prey interactions between triclad flatworms and larval black flies. Oikos 60: 187–196.
Hart, D. D. & C. M. Finelli, 1999. Physical–biological coupling in streams: the pervasive effects of flow on benthic organisms. Annual Reviews of Ecology and Systematics 30: 363–395.
Heede, B. H., 1981. Dynamics of selected mountain streams in the western United States of America. Zeitschrift fur Geomorphologie 25: 17–32.
Hoffmann, A. & V. H. Resh, 2003. Oviposition in three species of limnephiloid caddisflies (Trichoptera): hierarchical influences on site selection. Freshwater Biology 48: 1064–1077.
Holomuzki, J. R., P. C. Furey, R. L. Lowe & M. E. Power, 2013. Microdistributional variability of larval caddisflies in Mediterranean-climate streams in northern California. Western North American Naturalist 73: 261–269.
Hora, S. L., 1930. Ecology, bionomics, and evolution of the torrential fauna, with special reference to the organs of attachment. Philosophical Transactions of the Royal Society, Series B 218: 171–282.
Hynes, H. B. N., 1970. The Ecology of Running Waters. University of Toronto Press, Toronto.
Jacobsen, D., 2008. Low oxygen pressure as a driving factor for the altitudinal decline in taxon richness of stream macroinvertebrates. Oecologia 154: 795–807.
Judd, H. E., 1964. A study of bed characteristics in relation to flow in rough, high-gradient natural channels. Unpublished PhD Thesis, Utah State University, Logan.
Karlsson, O. M., J. S. Richardson & P. M. Kiffney, 2005. Modelling organic matter dynamics in headwater streams of south-western British Columbia, Canada. Ecological Modelling 183: 463–476.
Katano, I., H. Mitsuhashi, Y. Isobe, H. Sato & T. Oishi, 2005. Reach-scale distribution dynamics of a grazing stream insect, Micrasema quadriloba Martynov (Brachycentridae, Trichoptera), in relation to current velocity and periphyton abundance. Zoological Science 22: 853–860.
Katano, I., H. Doi, A. Houki, Y. Isobe & T. Oishi, 2007. Changes in periphyton abundance and community structure with the dispersal of a caddisfly grazer, Micrasema quadriloba. Limnology 8: 219–226.
Lancaster, J. & A. G. Hildrew, 1993. Flow refugia and the microdistribution of lotic macroinvertebrates. Journal of the North American Benthological Society 12: 385–393.
Lloyd, J. T., 1921. The biology of North America caddis fly larvae. Bulletin of the Lloyd Library 21: 1–124.
Lytle, D. A. & N. L. Poff, 2004. Adaptation to natural flow regime. TREE 19: 94–100.
Manuel, K. L. & T. C. Folsom, 1982. Instar sizes, life cycles, and food habits of five Rhyacophila (Trichoptera: Rhyacophilidae) species from the Appalachian Mountains of South Carolina, U.S.A. Hydrobiologia 97: 281–285.
McCune, B. & J. B. Grace, 2002. Analysis of Ecological Communities. MjM Software Design, Gleneden Beach.
Mérigoux, S. & S. Dolédec, 2004. Hydraulic requirements of stream communities: a case study on invertebrates. Freshwater Biology 49: 600–613.
Merritt, R. W., K. W. Cummins & M. B. Berg (eds), 2008. An Introduction to the Aquatic Insects of North America. Kendall-Hunt, Dubuque.
Mielke, P. W. & K. L. Berry, 2001. Permutation Methods: A Distance Function Approach. Springer Series in Statistics, Berlin.
Montgomery, D. R. & J. M. Buffington, 1997. Channel-reach morphology in mountain drainage basins. GSA Bulletin 109: 596–611.
Montgomery, D. R., E. M. Beamer, G. R. Pess & T. P. Quinn, 1999. Channel type and salmonid spawning distribution and abundance. Canadian Journal of Fisheries and Aquatic Sciences 56: 377–387.
Muehlbauer, J. D. & M. W. Doyle, 2012. Knickpoint effects on macroinvertebrates, sediment, and discharge in urban and forested streams: urbanization outweighs microscale habitat heterogeneity. Freshwater Science 31: 282–295.
Palmer, T. M., 1995. The influence of spatial heterogeneity on the behavior and growth of two herbivorous stream insects. Oecologia 104: 476–486.
Patterson, J. W. & R. L. Vannote, 1979. Life history and population dynamics of Heteroplectron americanum. Environmental Entomology 8: 665–669.
Peck, J. E., 2010. Multivariate Analysis for Community Ecologists: Step-by-Step Using PC-ORD. MjM Software Design, Gleneden Beach, 162 pp.
Poepperl, R., 2000. The filter feeders Hydropsyche angustipennis and H. pellucidula (Trichoptera: Hydropsychidae) in a Northern German lowland stream: microdistribution, larval development, emergence pattern, and secondary production. Limnologica: Ecology and Management of Inland Waters 30: 65–72.
Poff, N. L., 1997. Landscape filters and species traits: towards mechanistic understanding and prediction in stream ecology. Journal of the North American Benthological Society 16: 391–409.
Principe, R. E., G. B. Raffaini, C. M. Gualdoni, A. M. Oberto & M. C. Corigliano, 2007. Do hydraulic units define macroinvertebrate assemblages in mountain streams of central Argentina? Limnologica: Ecology and Management of Inland Waters 37: 323–336.
Purcell, A. H., C. Friedrich & V. H. Resh, 2002. An assessment of a small urban stream restoration project in northern California. Restoration Ecology 10: 685–694.
Sagnes, P., S. Mérigoux & N. Péru, 2008. Hydraulic habitat use with respect to body size of aquatic insect larvae: case of six species from a French Mediterranean type stream. Limnologica: Ecology and Management of Inland Waters 38: 23–33.
Scheuerlein, H., 1999. Morphological dynamics of step–pool systems in mountain streams and their importance for riparian ecosystems. In Jayawardena, A. W., J. M. Lee & Z. Y. Wang (eds), River Sedimentation: Theory and Applications. Balkema, Rotterdam: 205–210.
Smith, S. D., 1968. The Rhyacophila of the Salmon River drainage of Idaho with special reference to larvae. Annals of the Entomological Society of America 61: 655–674.
Smith, J. A. & A. J. Dartnall, 1980. Boundary layer control by water pennies (Coleoptera Psephenidae). Aquatic Insects 2: 65–72.
Statzner, B., 1981. The relation between “hydraulic stress” and microdistribution of benthic macroinvertebrates in a lowland running water system, the Schierenseebrooks (North Germany). Archiv für Hydrobiologie 91: 192–218.
Statzner, B., 1988. Growth and Reynolds number of lotic macroinvertebrates: a problem for adaptation of shape to drag. Oikos 51: 84–87.
Statzner, B. & D. Borchardt, 1994. Longitudinal patterns and processes along streams: modelling ecological responses to physical gradients. In Giller, P. S., A. G. Hildrew & D. G. Raffaelli (eds), Aquatic Ecology: Scale, Pattern and Process. Blackwell Scientific Publications, Oxford: 113–140.
Statzner, B. & B. Higler, 1986. Stream hydraulics as a major determinant of benthic invertebrate zonation patterns. Freshwater Biology 16: 127–139.
Statzner, B., N. Bonada & S. Dolédec, 2008. Predicting the abundance of European stream macroinvertebrates using biological attributes. Oecologia 156: 65–73.
Tullos, D. D., D. L. Penrose, G. D. Jennings & W. G. Cope, 2009. Analysis of functional traits in reconfigured channels: implications for the bioassessment and disturbance of river restoration. Journal of the North American Benthological Society 28: 80–92.
United States Department of Agriculture (USDA) Web Soil Survey [available on internet at http://websoilsurvey.sc.egov.usda.gov/]. Accessed 2 December 2014.
Usseglio-Polatera, P., M. Bournaud, P. Richoux & H. Tachet, 2000. Biomonitoring through biological traits of benthic macroinvertebrates: how to use species trait databases? Hydrobiologia 422: 153–162.
Vogel, S., 1994. Life in Moving Fluids: The Physical Biology of Flow, 2nd ed. Princeton University Press, Princeton.
Vogel, R. M. & C. N. Kroll, 1992. Regional geohydrologic–geomorphic relationships for the estimation of low-flow statistics. Water Resources Research 28: 2451–2458.
Wang, Z., C. Melching, X. Duan & G. Yu, 2009. Ecological and hydraulic studies of step–pool systems. Journal of Hydraulic Engineering 135: 705–717.
Weissenberger, J., H. Spatz, A. Emanns & J. Schwoerbel, 1991. Measurement of lift and drag forces in the m N range experienced by benthic arthropods at flow velocities below 1.2 m s−1. Freshwater Biology 25: 21–31.
Welsh, H. H. & L. M. Ollivier, 1998. Stream amphibians as indicators of ecosystem stress: a case study from California’s redwoods. Ecological Applications 8: 1118–1132.
Wichard, W., W. Arens & G. Eisenbeis, 2002. Biological Atlas of Aquatic Insects. Apollo Books, Stenstrup.
Wohl, E. E., S. Madsen & L. MacDonalds, 1997. Characteristics of log and clast bed-steps in step–pool streams of northwester Montana, USA. Geomorphology 20: 1–10.
Wolman, M. G., 1954. A method of sampling coarse river-bed material. American Geophysical Union Transactions 35: 951–956.
Yu, G. A., Z. Y. Wang, K. Zhang, X. H. Duan & T. C. Chang, 2010. Restoration of an incised mountain stream using artificial step–pool system. Journal of Hydraulic Research 48: 178–187.
Zhang, Y., 1996. Life history of Hydatophylax intermedius (Trichoptera, Limnephilidae) in Hokkaido, northern Japan. Aquatic Insects 18: 223–231.
Zimmermann, A. E., 2013. Step–pool channel features. In Shroder, J. F. & E. Wohl (eds), Treatise on Geomorphology, V 9, Fluvial Geomorphology. Academic, San Diego: 346–363.
Acknowledgments
We thank students Anibal Florez, Tobin Weatherson, Johnathan Baer, and Amanda Baca for assistance with field and laboratory work; Bob Wisseman, Aquatic Biology Associates, Inc., for identification of the benthic macroinvertebrates and metric calculation. We also thank Omar Abi-Chahine for graphic support and Kimberley Johnson, Tyrone Kelley, and Mike McCain (Six Rivers National Forest) for their assistance with the sampling permit and locating suitable field sites. Field research was conducted under US Forest Service Permit #GAS-34. The authors also thank Alexander Purcell, Patina Mendez, Raphael Mazor, and the three Reviewers whose advice helped improve this paper. This research was funded in part by the National Science Foundation Grant #1145469.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Marcelo S. Moretti
Rights and permissions
About this article
Cite this article
O’Dowd, A.P., Chin, A. Do bio-physical attributes of steps and pools differ in high-gradient mountain streams?. Hydrobiologia 776, 67–83 (2016). https://doi.org/10.1007/s10750-016-2735-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2735-5