Abstract
The Water Framework Directive 2000/60/EC (WFD) requires the analysis of biological elements of aquatic ecosystems to assess water quality. Diatoms are the component of the periphyton most commonly used to classify lotic environments. Within the context of the WFD the concept of ‘reference conditions’ was introduced and biological quality of watercourses is expressed as Ecological Quality Ratio (EQR). This study was carried out in Alto Adige/Südtirol (Province of Bolzano-Bozen, northern Italy), belonging to the Alpine eco-region, and to the hydro-ecoregion Inner Alps. During 2006–2009, epilithic diatoms were sampled from monitoring and reference sites of seven stream types. Diatom assemblages were analysed with TWINSPAN and CCA analyses to investigate species association and distribution in relation to stream characteristics. Altitude and geology resulted to be the most important factors influencing diatom assemblage composition, and were used to describe new stream types. Indicator species analysis was used to characterize reference assemblages. The biological quality of watercourses was assessed using different diatom indices: Specific Pollution sensitivity Index (IPS), Eutrophication and Pollution Index with Diatoms (EPI-D), Trophic Index (TI). We tested also the Intercalibration Common Metric index (ICM).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The European Water Framework Directive 2000/60/EC (WFD) (Council of the European Communities, 2000) recommends to member states to evaluate the ecological status of running waters through the analyses of the biological elements of the aquatic ecosystem. Benthic diatoms are the most frequently used algal group, due to their numerical dominance in the periphyton composition, and to the advanced scientific knowledge about species ecological preferences. In several European countries diatom indices were developed or existent ones were modified to be compliant with the WFD (for a summary of different national indices see Ector & Rimet, 2005).
In Italy, an index compliant with the WFD was not developed, and the Intercalibration Common Metric (ICM) index was proposed to implement the directive (Mancini & Sollazzo, 2009; van de Bund, 2009).
The WFD requires that for each water body type, reference conditions have to be established, representing that body type at high ecological status. The definition of surface water body types was performed in Europe for different geographic scales: ecoregions (Annex XI), hydro-ecoregions and water body types, with the aim to identify geographic areas with similar environmental conditions, which could be characterized by similar biotic assemblages. This classification could be defined as ‘a priori’, since water body types are defined according to abiotic parameters, without considering biotic communities.
But at the end of the intercalibration exercise it was found that differences between water body types were not always reflected by different diatom assemblages, and in some geographic areas typologies were redefined ‘a posteriori’, on the basis of diatom species ordination.
In Italy, 21 hydro-ecoregions were established (Buffagni et al., 2006; Wasson et al., 2006), and the identification of different stream types following abiotic parameters suggested by Buffagni et al. (2006), such as hydro-ecoregion, distance from source and regime (glacial or not glacial), led to the definition of tens of typologies for each Italian region, implying also the necessity to find their reference sites. This fragmentation can be considered useful and necessary only if characteristic biotic communities validate the typologies. This means that the ‘a priori’, abiotic classification of water bodies has to be verified: different stream typologies could be characterized by the same diatom assemblages if the selected abiotic factors are not the ones influencing diatom communities.
Diatoms of Alto Adige/Südtirol (N-E Italy) were mainly investigated in the nineteenth century (Grunow, 1860; Milde, 1864a, b; Schröder, 1895) and only few other studies were recently carried out (Cappelletti et al., 2007; Beltrami et al., 2008b, 2009; Lösch, 2008), while a first application of diatoms for biological monitoring started in 2007 (Lösch & Alber, 2009).
The aim of this study was (i) to describe diatom assemblages of running waters flowing in Alto Adige/Südtirol; (ii) to verify if there is a correspondence between established surface body types and diatom assemblages; (iii) to describe diatom assemblages of reference conditions for different stream types; (iv) to evaluate biological quality of sampled sites with different diatom indices; and (v) to test the application of the ICM for the first time.
Materials and methods
Study area
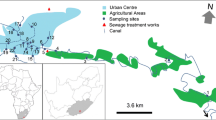
Alto Adige/Südtirol (Province of Bolzano-Bozen) is located in the North-East of Italy in the Alpine region bordering Austria. It belongs to the ecoregion Alps, hydro-ecoregion 3—Inner Alps (HER 3). Three intercalibration river types were found: Alpine calcareous (A1), Alpine siliceous (A2), and Central (C) (Buffagni et al., 2008). The Environmental Agency (EA) classified water body types in nine stream types (EA types) according to origin-flow regime and distance from source (Table 1). Reference sites were selected for each EA type, following the guidelines proposed by Buffagni et al. (2007). A total of 56 sites were investigated (Fig. 1), belonging to seven EA types (Table 1): 11 reference sites and 45 sites belonging to the monitoring network of the EA.
Environmental variables
Water samples for chemical analyses were collected by the EA of Bolzano-Bozen, stored in thermic boxes and brought to the laboratory. Temperature was measured in the field. Laboratory analyses were carried out following standard methods (ISO, 1984; UNI, 1994, 2004; APHA, 1998; APAT-IRSA/CNR, 2003; APPA BZ, 2004) for the measurement of pH, conductivity, % oxygen saturation, carbonate hardness, total suspended solids (TSS), biological oxygen demand (BOD5), orthophosphates (P-PO4 3−), total phosphorus (TP), ammonium (NH4 +), total nitrogen (TN), and Escherichia coli contamination (E. coli).
Diatom sampling, laboratory analyses and bio-indication methods
Benthic diatoms were collected from 2006 to 2009. Monitoring sites were sampled once, while reference sites were sampled in different seasons: a total of 67 samples were collected.
Sampling and samples processing were carried out following European standard methods (Kelly et al., 1998; European Committee for Standardization, 2003). Slides were analysed with a light microscope at 1,000× magnification, and diatoms were identified at the lowest possible taxonomic level following Krammer & Lange-Bertalot (1986, 1988, 1991a, b), Krammer (1997a, b, 2002, 2003), Reichardt (1999), and Lange-Bertalot (2001).
For the calculation of diatom indices at least 400 valves were counted for each slide (European Committee for Standardization, 2004). The software OMNIDIA 4.2 (available at http://omnidia.free.fr) was used to calculate the following indices: Specific Pollution sensitivity Index (IPS; Cemagref, 1982), Eutrophication and Pollution Index with Diatoms (EPI-D; Dell’Uomo, 2004), and Austrian Trophic Index (TI; Rott et al., 1999). These indices were chosen since they are widely used in Europe, and because they were considered the most reliable for the geographic region analysed in this study.
The ICM (Kelly et al., 2009) was calculated using the formula:
The calculation of EQR_IPS and EQR_TI requires reference values for IPS and TI. Mancini & Sollazzo (2009) proposed some values based on their data set (TI* and IPS*), and limits of quality classes were established by the intercalibration exercise and differ for the typologies A1, A2, and C (Table 2; Mancini & Sollazzo, 2009; van de Bund, 2009). In this study, we tested also reference values obtained from our reference sites (median; TI** and IPS** in Table 8).
Data analyses
In order to verify if specific diatom assemblages correspond to the stream types defined ‘a priori’ (EA types), a divisive hierarchical classification (TWINSPAN) was performed with PC-ORD (pseudo species cut levels: 0.00; 0.02; 0.05; 0.1; 0.2; McCune & Mefford, 1999). This classification of sites is purely based on diatom assemblages, without considering environmental variables. The significance of the identified groups was tested with the multi-response permutation procedure (MRPP, Mielke, 1984; Mielke & Berry, 2001) performed with PC-ORD. The Sørensen–Bray Curtis distance was used as the distance measure between matrices.
Groupings obtained were then compared with results of multivariate analyses, performed to explore relationships between diatoms and environmental variables. The species matrix was reduced by the exclusion of rare species (relative abundance <1%) and species abundance was log transformed. Multivariate analyses were performed using CANOCO 4.0 (ter Braak & Smilauer, 1998). The CCA ordination was selected according to the length of the environmental gradient (>3), as obtained from preliminary detrended correspondence analyses (DCA). A manual forward selection based on Monte Carlo permutation tests was performed to retain only the most important variables (P < 0.05). The significance of eigenvalues of the CCA was tested by Monte Carlo permutation tests (n = 1,000).
A non-parametric Kruskal–Wallis test with multiple comparisons of ranks was performed with the software STATISTICA 8, in order to evaluate the environmental parameters statistically different between the groups.
According to results obtained, new stream types were established and were used as grouping variable for the indicator species analysis (ISA, Dufrêne & Legendre, 1997; PC-ORD software), performed to identify reference assemblages, using species abundance of reference samples.
Results
The main physico-chemical variables, grouped according to EA types, are summarized in Table 3. Sites cover a wide altitudinal range (210–1,620 m a.s.l.) and pH and conductivity values (respectively, from 7 to 8.4 and from 44 to 648) are characteristic of siliceous and calcareous substrata. The trophic status ranges from oligotrophic to poly-eutrophic level (TP: 0.01–0.23 mg l−1), while BOD5 and E. coli contamination reveal the presence of organic pollution mainly due to farming activity in the surrounding area.
Diatoms identified and ecological preferences
A total of 177 species were identified. Species recorded in more than 3% of samples are listed in Appendix 1—Supplementary Material. Achnanthidium minutissimum (Kützing) Czarnecki was present in every sample and often dominant in abundance, reaching up to 83% of relative abundance. Achnanthidium pyrenaicum (Hustedt) H. Kobayasi, Encyonema minutum (Hilse) D.G. Mann and Fragilaria vaucheriae (Kützing) J.B. Petersen were present in more than 75% of sites. The reference site REF 8 showed the presence of species such as Brachysira neoexilis Lange-Bertalot, Encyonopsis minuta Krammer & E. Reichardt and Fragilaria delicatissima (W. Smith) Lange-Bertalot, common in lake benthic communities. For this reason, we decided to consider this site not reliable as reference site, as its community was probably influenced by the upstream lake. Anyway, it was not excluded in the general analyses, and was considered as a monitoring site.
It is also worth mentioning the presence of Didymosphenia geminata (Lyngbye) M. Schmidt in 16 sites, including also reference sites.
Among the rare species, present only at few sites with few specimens, some are listed in the German Red List (Lange-Bertalot & Steindorf, 1996) in the endangered categories (Table 4). Other taxonomically interesting species, recently described from European watercourses, were also found: Achnanthidium atomoides O. Monnier, Lange-Bertalot & Ector (Monnier et al., 2004), A. temniskovae Ivanov & Ector (Ivanov & Ector, 2006; Beltrami et al., 2009), Nitzschia alicae Hlúbiková & Ector, and N. puriformis Hlúbiková & Ector (Hlúbiková et al., 2009).
Diatom assemblages
TWINSPAN classification led to the separation of samples into four main groups, and their principal physical and chemical characteristics are summarized in Table 5. Groups 1 and 2 are characterized by Alpine and mid-altitude sites, respectively, calcareous and siliceous, with low nutrient contents. Group 3 is mainly represented by low altitude sites, with medium–high conductivity and hardness, and showing nutrient enrichment. Group 4 comprises low-land ditches, with very high saline contents (conductivity and hardness), and high nutrient concentration.
In each group different EA types can be found. Group 4 was almost only composed of ditches (EA type 0). MRPP performed on TWINSPAN groups was highly significant (A: 0.08, P = 0.000). These results were compared with the classification obtained by multivariate analyses.
The CCA explained 19.8% of total variance in species assemblages, and the first three axes accounted for 79% of it. By the forward selection out of all the available environmental variables, altitude, conductivity, pH and P-PO4 3− were selected as the most significant ones. The Monte Carlo test was highly significant for the first and for all canonical axes (P < 0.001). The first axis was correlated with nutrient enrichment (positively) and with altitude (negatively); the second axis was negatively correlated with pH (Fig. 2a). Samples were represented in the scatter plot by symbols referring to EA types and TWINSPAN groups (Fig. 2b, c). There is a good correspondence between EA type 0 (ditches) and TWINSPAN Group 4, and they showed a positive correlation with P-PO4 3− and conductivity. The other EA types were not distinguished along the axes.
Ordination plot based on Canonical Correspondence Analyses (CCA, λ1: 0.344, λ2: 0.184). a Scatter plot of environmental variables selected by forward selection and diatom species (minimum fit: 10). For diatom species codes, see Appendix 1—Supplementary material; b scatter plot of sites represented according to EA types. Type 0: +; Type 1: ○; Type 2: □; Type 7: ∆; Type 8: ●; Type 14: ∇; Type 18: ▼; c scatter plot of sites represented according to TWINSPAN groups. Group 1: □; Group 2: ∆; Group 3: ◊; Group 4: ○; filled symbols: reference sites
On the contrary, TWINSPAN Groups 1 and 2 were positively correlated with altitude and negatively with P-PO4 3−. Groups 1 and 3 had a positive relation with pH.
In Fig. 2c, reference sites (black symbols) are generally located on the left side of monitoring sites belonging to their group. In Group 1, the reference samples located on the right part of the group are references of the low-land typology, and are indeed very close to Group 3.
CCA scatter plot could also be used to highlight some incongruences of the TWINSPAN classification (e.g. squares among triangles).
Species distribution in relation to chemical variables is shown in Fig. 2a, where the species with a minimum fit of 10 are shown. Achnanthidium pyrenaicum, Cymbella compacta Oestrup, C. excisa Kützing, Delicata delicatula (Kützing) Krammer, Diatoma moniliformis Kützing, Didymosphenia geminata, Gomphonema pumilum (Grunow) E. Reichardt & Lange-Bertalot, and G. olivaceum (Hornemann) Brébisson were associated with high pH and altitude, and low nutrient concentrations. Psammothidium daonense (Lange-Bertalot) Lange-Bertalot, A. subatomus (Hustedt) Lange-Bertalot, Diatoma mesodon (Ehrenberg) Kützing, Fragilaria arcus (Ehrenberg) Cleve, G. olivaceum var. olivaceoides (Hustedt) Lange-Bertalot, and Tetracyclus rupestris (Braun) Grunow were highly correlated with low pH and were more abundant in high altitude sites. Achnanthidium atomoides, Cocconeis euglypta Ehrenberg, C. lineata Ehrenberg, Navicula cryptotenella Lange-Bertalot, N. gregaria Donkin, N. tripunctata (O.F. Müller) Bory were associated with high pH but and increasing values of P-PO4 3−. Planktonic species as Cyclotella meneghiniana Kützing, C. distinguenda Hustedt, C. costei Druart & F. Straub, and pollution tolerant species such as Eolimna minima (Grunow) Lange-Bertalot, and Navicula veneta Kützing were more abundant in ditches, characterized by high conductivity and P-PO4 3− values.
Differences in the physical and chemical features of the four TWINSPAN groups were statistically verified with Kruskal–Wallis multiple comparisons (Table 6). Group 2 differed from all the others by pH and hardness, parameters that suggest a different geology of the catchments, siliceous for Group 2, calcareous (or alluvial) for the other groups. Groups 1 and 2 shared a similar altitudinal range, different from that of Groups 3 and 4. Group 2 differed from Groups 3 and 4 also for the nutrient concentration.
Some of the frequent, endangered or taxonomically interesting species mentioned above are shown in Fig. 3.
Some characteristic diatom species (scale bar: 10 μm). 1: Eucocconeis laevis; 2, 3: Nupela lapidosa; 4: Achnanthidium pyrenaicum; 5: A. minutissimum; 6–7: A. temniskovae, raphe valve and raphe-less valve; 8, 9: A. lineare, raphe valve and raphe-less valve; 10, 11: A. atomoides, raphe valve and raphe-less valve; 12, 13: Tetracyclus rupestris, valve and septum; 14, 15: A. subatomus, raphe valve and raphe-less valve; 16: Aulacoseira alpigena; 17: Cyclotella meneghiniana; 18: Cocconeis placentula s.l.; 19: C. lineata; 20: C. euglypta; 21: Gomphonema pumilum s.l.; 22: Cyclotella costei; 23: C. distinguenda; 24: Encyonema silesiacum; 25: Delicata delicatula; 26: E. minutum; 27: Gomphonema olivaceum; 28: Navicula veneta; 29: N. cryptotenella; 30: Brachysira neoexilis and Encyonopsis minuta; 31: N. gregaria; 32: N. tripunctata; 33: Reimeria sinuata; 34: Diatoma mesodon; 35: Nitzschia fonticola; 36: N. inconspicua; 37: Cymbella neocistula; 38: C. compacta; 39: C. excisa; 40: Fragilaria arcus; 41: F. vaucheriae s.l.; 42: N. alicae; 43: N. dissipata; 44: N. puriformis
Reference communities
The analysis of the floristic list of reference samples was used to identify ‘reference species’ and specific assemblages for the typologies identified with TWINSPAN and CCA analyses: calcareous alpine and mid-altitude sites; siliceous alpine and mid-altitude sites; low-land and low altitude sites; low-land ditches.
Achnanthidium minutissimum was present in all samples. Achnanthidium pyrenaicum, Diatoma mesodon, Encyonema silesiacum (Bleisch) D.G. Mann, and Fragilaria arcus were very frequent in Groups 1, 2 and 3 (more than 60% of the sites of each group), but were absent in Group 4 (ditches). ISA was used to identify characteristic species for each group. It was not performed for ditches typology, as only one sample was available as reference of that group. For each group, the indicator species (P < 0.05), frequent species (recorded in more than 60% of sites of each group) and some peculiar species (exclusively found in a single group in at least 30% of sites) were listed (Table 7). Ditches’ community was described by the species found with a relative abundance >2%.
Water quality and ecological status assessment
Results obtained by the application of diatom indices were presented in Tables 8 and 9. According to IPS and EPI-D, sites were assessed as of good or very good quality (I and II class); only few sites, generally belonging to the ditch typology, were more polluted (IV class). TI has a finer scale and sometimes was stricter regarding classification of sites. For example, the class I of IPS corresponded to TI ranging from oligotrophic to mesotrophic–eutrophic. The most impacted sites (classes III of IPS and IV of EPI-D) were assessed as eutrophic–polytrophic by TI.
Regarding reference sites (Table 9), their quality was assessed by all diatom indices as very good. Also in this case, the worst judgement was given by TI for the EA Type 18 (low-land).
For ICM calculation, we had at first to divide our samples in the intercalibration types present in the hydro-ecoregion 3: A1, A2, and C, for which reference values of IPS and TI are available. The parameters shown in Table 2 were modified on the base of the classification results, and we used the following criteria:
-
A1: calcareous hill-mountain streams, altitude >500 m, pH >8, conductivity >150 μS cm−1, hardness >10°F;
-
A2: siliceous hill-mountain streams, altitude >500 m, pH <8, conductivity <200 μS cm−1, hardness <8°F;
-
C: low-land streams/rivers and ditches, altitude <400 m.
Results of the application of ICM are summarized in Table 8. Results obtained using reference values suggested by Mancini & Sollazzo (2009) (TI* and IPS*), and the median values of our reference sites (TI** and IPS**) can be compared. To calculate the Ecological Quality Ratio (EQR) of ditches, we compared the IPS and TI values of C type with the values of the ditch reference site.
Discussion
Interesting species distribution
The development of assessment methods compliant with the European WFD needs at first a good knowledge of species distribution and ecology.
This study revealed the presence in Alto Adige/Südtirol watercourses of some interesting, recently described species (Fig. 3): Achnanthidium atomoides, A. temniskovae, Nitzschia alicae, and N. puriformis.
Achnanthidium temniskovae was recorded only in the first two sites along Adige river, characterized by a mesotrophic status, while A. atomoides was generally present in low-land streams in meso-eutrophic conditions. Nitzschia alicae was typically found in siliceous reference sites (high water quality) at about 800 m a.s.l. Nitzschia puriformis had a wider distribution and was found in oligotrophic as well as in meso-eutrophic conditions, both in calcareous and siliceous areas. The presence of these species in Alto Adige/Südtirol gave additional data on their distribution and ecology in Europe (Hlúbiková et al., 2009).
Also, the presence of the colonial diatom Didymosphenia geminata, already recorded in the bordering Province of Trento (Beltrami et al., 2008a, c), has to be highlighted as it is studied in many countries as a possible invasive species (Blanco & Ector, 2009; Whitton et al., 2009). Its first record in Alto Adige/Südtirol was noticed in 2002 (Cappelletti et al., 2007; Beltrami et al., 2008b). Since 1999 a total of 77 sites were sampled, and D. geminata was observed in 38 of them. In particular, in 16 of them it was not present during the first survey in 1999–2000 and it could be supposed that it is currently spreading in the Province of Bolzano-Bozen, but usually with low relative abundances.
Diatom assemblages: ‘a priori’ vs ‘a posteriori’ classification
The TWINSPAN analysis was really useful to verify the ‘a priori’ classification of water bodies, since environmental parameters were not considered, and sites were grouped only according to assemblage similarity (biotic approach). Our results showed that the EA classification was not reflected in the CCA ordination, while TWINSPAN groups were more consistent with it (Fig. 2b, c). CCA and Kruskal–Wallis multiple comparisons were used to detect which parameters were more significant to discriminate the new groups. In particular, the CCA (Fig. 2a) revealed that the main factors were related to altitude, geology (pH), and nutrient enrichment (P-PO4 3−). The parameters chosen to identify EA types, instead, did not seem to discriminate diatom assemblages: distance from source and suspended solids (a proxy of glacial regime) were not selected by forward selection. Altitude is a parameter indirectly correlated with several others, such as temperature, current velocity, human impact, and is an important structuring parameter for diatom communities (Rimet et al., 2003). The same could be stated for distance from source. But in our results only altitude was significant, and for example in Group 3 sites with highly variable distance from source (8–118 km), but similar altitude (212–520 m a.s.l.) were found.
Mountain streams (Groups 1 and 2) are characterized by adnate species or attached to the substratum by short stalks (Achnanthidium, Diatoma, Fragilaria, small Gomphonema species), typical of turbulent waters and unstable environments (pioneer species) (Rimet et al., 2007). On the contrary low-land types (Groups 3 and 4) showed a high portion of the genera Navicula and Nitzschia with motile species, generally associated with finer substratum, steady flows, and more mature communities. Both altitude and distance from source could represent this ‘up stream–down stream’ gradient, but we can suppose that in the Alpine region turbulence and substratum size are more correlated to altitude rather than distance from source, while probably in low-land regions distance from source could be more significant. Incongruence between abiotic and biotic typifications of watercourses was observed by Ní Chatháin et al. (2004), and also their results showed that conductivity and alkalinity were the most important factors influencing communities of benthic diatoms.
Geology is considered one of the most important parameter influencing diatom species distribution (Rimet et al., 2004; Tison et al., 2005; Grenier et al., 2006; Rimet, 2009). One of the main bias in the classification of river types of Alto Adige/Südtirol, has been to consider all the province area as belonging to the hydro-ecoregion Inner Alps (siliceous), while some dolomitic areas are well represented, and also low-land types (Groups 3 and 4), running in alluvial catchments, have quite high pH, hardness and conductivity values.
The groups obtained with our analyses showed to be much more consistent with the intercalibration types A1, A2, and C (Buffagni et al., 2008; van de Bund, 2009). Even if the ditches (Group 4) could be considered together with low-land streams (Group 3) in the category C, we preferred to keep them separated. In fact, both TWINSPAN and multivariate analyses showed that they were quite distant from Group 3 and they were characterized by very high hardness and conductivity, and by a slow flow, as suggested by the presence of planktonic centric species (Cyclotella meneghiniana, C. distinguenda, C. costei).
Our results confirmed that altitude and geology are the main environmental factors influencing diatom communities (Tison et al., 2005). Broader categories of running waters (i.e. hydro-ecoregions, intercalibration typologies) could be sufficient in reflecting diatom variability, but for Alto Adige/Südtirol hydro-ecoregions should be revised considering the presence of calcareous substrata and low-land watercourses.
Reference communities
Reference assemblages characteristic of the typologies identified in this study are summarized in Table 7.
Diatoma mesodon and Fragilaria arcus, considered acidophilous by Rott et al. (1999) and, respectively, neutrophilous and alkaliphilous by Van Dam et al. (1994), were present in all reference samples, except for ditches. Their distribution seems to be more related to high water quality and fast flowing waters, rather than geology, and in fact they are typically found in many mountain streams (Round, 1991; Cantonati, 1998; Rimet et al., 2004; Gomà et al., 2005; Levkov et al., 2005; Tison et al., 2005; Cantonati et al., 2006; Bona et al., 2007). These two species could be considered as reference species for all stream types, even if, considering all the samples, they were more frequent in siliceous watercourses (Fig. 2a).
Calcareous mountain streams were characterized by the species Achnanthidium lineare W. Smith and Gomphonema pumilum, and among the frequent species we can mention alkaliphilous oligo-mesotrophic (Van Dam et al., 1994; Rott et al., 1999) ones such as Cymbella compacta and G. olivaceum. Achnanthidium pyrenaicum was often dominant in relative abundance.
In siliceous mountain streams, the indicator species A. minutissimum was generally dominant, but the taxonomy of this species is so complex that we cannot exclude the presence of some related species. Common was the presence of Encyonema minutum, Fragilaria vaucheriae s.l. and F. rumpens (Kützing) Carlson. Many acidophilous oligosaprobic/oligotrophic species were exclusive of this group: G. olivaceum var. olivaceoides, Nitzschia alicae, and more rarely A. subatomus, Aulacoseira alpigena (Grunow) Krammer, Diatoma hyemalis (Roth) Heiberg, Eucocconeis laevis (Oestrup) Lange-Bertalot, Meridion circulare (Greville) C. Agardh, Psammothidium daonense, and Tetracyclus rupestris.
The reference assemblage of low-land streams was characterized by more tolerant and alkaliphilous species: Cocconeis euglypta, C. pediculus Ehrenberg, D. moniliformis Kützing, D. vulgaris Bory, F. vaucheriae s.l., Navicula antonii Lange-Bertalot, N. cryptotenella, N. gregaria, N. lanceolata (C. Agardh) Ehrenberg, N. tripunctata, Nitzschia dissipata (Kützing) Grunow, N. inconspicua Grunow, and Rhoicosphenia abbreviata (C. Agardh) Lange-Bertalot. This group had many indicator species (Table 7) as its species composition was very different from the one of mountain watercourses, and almost all the indicator species were indeed exclusively found in this group.
In Alto Adige/Südtirol, A. minutissimum, F. rumpens, G. olivaceum, G. parvulum Kützing s.l., G. pumilum s.l., N. fonticola Grunow are meso-eutraphentic species typical of ditches at high quality status.
Many species found in reference sites are listed in the German Red List of diatoms (Lange-Bertalot & Steindorf, 1996; Table 4) and it highlights even more the necessity to preserve the watercourses in pristine conditions and high quality.
The defined reference assemblages are in accordance with studies of other authors regarding diatoms of mountain streams (Rimet et al., 2003, 2007; Schaumburg et al., 2004; Gomà et al., 2005; Tison et al., 2005). Anyway more sites should be investigated, especially for the calcareous areas.
Water quality and diatom indices
Monitoring sites were assessed with different diatom indices: IPS, EPI-D, TI, and ICM (Tables 8, 9). IPS and EPI-D were generally in good accordance, and the majority of sites were assessed as of class I or II. Only three sites were considered of class III (moderate quality) by IPS, corresponding to a III–IV or IV for EPI-D (moderate to bad quality). TI was more sensitive and distinguished different trophic levels, ranging from oligotrophic to eu-polytrophic. In some cases, TI classification was much more rigorous than IPS and EPI-D, assessing as meso-eutrophic sites considered of class I by the other two indices. These discrepancies were also found in other studies (Picińska-Fałtynowicz, 2007; Beltrami et al., 2009), and indeed the indices could be considered as complementary: TI was more sensitive at low levels of impact, IPS at higher pressures. For this reason, during the intercalibration exercise member states decided to integrate IPS and TI results in a multimetric index, the ICM (Kelly et al., 2009). Results of ICM calculation strictly depend on the values of IPS and TI chosen as reference for each type. The values obtained by our data set were of better quality than those proposed by Mancini & Sollazzo (2009), and so our ICM (ICM**) was stricter, and often more in accordance with the TI trophic status. In particular, in the C typology, ICM* assessed as high quality also sites of II or III IPS class, eutrophic or eu-polytrophic, while according to ICM** they were ‘good’. A higher disagreement was found in ditch typology: using TI** and IPS** values referring to our reference site, ICM** was always lower than ICM*, leading to worst quality judgments.
These results highlight the necessity of a more detailed study of reference sites, and a good selection of them. This is particularly difficult for low-land rivers where reference conditions are rare and often the ‘least impacted’ sites are chosen instead. A natural eutrophication along the longitudinal gradient can be accepted, but if reference sites are eutrophic, only the most impacted situations would be detected. For example in our data set, too many sites of C typology have ICM values higher than 1. This means that their quality was better than their reference and it could suggest that the references were not well chosen.
The importance of geology in structuring diatom assemblages will not emerge if the water quality assessment will be carried out only using trophic indices. Considering that the WFD requires the evaluation of ‘taxonomic composition and abundance of aquatic flora’, an index really compliant with WFD could take in consideration, for example, also the percentage of reference species found at monitoring sites, in relation to their ecoregional and geological preferences. This methodology is already used in Austria and Germany, both for algae and macrophytes (Schaumburg et al., 2004; Pfister & Pipp, 2009), but it needs a good data set of diatom reference assemblages.
Conclusions
The analyses of diatoms of Alto Adige/Südtirol revealed that the Italian stream typologies identified using abiotic approaches are not validated by diatom assemblages (at least for the Alpine typologies). Altitude and geology are the main factors structuring benthic diatom communities, and we suggest that stream types should be defined according to these parameters. In our study, four main types were identified: calcareous hill-mountain streams, siliceous hill-mountain streams, low-land watercourses, and low-land ditches. The definition of reference assemblages for each group could be a first step toward the formulation of a new metric that could take in consideration the presence/absence of reference species, following the example of other European countries. This study is also a first application of the ICM in Italy. Some problems occurred: first of all, reference values of IPS and TI, as well as class boundaries refer to intercalibration types, which are not clearly defined. Based on our results, we proposed some values of conductivity, pH, hardness to classify sites in A1, A2 and C types, but their reliability should be verified also in other regions. Second, the selection of good diatom reference sites can be difficult especially for low-land river types, where pristine conditions cannot be found and are often replaced by ‘least impacted’ sites. Attention must be paid to reference sites selection because the decision to use impacted sites as reference sites could lead to the overvaluation of the ecological status of monitoring sites.
References
APAT-IRSA/CNR, 2003. Metodi analitici per le acque. APAT Manuali e Linee Guida 29/2003. Roma: 1153 pp.
APHA, 1998. Standard Methods for the Examination of Water and Wastewater, 20th ed. American Public Health Association, Washington, DC: 1220 pp.
APPA BZ, 2004. Standard Operating Procedure SOP-D5.2-016. Bozen: 7 pp.
Beltrami, M. E., S. Blanco, F. Ciutti, C. Cappelletti, C. Monauni, S. Pozzi, F. Rimet & L. Ector, 2008a. Distribution and ecology of Didymosphenia geminata (Lyngbye) M. Schmidt (Bacillariophyta) in Trentino watercourses (Northern Italy). Cryptogamie Algologie 29: 141–160.
Beltrami, M. E., C. Cappelletti & F. Ciutti, 2008b. Didymosphenia geminata (Lyngbye) M. Schmidt (Bacillariophyta) in the Danube basin: new data from the Drava river (northern Italy). Plant Biosystems 142: 126–129.
Beltrami, M. E., C. Cappelletti, F. Ciutti, L. Hoffmann & L. Ector, 2008c. The diatom Didymosphenia geminata: distribution and mass occurrence in the province of Trento (Northern Italy). Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 30: 593–597.
Beltrami, M. E., F. Ciutti, C. Cappelletti, E. Rott & L. Ector, 2009. Epilithic assemblages of a large river in northern Italy (Adige River). Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 30: 915–918.
Blanco, S. & L. Ector, 2009. Distribution, ecology and nuisance effects of the freshwater invasive diatom Didymosphenia geminata (Lyngbye) M. Schmidt: a literature review. Nova Hedwigia 88: 347–422.
Bona, F., E. Falasco, S. Fassina, B. Griselli & G. Badino, 2007. Characterization of diatom assemblages in mid-altitude streams of NW Italy. Hydrobiologia 583: 265–274.
Buffagni, A., M. Munafò, F. Tornatore, I. Bonamini, A. Didomenicantonio, L. Mancini, A. Martinelli, G. Scanu & C. Sollazzo, 2006. Elementi di base per la definizione di una tipologia per i fiumi italiani in applicazione della direttiva 2000/60/EC. IRSA-CNR Notiziario dei Metodi Analitici 1: 2–19.
Buffagni, A., S. Erba, C. Mignuoli, G. Scanu, C. Sollazzo & R. Pagnotta, 2007. Criteri per la selezione di siti di riferimento fluviali per la Direttiva 2000/60/CE. IRSA-CNR. Brugherio: 27 pp.
Buffagni, A., S. Erba & R. Pagnotta, 2008. Definizione dello stato ecologico dei fiumi sulla base dei macroinvertebrati bentonici per la 2000/60/EC (WFD): il sistema di classificazione MacrOper per il monitoraggio operativo. IRSA-CNR Notiziario dei metodi Analitici, Numero speciale 2008: 24–46.
Cantonati, M., 1998. Diatom communities of springs in the Southern Alps. Diatom Research 13: 201–220.
Cantonati, M., R. Gerecke & E. Bertuzzi, 2006. Springs of the Alps—sensitive ecosystems to environmental change: from biodiversity assessments to long-term studies. Hydrobiologia 562: 59–96.
Cappelletti, C., F. Ciutti, M. E. Beltrami, R. Alber & A. Mutschlechner, 2007. Analisi della comunità delle diatomee epilitiche di sette corsi d’acqua dell’Alto Adige. Gredleriana 7: 127–140.
CEMAGREF, 1982. Etude des Méthodes Biologiques d’Appréciation Quantitative de la Qualité des Eaux. Ministère de l’Agriculture, CEMAGREF, Division Qualité des Eaux, Pêche et Pisciculture, Lyon: 218 pp.
Council of the European Communities, 2000. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. Official Journal of the European Communities L 327: 1–73.
Dell’Uomo, A., 2004. L’indice diatomico di eutrofizzazione/polluzione (EPI-D) nel monitoraggio delle acque correnti. Linee guida. APAT Agenzia per la protezione dell’ambiente e Servizi tecnici, Roma: 101 pp.
Dufrêne, M. & P. Legendre, 1997. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecological Monographs 61: 53–73.
Ector, L. & F. Rimet, 2005. Using bioindicators to assess rivers in Europe: an overview. In Lek, S., M. Scardi, P. F. M. Verdonschot, J.-P. Descy & Y.-S. Park (eds), Modelling Community Structure in Freshwater Ecosystems. Springer, Berlin, Heidelberg, New York: 7–19.
European Committee for Standardization, 2003. Water Quality—Guidance Standard for the Routine Sampling and Pretreatment of Benthic Diatoms from Rivers. European Standard EN 13946. European Committee for Standardization, Brussels: 14 pp.
European Committee for Standardization, 2004. Water Quality—Guidance Standard for the Identification, Enumeration and Interpretation of Benthic Diatom Samples from Running Waters. European Standard EN 14407. European Committee for Standardization, Brussels: 11 pp.
Gomà, J., F. Rimet, J. Cambra, L. Hoffmann & L. Ector, 2005. Diatom communities and water quality assessment in mountain rivers of the upper Segre basin (La Cerdanya, Oriental Pyrenees). Hydrobiologia 551: 209–225.
Grenier, M., S. Campeau, I. Lavoie, Y. S. Park & S. Lek, 2006. Diatom reference communities in Québec (Canada) streams based on Kohonen self-organizing maps and multivariate analyses. Canadian Journal of Fisheries and Aquatic Science 63: 2087–2106.
Grunow, A., 1860. Ueber neue oder ungenügend gekannte Algen. Erste Folge. Diatomaceen, Familie Naviculaceen. Verhandlungen der Kaiserlich-Königlichen Zoologisch-Botanischen Gesellschaft in Wien 10: 503–582.
Hlúbiková, D., S. Blanco, E. Falasco, J. Gomà, L. Hoffmann & L. Ector, 2009. Nitzschia alicae sp. nov. and N. puriformis sp. nov., new diatoms from European rivers and comparison with the type material of N. sublinearis and N. pura. Journal of Phycology 45: 742–760.
ISO, 1984. Water quality. Determination of Ammonium. Part 1: Manual Spectrometric Method. ISO 7150-1. International Standardization Organization, Geneva.
Ivanov, P. & L. Ector, 2006. Achnanthidium temniskovae sp. nov., a new diatom from the Mesta River, Bulgaria. In Ognjanova-Rumenova, N. & K. Manoylov (eds), Advances in Phycological Studies. Pensoft Publisher & University Publishing House, Sofia, Moscow: 147–154.
Kelly, M. G., A. Cazaubon, E. Coring, A. Dell’Uomo, L. Ector, B. Goldsmith, H. Guasch, J. Hürlimann, A. Jarlman, B. Kawecka, J. Kwandrans, R. Laugaste, E.-A. Lindstrøm, M. Leitao, P. Marvan, J. Padisák, E. Pipp, J. Prygiel, E. Rott, S. Sabater, H. van Dam & J. Vizinet, 1998. Recommendations for routine sampling of diatoms for water quality assessment in Europe. Journal of Applied Phycology 10: 215–224.
Kelly, M., C. Bennett, M. Coste, C. Delgado, F. Delmas, L. Denys, L. Ector, C. Fauville, M. Ferréol, M. Golub, A. Jarlman, M. Kahlert, J. Lucey, B. Ní Chatháin, I. Pardo, P. Pfister, J. Picinska-Faltynowicz, J. Rosebery, C. Schranz, J. Schaumburg, H. van Dam & S. Vilbaste, 2009. A comparison of national approaches to setting ecological status boundaries in phytobenthos assessment for the European Water Framework Directive: results of an intercalibration exercise. Hydrobiologia 621: 169–182.
Krammer, K., 1997a. Die cymbelloiden Diatomeen. Eine Monographie der weltweit bekannten Taxa. Teil 1. Allgemeines und Encyonema part. Bibliotheca Diatomologica 36: 1–382.
Krammer, K., 1997b. Die cymbelloiden Diatomeen. Eine Monographie der weltweit bekannten Taxa. Teil 2. Encyonema part., Encyonopsis und Cymbellopsis. Bibliotheca Diatomologica 37: 1–469.
Krammer, K., 2002. Cymbella. In Lange-Bertalot, H. (ed.), Diatoms of Europe. Diatoms of the European Inland Waters and Comparable Habitats, Vol. 3. A.R.G. Gantner Verlag K.G, Ruggell: 584 pp.
Krammer, K., 2003. Cymbopleura, Delicata, Navicymbula, Gomphocymbellopsis, Afrocymbella. In Lange-Bertalot, H. (ed.), Diatoms of Europe. Diatoms of the European Inland Waters and Comparable Habitats, Vol. 4. A.R.G. Gantner Verlag K.G, Ruggell: 530 pp.
Krammer, K. & H. Lange-Bertalot, 1986. Bacillariophyceae 1. Teil: Naviculaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa. Gustav Fischer Verlag, Stuttgart: 876 pp.
Krammer, K. & H. Lange-Bertalot, 1988. Bacillariophyceae 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa. Gustav Fischer Verlag, Stuttgart: 596 pp.
Krammer, K. & H. Lange-Bertalot, 1991a. Bacillariophyceae. 3. Teil: Centrales Fragilariaceae, Eunotiaceae. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa. Gustav Fischer Verlag, Stuttgart: 576 pp.
Krammer, K. & H. Lange-Bertalot, 1991b. Bacillariophyceae 4. Teil: Achnanthaceae. Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema. Gesamtliteraturverzeichnis Teil 1–4. In Ettl, H., G. Gärtner, J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa. Gustav Fischer Verlag, Stuttgart: 437 pp.
Lange-Bertalot, H., 2001. Navicula sensu stricto. 10 genera separated from Navicula sensu lato. Frustulia. In Lange-Bertalot, H. (ed.), Diatoms of Europe. Diatoms of the European Inland Waters and Comparable Habitats, Vol. 2. A.R.G. Gantner Verlag K.G, Ruggell: 526 pp.
Lange-Bertalot, H. & A. Steindorf, 1996. Rote Liste der limnischen Kieselalgen (Bacillariophyceae) Deutschlands. Schriften-Reihen für Vegetationskunde 28: 633–677.
Levkov, Z., S. Krstic, T. Nakov & L. Melovski, 2005. Diatom assemblages on Shara and Nidze mountains, Macedonia. Nova Hedwigia 83: 501–537.
Lösch, B., 2008. Makrozoobenthos und Diatomeen im Schlerngebiet (Südtirol). Gredleriana 8: 175–188.
Lösch, B. & R. Alber, 2009. La qualità biologica dei corsi d’acqua in Alto Adige. Indagini eseguite nel periodo 2005–2009. Annali del Laboratorio Biologico Provinciale n. 18. Provincia Autonoma di Bolzano-Alto Adige: 319 pp.
Mancini, L. & C. Sollazzo (eds), 2009. Metodo per la valutazione dello stato ecologico delle acque correnti: comunità diatomiche. Istituto Superiore di Sanità (Rapporti ISTISAN 09/19), Roma: 32 pp.
McCune, B. & M. J. Mefford, 1999. Multivariate Analysis of Ecological Data, version 4. MjM Software Design, Gleneden Beach, Oregon, USA: 237 pp.
Mielke, P., 1984. Meteorological applications of permutation techniques based on distance functions. In Krishnaiah, P. R. & P. K. Sen (eds), Handbook of Statistics. Elsevier Science Publisher, Amsterdam: 813–830.
Mielke, P. & K. J. Berry, 2001. Permutation Methods: A Distance Function Approach. Springer Series in Statistics: 344 pp.
Milde, J., 1864a. Ein Sommer in Süd-Tirol. Botanische Skizze. Botanische Zeitung 22. Jahrgang. Zweite Beilage 17–19: 1–22.
Milde, J., 1864b. Zur Cryptogamen—Flora Süd-Tirols: Dritter Artikel. Botanische Zeitung 22. Jahrgang. Dritte Beilage 43–44: 1–18.
Monnier, O., H. Lange-Bertalot, F. Rimet, L. Hoffmann & L. Ector, 2004. Achnanthidium atomoides sp. nov., a new diatom from the Grand-duchy of Luxembourg. Vie et Milieu-Life and Environment 54: 127–136.
Ní Chatháin, B., T. J. Harrington, J. McCarthy, M. Kelly-Quinn, C. Bradley & J. R. Baars, 2004. Preliminary results from the investigation of benthic diatoms from potential reference river sites in Ireland. Oceanological and Hydrobiological Studies 33: 3–15.
Pfister, P. & E. Pipp, 2009. Fliessgewässer Qualitätselement Phytobenthos: Felderhebung, Probenahme, Probenaufarbeitung und Ergebnisermittlung. Leitfaden zur Erhebung der Biologischen Qualitätselemente Teil A3—Phytobenthos. Bundesministerium für Land-und Forstwirtschaft, Umwelt und Wasserwirtschaft, Wien: 92 pp.
Picińska-Fałtynowicz, J., 2007. Epilithic diatoms as indicators of water quality and ecological status of streams of Sudety Mountains (South-Western Poland). In Ács, É., K. T. Kiss & J. Padisák (eds), Proceedings of 6th International Symposium on Use of Algae for Monitoring Rivers, Hungary, Balatonfüred, 12–16 Sept. 2006. Archiv für Hydrobiologie Supplement 161, Large Rivers 17: 287–305.
Reichardt, E., 1999. Zur Revision der Gattung Gomphonema. Iconographia Diatomologica 8: 1–203.
Rimet, F., 2009. Benthic diatom assemblages and their correspondence with ecoregional classifications: case study of rivers in north-eastern France. Hydrobiologia 636: 137–151.
Rimet, F., L. Tudesque, V. Peeters, H. Vidal & L. Ector, 2003. Assemblages-types de diatomées benthiques des rivières non-polluées du bassin Rhône-Méditerranée-Corse (France). Bulletin de la Société des Sciences Naturelles de l’Ouest de la France, 2ème Supplément Hors Série, 272–287.
Rimet, F., L. Ector, H. M. Cauchie & L. Hoffmann, 2004. Regional distribution of diatom assemblages in the headwater streams of Luxembourg. Hydrobiologia 520: 105–117.
Rimet, F., J. Gomà, J. Cambra, E. Bertuzzi, M. Cantonati, C. Cappelletti, F. Ciutti, A. Cordonier, M. Coste, F. Delmas, J. Tison, L. Tudesque, H. Vidal & L. Ector, 2007. Benthic diatoms in western European streams with altitudes above 800 m: characterisation of the main assemblages and correspondence with ecoregions. Diatom Research 22: 147–188.
Rott, E., H. Van Dam, P. Pfister, E. Pipp, K. Pall, N. Binder & K. Ortler, 1999. Indikationslisten für Aufwuchsalgen. Teil 2: Trophieindikation sowie Geochemische Präferenz; Taxonomische und Toxikologische Anmerkungen. Bundesministerium für Land- und Forstwirtschaft, Wien: 248.
Round, F. E., 1991. Diatoms in river water-monitoring studies. Journal of Applied Phycology 3: 129–145.
Schaumburg, J., C. Schranz, J. Foerster, A. Gutowski, G. Hofmann, P. Meilinger, S. Schneider & U. Schmedtje, 2004. Ecological classification of macrophytes and phytobenthos for rivers in Germany according to the Water Framework Directive. Limnologica 34: 283–301.
Schröder, J. L. B., 1895. Über Algen, insbesondere Desmidiaceen und Diatomaceen aus Tirol. In: 72. Jahresbericht der schlesischen Gesellschaft für vaterländische Cultur im Jahre 1894. II. Breslau: 42–47.
ter Braak, C. J. F. & P. Smilauer, 1998. CANOCO Reference Manual and User’s Guide to Canoco for Windows: Software for Canonical Community Ordination (version 4). Microcomputer Power, Ithaca, NY, USA: 352 pp.
Tison, J., Y.-S. Park, M. Coste, J. G. Wasson, L. Ector, F. Rimet & F. Delmas, 2005. Typology of diatom communities and the influence of hydro-ecoregions: a study on the French hydrosystem scale. Water Research 39: 3177–3188.
UNI, 1994. Qualità dell’acqua. Determinazione dell’ossigeno disciolto. Metodo iodometrico. UNI-EN 25813: 1994. UNI Ente Nazionale Italiano di Unificazione, Milano: 10 pp.
UNI, 2004. Qualità dell’acqua. Determinazione del fosforo. Metodo spettrometrico all’ammonio molibdato. UNI-EN-ISO 6878: 2004. UNI Ente Nazionale Italiano di Unificazione, Milano: 21 pp.
Van Dam, H., A. Mertens & J. Sinkeldam, 1994. A coded checklist and ecological indicator values of freshwater diatoms from The Netherlands. Netherlands Journal of Aquatic Ecology 28: 117–133.
van de Bund, W., 2009 (ed.). Water Framework Directive Intercalibration Technical Report. Part 1: Rivers. Draft. Joint Research Community, Scientific and Technical Reports. Office for Official Publications of the European Communities, Luxembourg: 179 pp. Available at: http://publications.jrc.ec.europa.eu/repository/handle/111111111/294.
Wasson, J. G., A. Chandesris, A. Garcia Bautista, H. Pella & B. Villeneuve, 2006. Combined Pressures and Geographical Context: Hydro-Ecoregions Framework. Deliverable 14, WP4, Activity 5, EC co-funded project REBECCA.
Whitton, B. A., N. T. W. Ellwood & B. Kawecka, 2009. Biology of the freshwater diatom Didymosphenia: a review. Hydrobiologia 630: 1–37.
Acknowledgments
Authors are grateful to Dr. U. Obertegger (Fondazione E. Mach) for her precious revision and suggestions during statistical analysis, and to Laboratorio Analisi Acqua—Environmental Agency of Bolzano-Bozen for supplying chemical data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: L. Ector, D. Hlúbiková & L. Hoffmann / Proceedings of the 7th International Symposium “Use of Algae for Monitoring Rivers”, Luxembourg, November 23–25, 2009
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Beltrami, M.E., Ciutti, F., Cappelletti, C. et al. Diatoms from Alto Adige/Südtirol (Northern Italy): characterization of assemblages and their application for biological quality assessment in the context of the Water Framework Directive. Hydrobiologia 695, 153–170 (2012). https://doi.org/10.1007/s10750-012-1194-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-012-1194-x