Abstract
There is increasing interest in the restoration of urban river systems because they are affected by multiple point- and diffuse-source impairments. In South Wales, these issues are exemplified by the rivers Taff and Ely, which have a well-documented history of pollution. We developed a network of river-monitoring sites to capture as many sources of impairment as possible and to provide an integrated assessment of basin-wide conditions using established, recently revised and new methods to assess ecological status as required by the Water Framework Directive (WFD). Most of the 34 river sites, except those in upstream areas, had moderate or poor ecological status and low species richness. There were significant correlations between the UK Trophic Diatom Index (TDI), phosphate and nitrate concentrations, but the strongest correlation was with sodium concentrations. Sodium also differentiated best between ecological status classes. The UK TDI, Ecological Quality Ratios and the Indice de Polluosensibilité Spécifique (IPS) correlated strongly, but the IPS reflected chemical conditions less well. There were also possible metal impacts including some upstream sites despite apparently good or high ecological status. These data reveal strong capacity in diatoms for reflecting stressors that affect urban river networks. We recommend (i) further assessments including geographically comprehensive and repeated surveys to underpin local decision-making and (ii) further refinement of the tools currently in use for WFD purposes through the development of specific diagnostic indices or multi-metric methods for a comprehensive assessment of complex catchments affected by multiple impairments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There is increasing interest in the restoration and sustainable management of urban river systems for three principal reasons. First, urban areas are characterised by multiple physical and chemical sources of impairment (Ormerod et al., 2010). These include domestic and industrial wastewater, and surface runoff of nutrients, fertilisers, pesticides, organic contaminants and metals from both point and diffuse sources (Mallin et al., 2009). Urban river channels are also highly modified, for example, for flood-risk management, while basin hydrology reflects extensive hard surfaces that affect runoff character (Daenport et al., 2001; Paul & Meyer, 2001; Mallin et al., 2009). Such changes also make urban rivers more sensitive to climate-change effects, and evidence suggests that they are heating more rapidly than other rivers (Kaushal et al., 2010). Secondly, urban locations represent areas of disproportionate importance to people. Throughout the world, increasing numbers of people now not only live in cities and are at risk from the adverse effects of impairment (Duh et al., 2008), but also derive benefit where rivers can be restored and enhanced for overall environmental quality (Platt et al., 1994; Duigan et al., 2009; Alberti, 2010).
In many developed countries and in industrialised areas, some pollution problems have been solved, and ecological quality has substantially improved (Ormerod & Jüttner, 2009). In many other cases, however, legacy infrastructures from past engineering works, as well as continued sewage outfalls, chemical discharges, pesticides, mine drainage, acidification, litter, agricultural runoff, landfill leachate, sediment delivery, etc., are continuing problems affecting recovery from upstream to downstream areas (Walsh & Wepener, 2009; Jüttner et al., 2010). So far there are few published case studies devoted to monitoring networks that have been developed specifically in urban river systems, which can effectively (i) appraise status and diagnose a wide range of potential impairments and multiple stressors, (ii) guide management actions, and (iii) act as baseline against which to judge restoration outcomes. The Water Framework Directive (WFD, Council of the European Communities, 2000) requires the assessment of ecological status using several groups of biological indicators representative of different trophic levels in a complex biological community. Urban monitoring networks could be particularly effective if several metrics assessing different ecosystem components such as diatoms and macroinvertebrates were used simultaneously (Carvalho et al., 2011). They sometimes respond to different problems and can independently reveal multiple pressures such as changes in habitat structure and water chemistry (Soininen & Könönen, 2004; Torrisi et al., 2010). Diatoms appear to be more responsive to eutrophication and organic pollution and also respond quickly to chemical change. They can therefore detect intermittent as well as chronic effects while providing detailed information about variability over time, and provide added value over existing methods using other organisms (Griffith et al., 2005; Hering et al., 2006; Passy, 2006). The diversity of diatom species, and the array of ecological conditions to which they respond (e.g. nutrients, Hall & Smol, 1999; metals, Hirst et al., 2002; organic pollution, Watanabe et al., 2005; sediments, Dickman et al., 2005; acidification, Lewis et al., 2007) might also give them potentially large capacity to diagnose impairments amongst the multiple sources affecting urban rivers (Wang et al., 2005; Ormerod et al., 2010). Moreover, the widespread, global distribution of species in polluted waters could provide effective parallel data in different global regions.
One of the key needs in assessing status and trends in urban rivers is to have appropriate monitoring and assessment methods in place in well-designed networks. For benthic diatoms, methods are developing rapidly (Lavoie et al., 2006; Chessman et al., 2007; Kelly et al., 2008), but their performance must be assessed in individual case studies and catchments, comparing index values and classification against actual biological and chemical conditions (Hamsher et al., 2004). Potential failure to detect multiple impacts of a different nature can then be addressed by either modification of the diatom methods or additional use of other indicator groups (Griffith et al., 2005).
The catchments of the rivers Taff and Ely cover part of an area of South Wales that was famous for its coal, iron and steel industries from the eighteenth to the twentieth centuries (John & Williams, 1980). These not only laid the foundations of rapid urban development but also led to a well-documented history of pollution (e.g. Scullion & Edwards, 1980). Previous studies showed that a range of impairments can affect river quality reflected by changes in benthic diatom assemblage composition with a shift to pollution-tolerant species and in some cases low species diversity (Gómez, 1998; Hirst et al., 2002; Jüttner et al., 2003; Kim et al., 2008). In this study, we aimed to develop a geographically extensive network of river monitoring sites sufficient to capture as many sources of impairment as possible and provide a preliminary assessment of basin-wide conditions using the recently revised UK Trophic Diatom Index (TDI), a new method to assess ecological status as required by the WFD, and a pollution index Indice de Polluosensibilité Spécifique (IPS) from France. We tested the applicability of these metrics against actual biological and chemical conditions in the Taff and Ely catchments. Specifically, we determined (i) how diffuse and point-source pollutions affected ecological status by investigating diatom assemblages and water chemistry in headwater areas, above and below major settlements, river confluences and effluents from sewage treatment works, and (ii) how ecological status classes, diatom indices, assemblage composition and diversity corresponded to the chemical conditions and to what extent they reflected chemical gradients.
Methods
Study area
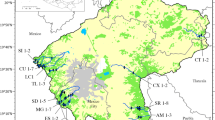
The area of investigation included the densely populated, industrialised and agricultural catchments of the rivers Taff and Ely including all major tributaries such as the Taff Bargoed, Taff Fechan, Taff Fawr, Cynon, Nant Clydach, Rhondda, Rhondda Fach, Rhondda Fawr in the Taff catchment, and the Nant Clun, Nant Mychydd in the Ely catchment. The area is located in South Wales, UK, between the Brecon Becon Mountains and the City of Cardiff where both rivers flow into a newly created lake Cardiff Bay which occupies their former estuaries (Crompton, 2002). In October and November 2004, samples were collected at 34 sites above and below major confluences, urban settlements and discharge points of sewage treatment works, and in the upper catchments upstream of major settlements.
Water chemistry
Conductivity, pH and temperature were measured at each river site (pH/Cond 340i, WTW, Burmarc, Bordon, UK), and water samples were taken for chemical analysis and 0.45 μm filtered. Photometric measurements for NO3-N (d.l. 0.1 mg l−1), NO2-N (d.l. 0.005 mg l−1), NH4-N (d.l. 0.01 mg l−1) and PO4-P (d.l. 0.01 mg l−1) were carried out on the day of collection using WTW test kits (NO3 09713, NO2 14776/1, NH4 14752, PO4 14848 and Photolab S12-A, Burmarc, Bordon, UK). Other major cations and anions were analysed by inductively coupled plasma atomic emission spectrometry (ICP-AES, Ca, Mg, Fe [d.l. 0.001 mg l−1], Na, K [d.l. 0.05 mg l−1], Sr [d.l. 0.0001 mg l−1], Ba [d.l. 0.0005 mg l−1], Al [d.l. 0.006 mg l−1], Si [d.l. 0.03 mg l−1]) and inductively coupled plasma mass spectrometry (Mn, d.l. 0.0005 mg l−1) on acidified water samples, and by ion chromatography (SO4, Cl [d.l. 0.01 mg l−1], F [d.l. 0.001 mg l−1], Department of Mineralogy, The Natural History Museum, London, UK). Alkalinity measurements were provided by the Environment Agency.
Diatoms
Diatoms were collected with toothbrushes from at least five stones in riffle areas at each river site following standard procedures (Kelly et al., 1998). Samples were preserved in ethanol and processed using standard methods (hot hydrogen peroxide oxidation) and mounted in Naphrax (Krammer & Lange-Bertalot, 1986–1991). Diatoms were identified and a minimum of 500 valves counted at ×1,000 magnification using a Nikon Eclipse E600 microscope equipped with differential interference contrast (DIC). The relative abundances of species were calculated. Identifications were based on Krammer & Lange-Bertalot (1986–1991), Krammer (1997a, b, 2002), Reichardt (1999) and Lange-Bertalot (2001).
Data analysis
Diversity H (Shannon diversity index, log10), evenness E (Shannon, also termed J) and species richness S were calculated using the software PRIMER 6. Species richness was calculated using rarefaction adjusted to a samples size of 500 valves. To assess the ecological status of river sites established, recently revised and new metrics for rivers were calculated using all diatom species found. They included the TDI and Ecological Quality Ratios (EQR), methods developed to monitor trophic status and ecological status in UK rivers (Kelly et al., 2007, 2008; DARLEQ—Diatom Assessment of River and Lake Environmental Quality). EQRs were calculated to assess the deviation of diatom assemblages from reference conditions and to determine ecological status classes as defined by the WFD (Council of the European Communities, 2000; Kelly et al., 2007). Uncertainty analysis to assess the risk of misclassification was performed on DARLEQ following Ellis & Adriaenssens (2006). Because the Taff and Ely catchments are affected by complex pressures including impacts from agriculture and the release of sewage, we also used the IPS (= Specific Pollution Sensitivity Index, SPI; Cemagref, 1982; Prygiel et al., 1999; OMNIDIA version 3). This index was developed to evaluate general water quality taking into account organic pollution and nutrients.
Analysis of variance and Tukey’s multiple comparison test (ANOVA, Minitab 14 and Excel 2003) were used to test differences in water chemistry, species richness, diversity, evenness, TDI, EQR and IPS between site groups. Regression analysis (Minitab 14) was applied to assess relationships between TDI, EQR, IPS and water chemistry.
To investigate variations in diatom assemblage composition and differences in water chemistry between river sites non-metric multidimensional scaling ordination (MDS) was used (PRIMER 6). Water chemistry data were analysed after log + 1 transformation using a Bray–Curtis similarity matrix, and diatom data were analysed after square-root transformation and using Euclidean distance as a resemblance measure. ANOSIM and SIMPER (PRIMER 6) were used to test whether differences between a priori defined groups (ecological status classes) were significant and which diatom species contributed to the separation between groups.
Results
In the catchments of the rivers Taff and Ely, a total of 23 (68%) of 34 sites were classified as moderate (7, 21%) or poor (16, 47%), and 11 (32%) had good or high ecological status (Fig. 1; Table 1). In the Ely catchment, a higher percentage of river sites had unfavourable ecological status with 11 sites classified as moderate or poor and only one site in the upper catchment classified as high. All sites in the upper catchments of the river Taff and its tributaries had good or high ecological status, but most sites in the middle and lower catchments were moderate or poor. Only above and below the confluence of the Taff Bargoed and the Taff was the difference in river quality large as indicated by the TDI, EQR and status class with high ecological status in the Taff Bargoed (T11) and poor status in the Taff (T9). In mid-catchment urban areas above and below main confluences, the ecological status was either moderate or poor, except at T3 and T21 in the Rhondda catchment and at T13 on the Taff. These sites had good ecological status, but the confidence of prediction was much lower than elsewhere. There was no impact of effluents from sewage treatment works on ecological status except in one case in the upper Ely catchment where the class changed from moderate to poor, elsewhere the status classes were already bad upstream of the works’ discharge. The likelihood of misclassification at the good/moderate boundary was low with only three sites potentially misclassified with a risk of >30%. All sites with high ecological status were classified with 100% confidence and those with poor ecological status on average with 70%.
Differentiating between ecological status classes, TDI values and EQR were on average (±SD) 72.4 (1.7)/0.45 (0.03) for poor, 61.0 (4.3)/0.62 (0.06) for moderate, 47.0 (3.2)/0.85 (0.04) for good and 32.0 (6.4)/1.0 (0.01) for high ecological status (Table 1), with expected TDI scores ranging from 35.1 to 39.8. The percentage of organic-tolerant taxa was on average (±SD) 67.7 (25.3) for poor, 52.7 (9.5) for moderate, 18.7 (20.3) for good and 5.9 (5.8) for high ecological status, differing significantly above and below the good/moderate boundary but not between poor and moderate or good and high ecological status classes (F (3,30) 17.7, P < 0.001).
IPS values varied between 16.3 and 19.9 (median 18.1, except at T15 with 14.0) in the upper Taff catchments, and between 9.1 and 15.5 (median 13.9, except at T13 with 18.4) in the middle and lower catchments. In the Ely catchment, IPS values varied between 12.3 and 15.2 (median 14.0). There were highly significant relationships between the TDI and IPS (R 2 47.2%, F (1,32) 28.6, P < 0.001), and between EQR and IPS (R 2 40.6%, F (1,32) 21.9, P < 0.001). IPS values (mean, ±SD) were significantly higher at sites with high ecological status but did not differ significantly between the other classes including at the good/moderate boundary (high 17.5, 1.9; good 14.7, 3.5; moderate 14.5, 0.6; poor 13.5, 0.8; F (3,30) 8.8, P < 0.001).
Diatom diversity in both catchments was very low. Although 74 taxa were found only 7 were common and abundant including Navicula gregaria Donkin, N. lanceolata (C. Agardh) Ehrenberg, Achnanthidium minutissimum (Kützing) Czarnecki, Fragilaria capucina cf. var. rumpens (Kützing) Lange-Bertalot, Cocconeis placentula Ehrenberg, Amphora pediculus (Kützing) Grunow, Reimeria sinuata (Gregory) Kociolek & Stoermer and Rhoicosphenia abbreviata (C. Agardh) Lange-Bertalot. Diversity H′, evenness and species richness were not significantly different between ecological status classes (Fig. 2; Table 2). Diversity and evenness tended to be higher in the moderate and good classes and species richness in the moderate class.
MDS ordination separated sites in different ecological status classes, and most clearly those with high or good ecological status, TDI values <50, organic pollution-tolerant taxa <20% and IPS values >16 from those with moderate or poor ecological status, higher TDI and lower IPS values, and more pollution-tolerant taxa (Fig. 3). Differences in species composition between ecological status classes were significant, in particular between poor or moderate and other classes, but not between good and high ecological status classes (ANOSIM P < 0.001, R = 0.63, Table 3). Navicula gregaria, N. lanceolata and A. pediculus were most abundant at sites with moderate or poor ecological status. Navicula gregaria was more abundant and A. minutissimum and R. sinuata were less abundant at sites with poor ecological status than at sites with moderate ecological status. At the good/moderate boundary C. placentula, R. sinuata and R. abbreviata were more abundant at sites with good ecological status. Achnanthidium minutissimum and F. capucina cf. var. rumpens were the most abundant species at sites with high ecological status.
MDS ordination of diatom assemblages at Taff and Ely river sites, South Wales, UK, of different ecological status classes (symbols are those used in Fig. 1), and characteristic species; the size of the species symbols represent relative abundances
MDS ordination using water chemistry data separated most river sites with poor from those with moderate ecological status, and sites with poor or moderate from those with good or high ecological status (Fig. 4; Table 4). Differences between sites with poor and other ecological status, and between moderate and high ecological status were significant, but there was no significant difference at the good/moderate boundary or between sites with good and high ecological status (ANOSIM P < 0.001, R = 0.51, Table 3). Of all the solutes measured, the concentrations of Na and K distinguished best between classes, but the differences were only significant between sites with poor ecological status and others. Concentrations of PO4-P and NO3-N were on average higher at sites with poor ecological status, but varied considerably, than at sites with moderate, good or high status (Fig. 5). However, at most sites with poor ecological status, PO4-P was below 0.10 mg l−1. At sites with moderate, good or high ecological status, PO4-P was mostly below the detection limit of 0.01 mg l−1. All sites with poor ecological status in the lower Ely catchment had significantly higher conductivity and concentrations of PO4-P, Na, K, Ca, SO4 and Ni than sites in the upper Ely catchment and in the Taff catchment with poor or moderate ecological status, and higher NO3-N concentrations than sites with good or high ecological status.
There were significant correlations between Na, K, nutrients and diatom metrics. The UK metrics were more strongly related to gradients in water chemistry than the metric from France and the strongest correlations were found for Na, followed by K, NO3-N and PO4-P (Table 5).
There were sites which differed considerably in chemistry from others. The site E6, a small stream located downstream of a sewage treatment works discharge, had the highest concentrations of PO4-P, NO3-N, NO2-N, NH4-N and K. Some sites had higher concentrations in metals such as Cu (T11 and 12, E5 and 8, ≥0.004 mg l−1 vs. <0.002 mg l−1 at most other sites), Fe (T11, 12 and 17, E6, 7 and 9, >0.25 mg l−1 vs. ≤0.18 mg l−1 at other sites), Ni (T11 and 12, E1–3 and 5–8, >0.004 mg l−1 vs. ≤0.003 mg l−1 at most other sites), Zn (T1, 12, 17 and 19, E1–3 and 8, >0.007 mg l−1 vs. ≤0.005 mg l−1 at most other sites). Of these sites, only T17 was clearly separated from others in the ordination of diatom assemblages.
Discussion
Assessment of ecological status
According to the data collected, in autumn of 2004, a minority of sites that were within and downstream of urban areas in the Taff catchment, and only one site in the Ely catchment, had good ecological status. These prevailing conditions are clearly important given that good status should be reached by 2015 according to the WFD (Council of the European Communities, 2000). As in the case of many urban catchments, various sources of impairment, such as sewage from qualified discharge, sewer overflow and urban run-off contribute to unsatisfactory conditions where good ecological status is not reached. Improvements, and restoration over extensive segments of this catchment appear to be needed to achieve better ecological conditions, and therefore effective monitoring should underpin management and assess progress. One issue with these data is that assessments were limited to a relatively small number of sites and mostly to one sampling. There is uncertainty about how representative one-off assessments are in revealing longer-term quality. Ideal sampling frequencies to detect variations and trends will also vary between river catchments (Lewis et al., 2007). During a 2-year study from 2004 to 2006 at Cardiff Bay and two of the Taff and Ely sites (T1, E1) with repeated sampling, there was variability in diatom composition between seasons, but no overall change of status with respect to the good/moderate boundary (Jüttner et al., 2010). T1 and E1 were located in the City of Cardiff, and the ecological status varied between poor (4×) and moderate (3×) at the Taff, except once when it was high. At the Ely, ecological status was poor (4×), moderate (3×) and bad once. Samples close to class boundaries are at higher risk of misclassification (Kelly et al., 2009a). In the Taff and Ely catchments, there was generally a low risk of misclassification at the good/moderate boundary. However, variability at the Cardiff river sites suggests that depending on circumstances sites on either side of the boundary might benefit from more frequent monitoring to assess the risk of misclassification and statistical power of particular upstream–downstream comparisons. Elsewhere, the variability of water quality index values in a stream catchment affected by urban pollution was also relatively small (Bere & Tundisi, 2011), but other studies showed that there can be considerable variation between samples collected from the same site at different times (Kelly, 2002; Kelly et al., 2009b). However, within-site variation differed between sites and repeated sampling over several years was recommended to assess the frequency of monitoring required on a case-by-case basis and to underpin decision making for additional investments to improve water quality. In South Wales, multiple samplings during different seasons and application of these methods to other catchments, some of which already monitored by the Environment Agency, would put the current results from the Taff and Ely catchments into context and expand the current network to other heavily impacted areas.
Ecological status, diatom assemblages and environmental conditions
Ecological status classes as defined by the WFD were assessed by comparing the actual biota with those expected in waters subject to no or minimal disturbance. The dominant species in the Taff and Ely catchments included some of those widely found and used for the assessment of ecological status classes in the UK (Kelly et al., 2008). The most abundant species Navicula gregaria followed by N. lanceolata have TDI indicator values of 4 (favoured by high concentrations of nutrients) and dominated at sites classified as moderate or poor. However, other species most abundant in these classes elsewhere such as Eolimna minina (Grunow) Lange-Bertalot, N. tripunctata (O.F. Müller) Bory and Planothidium lanceolatum (Brébisson) Round & Bukhtiyarova had very low relative abundances. There was a clear difference in diatom assemblage composition on either side of the good/moderate boundary shown independently by ordination and an ANOVA-type procedure. Most abundant at sites with good or high status were the groups of Achnanthidium minutissimum, Cocconeis placentula and Fragilaria capucina Desmazières. These taxa groups represent considerable taxonomic problems and contain varieties or separate species which differ from each other in their ecology (e.g. Hürlimann & Straub, 1991). In practice, these taxa are often difficult to separate and detailed taxonomic studies are essential to provide guidance for consistent identification. Potapova & Hamilton (2007) identified six morphological groups in the A. minutissimum species complex in North American river samples which clearly differed in their ecology. It can be assumed that such refined taxonomy if extended to other difficult groups would aid the determination of ecological status. However, this would require a high level of training and harmonisation across analysts to ensure that the analytical precision is maintained.
Species richness was very low in the Taff and Ely catchments, and particularly at sites with poor ecological status where, although not significantly lower than at other sites, in most cases only 2–3 taxa accounted for over 70% relative abundance. There is no simple relationship between diversity and pollution, and measures of richness are often regarded as unsuitable indicators of such impacts. They did also not differentiate between status classes in our study. However, bell-shaped curves with higher richness or diversity at intermediate enrichment and very low species richness at severely polluted sites are often found, and the latter can be due to impacts of metals, pesticides and their toxic effects, and to severe organic pollution (Nather Khan, 1991; Lobo et al., 1995; Gómez, 1999). Possible explanations for low species richness in our study include the background of long-term degradation in the Taff and Ely catchments, the multiple combination of stressors, such as urbanisation, sewage, metals, and the possibility that unmeasured factors—such as pesticides, chemical discharges, landfill leachate—might be involved. It is also possible that within these catchments environmental gradients were not sufficiently strong and in particular differences between important water chemistry parameters at most sites might have been too weak to be reflected by clear differences in species richness (Heino et al., 2009).
Chemistry mostly differed only between ecological status class poor and other status classes but not between good and high ecological status and in contrast to diatoms not at the good/moderate boundary. Concentrations of nutrients were low at sites with moderate, good or high status and a detection limit of 0.01 mg l−1 for PO4-P might be too high to differentiate between good and high ecological status. Ordination of sites based on water chemistry broadly reflected the ordination based on diatom assemblages. However, some sites e.g. E6, E7, T11, T13 and T14 clearly differed in chemistry but E6 and E7, and T11, T13 and T14, had similar diatom assemblages. Poor correlation between biotic indices and environmental variables can be due to several factors (Hill et al., 2000). Diatom assemblages reflect chemical conditions over several days to weeks (Hirst et al., 2004; Rimet et al., 2009) and chemical conditions on the day of collection might have differed from the average conditions that were reflected by assemblages over the preceding weeks (Iserentant & Blancke, 1986). Stronger relationships might have been found if biotic indices had been correlated with average values of several measurements or with time-weighted averages if such measurements had been taken before the diatom survey (Blanco et al., 2007). Alternatively, other unmeasured factors might have had overriding importance for the assemblages or diatoms were not sensitive to specific chemical conditions at these sites.
Although there were significant relationships between NO3-N, PO4-P and TDI the strongest correlation and best differentiation between status classes was with Na and K. Strong correlations between Na and TDI were also found elsewhere where PO4-P concentrations were low or non-detectable (Jüttner et al., 2003). Jenkins et al. (1995) proposed that higher Na concentrations in streams draining agricultural catchments might reflect higher weathering rates on agricultural land. This suggests that Na might be used instead of nutrients (which are also subject to larger short-term variation and therefore more difficult to measure accurately) in freshwaters without excessive salt pollution in developments of monitoring tools.
In France, the IPS index was significantly correlated with organic pollution and eutrophication (Prygiel & Coste, 1993), and this index might be regarded as an appropriate metric in urban streams which are affected by both types of pollution. In our study, the TDI, EQRs and IPS were significantly correlated, but the IPS reflected chemical conditions less well. There were no significant correlations between IPS and PO4-P, and the correlations with Na, K and NO3-N were weaker than those with TDI and EQR. The IPS did not differ at the good/moderate boundary but only between the high status class and the others in contrast to the significant difference between good and moderate status in the percentage of pollution-tolerant taxa as defined in the UK method. Although the metrics available in DARLEQ are optimised for the low/moderate pressure range (Kelly, pers. comm.), they perform better in polluted, urban streams in the UK than the index from France. This shows the advantage of applying methods which were developed locally or in geographically similar regions rather than using indices from different geographical areas (Poulíčková et al., 2004; Martín et al., 2010).
Some of the sites in the Taff and Ely catchments with apparently high ecological status might be affected by other stressors such as metals as indicated by their water chemistry. Stressors from different sources, such as sewage treatment, combined sewer overflows, industry including mining, diffuse pollution due to surface run-off from urban and agricultural areas including siltation, and impacts due to habitat change, such as channel modifications, all of which are relevant to the Taff and Ely catchments, can affect diatom assemblages in different ways. Multiple stressors can be monitored by applying specific indices designed to detect their specific effects (Ziemann, 1982; Andrén & Jarlman, 2008), through a combined index for ecological quality (Fore & Grafe, 2002), or by recording particular features in diatoms such as morphological deformations (Falasco et al., 2009). Some stressors such as habitat quality might be monitored more efficiently by other biological groups (Griffith et al., 2005). For stressors other than nutrients, specific diatom indices are not yet available in the UK. Developing tools combining assessments with different taxonomic groups and using functional as well as structural changes in communities as metrics might be the most comprehensive way of evaluating ecological conditions in complex catchments such as those in South Wales (Kelly et al., 2009b). However, the costs of developing and testing new methods are high. They would need to be justified by strong evidence that multiple pressures impair ecological functioning and by establishing the obvious gaps in existing methods to address them. In the meantime, using the currently available diatom-based methods but in a broader programme of geographically comprehensive and repeated surveys would establish a strong capacity for local decision-making.
Conclusions
An urban monitoring network was established for an overall assessment of ecological status in rivers of two complex catchments affected by multiple impairments using diatom-based methods from the UK and from France. Differences in diatom assemblage composition between ecological status classes and in particular at the good/moderate boundary were significant. However, water chemistry did not differentiate between moderate and good ecological status. Thus, diatom-based methods appear to be more sensitive in characterising ecological conditions than chemical analysis alone. Comparison of the UK and French metrics showed that the UK metrics better reflected actual chemical conditions. This confirms that locally developed monitoring tools should be favoured over methods from other geographical areas. Possible impacts by metals at some sites were largely not reflected by diatom assemblages. We suggest that currently available methods could be developed further to diagnose and untangle different effects of multiple stressors in river catchments, and to underpin measures of restoration. The frequency of monitoring required to detect catchment specific variability in environmental conditions should be determined by repeated surveys and adjusted when already existing problems will be complicated further due to climate change. Through surveillance monitoring this study linked ecological status to particular pressures in the rivers Taff and Ely and could aid the UK Environment Agency’s River Management Plan developing appropriate management strategies to achieve good ecological status in this catchment (Environment Agency, 2008).
References
Alberti, M., 2010. Maintaining ecological integrity and sustaining ecosystem function in urban areas. Current Opinion in Environmental Sustainability 2: 178–184.
Andrén, C. & A. Jarlman, 2008. Benthic diatoms as indicators of acidity in streams. Fundamental and Applied Limnology/Archiv für Hydrobiologie 173: 237–253.
Bere, T. & J. G. Tundisi, 2011. Influence of land-use patterns on benthic diatom communities and water quality in the tropical Monjolinho hydrological basin, São Carlos-SP, Brazil. Water SA 37: 93–102.
Blanco, S., E. Bécares, H.-M. Cauchie, L. Hoffmann & L. Ector, 2007. Comparison of biotic indices for water quality diagnosis in the Duero Basin (Spain). Archiv für Hydrobiologie Supplement 161, Large Rivers 17: 267–286.
Carvalho, L., R. Cortes & A. A. Bordalo, 2011. Evaluation of the ecological status of an impaired watershed by using a multi-index approach. Environmental Monitoring and Assessment 174: 493–508.
Cemagref, 1982. Etude des Méthodes Biologiques d’Appréciation Quantitative de la Qualité des Eaux. Rapport Division Qualité des Eaux Lyon – Agence financière de Bassin Rhône-Méditerranée-Corse, Pierre Bénite. Cemagref, Lyon: 218 pp.
Chessman, B. C., N. Bate, P. A. Gell & P. Newall, 2007. A diatom species index for bioassessment of Australian rivers. Marine and Freshwater Research 58: 542–557.
Council of the European Communities, 2000. Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. Official Journal of the European Communities L327: 1–72.
Crompton, D., 2002. Cardiff Bay Barrage. Proceedings of the Institution of Civil Engineers – Water & Maritime Engineering 154: 81–88.
Daenport, A. J., A. M. Gurnell & P. D. Armitage, 2001. Classifying urban rivers. Water Science & Technology 43: 147–155.
Dickman, M. D., M. R. Peart & W. W.-S. Yim, 2005. Benthic diatoms as indicators of stream sediment concentration in Hong Kong. International Review of Hydrobiology 90: 412–421.
Duh, J.-D., V. Skandas, H. Chang & L. A. George, 2008. Rates of urbanisation and the resiliency of air and water quality. Science of the Total Environment 400: 238–256.
Duigan, C., T. Hatton-Ellis, J. Latham, S. Campbell & B. Mathews, 2009. River conservation in Wales: a synthesis. In Williams, D. D. & C. A. Duigan (eds), The Rivers of Wales. A Natural Resource of International and Historical Significance. Backhuys Publishers/Leiden & Margraf Publishers, Weikersheim: 235–268.
Ellis, J. & V. Adriaenssens, 2006. Uncertainty estimation for monitoring results by the WFD biological classification tools. WFD Report GEHO1006BLOR_E_P. Environment Agency, Bristol. http://publications.environment-agency.gov.uk/PDF/GEHO1006BLOR-E-E.pdf. Accessed 2 Apr 2011.
Environment Agency, 2008. A consultation on the Draft River Basin Management Plan, Severn River Basin District. http://www.environment-agency.gov.uk/static/documents/Research/06_Severn_RBMP_Consultation_main_document.pdf. Accessed 2 Apr 2011.
Falasco, E., F. Bona, G. Badino, L. Hoffmann & L. Ector, 2009. Diatom teratological forms and environmental alterations: a review. Hydrobiologia 623: 1–35.
Fore, L. S. & C. Grafe, 2002. Using diatoms to assess the biological condition of large rivers in Idaho (U.S.A.). Freshwater Biology 47: 2015–2037.
Gómez, N., 1998. Use of epipelic diatoms for evaluation of water quality in the Matanza-Riachuelo (Argentina), a Pampean plain river. Water Research 32: 2029–2034.
Gómez, N., 1999. Epipelic diatoms from the Matanza-Riachuelo river (Argentina), a highly polluted basin from the pampean plain: biotic indices and multivariate analysis. Aquatic Ecosystem Health & Management 2: 301–309.
Griffith, M. B., B. H. Hill, F. H. McCormick, P. R. Kaufmann, A. T. Herlihy & A. R. Selle, 2005. Comparative application of indices of biotic integrity based on periphyton, macroinvertebrates, and fish to southern Rocky Mountain streams. Ecological Indicators 5: 117–136.
Hall, R. I. & J. P. Smol, 1999. Diatoms as indicators of lake eutrophication. In Stoermer, E. F. & J. P. Smol (eds), The Diatoms: Applications for the Environmental and Earth Sciences. Cambridge University Press, Cambridge: 128–168.
Hamsher, S. E., R. G. Verb & M. L. Vis, 2004. Analysis of acid mine drainage impacted streams using a periphyton index. Journal of Freshwater Ecology 19: 313–324.
Heino, J., J. Ilmonen, J. Kotanen, H. Mykrä, L. Paasivirta, J. Soininen & R. Virtanen, 2009. Surveying biodiversity in protected and managed areas: algae, macrophytes and macroinvertebrates in boreal forest streams. Ecological Indicators 9: 1179–1187.
Hering, D., R. K. Johnson, S. Kramm, S. Schmutz, K. Szoszkiewicz & P. F. M. Verdonschot, 2006. Assessment of European streams with diatoms, macrophytes, macroinvertebrates and fish: a comparative metric-based analysis of organism response to stress. Freshwater Biology 51: 1757–1785.
Hill, B. H., A. T. Herlihy, P. R. Kaufmann, R. J. Stevenson, F. H. McCormick & C. B. Johnson, 2000. Use of periphyton assemblage data as an index of biotic integrity. Journal of the North American Benthological Society 19: 50–67.
Hirst, H., I. Jüttner & S. J. Ormerod, 2002. Comparing the responses of diatoms and macro-invertebrates to metals in upland streams of Wales and Cornwall. Freshwater Biology 47: 1752–1765.
Hirst, H., F. Chaud, C. Delabie, I. Jüttner & S. J. Ormerod, 2004. Assessing the short-term response of stream diatoms to acidity using inter-basin transplantations and chemical diffusing substrates. Freshwater Biology 49: 1072–1088.
Hürlimann, J. & F. Straub, 1991. Morphologische und ökologische Charakterisierung von Sippen um den Fragilaria capucina-Komplex sensu Lange-Bertalot 1980. Diatom Research 6: 21–47.
Iserentant, R. & D. Blancke, 1986. Sensibilité des peuplements de diatomées aux changements de qualité de l’eau. Un essai de transplantation en eau courante [A transplantation experiment in running water to measure the response rate of diatoms to changes in water quality]. In Ricard, M. (ed.), Proceedings of the Eighth International Diatom Symposium, Paris, August 27–September 1, 1984. Koeltz Scientific Books, Koenigstein: 347–354.
Jenkins, A., W. T. Sloan & B. J. Cosby, 1995. Stream chemistry in the middle hills and high mountains of the Himalayas, Nepal. Journal of Hydrology 166: 61–79.
John, A. H. & G. Williams (eds), 1980. Glamorgan County History. Volume 5. Industrial Glamorgan from 1700 to 1970. Glamorgan County History Trust, Cardiff.
Jüttner, I., S. Sharma, B. M. Dahal, S. J. Ormerod, P. J. Chimonides & E. J. Cox, 2003. Diatoms as indicators of stream quality in the Kathmandu Valley and Middle Hills of Nepal and India. Freshwater Biology 48: 2065–2084.
Jüttner, I., P. J. Chimonides & S. J. Ormerod, 2010. Using diatoms as quality indicators for a newly-formed urban lake and its catchment. Environmental Monitoring and Assessment 162: 47–65.
Kaushal, S. S., G. E. Likens, N. A. Jaworski, M. L. Pace, A. M. Sides, D. Seekell, K. T. Belt, D. H. Secor & R. L. Wingate, 2010. Rising stream and river temperatures in the United States. Frontiers in Ecology and the Environment 8: 461–466.
Kelly, M. G., 2002. Role of benthic diatoms in the implementation of the Urban Wastewater Treatment Directive in the River Wear, North-East England. Journal of Applied Phycology 14: 9–18.
Kelly, M. G., A. Cazaubon, E. Coring, A. Dell’Uomo, L. Ector, B. Goldsmith, H. Guasch, J. Hürlimann, A. Jarlman, B. Kawecka, J. Kwandrans, R. Laugaste, E.-A. Lindstrøm, M. Leitao, P. Marvan, J. Padisák, E. Pipp, J. Prygiel, E. Rott, S. Sabater, H. van Dam & J. Vizinet, 1998. Recommendations for the routine sampling of diatoms for water quality assessments in Europe. Journal of Applied Phycology 10: 215–224.
Kelly, M. G., S. Juggins, H. Bennion, A. Burgess, M. Yallop, H. Hirst, L. King, B. J. Jamieson, R. Guthrie & B. Rippey, 2007. Use of diatoms for evaluating ecological status in UK freshwaters. Science Report: SC030103. Environment Agency, Bristol. http://www.wfduk.org/LibraryPublicDocs/diatoms_sc030103. Accessed 28 Mar 2011.
Kelly, M., S. Juggins, R. Guthrie, S. Pritchard, J. Jamieson, B. Rippey, H. Hirst & M. Yallop, 2008. Assessment of ecological status in U.K. rivers using diatoms. Freshwater Biology 53: 403–422.
Kelly, M., H. Bennion, A. Burgess, J. Ellis, S. Juggins, R. Guthrie, J. Jamieson, V. Adriaenssens & M. Yallop, 2009a. Uncertainty in ecological status assessments of lakes and rivers using diatoms. Hydrobiologia 633: 5–15.
Kelly, M. G., A. Haigh, J. Colette & A. Zgrundo, 2009b. Effect of environmental improvements on the diatoms of the River Axe, southern England. Fottea 9: 343–349.
Kim, Y. S., J. S. Choi, J. H. Kim, S. C. Kim, J. W. Park & H. S. Kim, 2008. The effects of effluent from a closed mine and treated sewage on epilithic diatom communities in a Korean stream. Nova Hedwigia 86: 507–524.
Krammer, K., 1997a. Die cymbelloiden Diatomeen. Eine Monographie der weltweit bekannten Taxa. Teil 1. Allgemeines und Encyonema Part. Bibliotheca Diatomologica 36: 1–382.
Krammer, K., 1997b. Die cymbelloiden Diatomeen. Eine Monographie der weltweit bekannten Taxa. Teil 2. Encyonema part., Encyonopsis and Cymbellopsis. Bibliotheca Diatomologica 37: 1–469.
Krammer, K., 2002. Cymbella. In Lange-Bertalot, H. (ed.), Diatoms of Europe. Diatoms of the European Inland Waters and Comparable Habitats, Vol. 3. A.R.G. Gantner Verlag K.G., Ruggell.
Krammer, K. & H. Lange-Bertalot, 1986–1991. Bacillariophyceae 1. Teil: Naviculaceae, 876 pp.; 2. Teil: Bacillariaceae, Epithemiaceae, Surirellaceae, 596 pp.; 3. Teil: Centrales, Fragilariaceae, Eunotiaceae, 576 pp.; 4. Teil: Achnanthaceae. Kritische Ergänzungen zu Navicula (Lineolatae) und Gomphonema, 437 pp. In Ettl, H., J. Gerloff, H. Heynig & D. Mollenhauer (eds), Süßwasserflora von Mitteleuropa Band 2/1–4. G. Fischer Verlag, Stuttgart.
Lange-Bertalot, H., 2001. Navicula sensu stricto. 10 genera separated from Navicula sensu lato. Frustulia. In Lange-Bertalot, H. (ed.), Diatoms of Europe. Diatoms of the European Inland Waters and Comparable Habitats, Vol. 2. A.R.G. Gantner Verlag K.G., Ruggell.
Lavoie, I., S. Campeau, M. Grenier & P. J. Dillon, 2006. A diatom-based index for the biological assessment of eastern Canadian rivers: an application of correspondence analysis (CA). Canadian Journal of Fisheries and Aquatic Sciences 63: 1793–1811.
Lewis, B. R., I. Jüttner, B. Reynolds & S. J. Ormerod, 2007. Comparative assessment of stream acidity using diatoms and macroinvertebrates: implications for river management and conservation. Aquatic Conservation: Marine and Freshwater Ecosystems 17: 502–519.
Lobo, E. A., K. Katoh & Y. Aruga, 1995. Response of epilithic diatom assemblages to water pollution in rivers in the Tokyo Metropolitan area, Japan. Freshwater Biology 34: 191–204.
Mallin, M. A., V. L. Johnson & S. H. Ensign, 2009. Comparative impacts of stormwater runoff on water quality of an urban, a suburban, and a rural stream. Environmental Monitoring and Assessment 159: 475–491.
Martín, G., J. Toja, S. E. Sala, M. de los Reyes Fernández, I. Reyes & M. A. Casco, 2010. Application of diatom biotic indices in the Guadalquivir River Basin, a Mediterranean basin. Which one is the most appropriated? Environmental Monitoring and Assessment 170: 519–534.
Nather Khan, I. S. A., 1991. Effects of urban and industrial wastes on species diversity of the diatom community in a tropical river, Malaysia. Hydrobiologia 224: 175–184.
Ormerod, S. J. & I. Jüttner, 2009. Pollution effects on Welsh rivers: a damaged past, an uncertain future? In Williams, D. D. & C. A. Duigan (eds), The Rivers of Wales. A Natural Resource of International and Historical Significance. Backhuys Publishers/Leiden & Margraf Publishers, Weikersheim: 181–203.
Ormerod, S. J., M. Dobson, A. G. Hildrew & C. R. Townsend, 2010. Multiple stressors in freshwater ecosystems. Freshwater Biology 55(Suppl. 1): 1–4.
Passy, S. I., 2006. Diatom community dynamics in streams of chronic and episodic acidification: the roles of environment and time. Journal of Phycology 42: 312–323.
Paul, M. J. & J. L. Meyer, 2001. Streams in the urban landscape. Annual Review of Ecology and Systematics 32: 333–365.
Platt, R. H., R. A. Rowntree & P. C. Muick (eds), 1994. The Ecological City: Preserving and Restoring Urban Biodiversity. University of Massachusetts Press, Amherst.
Potapova, M. & P. B. Hamilton, 2007. Morphological and ecological variation within the Achnanthidium minutissimum (Bacillariophyceae) species complex. Journal of Phycology 43: 561–575.
Poulíčková, A., M. Duchoslav & M. Dokulil, 2004. Littoral diatom assemblages as bioindicators of lake trophic status: a case study from perialpine lakes in Austria. European Journal of Phycology 39: 143–152.
Prygiel, J. & M. Coste, 1993. The assessment of water quality in the Artois-Picardie water basin (France) by the use of diatom indices. Hydrobiologia 269–270: 343–349.
Prygiel, J., M. Coste & J. Bukowska, 1999. Review of the major diatom-based techniques for the quality assessment of rivers – state of the art in Europe. In Prygiel, J., B. A. Whitton & J. Bukowska (eds), Use of Algae for Monitoring Rivers III. Agence de l’Eau Artois-Picardie, Douai: 224–238.
Reichardt, E., 1999. Zur Revision der Gattung Gomphonema. Die Arten um G. affine/insigne, G. angustatum/micropus, G. acuminatum sowie gomphonemoide Diatomeen aus dem Oberoligozän in Böhmen. Iconographia Diatomologica 8: 1–203.
Rimet, F., L. Ector, H.-M. Cauchie & L. Hoffmann, 2009. Changes in diatom-dominated biofilms during simulated improvements in water quality: implications for diatom-based monitoring in rivers. European Journal of Phycology 44: 567–577.
Scullion, J. & R. W. Edwards, 1980. The effects of coal industry pollutants on the macroinvertebrate fauna of a small river in the South Wales coalfield. Freshwater Biology 10: 141–162.
Soininen, J. & K. Könönen, 2004. Comparative study of monitoring South-Finnish rivers and streams using macroinvertebrate and benthic diatom community structure. Aquatic Ecology 38: 63–75.
Torrisi, M., S. Scuri, A. Dell’Uomo & M. Cocchioni, 2010. Comparative monitoring by means of diatoms, macroinvertebrates and chemical parameters of an Apennine watercourse of central Italy: the river Tenna. Ecological Indicators 10: 910–913.
Walsh, G. & V. Wepener, 2009. The influence of land use on water quality and diatom community structures in urban and agriculturally stressed rivers. Water SA 35: 579–594.
Wang, Y.-K., R. J. Stevenson & L. Metzmeier, 2005. Development and evaluation of a diatom-based Index of Biotic Integrity for the Interior Plateau Ecoregion, USA. Journal of the North American Benthological Society 24: 990–1008.
Watanabe, T., T. Ohtsuka, A. Tuji & A. Houki, 2005. Picture Book and Ecology of the Freshwater Diatoms. Uchida Rokakuho Publishing, Tokyo.
Ziemann, H., 1982. Indikatoren für den Salzgehalt der Binnengewässer – Halobiensystem. In Breitig, G. & W. von Tümpling (eds), Ausgewählte Methoden der Wasseruntersuchung. Band II. Biologische, mikrobiologische und toxikologische Methoden. VEB G. Fischer Verlag, Jena: 89–95.
Acknowledgments
We would like to thank Gary Jones, Department of Mineralogy, Natural History Museum, London, for part of the chemical analyses, and Martyn Kelly and two anonymous reviewers for their helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: L. Ector, D. Hlúbiková & L. Hoffmann / Proceedings of the 7th International Symposium “Use of Algae for Monitoring Rivers”, Luxembourg, November 23–25, 2009
Rights and permissions
About this article
Cite this article
Jüttner, I., Chimonides, P.J. & Ormerod, S.J. Developing a diatom monitoring network in an urban river-basin: initial assessment and site selection. Hydrobiologia 695, 137–151 (2012). https://doi.org/10.1007/s10750-012-1123-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-012-1123-z