Abstract
We evaluated habitat changes of tributary (drowned river mouth) wetlands in the upper St. Lawrence River with analysis of pre-and post-regulation water levels and historical vegetation reconstruction from aerial photographs. In addition, the germination response of transplanted wetland soil was compared to understand responses to moist versus saturated hydrology. Typha stem density was sampled in reference sites under the influence of water-level regulation by the International Joint Commission (IJC) and compared to treatment sites where installed control structures held levels (<0.7 m) higher. Major hydrologic changes due to regulation included a reduction of inter-annual variability with a reduction in peak levels and periodic lows, leading to a dampening of 30–40 year water-level cycles. Wetland photo interpretation indicated that flooded and mixed habitat categories were apparent at all sites for pre-regulation in 1948, but post-regulation photos (1972 and 1994) showed encroachment of robust emergent (Typha angustifolia, T. latifolia, and T. x glauca) in these habitats. Vegetation surveys (7 years, 2001–2007) indicated that reference and treatment sites were dominated by Typha, but mean stem densities were not statistically different. Typha stem density, however, declined in response to decreased summer water level. Periodic summer low water levels coupled with higher winter levels (that promote muskrat activity) were hypothesized to have the greatest effect on reducing Typha density. Seed-bank analysis indicated that a greater diversity of plant species germinated in mesic (moist) conditions than in the saturated treatment (flooded), where Typha was the dominant seedling component. Altered hydrologic regimes and invasive Typha have had a substantial effect on habitat structure within coastal wetlands and inferences from local management of levels provide useful guidance for future system-wide regulation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The natural flow regime of a large river ecosystem encompasses all hydrologic relationships, including the magnitude, duration, and periodicity of water levels, and has a major influence on the physical, chemical, and biological environment (Poff et al., 1997). Water level and fluctuation largely determine plant assemblage composition, survival, and development (Keddy, 2000). Consequently, wetland plant communities are largely determined by flooding depth, duration, intensity, and frequency (van der Valk, 1981; Keddy, 2000; Baldwin et al., 2001; Euliss et al., 2004; Batzer & Sharitz, 2007). Interruption of natural flow has disrupted aquatic systems worldwide (Dynesius & Nilsson, 1994), and strong effects are purported for US rivers (Revenga et al., 2000). Managing river flows to maintain the ecological integrity, as well as to provide water for utilitarian purposes, is of significant societal interest (Richter et al., 2003). Water-level control of Lake Ontario and the upper St. Lawrence River began in 1960 with the completion of the Robert Moses-Saunders Power Dam, constructed as part of the St. Lawrence Seaway and Hydropower Project. Owned by the United States and Canada, operation of the dam is controlled by policies of the International Joint Commission (IJC), formed by the Boundary Water Treaty Act of 1909. The IJC implements the United States Army Corps of Engineers (ACOE) Plan 1958D with deviations (1958DD) under its 1956 Orders of Approval to regulate water levels between 74.2 and 75.4 m above sea level for Lake Ontario.
Coastal wetland ecosystem change is believed to be strongly associated with water level controls in the Great Lakes (Keddy & Reznicek, 1986; Wilcox & Xie, 2007, 2008; Wilcox et al., 2008) and the response of robust emergent Typha to hydrologic alteration is a key feature in understanding the effects of disturbance on wetlands to assess their current state and future condition. Prior to European settlement, natural forces were the mechanisms driving long-term vegetation dynamics. Since the early 1800s, human activity has created disturbances capable of altering Lake Ontario wetland ecosystems, including cumulative effects of deforestation, agriculture, changes in fire regimes, exotic and/or invasive species introductions, shoreline development, dredging and channel alteration, and system-wide effects of water-level regulation (Whillans, 1982; Wilcox, 1995; Whillans, 1996).
In context with potential changes in long-term succession processes of upper St. Lawrence River coastal wetlands, recent alteration of disturbance regimes has likely had significant cascading ecological effects. Keddy (2000) reviewed how fluctuating hydrology affects key biological components of wetlands. Research on the effects of fluctuating water levels on ecosystem processes has recently received more attention in the Great Lakes basin (Keddy & Reznicek, 1986; Hudon, 1997; Wilcox & Nichols, 2008) and elsewhere (David, 1996; Tremolieres et al., 1998; Jansson et al., 2000). Effects of hydrologic management on wetland plant communities have also received focus in the recent IJC Lake Ontario–St. Lawrence River Water Level Study. Models to predict water-level regulation effects were developed from a detailed study of plant community response for wetland geomorphic types including drowned river mouths (Wilcox & Xie, 2007). The authors’ interpretation of modeling results suggests that for years with low supply, unregulated water levels, with periodic low levels will produce the most meadow-marsh habitat (Wilcox & Xie, 2008). Although effects on Typha were included in the model (Wilcox et al., 2005), the meadow-marsh community was selected as a meaningful indicator and model results for Typha were not completely presented in model output (Wilcox & Xie, 2007, 2008).
We employed a multifaceted approach to address the question of wetland change and Typha responses that included a historical wetland interpretation where we examined system-wide alterations in hydropattern and interpreted changes in vegetation using historic aerial photographs. The ecological response of wetland vegetation was compared between manipulated treatment marshes (that held water-level higher and prevented managed fall drawdown), to reference marshes that fluctuated according to IJC regulation. Potential ecological outcomes of water levels were evaluated for the wetland plant community, with a focus on Typha and the wetland seed bank. Our specific objectives were to (1) analyze water levels pre- and post- hydrologic alteration to identify specific water-level and hydropattern changes during the period of record; (2) interpret aerial photographs of coastal wetlands to identify changes in general wetland habitat types; and (3) determine the potential effects of water level management practices on wetland plant establishment and persistence of Typha.
Study area
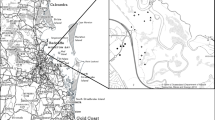
St. Lawrence River is the only natural outlet to the Great Lakes and flows 965 km from Lake Ontario to the Gulf of St. Lawrence. As the tenth largest river in the world in flow, it represents a principal North American river draining an area greater than 764,000 sq km (Mills et al., 1981). Average annual discharge at Cornwall, Ontario was 7,960 m3 s−1 from 1960 to 1990 (Water Survey of Canada, 1990). The upper river extends from the mouth of Lake Ontario, 150 km downstream to the Robert Moses Saunders Power Dam. The study was conducted within the Thousand Islands Region of the upper river. The Thousand Islands are a complex archipelago of over 1,800 islands in a heterogeneous mix of deep (<73 m) riverine environments, shoals, rocky shorelines, and bays with extensive littoral communities and nearshore coastal emergent marshes, many of which are fed by small- to medium-sized tributaries. Tributary wetland study sites included two areas within the NYSDEC French Creek Wildlife Management Area in Clayton, New York: French Creek (13.97 ha) that was split into upper and lower sections for photointerpretation and the Carpenters Branch of French Creek (9.03 ha). Little Cranberry Creek (3.05 ha), Cranberry Extension (3.36 ha), and Cranberry Marsh (35.63 ha) study sites drain into the Goose Bay study site used in photointerpretation and are associated with the NYSDEC Cranberry Creek Wildlife Management Area, near Alexandria Bay New York. The Chippewa Tributary (4.26 ha) site is a small tributary that drains to Chippewa Bay (Fig. 1).
Study sites in the Thousand Islands Region of the upper St. Lawrence River in Clayton and Alexandria Bay, New York, including French Creek and Carpenters Branch, Little Cranberry Creek, Cranberry Extension, Cranberry Marsh, and Chippewa Tributary. Governors Island is the location of the Thousand Islands Biological Station
Methods
Lake Ontario–St. Lawrence River water levels
Water-level data from Lake Ontario (1900–2002), and the upper St. Lawrence River (1970–2002) were used to compare pre- and post regulation differences in timing, magnitude, and periodicity of water levels. Because water-level data from the upper St. Lawrence River were not available prior to 1970, data from Lake Ontario at Oswego, New York (NOAA gauging station), about 70 km from the mouth of the St. Lawrence River, were used. We present regression and correlation analyses between three Lake Ontario stations (Lake): Rochester (1970–2002), Oswego (1970–2002), and Cape Vincent (1970–2002), and two St. Lawrence River stations (River): Alexandria Bay (1983–2002) and Ogdensburg (1970–2002), to validate use of Lake Ontario water-level data to represent River fluctuations.
Simulation of St. Lawrence River water levels with the regulation influence removed (natural predicted) were compared to pre- and post-regulation monthly levels to evaluate effects. Natural predicted levels were determined through development of a regression model for pre-regulation Lake Ontario against Lake Erie (1900–1955). Lake Erie is an unregulated Great Lake that connects to Lake Ontario via the Niagara River and Welland Canal system. Lake Ontario receives over 80% of its inflow from Lake Erie (Environmental Protection Agency and Government of Canada, 1995); therefore, we expected hydrologic characteristics (e.g., periodicity, fluctuations) to be similar between the lakes. The predictive relationship developed was applied to the post-regulation period 1960 to 2002 to reconstruct water levels of Lake Ontario independent of the influence of regulation and to the pre-regulation period 1900–1955 for model validation. Differences in mean Lake Ontario water level from pre- to post-regulation and between actual and natural predicted levels for spring (March–May), summer (June–August), fall (September–November), and winter (December-February) were visualized graphically and by calculation of the mean squared deviation between the periods. We also compared the natural predicted water-level to that simulated by the IJC hydrodynamic modeling for the LOSLR Water Levels study (Caldwell & Fay, 2002) for the post-regulation period using a simple linear regression fit of the data series and a two-tailed paired sample t test (Zar, 1999).
Historical wetland interpretation
Aerial photograph interpretation was used to examine vegetation changes between pre-regulation years (1941 and 1948) and post-regulation years (1972 and 1994). Photo interpretation was limited to available high quality images where habitat categories could be distinguished and included our study sites at Cranberry Marsh (1948, 1972, 1994); Little Cranberry Creek (1948, 1972, 1994), and their mouth at Goose Bay (1941, 1948, 1972, 1994), in addition to French Creek (1948, 1994) and Carpenters Branch (1948, 1994; Fig. 1). No imagery was interpreted for Chippewa Tributary and Cranberry Extension. All photos were obtained from the SUNY College of Environmental Science and Forestry (ESF) Mapping Sciences Laboratory Archives, and date of photo, scale, resolution, and lineage for each image was variable among years (Table 1). Mean monthly water levels were relatively low at 74.37 m for the 1941 photo and high for the remaining photos: 75.05 m elevation in 1948, 75.20 m in 1972, and 75.10 m in 1994. We digitized photographs at a 2000 dpi resolution and used ESRI ArcView™, version 3.3 (2002) to delineate general habitat categories. The photographs were geospatially referenced (rubbersheeting) to the place all images at approximately the same resolution with 2003 natural color digital orthroimagery (30 cm raster) provided by the New York State GIS Clearinghouse (http://www.nysgis.state.ny.us). These digital images have been orthorectified and geospatially referenced to UTM units at a horizontal positional accuracy listed at ±4 ft.
Wetlands on images were delineated into six broad habitat categories: open water (channel), robust emergent (Typha-dominated), flooded vegetation (flooded), mixed (patchy areas of visibly flooded Typha), wet meadow, and shrub. Habitat categories from 1994 photographs were ground-verified from independent vegetation surveys (Halpern, unpublished data), as well as through vegetation maps created in 1974 (Geis & Kee, 1977). For each wetland, we calculated the proportion of surface area covered by each habitat category and compared these proportions between years by comparing polygon area.
Water-level manipulation
Six tributary sites (i.e., altered as treatments and IJC-regulated) were compared to quantify the ecological effects of water-level management on plant community composition and structure. Study sites were selected to be representative of typical drowned river mouth wetlands in the region and for treatment sites that they were candidates for manipulation of water levels with water-control structures. Cranberry Marsh (water-control structure installed in 1998) and Carpenters Branch (water-control structure installed in September 2002) were manipulated (treatment sites) to mimic higher water levels. At Cranberry Marsh, levels were increased from 1999 until 2005 when water-level control ceased. Water-level management was initiated on Carpenters Branch in fall 2002 and continued through the study period. Reference sites exposed to ambient St. Lawrence River water levels (regulated by the IJC) included Chippewa Tributary, Little Cranberry Creek, Cranberry Extension, and French Creek located in the Town of Clayton, NY (Fig. 1). Water levels for treatment sites were maintained between 74.9 and 75.7 m elevation (mean = 75.4, SD = 0.18), compared to IJC regulated levels (mean = 74.7, SD = 0.18). Water levels of reference sites were measured using the NOAA gauging station at Alexandria Bay, NY.
Typha stem density
In order to evaluate existing Typha structure, we established permanent vegetation sampling transects within each study site along an elevation gradient (+2.5; +1.0; 0.0; −0.5; −1.0; −1.5; −2.5 feet); these elevations correspond to 75.80; 75.34; 75.04; 74.89; 74.73; 74.58; 74.28 m IGLD 1985, respectively, extending from upland habitat to open channel using a laser plane to establish sampling points. Sampling points were staked, marked with flagging, and locations recorded with GPS. Vegetation measurements at each sampling point were made within a 1-m2 quadrat at each site from 2001 to 2007, except for Cranberry Marsh, which was not sampled in 2001. While plant species composition and structure were quantified, we focus here on counts of Typha stem density which includes T. latifolia L., T. angustifolia L., and the hybrid T. x glauca Godr. as Typha sp.
We were interested in evaluating the effect of local water-level regulation on Typha density and in determining the distribution of Typha across the elevation gradient. To address the site-specific water-level-regulation effects, we explored two different ANOVA models. First, we compared Typha density across management treatments by coding sites/years as managed (Cranberry Marsh 2002–2004 and Carpenter’s Branch 2004–2007) or non-managed (reference sites, Cranberry Marsh 2005–2007, and Carpenter’s Branch 2001–2003) and included elevation, site, and year as blocking variables to partition and account for non-management-related variation and dependence on time. Next, to address the non-uniform distribution of samples across treatments (Table 2), we tested for differences in Typha density across sites, years, and their interaction blocking by elevation. Then, to isolate and explore patterns of Typha zonation across the elevation gradient, we blocked by site and year and tested for significant differences among elevations. In all analyses, we used Type III sum of squares ANOVA (SAS Institute, 1998, a priori significance level of α = 0.05) to evaluate model interactions and main effects, then used least significant means post hoc pairwise comparisons, which account for variation controlled by blocking variables in the full model, to determine specific differences within treatment variables. In addition, mean stem density at reference sites was plotted against peak growing season water level (June–August) and fitted with simple regressions to examine potential relationships.
Seed-bank germination
A seed-bank germination trial was conducted to evaluate plant community germination and establishment in response to changes in wetland hydrology. Composite soil samples (n = 29) were collected along previously described transects during spring 2002. Sample cores were excavated using an aluminum can opened at both ends (8.25 cm-diameter by 10 cm-depth) within a 2 m2 quadrat along five Cranberry Extension transects, eight transects at Little Cranberry Creek, Chippewa Tributary, and Carpenters Branch. Multiple cores were taken within transects across elevations and were composited, homogenized, and a subsample of 4,000 cm3 transported to the greenhouse for germination. Subsamples were divided into two equal treatments including mesic (moist) and saturated following Warr et al. (1993). Saturated and mesic treatments were selected to represent conditions of the wetland moisture gradient that may be encountered under differing water levels. Subsamples were watered every 2–3 days in the greenhouse for 15 months, until no new species germinated. Specimens were identified according to Gleason & Cronquist (1991), counted, and removed. Unidentified seedlings were transplanted and allowed to grow and flower to assist in their identification. Taxa stem counts from each subsample (each representing 200 cm2 of wetland surface) were multiplied by a factor of 50 to estimate density (stems/m2). We determined the total number of germinants, species richness (S), and Simpson’s Evenness (E D or ‘equitability’) for each transect and tested for differences among treatment (mesic vs. saturated) using analysis of variance and blocking by site to control for inter-site variability. Transects represented individual replicates of each treatment within sites. Several variations of Simpson’s evenness were available; here, we used the following calculation:
where p 2i is the proportion of the number of germinants of each taxa to the total number of germinants in the sample squared. Evenness (‘equitability’) in this form of Simpson’s index takes a value between 0 and 1, where 0 is complete monoculture and 1 is perfect evenness.
Typically, field sampling data of this type are rarefied to compensate for differences in the number of organisms sampled. Since substrate samples were of equal volume (eliminating area effects), competitive interactions were removed, all seeds within samples had equal and ample opportunity to germinate, and all germinants were counted, rarefaction was unnecessary. Taxa were grouped into seven “functional guilds” based on life history characteristics: graminoid emergents, grasses, meadow sedges, herbs, ferns, submerged aquatic vegetation (SAV), and floating-leaved for a graphical representation of seed bank response. The “graminoid emergent” guild included cattail (Typha), iris (Iris), bulrushes (Schoenoplectus), bur-reed (Sparganium), aquatic sedges (Carex), and spike-rushes (Eleocharis).
Results
Lake Ontario–St. Lawrence River water levels
As expected, water levels recorded at gauging stations were highly correlated and had similar regression slopes that indicated strong synchrony among sites (Table 3). Although most slopes were greater than 1.0, the magnitude of difference was generally small, with patterns observed in Lake Ontario similar to those in the upper St. Lawrence River. Pre-regulation Lake Ontario water levels also demonstrated a strong similarity to those of Lake Erie for 1900–1955 (R 2 = 0.90), justifying our development of a natural predicted water level from this relationship (Fig. 2). The natural-predicted water level simulation had a close relationship to that developed with hydrodynamic modeling as part of the LOSLR water level study and paired mean differences were not statistically different from zero (t = −0.834, df = 432, P = 0.404). A simple regression fit of the water level prediction datasets also show a close relationship (R 2 = 0.92) and slope approximates a 1:1 relationship (Fig. 2). Changes in mean actual annual Lake water level from pre- to post-regulation showed a 7 cm increase, while natural predicted level had a much greater increase of 40 cm, a difference of 33 cm. In comparison to natural predicted levels, actual levels were much lower for all seasons in the post-regulation period, due to the prevalence of high supplies. For actual levels, the difference was most pronounced for spring and summer (Fig. 3).
Pre-regulation mean monthly water levels from Lake Ontario at Oswego, NY plotted against levels data from unregulated Lake Erie at Cleveland, OH (m IGLD 1985) for 1900 to 1955 (top panel). Data were obtained from the NOAA Center for Operational Oceanographic Products and Services (www.tidesandcurrents.noaa.gov). Natural-predicted water levels generated from the above relationship and applied to the post-regulation period plotted against the IJC Water-Levels Study hydrodynamic simulation the same period (bottom panel)
The variability of mean actual levels decreased from pre- to post-regulation, where natural predicted levels showed more variation than actual levels in the post-regulation period for all seasons. The actual pre-regulation (1900–1955) seasonal range in water levels was 59.7 cm (SD = 18.6) and similar to those predicted by the regression relationship; natural-predicted levels of 58.7 (SD = 15.8). In contrast, the natural predicted (69.1 cm, SD = 19.6) post-regulation (1960–2002) seasonal range was 16.4 cm greater than actual levels (52.7 cm, SD = 15.8). Interannual variability as measured by mean squared deviation showed a decrease pre- to post-regulation (1.14 vs. 0.34 m2) for actual levels compared to deviation for natural predicted pre- and post-regulation levels (1.01 and 1.41 m2). The natural 30–40 year water level periodicity of extreme lows and internal 7–10 year cycles from lows to highs are absent in the actual post-regulation period, whereas natural predicted levels clearly show these fluctuations (Fig. 4).
Historical wetland interpretation
Differences were observed between pre- and post-regulation wetland habitat categories delineated from historical aerial photographs. In 1948, most wetlands were dominated by flooded, mixed, or wet meadow habitats, where shallow emergent, floating-leaved, and submersed aquatic plants likely occurred (Fig. 5). Robust emergent vegetation increased in all wetlands between 1948 and 1994. Similarly, mixed habitat type (flooded emergent) decreased at all sites between 1948 and 1994. The open water category, indicative of the channel, also increased between 1948 and 1994 at all wetlands. This is may be an artifact of seasonal differences between aerial photographs used (May vs. September) or a response to maintained higher water levels. The wet meadow habitat category decreased in two sites (French Creek downstream and Carpenters Branch) and increased in two sites (French Creek upper and Little Cranberry Creek).
Goose Bay was the only site where photographs were interpreted for 4 years (two pre-regulation: 1941 and 1948 and two post-regulation: 1972 and 1994). In 1941, during low water levels (74.37 m IGLD 1985), the robust emergent category contributed the highest relative proportion of the wetland area (93%). In the 1948 photointerpretation, robust emergent vegetation, although still dominant, had decreased to 32%, with flooded, mixed, and wet meadow categories contributing to overall wetland plant community. Post-regulation photographs (i.e., 1972 and 1994) indicated an increase in robust emergent category from 1948 (32%) to 1972 (77%), with no discernible difference in 1994 (77%) compared to 1972.
Typha stem density
We did not observe a statistically significant difference in Typha stem density due to local water-level management in Cranberry Marsh (2002–2004) and Carpenter’s Branch (2004–2007) relative to reference sites and the unmanaged periods of managed sites (full model: F 4,1082 = 5.33, P < 0.0001, management effect: F 1,1082 = 0.37, P = 0.5417). Finer scale analysis of site × year differences in Typha stem density (with 2001 omitted because Cranberry Marsh was not sampled; full model: F 36,971 = 3.38, P < 0.0001) resulted in a non-significant interaction between year and site (F 25,971 = 0.95, P = 0.5328) and significant main effects (site: F 5,971 = 7.67, P < 0.0001, year: F 5,971 = 11.04, P < 0.0001). Differences among sites were not consistent with water level treatments (also inferred by the non-significant interaction) but, instead, showed strong geographic trends (Fig. 6). The western-most (upstream) sites, French Creek and Carpenter’s Branch both had significantly lower Typha density than any of the other sites (13.68 ± 0.89 stems m−2 SE and 14.91 ± 0.94 stems m−2, respectively). Little Cranberry Creek had the highest density of Typha (20.59 ± 0.78 stems m−2), but aside from French Creek and Carpenter’s Branch, was only significantly greater than Cranberry Marsh (17.45 ± 0.80 stems m−2; Fig. 6, P < 0.04 in all significant pairwise comparisons). During our study period there was a statistically significant increase in Typha density from 2002 (18.12 ± 0.97 stems m−2) to 2003 (20.83 ± 1.03 stems m−2), a non-significant decrease through 2005, and then 2006 (13.65 ± 0.80 stems m−2) and 2007 (13.98 ± 0.67 stems m−2) had a significant decrease in Typha density (Fig. 6, P < 0.02 for all significant pairwise comparisons).
Comparison of Typha mean stem density for drowned river mouth wetlands at manipulated sites at Carpenters Branch (CB) in Clayton, NY and Cranberry Marsh (CM) in Alexandria Bay, NY and reference wetlands; Cranberry Extension (CE), Chippewa Tributary (CT), Little Cranberry Creek (LCC), and French Creek (FC). Error bars represent the standard error around the mean. The number of annual site-specific transects sampled are listed in Table 2. No significant differences in Typha stem density occurred for FC and C, these sites were statistically different (α = 0.05) from all other sites, and CM was different than LC. Between years, 2002 was statistically different than 2003; 2006 and 2007 were different than all other years compared
We found statistically significant differences in Typha density among elevation zones (full model F 6,1082 = 12.93, P < 0.0001, elevation: F 4,1082 = 14.40, P < 0.0001). Maximum density occurred at the 74.58 m IGLD elevation (20.99 stems•m−2 ± 0.77) and, with the exception of 74.73 m IGLD (19.20 stems•m−2 ± 0.66), was significantly greater than at all other elevations. The lowest density of Typha occurred at the extremes; downslope along the edge of the water channel (74.28 m IGLD, 12.39 stems•m−2 ± 1.00) and upslope along the riparian interface (75.04 m IGLD, 13.71 stems•m−2 ± 0.91). At reference sites, simple linear regressions revealed a relationship between Typha density and mean growing season water level (June–August; Fig. 7). A pattern of increasing stem density with increasing water level was observed at three sites. The French Creek site deviated from this pattern in 2002, where the lowest stem density was associated with a relatively high summer water level.
Seed bank germination
A total of 78 taxa germinated from the seed bank trial. Five taxa did not flower and remained unidentified to the species level (Carex spp. 1, 2, and 3, and aster 1 and 2). Across all sites, the mesic treatment resulted in significantly greater species richness (mean = 13.7 species per sample and 67 total species across all mesic samples) and total number of germinants (mean = 1,758/sample) than the saturated treatment (mean = 8.21 species per sample and 48 total species across all mesic samples and 938 germinants per sample on average, species richness F 1,57 = 40.72, P < 0.0001 and total number of germinants F 1,57 = 12.48, P = 0.0008) due to the large number of herbs that germinated under mesic conditions. In contrast, evenness, although relatively low in both treatments (mean 0.30 across all mesic samples and 0.38 in saturated samples), was significantly greater under saturated conditions (F 1,57 = 5.56, P = 0.0220, Table 4).
Calamagrostis canadensis (Michx.) P. Beauv. was the dominant component of the seed bank in the mesic treatment for Cranberry Extension (3,240 stems/m2, SD = 371), Little Cranberry Creek (3,569 stems/m2, SD = 597), Chippewa Tributary (2,644 stems/m2, SD = 579), and Carpenters Branch (5,600 stems/m2, SD = 438; Fig. 8). Within Cranberry Extension, the most dominant component of the seed bank was Sagittaria latifolia Willdenow in the saturated treatment (1,050 stems/m2, SD = 321). Typha was dominant in the saturated treatment (1,031 stems/m2, SD = 150) at Little Cranberry Creek and Chippewa Tributary (1,863 stems/m2, SD = 192). Schoenoplectustabernaemontani (C.C. Gmelin) Palla was the most common at Carpenters Branch (525 stems/m2, SD = 175) under saturated conditions. At the Cranberry Extension, arrowhead (Sagittaria latifolia) from the herb group and rice-cut grass (Leersia orzyoides (L.) Sw.) from the grass group had greatest density under saturated conditions. Several non-native species germinated from seed-bank samples, including Lythrum salicaria L. (purple loosestrife) in Chippewa Tributary samples, and Hydrocharis morsus-ranae L. (European frogsbit) and Rorippa nasturtium-aquaticum L. (watercress) were present at all four sites.
Densities of seedlings (stems/m2) germinated from the Cranberry Extension, Little Cranberry, Chippewa Tributary seed bank under saturated (black) and mesic (white) conditions. The species are grouped into categories of graminoid emergents, grasses, meadow sedges, herbs, ferns, submerged aquatic vegetation (SAV), and floating-leaved species. Only taxa with densities greater than 10 stems/m2 are listed
Discussion
All hydrologic characteristics considered (magnitude, duration, and periodicity of water levels) demonstrated major alterations relative to predictions of unmanaged levels. Lake Michigan and Huron water levels are known to fluctuate as quasi-periodic cycles of 30–35 years and 160 years (Baedke & Thompson, 2000). Cohen & Robinson (1975) demonstrated with Fourier analysis 1-year seasonal and 8-, 11-, 22-, and 36-year low frequency, long-term patterns in a study of cyclic water-level fluctuations in Lake Ontario from 1860 to 1971. Long-term predictions made from the analysis did not, however, consider the effect of regulation. During pre-regulation, levels and outflows had a more direct linear relationship compared to the regulation period (International Lake Ontario Study Board, 2006). Controlled releases guided by the 1956 IJC Orders of Approval have altered this natural relationship via regulation. The natural hydraulic constraints on the system through its channel morphometry were permanently altered by removal of natural limestone sills that served as a controlling point near Ogdensburg, NY and Prescott, Ontario. We did not repeat the earlier analyses, but it was clear from water-level reconstruction that regulation has dampened hydropattern features, especially longer term cycles and reduction of within-year or seasonal variability through management against extremes. It is also clear that mean annual levels have been on the rise, and three peak high levels in 1973, 1986, and 1997 (~76.0 m IGLD) would have been more extreme in absence of regulation.
Concomitant with the observed hydrologic alterations, changes in wetland habitat categories were observed. The aerial photographs and digital images indicated that wetlands changed in vegetative composition. Photograph interpretation showed a reduction in flooded and mixed-vegetation, and a post-regulation dominance of these areas by Typha. A pattern of Typha encroachment was well-demonstrated in this geographic region by Wilcox et al. (2008) and Cooper et al. (2008). Wilcox et al. (2008) completed a detailed interpretation via imagery over five decades for multiple wetland geomorphic types including drowned river mouths representative of our study sites. Findings regarding wetland habitat changes before and after regulation were similar to our imagery interpretations. Wilcox et al. (2008), however, go further with inferences regarding wetland zonation effects due to flooding/dewatering history at specific elevations and the mechanism of upslope invasion of Typha with high water levels in relation to loss of sedge meadow habitat.
A detailed pollen analysis from a wetland core (10 m) extracted, dated, and interpreted from Goose Bay at the mouth of Cranberry Creek indicated a pronounced dominance of robust emergent Typha angustifolia/sparganium pollen in the recent sediment record that replaced sedge pollen as the dominant type c. 1900 A.D, well prior to the influence of lake-level regulation (Beland, 2003). Increases in sediment bulk density suggest that anthropogenic land-use changes during the 19th century profoundly changed wetland structure and were a causative factor in converting the wetland from a sedge to Typha dominated marsh. Prior to land conversion, wetland development proceeded in a balance of natural influences of autogenic succession and water depth controlled by isostatic rebound (Beland, 2003). Although this investigation did not fully differentiate between the forms of Typha, the current water level regime has been shown to increase T. x glauca invasion at higher wetland elevations, while T. angustifolia, which is tolerant of deeper waters (Grace & Wetzel, 1992), has invaded deeper waters. Typha latifolia has not been invasive (Wilcox et al., 2008), but also showed an increase in pollen levels in sediments c. 1930.
Seed-bank evidence does indicate that increased water levels and a lack of variability since regulation may contribute to the continued Typha dominance of the wetland. Our findings were consistent with expectations following relationships defined by Keddy & Reznicek (1986). The four seed bank study sites were similar in taxa composition when the seed bank was allowed to germinate under mesic conditions, with Calamagrostis canadensis as the most common species at all sites and a diversity of less common herb taxa at all sites. Under saturated conditions, graminoid emergents, primarily Typha, were dominant at two sites. If water levels continue to remain relatively high, without periodic lows during the growing season, our results indicate that Typha could dominate seed germination, suppressing the sedge meadow, herb, and fern guilds. A compounding factor is the abundant litter produced by Typha which can further suppress the existing seed bank (Farrer & Goldberg, 2009). The greatest effects will likely be experienced by annuals that rely on seed germination for propagation. Alternatively, Typha productivity within well-established mats probably persists through perennial shoots, but seed-bank germination could be important following physical disturbances and in upslope migration of Typha where rhizomes do not exist.
No statistically discernable relationship of Typha stem density between treatment and reference wetlands was observed despite the planned alterations in levels. A relationship of increasing Typha stem density with increasing summer growing season water levels was observed for reference sites. Gathman et al. (2005), in a study of plant community response in coastal wetlands of Lake Huron, observed a similar increase in Typha angustifolia during a period of flooding and observed rapid changes in overall emergent stem density associated with flooding depth. From our data in reference sites, it appears that with moderately higher growing-season water levels, Typha growth was facilitated. It is possible in treatment locations, where water levels were held higher, stem densities would have been expected to increase relative to reference areas. Maximum altered water levels for treatment sites were below those reported to cause Typha die back (Grace & Harrison, 1986; Sodja & Solberg, 1993). In addition, extensive floating Typha mats with buoyant rhizomes such as in our study areas may make stands more resilient to such flooding disturbances (Hogg & Wein, 1988; Krusi & Wein, 1988). Treatment and reference wetlands therefore may have arrived at a similar vegetation state because of independent mechanisms, and Typha density could have been greater for treatment sites if levels were not also maintained higher over winter months.
The potential for increased production of Typha in treatment wetland water depths may have been offset by increased herbivory and disturbance by overwintering muskrats (Ondatra zibethicus L.) in treatment sites. Many investigators have demonstrated that water levels and food presence are the most important variables influencing muskrat abundance (Bellrose & Brown, 1941; Errington, 1961; Thurber et al., 1991; Clark & Kroeker, 1993). Muskrats are also well-known for their preference of Typha as a forage species, and it is considered the most important plant associated with house and mound construction (Takos, 1947; Bellrose, 1950; Bishop et al., 1979; Lacki et al., 1990; Clark, 1994, 2000). Research conducted in our study areas during a similar period (2001–2006; Toner, 2006) as a companion study indicated that muskrats can remove significant quantities of Typha biomass. Consistent with this hypothesis, muskrat activity and house density was low or absent from the reference sites and was not expected to have a significant effect on Typha stem density (Toner et al., in press). The study indicated that regulated low fall levels prevented muskrat access to floodplains and their distribution was generally limited to channel edges. Conversely, Typha consumption estimates by muskrats for Cranberry Marsh reached levels of 28% of the standing biomass, and house density was highest for Carpenters Branch following treatment (Toner, 2006).
The observed strong geographic trend of lower cattail stem density in all wetlands (treatment and control) in the French Creek area (including Carpenter’s Branch) versus the Cranberry Marsh area may be explained by differences in local watershed conditions. French Creek sites (including Carpenters Branch) have an abundance of groundwater inputs known as the Frontenac Springs that may slightly dampen effects of lake-level management and influence soil moisture conditions. Many other factors, such as nutrients (Woo & Zedler, 2002), competition, disturbance, and the suppression of the seed bank by Typha litter (Farrer & Goldberg, 2009) also could have influenced Typha and were not considered in our study.
Management considerations
Our natural-predicted water levels indicated a mean annual water level 33 cm higher than that observed with regulation. These conditions would likely have increased muskrat disturbance effects within the wetlands. At the same time, the higher overall water levels in the absence of regulation plausibly could have increased open water habitats at lower elevations and increased Typha movement upslope further into wet-meadow and woody plant zones. The observed relationship of higher Typha stem density to increased water levels during the growing season months of June through August suggests that when higher water levels are present during the growing season, Typha density will likely increase, especially if a fall drawdown limits muskrat and physical disturbance. High water levels also create conditions known to damage the Typha mats (Geis & Kee, 1977) and cause downstream transport of cattail mats. For example, an estimated 8 ha Typha island is known to have broken away in Eastern Lake Ontario during a high water event in 1973 (Author, personal communication). These floating mats create opportunities for vegetation change due to exposure of underlying sediments and seed banks. Conversely it is known that floating mats may also damage suitable habitats and colonize new areas. In our drowned river mouth study areas, floating mats generally appear to rise and settle in place with changing water levels and major changes in mat structure were not observed during the study period.
Water-level regulation in the system has dampened natural fluctuations in periodicity between highs and lows that are believed to regulate both disturbance and wetland zonation processes ultimately selecting for dense Typha stands (Grace, 1989; Keough et al., 1999). The demonstrated loss of decadal and annual variability in water levels also appears to constrain germination (Keddy & Reznicek, 1986) and persistence of diverse wetland plant communities (Wilcox et al., 2008). Based on our data, more natural fluctuations including low growing season levels would promote greater diversity and germination of sedges and grasses within wet meadow habitats, and reduce Typha stem densities. Periodic fall and winter high levels would promote muskrat disturbance and herbivory within Typha mats. Comparisons of treatment and reference wetlands in the Great Lakes serve as model systems and provide useful inferences to guide water-level-regulation policy targeting environmental benefit. To achieve improved results for wetlands, regulators should consider the dynamics of Typha and other invasive species in the planning process.
References
Baedke, S. J. & T. A. Thompson, 2000. A 4,700 year record of lake level and isostasy for Lake Michigan. Journal of Great Lakes Research 26: 416–426.
Baldwin, A. H., M. S. Egnotovich & E. Clarke, 2001. Hydrologic change and vegetation of tidal freshwater marshes: field, greenhouse, and seed-bank experiments. Wetlands 21(4): 519–531.
Batzer, D. P. & R. R. Sharitz, 2007. Ecology of Freshwater and Estuarine Wetlands. University of California Press, Berkeley, CA, USA.
Beland, M., 2003. Holocene vegetation changes in a St. Lawrence River coastal wetland and surrounding uplands: effects of climate change and anthropogenic disturbance. Master’s Thesis, State University of New York, College of Environmental Science and Forestry, Syracuse, New York.
Bellrose, F. C., 1950. The relationship of muskrat populations to various marsh and aquatic plants. Journal of Wildlife Management 14: 299–315.
Bellrose, F. C. & L. G. Brown, 1941. The effect of fluctuating water levels on the muskrat population of the Illinois River Valley. Journal of Wildlife Management 5: 206–212.
Bishop, R. A., R. D. Andrews & R. J. Bridges, 1979. Marsh management and its relationship to vegetation, waterfowl, and muskrats. Proceedings of the Iowa Academy of Science 86: 50–56.
Caldwell, R., & D. Fay, 2002. Lake Ontario Pre-project outlet hydraulic relationship final report. Lake Ontario-St. Lawrence River Water Levels Study Report, International Joint Commission.
Clark, W. R., 1994. Habitat selection by muskrats in experimental marshes undergoing succession. Canadian Journal of Zoology 72: 675–680.
Clark, W. R., 2000. Ecology of muskrats in prairie wetlands. In Murkin, H. R., A. G. van der Valk & W. R. Clark (eds), Prairie Wetland Ecology: The Contribution of the Marsh Ecology Research Program. Iowa State University Press, Iowa: 287–313.
Clark, W. R. & D. W. Kroeker, 1993. Population dynamics of muskrats in experimental marshes at Delta, Manitoba. Canadian Journal of Zoology 71: 1620–1628.
Cohen, B. P. & J. E. Robinson, 1975. Cyclic fluctuations in water levels of Lake Ontario. Computers & Geosciences 1: 105–108.
Cooper, J. E., J. V. Mead, J. M. Farrell & R. G. Werner, 2008. Coexistence of pike (Esox lucius) and muskellunge (E.masquinongy) during early life and the implications of habitat change. Hydrobiologia 601: 41–53.
David, P. G., 1996. Changes in plant communities relative to hydrologic conditions in the Florida Everglades. Wetlands 16: 1–23.
Dynesius, M. & C. Nilsson, 1994. Fragmentation and flow regulation of river systems in the northern third of the world. Science 266: 753–762.
Environmental Protection Agency and the Government of Canada. 1995. The Great Lakes: An Environmental Atlas and Resource Book, 3rd Ed. Great Lakes National Program Office, Chicago, Ill: 49 pp.
Errington, P. L., 1961. Muskrats and Marsh Management. Wildlife Management Institution Publication. University of Nebraska Press, Lincoln: 183 pp.
Euliss N. H. Jr., J. W. LaBaugh, L. H. Fredrickson, D. M. Mushet, M. K. Laubhan, G. A. Swanson, T. C. Winter, D. O. Rosenberry & R. D. Nelson, 2004. The wetland continuum: a conceptual framework for interpreting biological studies. Wetlands 24: 448–458.
Farrer, E. C. & D. E. Goldberg, 2009. Litter drives ecosystem and plant community changes in cattail invasion. Ecological Applications 19: 398–412.
Gathman, J. P., D. A. Albert & T. M. Burton, 2005. Rapid plant community response to water level peak in northern Lake Huron coastal wetlands. Journal of Great Lakes Research 31: 160–170.
Geis, J. W. & J. L. Kee, 1977. Coastal wetlands along Lake Ontario and St. Lawrence River in Jefferson County, New York. State University of New York, College of Environmental Science and Forestry, Syracuse, New York.
Gleason, R. A. & A. Cronquist, 1991. Manual of Vascular Plants of Northeastern United States and Adjacent Canada, New York Botanical Garden. Bronx, NY.
Grace, J. B., 1989. Effects of water depth on Typha latifolia and Typha domingensis. American Journal of Botany 76: 762–768.
Grace, J. B. & J. S. Harrison, 1986. The biology of Canadian weeds. Typha latifolia L., Typha angustifolia L., and Typha x glauca Godr. Canadian Journal of Plant Science 66: 361–379.
Grace, J. B. & R. G. Wetzel, 1982. Niche differentiation between two rhizomatous plant species: Typha latifolia and Typha angustifolia. Canadian Journal of Botany 60: 46–57.
Hogg, E. H. & R. W. Wein, 1988. The contribution of Typha components to floating mat buoyancy. Ecology 69(4): 1025–1031.
Hudon, C., 1997. Impact of water level fluctuations on St. Lawrence River aquatic vegetation. Canadian Journal of Fisheries and Aquatic Sciences 54: 2853–2865.
International Lake Ontario Study Board. 2006. Options for managing Lake Ontario and St. Lawrence River water levels and flows. Final Report by the International Lake Ontario – St. Lawrence River Study Board, International Joint Commission.
Jansson, R., C. Nilsson, M. Dynesius & E. Andersson, 2000. Effects of river regulation on river-margin vegetation: a comparison of eight boreal rivers. Ecological Applications 10: 203–224.
Keddy, P. A., 2000. Wetland Ecology Principles and Conservation. Cambridge University Press, Cambridge, UK.
Keddy, P. A. & A. A. Reznicek, 1986. Great Lakes vegetation dynamics: the role of fluctuating water levels and buried seeds. Journal Great Lakes Research 12: 25–36.
Keough, J. R., T. A. Thompson, G. R. Guntenspergen & D. A. Wilcox, 1999. Hydrogeomorphic factors and ecosystem responses in coastal wetlands of the Great Lakes. Wetlands 19: 821–834.
Krusi, B. O. & R. W. Wein, 1988. Experimental studies on the resiliency of floating Typha mats in a freshwater marsh. Journal of Ecology 76: 60–72.
Lacki, M. J., W. T. Peneston, K. B. Adams, F. D. Vogt & J. C. Houppert, 1990. Summer foraging patterns and diet selection of muskrats inhabiting a fen wetland. Canadian Journal of Zoology 68: 1163–1167.
Mills, E. L., S. B. Smith & J. L. Forney, 1981. The St. Lawrence River in winter: population structure, biomass, and pattern of its primary and secondary food web components. Hydrobiolgia 79: 65–75.
Poff, N. L., J. D. Allan, M. B. Bain, J. R. Karr, K. L. Prestegaard, B. D. Richter, R. E. Sparks & J. C. Stromberg, 1997. A paradigm for river conservation and restoration. BioScience 47: 769–783.
Revenga, C., J. Brunner, N. Henninger, K. Kassem & R. Payne, 2000. Pilot Analysis of Global Ecosystems: Freshwater systems. World Resources Institute (WRI), Washington, DC.
Richter, B. D., R. Mathews, D. L. Harrison & R. Wigington, 2003. Ecologically sustainable water management: managing river flows for ecological integrity. Ecological Applications 13: 206–224.
SAS Institute Inc., 1998. SAS/STAT User’s Guide, Release 6.03 Edition. Cary, NC: 1028 pp.
Sodja, R. S., & K. L. Solberg, 1993. Management and control of cattails, No. 13.4.13 in the Waterfowl Management Handbook, Office of Information Transfer, US Fish and Wildlife Service, Ft. Collins, CO: 8 pp.
Takos, M. J., 1947. A semi-quantitative study of muskrat food habits. Journal of Wildlife Management 11: 331–339.
Thurber, J. M., R. O. Peterson & T. D. Drummer, 1991. The effect of regulated lake levels on muskrats, Ondatra zibethicus, in Voyageurs National Park, Minnesota. Canadian Field-Naturalist 105: 34–40.
Toner, J. A., 2006. Muskrat house abundance and cattail use in upper St. Lawrence River tributary wetlands: modeling the effects of water level regulation. Master’s Thesis, SUNY College of Environmental Science and Forestry, Syracuse, New York.
Toner, J.A., J. M. Farrell & J. V. Mead, 2010. Muskrat abundance responses to water-level regulation within freshwater coastal wetlands. Wetlands (in press).
Tremolieres, M., J. M. Sanchez-Perez, A. Schnitzler & D. Schmitt, 1998. Impact of river management history on the community structure, species composition and nutrient status in the Rhine alluvial hardwood forest. Plant Ecology 135: 59–78.
van der Valk, A. G., 1981. Succession in wetlands: a Gleasonian approach. Ecology 62: 688–696.
Warr, S. J., K. Thompson & M. Kent, 1993. Seed banks as a neglected area of biogeographic research: a review of literature and sampling techniques. Progress in Physical Geography 17: 329–347.
Water Survey of Canada, 1990. Historical Streamflow Summary, Ontario. Environment Canada, Water Survey of Canada, Ottawa, Ontario.
Whillans, T. H., 1982. Changes in marsh area along the Canadian shore of Lake Ontario. Journal of Great Lakes Research 8: 570–577.
Whillans, T. H., 1996. Historic and comparative perspectives on rehabilitation of marshes as habitat for fish in the lower Great Lakes Basin. Canadian Journal of Fisheries and Aquatic Sciences 53(Supplement 1): 58–66.
Wilcox, D. A., 1995. Wetland and aquatic macrophytes as indicators of anthropogenic hydrologic disturbance. Natural Areas Journal 15: 240–248.
Wilcox, D. A. & S. J. Nichols, 2008. The effect of water-level fluctuations on plant zonation in a Saginaw Bay, Lake Huron wetland. Wetlands 28: 487–501.
Wilcox, D. A. & Y. Xie, 2007. Predicting wetland plant community responses to proposed water-level-regulation plans for Lake Ontario: GIS-based modeling. Journal of Great Lakes Research 33(4): 751–773.
Wilcox, D. A. & Y. Xie, 2008. Predicted effects of proposed new regulation plans on sedge/grass meadows of Lake Ontario. Journal of Great Lakes Research 34(4): 745–754.
Wilcox, D. A., J. W. Ingram, K. P. Kowalski, J. E. Meeker, M. L. Carlson, Y. Xie, G. P. Grabas, K. L. Holmes & N. J. Patterson, 2005. Evaluation of water level regulation influences on Lake Ontario and Upper St. Lawrence River coastal wetland plant communities: final project report. International Joint Commission, Ottawa, ON and Washington, DC.
Wilcox, D. A., K. P. Kowalski, H. Hoare, M. L. Carlson & H. Morgan, 2008. Cattail invasion of sedge/grass meadows and regulation of Lake Ontario water levels: photointerpretation analysis of sixteen wetlands over five decades. Journal of Great Lakes Research 34(2): 301–323.
Woo, I. & J. B. Zedler, 2002. Can nutrients alone shift a sedge meadow towards the invasive Typha x glauca? Wetlands 22: 509–521.
Zar, J. H., 1999. Biostatistical Analysis, 4th Ed. Prentice-Hall, Englewood Cliffs: 663 pp.
Acknowledgments
This research was supported primarily by the Great Lakes Protection Fund Grant WR537 and a Federal Aid in Sportfish Restoration Grant FA-5-R administered by the New York State Department of Environmental Conservation (NYSDEC) and grants from the International Joint Commission Lake Ontario-St. Lawrence River Water Levels Study. We thank the numerous volunteers and significant others too numerous to list; without their time and patience, this research would not have been possible. This research is a contribution of the Thousand Islands Biological Station.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: M. Power, J. Marty, M. R. Twiss, J. Ridal, Y. de Lafontaine, J. M. Farrell / St. Lawrence River–Great Lakes Ecosystems: An Ecological Overview
Rights and permissions
About this article
Cite this article
Farrell, J.M., Murry, B.A., Leopold, D.J. et al. Water-level regulation and coastal wetland vegetation in the upper St. Lawrence River: inferences from historical aerial imagery, seed banks, and Typha dynamics. Hydrobiologia 647, 127–144 (2010). https://doi.org/10.1007/s10750-009-0035-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-009-0035-z