Abstract
The aim of this work was to analyse those characteristics of the Atlantic coast that are associated with distribution, abundance and breeding activity of Otaria flavescens at three ecological scales. We followed a research strategy that consists in looking at the variables that are associated with the pattern of distribution at regional and landscape scales, instead of following local population in time. We used bibliographic data of censuses carried out between 1946 and 1997 and a Geographic Information System (GIS) to integrate information proceeding from censuses and different environmental variables. At a regional scale, we found that the distribution of breeding colonies did not vary in the period of time analysed and was associated with the pattern of occupation of the coast and the tide width. There was a significant decrease in abundance between 1946 and 1997. In ‘north-central Patagonia’, the segment of coast with the highest number of sea lions in Argentina, distribution of colonies was associated with islands availability and negatively correlated with places were anthropogenic disturbance was high. At a local scale, breeding colonies were positive associated with slight slope coasts and negatively associated to rocky beaches. We identify those characteristics of the coast associated with distribution of breeding colonies of O. flavescens, which operate at different ecological and temporal scales.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the key information to generate actions in favour of species conservation is the identification of environmental factors that regulate abundance of animal populations (Frankham et al., 2002; Halpin et al., 2006). The most traditional method for studying these factors is to follow local population along several years, to estimate demographic parameters, and associate changes in variables such as mortality rate or fecundity with external or internal factors (Clutton-Brock & Coulson, 2003). We followed an alternative approach that implies comparing the distribution of a species at a large scale at two moments separated by a large period of time, and deduce potential factors from the associations between the distribution of animal abundances and the distribution of environmental factors.
The South American sea lion, Otaria flavescens (Shaw 1800), is distributed along approximately 10,000 km of the coast of South America, from Recifes das Torres (29°20′S, 49°43′W) in southern Brazil to Cape Horn in the extreme south of the Atlantic coast, and from Cape Horn to Zorritos (3°40′S, 80°34′W) in northern Peru in the Pacific Ocean (Riedman, 1990). In the Atlantic coast, the northernmost breeding grounds are along the costs of Uruguay (Isla de Lobos, Cabo Polonio and La Coronilla). Northern to the Uruguayan breeding grounds, in southern Brazil, there are only two non-breeding colonies where sub adult males predominate (Sanfelice et al., 1999). In Argentina, O. flavescens is the most abundant pinniped. It concentrates and breeds primarily in continental and island colonies of Patagonian region (Vaz-Ferreira, 1982; Crespo, 1988), from Punta Bermeja (41°08′S, 63°04′W) to Tierra del Fuego Island.
Since 1946, several studies tried to assess the population status of O. flavescens along the Atlantic coast. In Argentina, the first extensive census was carried out by Carrara (1952). The surveys were performed between 1946 and 1949, covering most of the 3500 km on the Patagonian coastline, from 38°30′S to 55°01′S (Carrara, 1952; Schiavini et al., 2004). Carrara found 77 O. flavescens colonies and estimated a population of 137,595 individuals. Approximately 40 years later, several authors (Crespo & Pedraza, 1991; Szapvievich, 1992; Reyes et al., 1996; Schiavini et al., 1999, 2004) made partial censuses of O. flavescens colonies in the Patagonian coast between 1989 and 1997. Altogether, these censuses covered almost the same stretch of coastline surveyed by Carrara (1952), from Banco Culebra (40°22′S, 61°59′W) to San Martín de Tours (55°01′S, 66°20′W). There are no systematic censuses published for the whole coast of Uruguay but population was estimated in 50,000 individuals for 1950 (Vaz-Ferreira, 1950) and 15,000 individuals for 1995, with a decreasing annual rate of 4.5% (Páez, 2005).
Our objective was to analyse the historical, ecological, topographic, and anthropogenic factors that are associated with distribution, abundance and breeding activity of sea lion at three ecological scales: (1) regional scale (along the continental coast of Uruguay and Argentina), (2) landscape scale (in ‘north-central Patagonia’, the segment of coast with the highest number of sea lions in Argentina), and (3) local scale, considering the topographic characteristics of the colonies.
Materials and methods
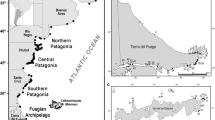
We conducted our study along the Atlantic coast of Uruguay and Argentina, covering approximately 5300 km of the distribution of sea lions, from Isla del Marco, Uruguay (34°20′S, 53°46′W) to San Martín de Tours Island, Tierra del Fuego, Argentina (55°01′S, 66°20′W) (Fig. 1).
Sea lions census data of breeding colonies were obtained from different bibliographic sources and personal communications. Breeding colonies are defined as the colonies in which pups were observed during the breeding season (Carrara, 1952). These data were processed to obtain two distribution data sets: (1) ‘past’ distribution, containing census data from Argentina between 1946 and 1949 and from Uruguay in 1950 and (2) ‘present’ distribution, containing census data from Argentina between 1989 and 1997 and from Uruguayan in 1995. Two dependent variables were defined: (1) ‘number of breeding sea lions’ and (2) ‘number of breeding colonies’. Only censuses in the breeding season were used. If there were more than one data for the same colony, mean values were used.
For study area characterisation and analyses of distribution and abundance, the 5300 km of Atlantic coast were divided into 53 segments of 100 km using a Geographic Information System (GIS) with ARCVIEW® software (license provided by PRODITEL group, Universidad de Luján, Argentina). Two of these segments were slightly longer than 100 km, one at the southern end of the study area (110 km) and the other prior to the interruption of marine coast by the Río de la Plata estuary (126 km). In each segment, the following variables were measured (information source between brackets):
-
(1)
Number of breeding sea lions (Vaz-Ferreira, 1950; Carrara, 1952; Crespo & Pedraza, 1991; Szapkievich, 1992; Reyes et al., 1996; Schiavini et al., 1999, 2004; Enrique Páez personal communication).
-
(2)
Number of breeding colonies (same sources as number of breeding sea lions).
- (3)
- (4)
- (5)
- (6)
-
(7)
Fish landings, mean landed tons of fish year-1 (only species that are part of sea lions diet: Nascimento et al., 1985; Rivero et al., 1999; Koen Alonso et al., 2000; Szteren and Páez, 2002; Szteren et al., 2004; Suárez et al., 2005), (landings: DINARA, 2006; SAGPA, 2006, data collected between 1992 and 2003 for Argentina and between 1994 and 2000 for Uruguay).
-
(8)
Number of artisan fishing vessels (Caille, 1996; DINARA, 2006).
-
(9)
Fish species richness, number of fish species in the segment. We only include species that are part of sea lions diet (Cousseau & Perrotta, 2000).
-
(10)
Harvesting, number of sea lions harvested between 1917 and 1960 for Argentina and between 1963 and 1991 for Uruguay, periods in which most of the commercial harvesting was done (Concessionaire registers; Carrara, 1952; Godoy, 1963; Videla, 1980; Crespo & Pedraza, 1991; DINARA, 2006).
- (11)
- (12)
-
(13)
Number of tourists (Tagliorette & Losano, 1996a; MTD, 2006; STPSC, 2006, data between 1995 and 2000 for Argentina and between 1997 and 2000 for Uruguay).
-
(14)
Sea surface temperature (SST) measured in a 100 km buffer from the coast (PODAAC-JPL-CIT-NASA, 2006).
-
(15)
Tide width in meters (SHN, 2006).
-
(16)
Primary productivity, estimated as mg of chlorophyll a (m3)−1, measured in a 100 km buffer from the coast (SIMBIOS-NASDA-OCTS project-NASA, GES DISC DAAC, 2006).
-
(17)
Mean distance to high (0.9 mg m−3 chlorophyll-a) and middle (0.5 mg m−3 chlorophyll-a) productivity waters, estimated over a digital map as the average distance to high and middle productive waters from each colony.
These data sets were used to build different geo-referenced shape files using GIS. Climatological monthly images of satellite-derived SST and Ocean Color (chlorophyll-a concentration) with a spatial resolution of 9.28 km were used. For SST, an image corresponding to the breeding season of January 1995 (were most of the censuses were carried out) was used. In the case of the Ocean Color, we used an image corresponding to the breeding season of January 1997, due to the unavailability of images for 1995. Comparisons with available images for the 1980’ decade did not shown differences in the general pattern of monthly chlorophyll-a concentration, suggesting that the 1997 image is a good representation of the previous years. The SST climatologies are a standard product known as the Pathfinder + Erosion monthly climatologies (Palacios, 2003). This product was computed with Advanced Very High Radiometer (AVHRR) measurements processed with version 4.0 of the Pathfinder Oceans algorithm. Ocean Color climatologies derived from the Ocean Color and Temperature Scanner (OCTS), which operated between November 1996 and June 1997.
We used five coastal segments identified from the census data (see Results) as sample units for the comparison at a regional level of the distribution and abundance of O. flavescens in relation to time (‘past’ versus ‘present’ distribution). For these analyses, total number of breeding colonies and total number of breeding sea lions in each of the five segments were compared using a Wilcoxon test. For the analysis of distribution of breeding colonies, we performed a contingency analysis.
We performed a principal components analysis (PCA) to analyse natural and anthropogenic variables and associate them with sea lions abundance and breeding colonies distribution in ‘north-central Patagonia’ (2100 km of coast). This segment was the largest with a continuous distribution of sea lions breeding colonies. For the analysis, we used 21 of the total fifty-three 100 km segments (segments 15–35). Variables with a factor score higher than 0.6 were considered to contribute high scores to the component (axis). We conducted simple regression analysis to determine the effect of environmental variables in sea lions abundance and breeding activity distribution. We used the first two factors of the PCA (which explain 42.68% of data variability) as independent variables, and both number of sea lions and number of breeding colonies as dependent variables. We calculated Spearman rank correlations coefficients between all these variables.
To determine which are the local (topographic) characteristics associated with breeding colonies in O. flavescens, we searched for a binomial linear model (Dobson, 1990) with the minimum number of factors to predict the binary dependent (response) variable ‘type of colony’ (breeding or non-breeding colony). This type of analysis allows including continuous and categorical variables. Factors included in the analysis were: ‘slope of the coast’ (slight or steep); ‘beach substratum’ (sand, rock or smooth pebble); ‘presence of cliffs’; ‘height of the cliffs’ and ‘presence of banks’. We used topographic data from 63 breeding and 53 non-breeding colonies obtained from the same sources of census data. Non-breeding colonies are defined as those colonies in which pups were not detected during the breeding season and are primarily composed by adult and juvenile males (Carrara, 1952).
Results
Breeding colonies distribution and abundance at a regional scale
From data collected in censuses between 1989 and 1997, we identified a total of 45 breeding colonies (Fig. 2) in three areas with breeding activity: (1) ‘Uruguay’ (226 km in length) along the coast of Uruguay, with a total of five breeding colonies and 13,680 individuals; (2) ‘north-central Patagonia’ (2100 km in length) along the north-central Patagonian coast, from Punta Bermeja (41°08′S, 63°04′W) to Islote Lobos (47°58′S, 65°54′W), with 30 breeding colonies and 35,740 individuals; and (3) ‘southern Tierra del Fuego’ (410 km in length), along the southern coast of Tierra del Fuego Province and Isla de los Estados, from Caleta Policarpo Oeste (54°38′S, 65°38′W) to Islote Les Eclaireurs (54°52′S, 68°06′W), with 10 breeding colonies and 1240 individuals (Fig. 2).
These three breeding areas, are separated by two segments without or with low breeding activity: (4) ‘Buenos Aires’ (1200 km in length), between ‘Uruguay’ and ‘north-central Patagonia’, without breeding colonies (Fig. 2); and (5) ‘southern Patagonia’ (1400 km in length), southern to 48°S, between ‘north-central Patagonia’ and ‘southern Tierra del Fuego’ breeding areas, with only one breeding colony with low breeding activity (763 individuals, 135 pups born in 1995) at Cerro Bayo (50°15′S, 68°37′W) and 7 pups counted in Monte León (50°19′S, 68°51′W) (Fig. 2).
In summary, the distribution of O. flavescens breeding colonies in the Atlantic coast presents three distinct areas with breeding activity separated by two large segments in which breeding activity is absent. We used these five segments as sample units for the comparison at a regional level of the distribution and abundance of O. flavescens in relation to time (‘past’ versus ‘present’ distribution) and to environmental and anthropogenic variables.
There were non-significant differences between the ‘present’ and the ‘past’ distribution of breeding colonies (Figs. 2–3) (contingency analysis: Chi Square = 6.980, p = 0.14). The total number of breeding colonies did not differed (Wilcoxon test: Z = −1.278, p = 0.2012), but the total number of breeding sea lions was different (Wilcoxon test: Z = −2.023, p = 0.043), between ‘past’ and ‘present’ (Figs. 2–3).
Distribution of natural and anthropogenic factors studied in the five segments was different (Fig. 4). ‘Uruguay’ was characterised by a high anthropogenic disturbance, high productivity and fish richness associated with temperate waters, a high proportion of segments with islands (all segments have more than one island near the coast), low tide width, the highest number of protected areas and a rich history of harvesting with almost 50,000 sea lions harvested in 29 years (Fig. 4). ‘Buenos Aires’ showed a similar pattern than ‘Uruguay’ in anthropogenic disturbance, productivity, fish richness and tide width, but differed in availability of islands and harvesting history (no commercial harvesting was carried out). Fewer protected areas were found in ‘Buenos Aires’ with a lower protected surface (Fig. 4). ‘North-central Patagonia’, ‘southern Patagonia’ and ‘southern Tierra del Fuego’ were characterised by a low anthropogenic disturbance, and a fish richness declining towards the south of the continent accompanying the diminution of sea surface temperature and productivity (Fig. 4). ‘Southern Tierra del Fuego’ had the highest protected surface, the highest concentration of islands and the highest harvesting activity (Fig. 4). Commercial harvesting was absent in ‘southern Patagonia’ and intermediate in ‘north-central Patagonia’ (Fig. 4). Tide width reached its highest amplitude in ‘southern Patagonia’, being intermediate in ‘north-central Patagonia’ and low in ‘southern Tierra del Fuego’ (Fig. 4).
Breeding colonies distribution and abundance in north-central Patagonia
The first factor of the PCA explained 21.77% of the variation of the data, and the second factor 20.91%. Factor 1 had positive value only on ‘SST’ (α = 0.87) and ‘fish richness’ (α = 0.68) and negative values on ‘protected surface’ (α = −0.72) and ‘protected areas’ (α = −0.71) (Fig. 5). Factor 2 had positive values on ‘people’ (α = 0.83), ‘harbours’ (α = 0.77), ‘cities’ (α = 0.73) and ‘fish landings’ (α = 0.64). There were no variables with negative values higher than the criterion (0.6) and the variable with the highest negative value was ‘islands’ (α = −0.33) (Fig. 5).
Factors 1 and 2 (top) of the PCA showing the arrangement of the environmental variables sampled along the ‘north-central Patagonia’ segment. Variables with high contribution (>0.6) to the positive quadrant of Factor 2 and variable with the highest negative value are labelled as follows: A = people; B = harbours; C = cities; D = fish landings and E = islands. Factors 1 and 2 (bottom) showing the arrangement of the twenty-one 100 km segments sampled along ‘north-central Patagonia’. Larger symbols indicate more breeding colonies
Regression analysis retained Factor 2, but not Factor 1 as predictor of distribution of sea lions breeding colonies (Table 1). These two factors failed as predictors of abundance of sea lions (Table 1). Most of the segments with breeding colonies (9 of 10) were located in the negative quadrants of Factor 2 (Fig. 5).
‘Number of breeding sea lions’ and ‘number of breeding colonies’ were positively correlated with ‘islands’ (Spearman, r s = 0.5932 and 0.5103, p < 0.05) and negatively correlated with ‘cities’ (Spearman, r s = −0.4148 and −0.4090, p < 0.05) and ‘people’ (Spearman, r s = −0.4617 and −0.4485, p < 0.05) (Table 2). No other correlations were found (Table 2).
In summary, significant Spearman rank correlation coefficients were obtained for variables with the highest positive and negative values in Factor 2. Consequently, islands, cities and people were considered to be associated with distribution of breeding colonies in Patagonia.
Local determinants
Data used in the binomial linear model were obtained for 63 breeding and 53 non-breeding colonies. The best model that predicted the binary dependent variable ‘type of colony’ included the effects of two of the five variables analysed, ‘slope of the coast’ and ‘beach substratum’ (Likelihood score 9.84, p = 0.043). A slight slope of the coast was associated with breeding colonies (B = 0.50, p = 0.013). Association between ‘type of colony’ and ‘beach substratum’ was not so clear. Breeding colonies were negatively associated with rock beaches (B = −0.65, p = 0.051).
Discussion
Breeding colonies distribution at a regional scale
At a regional scale, O. flavescens showed a patchy distribution of breeding activity that did not vary in the last 50 years. Breeding colonies aggregate in certain areas of the coast that are separated by hundreds and even thousands of kilometres from other clumps of colonies. Breeding activity is absent in two large segments of coast, ‘Buenos Aires’ (1200 km in length) and ‘southern Patagonia’ (1400 km in length). Other authors (Reyes et al., 1999; Dans et al., 2004; Reyes, 2004; Schiavini et al., 2004; Thompson et al., 2005) reported changes in the distribution of colonies at smaller or local scales.
The lack of sea lion breeding colonies in ‘southern Patagonia’ is probably due to the effect of the extreme variation in tide width, with a mean value of 12 m and a maximum of 18 m. This high tide width produces great fluctuations in the coastline location making difficult the sea lions access to water (primarily during the breeding season) and turning these places unsuitable for the settlement of a breeding colony. We have not found historical or palaeontologic records of the presence of large colonies, suggesting that this area has never sustained significant breeding activity.
In contrast, in the coast of Buenos Aires, breeding colonies of sea lions were abundant until the second half of the 19th century, especially around Mar del Plata town (Rodríguez & Bastida, 1998). Disappearance of these colonies in ‘Buenos Aires’ appears to be related with the large scale pattern of human settlement occurred at the end of that century, when the coastal zones were rapidly colonized by man and by the turn of the century, the sea lion colonies finally disappeared (Rodríguez & Bastida, 1998). These sea lion colonies were not subjected to commercial harvesting, and only transient aboriginal groups performed small local catches (Rodríguez & Bastida, 1998). No sea lion colonies were afterwards recorded in the area during the 20th century until the mid 1960s, when small non-breeding groups, composed only by male sea lions, established in the area of Mar del Plata (38°00′S, 57°33′W) and Quequén (38°32′S, 58°42′W) harbours (Rodríguez & Bastida, 1998). Breeding colonies were not re-established in Buenos Aires coast probably due to a combination of factors. First, most of the coast was used during the 20th century as summer holiday centres, i.e., the beaches were used by people in the peak of the breeding season of sea lions (Morello, 1983; Bertoncello, 1992). Second, there was a lack of remaining colonies in the region and the contagious pattern of breeding colony formation determines that new colonies are formed near old ones (Boyd, 1993; Cassini, 2000; Pomeroy et al., 2000). According to our data, there is a significant positive correlation between the number of ‘present’ and ‘past’ colonies in each 100 km segment of the coast where new colonies were formed (Fig. 6, Spearman, r s = 0.5775, p = 0.02). Third, the lack of islands near the colonies prevents the use of alternative areas for reproduction. Availability of islands appears to be an important environmental factor associated with distribution of Californian sea lions, Zalophus californianus (Lesson 1828). Aurioles-Gamboa & Zabala-Gonzalez (1994) found that 86% of the 29 colonies and 98.8% of the population in the Gulf of California, a highly disturbed environment, are placed in islands.
Abundance at a regional scale
Contrary to what occurred with the distribution of breeding colonies, overall numbers of sea lions decreased in the second half of the 20th century, primarily in ‘Uruguay’ and ‘southern Tierra del Fuego’. A similar decrease was found in Malvinas (Falkland) Islands where the 1995 pup counts were less than 2.7% of the counts in 1934–1937 (Thompson et al., 2005). In the last 20 years, a trend towards a systematic population increase was observed in population of northern and central Patagonia (Dans et al., 2004; Reyes, 2004), and a slight increase in Malvinas islands (Thompson et al., 2005). However, these increases are far to imply a recovery of the overall population to the numbers counted in the 1950s. In Uruguay, recent reports indicated a decrease rate of 4.5% (Páez, 2005) while there were no data in present trends for Southern Patagonia (Schiavini et al., 1999). Crespo & Pedraza (1991) proposed the hypothesis that the delay in recovering historical numbers could be caused by a process in which the marine ecosystem reached a new trophic equilibrium, with species like small cetaceans, elephant seals and marine birds occupying trophic and spatial niches left by sea lions in the Atlantic coast. However, evidence supporting or rejecting this hypothesis is still lacking.
Other sea lions populations experience overall reductions in population numbers. A decline of 12.7% between 1985 and 2003 has been reported in pup counts of Australian sea lion (Neophoca cinerea Péron 1816) from Seal Bay, South Australia (McKenzie et al., 2005). There seemed to be no obvious reasons for this decline. The authors suggested that one of the possible factors of the decline is food shortage for the females during lactation. The United States population of Steller’s sea lions declined 68% since 1970 and the causes of this decline remain uncertain (Merrick et al., 1997). However, the consensus among researchers was that the decline is a result of changes in the availability of preferred prey (Merrick et al., 1987; Loughlin & Merrick, 1989; Alverson, 1991; Springer, 1992). Trites & Donnelly (2003) analysed data related with nutritional stress (e.g. reduced body size, reduced productivity, high mortality of pups and juveniles) collected in Alaska for this species, and concluded that these data are consistent with the hypothesis that Steller’s sea lions in the declining regions were nutritionally compromised because of the relative quality of available prey.
Breeding colonies distribution and abundance in north-central Patagonia
Variables related with human disturbance were negatively associated with sea lion breeding colonies distribution. The relation between the absence of breeding colonies and urban areas indicates that human presence has a negative influence on sea lions distribution. In contrast, the low level of human settlements would be a desired characteristic for the establishment of sea lion breeding colonies. Only one variable, availability of islands, was positively associated with breeding colonies (Fig. 5, Table 2). Thus, breeding colonies would be associated with segments of coast with low anthropogenic disturbance unless islands are available.
‘Fish richness’, ‘SST’ and ‘primary productivity’ were not associated with present colonies distribution. Along ‘north-central Patagonia’ the number of fish species in which sea lions feed, SST and primary productivity are almost constant.
Distribution of breeding colonies and abundance were not associated with protected areas and tourism. Protected areas managers can face a dilemma between promoting tourism to earn revenue and promoting effective measures to conserve species, habitants and diversity (Goodwing & Leader-Williams, 2000). The root cause of this problem lies with the socio-economic reasons why many protected areas were established, often as a focus for wildlife-based tourism, rather than as a focus for biodiversity conservation (Leader-Williams et al., 1990). This appears to be the case for sea lions in the Patagonian region. The Patagonian coast shows an increasing tourist activity primarily motivated in the observation of fauna or singular ecosystems (Tagliorette & Losano, 1996b). In opinion polls made in the most important cities of Patagonia, 50–80% of total tourist were classified as eco-tourist and visited at least one protected area (Tagliorette & Losano, 1996a). In the segment of coast with the highest concentration of sea lions and breeding colonies (between 43°30’S and 45°40’S), there are only two small protected areas, Cabo Dos Bahías, with 160 ha of protected surface and Punta Tombo, a coastal area of 210 ha occupied for the greatest Magellan penguin, (Spheniscus magellanicus Forster 1781) colony in Patagonia. Moreover, most of the coastal protected areas in ‘north-central Patagonia’ are terrestrial areas over the coast, with a low surface of marine environments protected (Yorio, 2001). Only two marine protected areas are found in this zone, Isla Escondida, a fishing restricted area founded in 1983, which is spatially and temporally modified along the years, and Bajo Mazarredo, another fishing restricted area that protects shrimp breeding grounds. (Diegues et al., 1995). These areas represent management tools for fishing activities and were not considered in our analysis, but they could promote indirect effects on other components of the ecosystem.
Local determinants
Binomial linear model results indicated that breeding colonies were associated with slight slope coasts. This is in accordance with the prevailing idea, never tested until now, based in observations in the Uruguayan colonies (Vaz-Ferreira, 1965). However, Vaz-Ferreira (1965) postulated that sea lions avoid rocky beaches and prefer sand or small rock beaches. Model results did not show positive associations with a particular substratum but breeding colonies were negatively associated with rock beaches. We do not found in the literature other works that tried to address habitat preferences of O. flavescens. However, Stevens & Boness (2003) address this topic for A. australis in three colonies in Peru. They found that currently used sites for breeding were more likely to have stacked rocks, tide pools and abundant shade, and associated this findings with thermoregulation. Tide pools would be a valuable resource for sea lions thermoregulation but taking into account that male sea lions are almost three times bigger than fur seals; beaches composed by stacked rocks would be less accessible and an uncomfortable habitat for sea lions.
Conclusion and conservation considerations
We investigate the variables that are associated with distribution of O. flavescens breeding colonies at regional and landscape scales, instead of following local population in time. This method allows us identifying some characteristics of the coast that could be associated with distribution of breeding colonies, which operate at different ecological and temporal scales. Preliminary conclusions must be taken as working hypotheses for future studies on potential factors of population decline, rather than as management proposals. More research on population abundance and distribution is required in Southern Patagonia.
References
Alverson, D. L., 1991. Commercial Fisheries and the Steller Sea Lion (Eumetopias jubatus): the Conflict Arena. Fisheries Research Institute, University of Washington, Seattle.
ANP, Administración Nacional de Puertos, 2006. http://www.anp.com.uy/montevideo/default.asp.
Aurioles-Gamboa, D. & A. Zabala-Gonzalez, 1994. Ecological factors that determine distribution and abundance of California sea lion Zalophus californianus in the Gulf of California. Ciencias Marinas 20: 353–553.
Bertoncello, R., 1992. Configuración socio-espacial de los balnearios del Partido de la Costa (Provincia de Buenos Aires). Editorial Abril, Buenos Aires.
Boyd, I. L., 1993. Pup production and distribution of breeding Antarctic fur seals (Arctocephalus gazella) at South Georgia. Antarctic Science 5: 17–24.
Caille, G. M., 1996. La pesca artesanal en las costas de Patagonia: hacia una visión global. Informe Técnico N° 7 del Plan de Manejo Integrado de la Zona Costera Patagónica. Fundación Patagonia Natural, Puerto Madryn.
Carrara, I. S., 1952. Lobos marinos, pingüinos y guaneras de las costas del litoral maritimo e islas adyascentes de la República Agentina. Facultad de Ciencias Veterinarias, Universidad Nacional de La Plata, La Plata.
Cassini, M. H., 2000. A model on female breeding dispersion and the reproductive systems of pinnipeds. Behavioural Processes 51: 93–99.
ClearingHouse, 2006. http://www.cndg.clearinghouse.com.uy/datos/gratuitos.aspx.
Clutton-Brock, T. H. & T. Coulson, 2003. Comparative ungulate dynamics: the devil is in the detail. In Sibly, R. M., J. Hone & T. H. Clutton-Brock (eds), Wildlife Population Growth Rates. The Royal Society and Cambridge University Press, Cambridge: 249–268.
Cousseau, M. B. & R. G. Perrotta, 2000. Peces marinos de Argentina. Biología, Distribución, Pesca. INIDEP, Mar del Plata.
Crespo, E. A., 1988. Dinámica poblacional del lobo marino del sur Otaria flavescens (Shaw 1800), en el norte del litoral patagónico. PhD Thesis, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Buenos Aires, Argentina.
Crespo, E. A. & S. N. Pedraza, 1991. Estado actual y tendencia de la población de lobos marinos de un pelo (Otaria flavescens) en el litoral norpatagónico. Ecología Austral 1: 87–95.
Dans, S. L., E. A. Crespo, S. N. Pedraza & M. Koen-Alonso, 2004. Recovery of the South American sea lion (Otaria flavescens) population in northern Patagonia. Canadian Journal of Fisheries and Aquatic Science 61: 1681–1690.
Diegues, A., G. Harris & A. Castro Moreira, 1995. Marine region 9: South Atlantic. In Kelleher, G., C. Bleakley & S. Wells (eds), A Global Representative System of Marine Protected Areas, Vol. II. The Great Barrier Reef Marine Park Authority, The World Bank and The World Conservation Union (IUCN), Washington: 71–85.
DINAMA, Dirección Nacional de Medio Ambiente, 2006. http://www.dinama.gub.uy/modules.php?op=modloadandname=dinama/areas_naturalesandfile=areasprotegidas.
DINARA, Dirección Nacional de Recursos Acuáticos, 2006. http://www.dinara.gub.uy/Mamíferos-Marinos/Explotacion.htm.
Dobson, A. J., 1990. An Introduction to Generalized Linear Models. Chapman and Hall, New York.
Frankham, R., J. D. Ballou & D. A. Briscoe, 2002. Introduction to Conservation Genetics. Cambridge University Press, Cambridge.
Godoy, J. C., 1963. Caza de pinnípedos. Serie Evaluación de los Recursos Naturales de la República Argentina. Consejo Federal de Inversiones, Buenos Aires.
Goodwing, H. J. & N. Leader-Williams, 2000. Tourism and protected areas–distorting conservation priorities towards charismatic megafauna? In Entwistle, A. & N. Dunstone (eds), Priorities for the Conservation of Mammalian Diversity–Has the Panda had its day? Cambridge University Press, Cambridge: 257–275.
Halpin, P. N., A. J. Read, B. D. Best, K. D. Hyrenbach, E. Fujioka, M. S. Coyne, L. B. Crowder, S. A. Freeman & C. Spoerri, 2006. OBIS-SEAMAP: developing a biogeographic research data commons for the ecological studies of marine mammals, seabirds, and sea turtles. Marine Ecology Progress Series 316: 329–246.
IGM, Instituto Geográfico Militar, 2006. http://www.sig.igm.gov.ar/.
INAPE, Instituto Nacional de Pesca, 2000. Sinopsis de la biología y ecología de las poblaciones de lobos finos y leones marinos de Uruguay. Pautas para su manejo y administración. MGAP, Montevideo.
INDEC, Instituto Nacional de Estadísticas y Censos, 2006. http://www.indec.mecon.ar/.
INE, Instituto Nacional de Estadística, 2006. http://www.ine.gub.uy./fase1new/TotalPais/divulgacion_TotalPais.asp.
Koen-Alonso, M., E. A. Crespo, S. N. Pedraza, N. A. García & M. A. Coscarella, 2000. Food habits of the South American sea lion, Otaria flavescens, off Patagonia, Argentina. Fisheries Bulletin 98: 250–263.
Leader-Williams, N., J. Harrison & M. J. B. Green, 1990. Designing protected areas to conserve natural resources. Science Progress 74: 189–204.
Loughlin, T. R. & R. L. Merrick, 1989. Comparison of commercial harvests of walleye pollock and northern sea lion abundance in the Bering Sea and Gulf of Alaska. In Proceedings of the International Symposium of Biological Management. Walleye Pollock. Alaska Sea Grant Rep. 89-1. University of Alaska, Fairbanks, 679–700.
McKenzie, J., S. D. Goldsworthy, P. D. Shaughnessy & R. McIntosh, 2005. Understanding the Impediments to the Growth of Australian Sea Lions Populations. South Australian Research and Development Institute (Aquatic Science), Adelaide: 107. SARDI publication Number RD04/0171.
Merrick, R. L., T. R. Loughlin & D. G. Calkins, 1987. Decline in abundance of the northern sea lion, Eumetopias jubatus, in Alaska, 1956.86. Fisheries Bulletin 85: 351–365.
Merrick, R. L., M. K. Chumbley & G. V. Byrd, 1997. Diet diversity of Steller sea lions (Eumetopias jubatus) and their population decline in Alaska: a potential relationship. Canadian Journal of Fisheries and Aquatic Science 54: 1342–1348.
Morello, J., 1983. Ecología, preferencias turísticas y ambientes extraurbanos. Medio ambiente y turismo. CLASCO: Buenos Aires.
MTD, Ministerio de Turismo y Deporte, 2006. http://www.mintur.gub.uy/contactos.htm.
Nascimento, M. G., R. Bustamante & C. Oyarzun, 1985. Feeding ecology of the South American sea lion Otaria flavescens: food contents and food selectivity. Marine Ecology Progress Series 21: 135–143.
Páez, E., 2005. ¿Yo…? Otaria. ¿Y usted….? III Jornadas de Conservación y Uso Sustentable de la Fauna Marina. Montevideo, September 14–17 2005, 28.
Palacios, D. M., 2003. Oceanographic conditions around the Galápagos Archipelago and their influence on cetacean community structure. PhD Thesis, Oregon State University, Oregon, USA.
PODAAC-JPL-CIT-NASA, Physical Oceanography Distributed Active Archive Center–Jet Propulsion Laboratory–California Institute of Technology and National Aeronautics and Space Administration, 2006. http://www.podaac.jpl.nasa.gov/cgibin/dcatalog/fam_summary.pl?sst+pfsst.
Pomeroy, P. P., S. D. Twiss & C. D. Duck, 2000. Expansion of a grey seal (Halichoerus grypus) breeding colony: changes in pupping site use at the Isle of May, Scotland. Journal of Zoology, London 250: 1–12.
Reyes, L. M., 2004. Estado actual y tendencia de la población de lobos marinos de un pelo del centro de Patagonia. XIX Jornadas Argentinas de Mastozoología. SAREM. Puerto Madryn, November 8–11 2004, 92.
Reyes, L. M., E. A. Crespo & V. Szapkievich, 1996. Distribución y abundancia de lobos marinos de un pelo (Otaria flavescens) en el centro y sur de Chubut. Informe Técnico N° 10 del Plan de Manejo Integrado de la Zona Costera Patagónica. Fundación Patagonia Natural, Puerto Madryn.
Reyes, L. M., E. A. Crespo & V. Szapkievich, 1999. Distribution and population size of the southern sea lion (Otaria flavescens) in central and southern Chubut, Argentina. Marine Mammal Science 15: 478–493.
Riedman, M. L., 1990. The Pinnipeds. Seals, Sea Lions and Walruses. University of California Press, Los Angeles and Oxford University Press, Oxford.
Rivero, L., R. Bastida, D. Rodriguez & G. Westergaard, 1999. Hábitos tróficos de los lobos marinos de un pelo (Otaria flavescens) en el apostadero de Puerto Quequén–Argentina. Octavo Congreso Latinoamericano de Ciencias del Mar, Trujillo.
Rodríguez, D. & R. Bastida, 1998. Four hundred years in the history of pinniped colonies around Mar del Plata, Argentina. Aquatic Conservation: Marine and Freshwater Ecosystems 8: 721–735.
SAGPA, Secretaria de Agricultura, Ganadería, Pesca y Alimentos, 2006. http://www.sagpya.mecon.gov.ar/.
Sanfelice, D., V. Castro-Vasques & E. A. Crespo, 1999. Ocupação sazonal por duas espécies de Otariidae (Mammalia, Carnívora) da Reserva Ecológica Ilha dos Lobos, Rio Grande do Sul, Brasil. Iheringia (Zoology) 87: 101–110.
SAyDS, Secretaria de Ambiente y Desarrollo Sustentable, 2006. http://www2.medioambiente.gov.ar/bases/areas_protegidas/default.asp.
Schiavini, A., E. A. Crespo & V. Szapkievich, 1999. Estado de la población del lobo marino de un pelo (Otaria flavescens) en las provincias de Santa Cruz y Tierra del Fuego. Informe Técnico N° 40 del Plan de Manejo Integrado de la Zona Costera Patagónica. Fundación Patagonia Natural, Puerto Madryn.
Schiavini, A., E. A. Crespo & V. Szapkievich, 2004. Status of the population of South American sea lion (Otaria flavescens Shaw, 1800) in southern Argentina. Mammalian Biology 69: 108–118.
SHN, Servicio de Hidrografía naval, 2006. http://www.hidro.gov.ar/oceanografia/Tmareas/Form_Tmareas.asp.
SIMBIOS-NASDA-OCTS project-NASA, GES DISC DAAC, Goddard Space Flight Center (GSFC)-GSFC Earth Sciences Data and Information Services Center Distributed Active Archive Center, 2006. http://www.daac.gsfc.nasa.gov/data/datapool/OCTS/.
Springer, A. M., 1992. A review: Walleye pollock in the North Pacific. How much difference do they really make? Fisheries Oceanography 1: 80–96.
Stevens, M. A. & D. J. Boness, 2003. Influences of habitat features and human disturbance on use of breeding sites by a declining population of Southern fur seals (Arctocephalus australis). Journal of Zoology, London 260: 145–152.
STPSC, Subsecretaría de Turismo Provincia de Santa Cruz, 2006. http://www.scruz.gov.ar/turismo/.
Suárez, A. A., D. Sanfelice, M. H. Cassini & H. L. Cappozzo, 2005. Composition and seasonal variation in the diet of the South American sea lion (Otaria flavescens) from Quequén, Argentina. Latin American Journal of Aquatic Mammals 4: 163–174.
Szapvievich, V. B, 1992. Estimación de la población del lobo marino de un pelo Otaria flavescens (Shaw 1800) en el litoral patagónico mediante fotografías obtenidas en relevamientos aéreos. Magisterial Thesis, Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires, Buenos Aires, Argentina.
Szteren, D. & E. Páez, 2002. Predation by southern sea lions (Otaria flavescens) on artisanal fishing catches in Uruguay. Marine and Freshwater Research 53: 1161–1167.
Szteren, D., D. E. Naya & M. Arim, 2004. Overlap between pinniped summer diet and artisanal fishery catches in Uruguay. Latin American Journal of Aquatic Mammals 3: 119–125.
Tagliorette, A. & P. Losano, 1996a. Estudio de la demanda turística en las ciudades de la costa patagónica. Informe Técnico N° 24 del Plan de Manejo Integrado de la Zona Costera Patagónica. Fundación Patagonia Natural, Puerto Madryn.
Tagliorette, A. & P. Losano, 1996b. Demanda turística en áreas costeras protegidas de la Patagonia. Informe Técnico N° 25 del Plan de Manejo Integrado de la Zona Costera Patagónica. Fundación Patagonia Natural, Puerto Madryn.
Thompson, D., I. Strange, M. Riddy & C. D. Duck, 2005. The size and status of the population of Southern sea lions Otaria flavescens in the Falkland Islands. Biological Conservation 121: 357–367.
Trites, A. W. & C. P. Donnelly, 2003. The decline of Steller sea lions Eumetopias jubatus in Alaska: a review of the nutritional stress hypothesis. Mammal Review 33: 3–28.
Vaz-Ferreira, R., 1950. Observaciones sobre la Isla de Lobos. Revista de la Facultad de Humanidades y Ciencias 5: 145–176.
Vaz-Ferreira, R., 1965. Comportamiento antisocial en machos subadultos de Otaria byronia (de Blainville), (lobos marino de un pelo). Revista de la Facultad de Humanidades y Ciencias 22: 204–207.
Vaz-Ferreira, R., 1982. Otaria flavescens (Shaw), South American sea lion. In FAO, UNEP (eds), Mammals in the Seas. Small Cetaceans, Seals, Sirenians and Otters. FAO Fisheries Series, Rome: 477–495.
Videla P. H. B., 1980. Recursos Naturales de la Tierra del Fuego: Lobos Marinos. Dirección de Intereses Marinos, Ushuaia.
Yorio, P., 2001. Áreas marinas protegidas en la Argentina. Ciencia Hoy 64: 32–38.
Acknowledgements
To Leonardo Di Franco, for his help with satellite images analysis and use of Geographic Information Systems. We specially thank Enrique Páez who kindly gave us Uruguayan census data. Original data of harvesting in Argentina were kindly provided by José Machinea, Claudio Campagna and Alberto Montero. This study received financial support from Fundación Antorchas (HLC grant, Project A-13 672/1–3), Consejo Nacional de Investigaciones Científicas y Técnicas (PIP-02193 and PIP-05489). J. I. Túnez was supported by a PhD grant from Consejo Nacional de Investigaciones Científicas y Tecnológicas.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: K. Martens
Rights and permissions
About this article
Cite this article
Túnez, J.I., Cappozzo, H.L. & Cassini, M.H. Natural and anthropogenic factors associated with the distribution of South American sea lion along the Atlantic coast. Hydrobiologia 598, 191–202 (2008). https://doi.org/10.1007/s10750-007-9150-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-007-9150-x