Abstract
Plant transporters are crucial for nutrient acquisition, cellular homeostasis, and stress responses. They mediate the exchange of chemicals and signals inside and outside the biomembrane in plants. Therefore, the study of plant transporters is critical to understanding the mechanisms of plant stress tolerance, moreover, transporters have potential applications in crop breeding for improved stress tolerance. Here we review recent findings about different transporters family (ABC, MATE, NRAMP, KUP, COPT/Ctr, NPF, NRT, PHT, YSL, ZIP, STP, etc.), including their roles in abiotic stress tolerance, and plant growth and yield. Moreover, we highlight the roles of transporters in plant responses to abiotic stresses such as drought, waterlogging, salt and alkali, heavy metal stress, and nutrient deficiency. To clarify the mechanisms underlying the roles of transporters in various stress responses, we integrate information about transporter activity in plants under stress and thus reveal a link between nutrient accumulation and stress tolerance. Finally, we list genes with potential applications in genetic improvement of resistrance genes, and summarize and discuss these transporters for benefiting the research community.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Abiotic stresses such as drought and waterlogging, soil salinization and alkalization, heavy metal toxicity, and nutrient deficiency restrict crop yields and biomass production worldwide (Vahdati and Leslie 2013). Molecular biological studies have provided extensive evidence about the mechanisms of stress tolerance in plants, including the various signaling and transcription factors involved. Intriguingly. Transporters have vital roles in plant stress responses (Hirayama and Shinozaki 2010). Transporters are a class of membrane proteins that transport biomolecules across membranes. Numerous transporters help plants move metabolites, toxic pollutants, and nutrients during growth, and these transporters, especially nutritional transporters that transfer nutrients needed for plants growth are crucial for nutrient acquisition, development, cellular homeostasis, and stress responses (Bo et al. 2017). For example, carbohydrate transporters improve tolerance to drought (Ma et al. 2019a) and waterlogging stress (Phukan et al. 2018) by adjusting carbohydrates content. Mineral nutrition transporters respond to nutrient deficiencies by regulating the Macro-elements and Micro-elements in the cytoplasm, vacuoles and extracellular space (Li et al. 2019). Metal cation transporters can adjust the ion balance in the cells to achieve the effect of anti-salt (Chai et al. 2020) and anti-metal toxicity (Veraestrella et al. 2017). Recently, there have been many research results showing that transporters are closely related to plant stress resistance, growth and development. In this review, we summarize our current understanding of the roles of different transporters in plant stress responses and examine the link between transporter activities, nutrient acquisition, and stress tolerance.
Transporters in abiotic stress tolerance
Drought tolerance
Drought stress is one of the most important factors restricting plant growth. Plants respond to drought stress by closing their stomata, causing downstream metabolic perturbations (i.e. increased activity in the photorespiration pathway) that increase reactive oxygen production (Fang and Xiong 2014). In addition, plant hormones such as abscisic acid (ABA) activate various stress responses and the plant initiates root development (Fang and Xiong 2014).
Functional studies have verified the roles of many transporters in drought stress. For example, some sugar transporters allow cells to accumulate sugar and this change in osmotic pressure increases drought tolerance. For instance, in micropropagated apple plants (Malus × domestica), increased sorbitol transporter gene (MdSOT3 and MdSOT5) activity improved drought tolerance (Li et al. 2012). Also in apple, the sucrose transporter MdSUT2.2 is phosphorylated by the protein kinase MdCIPK22, which regulates its transcription activity under drought conditions, allowing cells to accumulate sugar and thus increasing drought tolerance (Ma et al. 2019a). Movement of sodium and potassium ions also helps alleviate drought stress by affecting the osmotic balance. For example, overexpressing the rice (Oryza sativa) high-affinity potassium transporter gene OsHAK1 enhanced drought tolerance and improved grain yield under drought conditions (Chen et al. 2017a).
The stress hormone abscisic acid (ABA) plays a key role in drought tolerance. When plants sense drought conditions, ABA functions as a long-distance signal to transmit information from the roots to the above-ground tissues of the plants. The ABA signal increases drought tolerance by inducing stomata to close, maintaining water balance in cells, and increasing root permeability to water (Zhang et al. 2006). Loss-of-function Arabidopsis thaliana mutants of a plasma membrane ABA uptake transporter, ABCG40, showed reduced sensitivity to ABA and reduced resistance to drought stress (Kang et al. 2010). Overexpressing the cell membrane ABA transporter gene AtABCG25 enhanced plant drought tolerance (Kuromori et al. 2016).
Controlling the movement of guard cells enhances drought tolerance in plants by decreasing water loss and many different transporters take part in this process, including auxin, nitrate, and ABA transporters (Jarzyniak and Michal 2014). In Arabidopsis thaliana, the Major Facilitator Superfamily transporter ZINC-INDUCED FACILITATOR-LIKE 1 (ZIFL1) affects polar auxin transport, but ZIFL1.3, a truncated form produced by alternative splicing, regulates stomatal closure to mediate drought tolerance (Remy et al. 2013). The nitrate transporter AtNRT1.1 (CHL1) functions in stomatal opening in Arabidopsis under drought stress (Guo et al. 2003). The Arabidopsis ABC transporter MULTIDRUG RESISTANCE-ASSOCIATED PROTEIN5 (AtMRP5) is an important component of guard cell function and water use (Klein et al. 2003). Mutation of AtMRP4, an ABC transporter, decreased stomatal opening and increased drought resistance in Arabidopsis (Klein et al. 2004). Arabidopsis mutants of AtDTX50, a DTX/MATE (Multidrug and Toxic Compound Extrusion) family member and ABA efflux transporter, showed lower stomatal conductance and were more tolerant to drought stress than the wild type (Zhang et al. 2014). A mutant of AtABCG22, an Arabidopsis ABC transporter, had increased water loss and lower leaf temperature than the wild type due to effects on stomatal regulation (Kuromori et al. 2011). The ABC transporter AtABCB14 regulates stomatal movement by affecting malate transport and increasing osmotic pressure in Arabidopsis (Lee et al. 2008). Lastly, REDUCED CULM NUMBER 1/OsABCG5, a half-size ABC subfamily G protein, is indispensable for stomatal closure and reducing water loss in rice (Oryza sativa) (Matsuda et al. 2016).
Many stress-related transporters have been isolated by mutant analysis. Now, various “omics” techniques have been effectively used to search for anti-stress factors in plants and have uncovered the roles of various transporters in stress tolerance. For example, a whole-genome oligomer microarray analysis identified several transporter genes as significantly responsive to drought stress in rice (Oryza sativa) (Junli et al. 2007). A recent study used genome-wide sequencing data to find new transporter genes related to drought tolerance in Populus (Bai et al. 2019). Solexa sequencing and qRT-PCR identified various transporter genes that were highly expressed in drought-treated Paulownia fortunei leaves (Dong et al. 2014). The regulation of different transporter genes also plays a role in the drought tolerance of Gossypium herbaceum, as revealed using microarray analysis, pyrosequencing, and qRT-PCR of roots (Ranjan et al. 2012). A putative ATP-binding cassette (ABC) transporter ATP-binding protein was identified by proteomic analysis of proteins extracted from Hippophae rhamnoides leaves (Xu et al. 2009). Nine rice ABC transporter genes that are significantly regulated by drought stress were discovered by analysis of anatomical data analysis and qRT-PCR (Nguyen et al. 2014). With the development of sequencing technologies, omics approaches will find more and more new transporters and improve our understanding of the mechanisms by which transporters respond to drought stress.
Waterlogging tolerance
Waterlogging induces hypoxia in plants. Anaerobic respiration caused by waterlogging leads to excessive reduction of the photosynthetic electron transport chain and the formation of reactive oxygen species (ROS) such as superoxide radicals and singlet oxygen (Blokhina et al. 2003). Initially, ROS and NO production can help adaptive responses such as formation of aerenchyma and adventitious roots, but excessive ROS cause severe oxidative damage under waterlogging stress (Planchet and Kaiser 2006; Steffens et al. 2012). Transporters also take part in the waterlogging stress response in plants. AtSWEET10, a SWEET transporter, is involved in enhancing carbohydrate availability and waterlogging tolerance in Arabidopsis. AtSWEET10 expression is activated by waterlogging due to the presence of a drought-responsive element (DRE) and a GCC box in the AtSWEET10 promoter. Heterologous expression assays showed that the AtSWEET10 DRE can be targeted by MaRAP2-4, a waterlogging-responsive ERF from Mentha arvenis (Phukan et al. 2018). ABC transporter subfamily G (ABCG) gene ABCG5 is highly expressed in seedling cotyledons, and under waterlogging condition, its activity is required to form the dense cuticle to resist waterlogging damage for the normal growth of plants (Lee et al. 2020).
Salt and alkali tolerance
Soil salinization and alkalization are major environmental hazards that limit plant growth and development. Exposure to salt affects plant water relations, inducing responses similar to those of drought stress. Moreover, high salt creates ionic stress, in which excess ions (Na+) disrupt cellular metabolism. Alkali stress affects many metabolic and physiological processes, such as photosynthesis, cell ionic balance, antioxidant systems, and osmotic absorption of water (Zhang and Mu 2010). In recent years, many genes related to salt stress have been discovered, and the molecular mechanism of salt stress has been gradually elucidated (Yang and Guo 2018). In addition to the harm caused by water stress, soil salinization and alkalization also directly affect plant growth and development.
Maintaining the proper balance of sodium and potassium is an effective strategy used by plants to survive salt stress (Ali et al. 2018). Indeed, transcriptomics approaches have identified various monovalent cation transporters that function in plant responses to salinity stress. The barley (Hordeum vulgare) sodium transporter and high-affinity potassium transporter HvHKT1;1 reduces Na+ accumulation in shoots and regulates ion concentrations in root cells, thereby improving salt tolerance (Yong et al. 2018). The sodium transporter HKT1;2 plays important roles in maintaining sodium/potassium homeostasis and salt tolerance in tomato (Solanum lycopersicum) (Noelia et al. 2017). The High-Affinity Potassium Transporter EpHKT1;2 in the halophyte Eutrema parvula is a key contributor to the halophyte features of this plant and helps determine its tolerance to Na+ ion toxicity (Ali et al. 2018).
The SOS (salt overly sensitive) pathway is induced by high salt upregulates transporters such as the high-affinity potassium transporter AtHKT1;1 in Arabidopsis to regulate ion homeostasis and thus enhance plant salt tolerance (Oh et al. 2009). The Na+/H+ transporter SOS1 and the protein kinase SOS2 act with the plasma membrane transporter PUT3 and affect its regulatory activity, thereby maintaining the balance of Na+ and K+ in the cell and affecting salt resistance in Arabidopsis (Chai et al. 2020).
Other transporters, including magnesium (Mg), ammonium and sucrose transporters, also affect salt tolerance. For example, the Mg transporter OsMGT1 transports Mg2+ in the root maturation zone and enhances OsHKT1;5 activity to restrict Na+ accumulation and improve salt tolerance in rice (Zhi et al. 2017). Heterologous overexpression of the Puccinellia tenuiflora ammonium transporter PutAMT1;1 in Arabidopsis thaliana can alleviate the ammonia toxicity induced by salt stress, thus improving the root salt tolerance in the later stage of seed germination (Bu et al. 2019). Overexpressing the sucrose transporter gene MdSUT2.2 enhanced salt tolerance in a MdCIPK13 phosphorylation-dependent manner in transgenic apple (Ma et al. 2019b). OsLCT1, a low-affinity cation transporter, affects salinity tolerance in rice (Nio et al. 2018).
Transporters in multiple plant abiotic stress responses
Stresses often occur together (for example, drought and heat) and transporters take part in multiple plant stress responses. The barley silicon transporters HvLsi1 and HvLsi2 regulate Si accumulation of the shoots of barley plants under K deficiency, thereby improving osmotic stress tolerance (Seyed et al. 2017). Overexpressing AtABCG36/AtPDR8, encoding an ABC transporter, increased drought and salt-stress resistance in Arabidopsis (Kim et al. 2010). In the monocot sorghum (Sorghum bicolor), the auxin influx carriers Auxin resistant 1/like aux1 (AUX⁄ LAX) and the auxin efflux carriers Pin-formed (PIN) and P-glycoprotein (PGP) are auxin transporters that function in plant responses to ABA, salt, and drought stress (Shen et al. 2010). ProT2 is a proline transporter involved in water and salt stress responses in Arabidopsis (Rentsch et al. 1996). In cucumber (Cucumis sativus), CsTST1 functions in responses to various abiotic stresses (cold, salt, and osmotic stress), and in response to the stress hormone ABA by affecting the distribution of carbohydrates (Huang et al. 2020).
Sucrose plays a key role in regulating guard cell movement, which affects gas exchange and water loss (Antunes et al. 2017). Heterologous overexpression of a rice monosaccharide transporter gene OsMST6 may increase soluble sugar content and osmotic pressure in cells, and enhance the drought and salt tolerance in Arabidopsis (Monfared et al. 2020). Heterologous overexpression of the Dianthus spiculifolius sugar transporter genes DsSWEET17 and DsSWEET12 in Arabidopsis increased root length and fresh weight and enhanced salt, osmotic, and oxidative stress tolerance (Zhou et al. 2018a; Zhou et al. 2018b). OsSUT2, a sucrose transporter identified in rice (Oryza sativa L. cv. Nipponbare), is essential for plant tolerance to drought and salinity stress (Ibraheem et al. 2011). Arabidopsis TONOPLAST MONOSACCHARIDE TRANSPORTER1 is required for cytosolic glucose homeostasis and plays an important role in stress responses (Wormit et al. 2006). Monosaccharide-facilitated diffusion transporters affect sugar accumulation in plant cells and help regulate osmotic pressure (Yamada et al. 2010). The sucrose transporters AtSUC2 and AtSUC4 play important roles in abiotic stress tolerance in Arabidopsis via an ABA-dependent pathway (Gong et al. 2015). Many sulfate transporters take part in drought and salinity tolerance in Arabidopsis and Medicago truncatula (Gallardo et al. 2014). Using these transporters to enhance plant resistance to salt and alkali stress will be useful in plant genetics and breeding for crop improvement.
Heavy metal stress
Heavy metal toxicity has become an increasingly serious problem due to human activity, which has increased soil heavy metals such as aluminum, cadmium, manganese, and cesium (Mohammed et al. 2011). Heavy metal hyperaccumulation and tolerance are important protective mechanisms used by plants to sequester toxic metal ions and thus improve plant survival. In tobacco (Nicotiana tabacum), metal ions are isolated into vacuoles by using transporters and exchanging with H+, and thus have roles in heavy metal hyperaccumulation and tolerance (Veraestrella et al. 2017). Therefore, transporters play important roles in plant responses to heavy metal stress.
Aluminum detoxification
Aluminum (Al) toxicity inhibits root elongation and plant productivity in acidic soils. The release of organic acids from roots is an effective method used by plants to alleviate Al toxicity, a process primarily regulated by transporters (Park et al. 2017). STAR1 and STAR2 form a bacterial-type ABC transporter complex with UDP-glucose transport activity that is required for the detoxification of Al in rice (Huang et al. 2009). The expression of the ABC transporter-like gene ALUMINUM SENSITIVE3 (ALS3) increases under high Al concentrations (above the toxicity threshold) in buckwheat (Fagopyrum esculentum), and ALS3 might promote Al detoxification (Reynallorens et al. 2015).
MATE transporters take up iron by secreting primary metabolites such as citric acid into the xylem and rhizosphere. Similarly, some MATE transporters are highly expressed in root tips and secrete citric acid to improve aluminum tolerance. The rice bean (Vigna umbellata) citrate transporters VuMATE2 and VuMATE1 function in tandem to induce citrate secretion, thereby enhancing Al resistance (Liu et al. 2018b). The organic acids combine with metal ions to form stable, non-toxic complexes responsible for transport, redistribution, or reduction of metal toxicity (Upadhyay et al. 2019; Magalhaes et al. 2007). An insertion in the rice citrate efflux transporter gene OsFRDL4 increased its promoter activity and improved its expression, thereby enhancing Al tolerance (Yokosho et al. 2016). Heterologous overexpression of a Brachypodium distachyon MATE gene enhanced Al tolerance in Setaria viridis (Ribeiro et al. 2017). Genome-wide analysis of MATE family genes in poplar (Populus) and soybean (Glycine max) confirmed their function in enhancing Al tolerance (Li et al. 2017b; Liu et al. 2016a); their homologs play similar roles in bread wheat (Triticum aestivum L.) (Garciaoliveira et al. 2018).
Like citrate, malate can combine with Al to reduce its toxicity by facilitating its transport. Heterologous expression of the Brassica oleracea ALMT gene BoALMT1 increased malate secretion and H+ efflux, thereby enhancing Al tolerance in Xenopus oocytes and Arabidopsis (Zhang et al. 2018b). Al retained in the root cell walls of plants is toxic, but this can be relieved by NIP1;2-induced accelerated Al-malate transport (Wang et al. 2018a). Overexpressing the plasma membrane-located organic acid transporter gene CsALMT1 increased malate secretion and improved Al tolerance in Camelina sativa L. (Park et al. 2017). Overexpressing Al-ACTIVATED MALATE TRANSPORTER9 (Sl-ALMT9), which is regulated by Sl-WRKY42, promoted malate accumulation in tomato (Solanum lycopersicum) fruit and enhanced Al tolerance (Ye et al. 2017).
Other transporters, including those that transport Al3+ or other ions, also affect Al tolerance. The rice NRAMP (natural resistance and macrophage protein) Al transporter (NRAT1) enhanced Al resistance in transgenic Arabidopsis by transporting Al from the root cell wall to the root cell for sequestration in the vacuole (Li et al. 2014). The absence of ZmPGP1, encoding an auxin efflux carrier P-glycoprotein belonging to the ABC transporter family, led to the accumulation of high levels of auxin and alleviated Al toxicity in maize (Zhang et al. 2018c). Tobacco (Nicotiana tabacum L.) NtSUT1, a plasma membrane-localized sucrose/H+ symporter, actively regulates sucrose uptake in the root apex, promotes root elongation, and enhances Al tolerance (Kariya et al. 2017). Heterologous overexpression of the Jerusalem artichoke (Helianthus tuberosus) NHX-type transporters HtNHX1 and HtNHX2 regulate K+/H+ fluxes and cell wall structure, thereby enhancing Al3+ tolerance in rice (Li et al. 2020).
Cadmium detoxification
Cadmium (Cd) is a toxic heavy metal that affects plant growth and development due to its effects on plant nutrient absorption and other physiological processes (Clarkson 1996). Cd transporters play important roles in resistance to Cd toxicity. Genome-wide analysis of mulberry (Morus sp.) revealed four transporter families with Cd transport activity, including zinc-regulated transporters, iron-regulated transporter-like proteins (ZIPs), NRAMPs, the heavy metal ATPases (HMAs), and the metal tolerance or transporter proteins (MTPs) families; perhaps these transporters could be exploited for Cd phytoremediation (Fan et al. 2018). Negative regulation of Cd absorption via inhibiting the activity of the Fe2+ transporter IRT1 improved Cd tolerance of plants (Guan et al. 2019; Xu et al. 2018).
Bioinformatics and high-throughput sequencing identified 84 differentially expressed ABC genes in Brassica napus that are responsive to Cd (Zhang et al. 2018d). Heterologous overexpression of the Miscanthus sacchariflorus yellow stripe-like transporter gene MsYSL1 in Arabidopsis enhanced plant resistance to Cd by mediating the reallocation of metal ions (Chen et al. 2018). AtABCC3, an ABC-type transporter whose expression is induced by Cd exposure, increases phytochelatin-mediated Cd tolerance in coordination with AtABCC1/AtABCC2 (Brunetti et al. 2015). Arabidopsis OLIGOPEPTIDE TRANSPORTER3, a phloem-specific iron transporter, facilitates iron recirculation from mature to developing tissues (Zhai et al. 2014). NRT1.8, a member of the nitrate transporter (NRT1) family, removes nitrate from xylem vessels and improves Cd2+ tolerance in Arabidopsis (Li et al. 2010). In rice, OsABCG36, a G-type ATP-binding cassette transporter, does not affect the accumulation of Cd in shoots, but it exports Cd or Cd conjugates from root cells to increase plant resistance to this heavy metal (Fu et al. 2019).
Manganese detoxification
Manganese (Mn) is an important cofactor for many antioxidant defense enzymes that take part in the disposal of superoxide radicals, a function that is critical for plant survival. However, high concentrations of Mn in acidic or insufficiently drained soil are toxic to plants. NRAMP2, a divalent metal transporter of the NRAMP family, is critical for plant photosynthesis and cellular redox homeostasis in Arabidopsis (Alejandro et al. 2017). NRAMP2 plays an important role in remobilization of Mn in the Golgi to facilitate root growth under Mn-deficient conditions (Gao et al. 2017). HAK/K+ uptake (KUP)/K+ transporters (KTs) also play important roles in responses to K+ deficiency and abiotic stress in pear seedling rootstock (Yan et al. 2018).The tonoplast-localized transporter MTP8.2 (Mn-CDF metal tolerance protein) enhances plant tolerance to Mn along with MTP8.1 by sequestering Mn in plant vacuoles of root and shoot (Takemoto et al. 2017). The membrane protein CHLOROPLAST MANGANESE TRANSPORTER1 (CMT1) plays important roles in maintaining Mn homeostasis in Arabidopsis chloroplasts (Eisenhut et al. 2018). NH4+ downregulates the expression of the Mn influx transporter gene OsNRAMP5 through rhizosphere acidification, thereby improving manganese detoxification in rice (Hu et al. 2019).
Cesium detoxification
The release of cesium into the environment has led to serious environmental concerns, because it can be taken up by crops and affect crop production. Knocking out of OsHAK1, encoding a potassium transporter, reduced cesium levels in rice without affecting potassium absorption or crop yields (Rai et al. 2017). K+ UPTAKE PERMEASE (KUP) family proteins significantly reduce the accumulation of cesium in Arabidopsis; these KUP transporters play complementary rather than redundant roles in this process (Adams et al. 2019).
Resistance to many types of heavy metal stress
Metal transporters play critical roles in maintaining metal homeostasis and reducing metal toxicity. TpNRAMP5, a metal transporter isolated from dwarf Polish wheat (Triticum polonicum L.), enhances the accumulation of Cd, Co, and Mn but not Zn or Fe (Peng et al. 2018). Heterologous expression of SnYSL3, encoding a YSL transporter from Solanum nigrum, led to the formation of a range of metal-nicotianamine complexes to improve metal stress resistance in Arabidopsis, especially resistance to Cd and Fe (Feng et al. 2017b). NcZNT1, a Zn transporter from Noccaea caerulescens, enhanced Zn and Cd tolerance when expressed in Arabidopsis (Lin et al. 2016). Many regulators affect the expression of transporter genes, such as the post-transcriptional regulation of BnNRAMP1b, a vital Cd, Zn, and Mn transporter, by miR167 (Meng et al. 2017). The uptake of heavy metals in rice involves pH-dependent metal transporters such as OsNRAMP5, which affect Cd and Mn resistance in rice (Zhang et al. 2019b). As heavy metal pollution in soils around the world becomes more and more serious, plants with high tolerance to heavy metals will be more adaptable to the environment and have a higher survival rate, so study and utilization of metal transporters will become more and more important. Plants hyperaccumulating heavy metals also have potential uses in bioremediation of heavy metal pollution.
The roles of transporters in surviving nutrient deficiency
Plants require 17 essential elements and 14 minerals for growth, and these are only obtained through absorption from the environment, especially the soil (Nath and Tuteja 2016). The evolution of roots improved the adaptability of plants to the terrestrial environment and helped plants absorb nutrients from the soil. The absorption and utilization of soil nutrients are critical for plant growth and yield (Shahzad et al. 2018). Moreover, understanding nutrient transport has key implications for biofortification efforts to improve the nutrition of plant-based foods. Nutrient receptors and transporters play important roles in nutrient uptake and signaling (Nath and Tuteja 2016).
Phosphate deficiency
Phosphorus (P) is an essential nutrient for plant growth and development, functioning as a core component of nucleic acids and a key element in energy transfer (Peret et al. 2014); inorganic phosphate (Pi) from the soil is the only source of phosphorus for plants. The remodeling of root system architecture (RSA) is influenced by Pi deficiency, which is important for plant growth (Vance et al. 2003). In Arabidopsis, an ABC transporter complex composed of ALS3 and AtSTAR1 cooperates with LOW PHOSPHATE ROOT1/2 (LPR1/2) to regulate Pi-deficiency responses and the active remodeling of RSA by modulating Fe homeostasis (Dong et al. 2017).
The phosphate transporter 1 (PHT1) transporter family plays a major role in the uptake of phosphate from soils in plants (Nussaume et al. 2011). Heterologous expression experiments have been used to examine transporter function and establish a foundation for improving P uptake in crops, thereby reducing the need for fertilizer. For example, the rice myeloblastosis (MYB) transcription factor OsMYB5P triggers the expression of AtPht1;3 to improve phosphate acquisition and regulate shoot development and RSA (Yang et al. 2018b). The Pteris vittata phosphate transporter PvPht1;2 enhances phosphorus accumulation without inducing the uptake of the toxic element arsenic when expressed in tobacco (Cao et al. 2018). Heterologous overexpression of the rice Pi transporter gene OsPT8 increased Pi concentrations and selenium accumulation in tobacco, paving the way for breeding selenium-enriched plants (Song et al. 2017).
Plants increase Pi uptake via symbiosis with mycorrhizal fungi, which receive fixed carbon from the plant in a mutually beneficial symbiotic relationship that improves the nutrient absorption capacity of the plants and alleviates abiotic stress. Mycorrhizal fungi induce expression of some Pi transporters to improve the Pi level of the host plant, and induce some transporters to maintain the mycorrhizal fungus–plant symbiotic relationship (Maclean et al. 2017). In maize, mycorrhiza formation induces the expression of the Pi transporter gene ZmPt9 to enhance plant Pi uptake (Liu et al. 2018a). The expression of the Pi transporter gene HcPT2 from the mushroom-forming fungus Hebeloma cylindrosporum is induced by the host plant and enhances the accumulation of Pi in both the mycelium and host plant (Becquer et al. 2018).
AtNPF (nitrate transporter 1/peptide transporter family) 7.3/AtNRT1.5 is a nitrate transporter that plays an important role in regulating the response to phosphorus deficiency in Arabidopsis (Cui et al. 2019). An ABC transporter complex formed by ALS3 and NAP3 plays a critical role in the sugar-dependent response of Arabidopsis to phosphate deficiency (Belal et al. 2015). Remodeling of RSA is an effective way to enhance plant tolerance of phosphate (Pi) deficiency, which can be improved by increasing the level of the ABC transporter complex encoded by ALS3, LOW PHOSPHATE ROOT1 (LPR1), and LPR2 (Dong et al. 2017). The PHT family protein PHT5 in the vacuolar membrane regulates cytoplasmic Pi homeostasis to enhance plant growth under Pi deficiency (Lin et al. 2018; Liu et al. 2016b). Finally, VACUOLAR PHOSPHATE TRANSPORTER1 (VPT1) functions in vacuolar Pi storage and Pi adaptation in Arabidopsis (Liu et al. 2015).
Iron deficiency
Iron (Fe) is an important micronutrient for plant growth and essential metabolic processes. Fe is involved in plant photosynthesis, respiration, nitrogen fixation, plant hormone and DNA biosynthesis (Briat 2005), and Fe transporters are very important in regulating iron homeostasis to adapt to different plant growth requirements and to cope with environmental changes (Conte and Walker 2011). The NRAMP family is also involved in iron transport. In the case of iron deficiency, AtNRAMP3 regulates metal transport in the vacuole, resulting in the accumulation of Mn, Zn, and Fe in Arabidopsis (Thomine et al. 2003). The proton-coupled metal ion transporter AtNRAMP6 may be involved in the transport of Fe2+ from the Golgi/trans-Golgi network to the cytoplasm, contributing to the homeostasis and reuse of Fe2+; and mutation of AtNRAMP6 inhibits lateral root growth under iron-deficient conditions in Arabidopsis (Li et al. 2019). The iron transporter OsIRT1 can effectively take up Fe from the soil under iron deficiency conditions in rice (Bughio et al. 2002). Heterologously expressed HvYS1 (a barley yellow stripe 1 Fe transporter) increased Fe uptake, Fe translocation from root to shoot, and Fe accumulation in rice seeds (Banakar et al. 2016). Increasing iron transport in endosperm by overexpressing the vacuolar iron transporter gene TaVIT2 improved iron homeostasis in wheat tissue (Connorton et al. 2017).
Potassium deficiency
Potassium (K+) is a major nutrient that plays important roles in plant responses to osmotic stress and basic plant metabolism. Potassium is involved in photosynthesis, protein synthesis, osmotic pressure regulation, stomatal movement, enzyme activity activation, and other physiological activities in plants; it plays an especially important role in plant stress tolerance (Leigh and Jones 1984). Potassium transporters are responsible for the uptake and transport of K+. OsHAK1 (Chen et al. 2015) and OsHAK5 (Yang et al. 2014) are important high-affinity K+ (HAK) transporters that are activated under K+ deficiency conditions. Heterologous expression of ApKUP3, a high-affinity K+ transporter gene from Alternanthera philoxeroides, increased K+ accumulation and enhanced drought tolerance in rice (Song et al. 2014). The K+ transporter GhKT2 participates in K+ acquisition, transport, and distribution in cotton (Gossypium hirsutum) (Wang et al. 2018b). GhHAK5a, a high-affinity potassium transporter in cotton, is important for K+ absorption by roots in plants under potassium deficiency (Wang et al. 2019b). In maize, ZmHAK5 and ZmHAK1 regulate the dynamic balance of K+ under low K+ conditions, affecting the absorption and distribution of K+, and improving K+ utilization efficiency (Qin et al. 2019).
Magnesium deficiency
Magnesium (Mg) is an essential mineral element for plant growth and development. CorA/MRS2/MGT-type Mg2+ transporters are very important in maintaining Mg homeostasis. For instance, AtMGT6 (Mao et al. 2014) and AtMGT7 (Gebert et al. 2009) are critical for maintaining normal physiological activities in Arabidopsis under low Mg conditions. ZmMGT10 is specifically expressed in maize roots, and increased Mg2+ uptake via roots to enhance plant tolerance under magnesium deficiency conditions (Li et al. 2017a). ZmMGT12 is a Mg2+ transporter that functions in Mg transport to chloroplasts in maize (Li et al. 2018a). The Mg transporter gene OsMGT1 is required for resistance to magnesium deficiency in rice (Zhang et al. 2019a).
Copper deficiency
Copper (Cu) takes part in a variety of physiological processes in plants, making this micronutrient important for plant growth and development. Cu deficiency affects plant fertility. The main symptoms are vegetative and reproductive growth disorders, young leaf deformity, and insufficient water transport (Burkhead et al. 2009). The reported Cu transporters are mainly from the COPT/Ctr protein family. Cu might be co-transported by COPT2, COPT3, and COPT4 with COPT6. COPT7 can act alone in different tissues to regulate Cu homeostasis in rice (Yuan et al. 2011). Cu deficiency can induce the expression of the transporter genes COPT2 (in roots) and HMA1 (in leaves), causing Cu to migrate and circulate among leaves in Brassica napus (Billard et al. 2014). Other transporters also regulate Cu homeostasis, Zn transporters ZIP2 and ZIP4 can transport Cu2+ in Arabidopsis, and ZIP2 expression is significantly increased under low copper conditions (Puig et al. 2010). OsYSL16, a member of the yellow-stripe like (YSL) family and a Cu-nicotianamine (Cu-NA) transporter, is important for Cu redistribution under Cu-limited conditions in rice (Zhang et al. 2018a).
Zinc deficiency
Zinc (Zn) is an essential micronutrient for plant growth and development. In addition to promoting Zn absorption by arbuscular mycorrhizal fungi, Zn transporters are involved in Zn deficiency tolerance in plants (Khatun et al. 2018). The ZIP transporter family plays an important role in the transport of Zn. Zn deficiency can induce OsZIP6 expression in roots and leaves of rice (Kavitha et al. 2015). Under zinc-deficiency conditions, group F TabZIP transcription factors bind to ZDREs (Zn-deficiency-response elements) of TaZIP promoters to enhance TaZIP expression and maintain Zn homeostasis in wheat (Evens et al. 2017). SaZIP4 from Sedum alfredii plays critical roles in Zn accumulation, as demonstrated in transgenic Arabidopsis (Yang et al. 2018a). The tonoplast-localized transporter OsHMA3 transports Zn to and from the vacuole to help rice plants cope with Zn toxicity or the lack of Zn (Cai et al. 2019).
The roles of transporters in plant growth and yield
Transporters are responsible of the absorption of nutrients and regulate plant growth and development. Manipulating the expression of OsAAP3, encoding an amino acid transporter, increased tiller formation and grain yield in rice (Lu et al. 2018). The tomato tonoplast dicarboxylate transporter (SITDT) helps remobilize malate and citrate in the vacuoles of fruit cells, which is important for their flavor and organoleptic quality of the fruit (Liu et al. 2017b).In grape (Vitis vinifera cv. Malbec) ripening stage, ABA and GA3 increase the expression of sugar transporter genes VvHT2, VvHT3 and VvHT6 in berries to promote berry ripening (Murcia et al. 2018). The Arabidopsis jasmonate transporter AtJAT1/AtABCG16 controls the distribution of the phytohormone jasmonate and regulates plant growth and stress responses (Li et al. 2017c). The maize MATE transporter Big embryo 1 is required for the production of intermediates or products of the CYP78A pathway and regulates lateral organ initiation in plants (Suzuki et al. 2015). Transporters also link malate transport and mineral nutrition. Overexpressing OsALMT4, encoding an ALMT, affected the efflux and compartmentation of malate and increased Mn toxicity symptoms in rice (Liu et al. 2017a). Overexpressing OsNRT2.3b, encoding a nitrate transporter, promoted Pi uptake and translocation and increased grain yield in rice (Feng et al. 2017a).
Nitrogen (N) is a key factor in plant growth and root extension. Upregulating a nitrate transporter gene improved nitrogen status and plant growth in winter wheat (Triticum aestivum L.) (Jiang et al. 2017). The nitrate transporter CsNRT2.1 improves nitrate uptake, root length, and lateral root number in cucumber (Li et al. 2018b). A hexose transport protein (HT), CSHT11, plays an important role in the development of pollens and pollen tubes in cucumber (Wen et al. 2020). The ABCC3-type transporter affects seed yield and quality in chickpea (Cicer arietinum) by regulating the transport of glutathione conjugates (Basu et al. 2019). The nodule-localized Pi transporter GmPT7 enhances nitrogen fixation and seed yield in soybean (Chen et al. 2019).
The tonoplast-localized sugar transporter CITST2, whose expression is regulated by the WRKY transcription factor SUSIWM1, is involved in sugar uptake and accumulation in the flesh cells of watermelon (Citrullus lanatus) fruit (Ren et al. 2018). Expressing the human UDP-galactose transporter gene hUGT1 increased the lignin content and hardness of leaves and stems in tobacco (Abedi et al. 2018). Genome-wide identification, expression, and functional analyses demonstrated that the sugar transporter (STP) gene family is involved in early tuber growth and monosaccharide distribution in cassava (Manihot esculenta) (Liu et al. 2018c). Sucrose transporters also function in cotton fiber elongation, which can be activated by suppressing the expression of the putative sterol carrier protein gene GhSCP2D (Zhang et al. 2017).
Pollen development is key for high yields of grain/seed and fruit crops. The tapetum plays an important role in pollen development. The ABCG/WBC-type ABC transporters play a crucial role in the transport of sporopollenin precursors for exine formation on the pollen surface (Choi et al. 2011). The ABC transporter AcABCG38 plays important role in pollen development in pineapple (Ananas comosus), suggesting it could be used to improve pineapple production (Chen et al. 2017b).
Arbuscular mycorrhizal (AM) symbiosis is important for plant growth and defense due to the exchange of signals exchange between plant roots and AM fungi. Finally, the N-acetylglucosamine (GlcNAc) transporter NOPE1 stimulates fungal pathogenesis, increases the expression of virulence genes, and functions in AM symbiosis (Nadal et al. 2017). Destroying the activity of the amino acid transporter OsLHT1 inhibits the growth and reproduction of rice (Wang et al. 2019a). By using transporters that improve plant growth and yield, future research can produce higher yields and improve quality.
Summary and conclusions
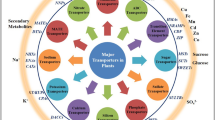
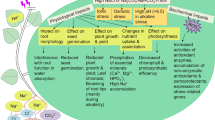
In this review, we provided an overview of the roles of transporters in plants under stress conditions. We summarized these transporters in Table S1, S2 and S3 and drawn a figure about the transporters transport substances under a variety of environmental stresses (Fig. 1). In conclusion, when plants are subjected to different external pressures, transporters will take different measures to protect the plants. Under salt stress, Na+ efflux by the role of the transporters in plasma membrane, and the transporters on the vacuole membrane and the vesicle membrane will cause Na+ to flow into them, thereby reducing the Na+ concentration in the cytoplasm and avoiding the cells from being poisoned by salt ions (Yang and Guo 2018). The transporters maintains the necessary material balance in the plant cells to response to stresses. Heavy metal stress triggers the transporters to store excess heavy metals in the vacuole, and nutrient deficiency causes transporters to transport nutrients in vacuole to cytoplasm. Similarly, under stress, plants will induce transporters to transport signal molecules, which will allow the expression of some stress-resistant genes to deal with adverse environments (Fig. 1). Transporters manage the acquisition of essential macro- and micronutrients while also play essential roles in preventing damage from environmental conditions such as high salt and heavy metals. Transporter families such as ABC transporters, which function in various parts of the plant, are involved in plant growth and development and the physiological and biochemical reactions required for stress resistance (Do et al. 2018). Transporters also interact with multiple metabolic pathways or regulate key genes to enhance stress resistance in plants. Therefore, the exploration of transporters is important for understanding plant stress resistance and plant growth, and for crop improvement.
References
Abedi T, Khalil MFM, Koike K, Hagura Y, Tazoe Y, Ishida N, Kitamura K, Tanaka N (2018) Expression of the human UDP-galactose transporter gene hUGT1 in tobacco plants’ enhanced plant hardness. J Biosci Bioeng 126(2):241–248
Adams E, Miyazaki T, Shin R (2019) Contribution of KUPs to potassium and cesium accumulation appears complementary in Arabidopsis. Plant Signal Behav 14(1):1554468
Alejandro S, Cailliatte R, Alcon C, Dirick L, Domergue F, Correia D, Castaings L, Briat J, Mari S, Curie C (2017) Intracellular distribution of manganese by the trans-Golgi Network Transporter NRAMP2 is critical for photosynthesis and cellular redox homeostasis. Plant Cell 29(12):3068–3084
Ali A, Khan IU, Jan M, Khan HA, Hussain S, Nisar M, Chung WS, Yun D (2018) The high-affinity potassium transporter EpHKT1;2 from the extremophile eutrema parvula mediates salt tolerance. Frontiers Plant Sci 9:1108–1108
Antunes WC, Danilo DMD, Pinheiro DP, Williams TCR, Loureiro ME (2017) Guard cell-specific down-regulation of the sucrose transporter SUT1 leads to improved water use efficiency and reveals the interplay between carbohydrate metabolism and K+ accumulation in the regulation of stomatal opening. Environ Exp Bot 135:73–85
Bai X, Xu J, Shao X, Luo W, Niu Z, Gao C, Wan D (2019) A novel gene coding γ-aminobutyric acid transporter may improve the tolerance of Populus euphratica to adverse environments. Frontiers Plant Sci 10:1083
Banakar R, Fernández ÁA, Abadía J, Capell T, Christou P (2016) The expression of heterologous Fe (III) phytosiderophore transporter HvYS1 in rice increases Fe uptake, translocation and seed loading and excludes heavy metals by selective Fe transport. Plant Biotechnol J 15:423–432
Basu U, Upadhyaya HD, Srivastava R, Daware A, Malik N, Sharma A, Bajaj D, Narnoliya L, Thakro V, Kujur A (2019) ABC transporter-mediated transport of glutathione conjugates enhances seed yield and quality in chickpea. Plant Physiol 180(1):253–275
Becquer A, Garcia K, Amenc L, Rivard C, Dore J, Trivessegura C, Szponarski W, Russet S, Baeza Y, Lassallekaiser B (2018) The Hebeloma cylindrosporum HcPT2 Pi transporter plays a key role in ectomycorrhizal symbiosis. New Phytol 220(4):1185–1199
Belal R, Tang R, Li Y, Mabrouk Y, Badr E, Luan S (2015) An ABC transporter complex encoded by aluminum sensitive 3 and NAP3 is required for phosphate deficiency responses in Arabidopsis. Biochem Biophys Res Commun 463(1):18–23
Billard V, Ourry A, Maillard A, Garnica M, Coquet L, Jouenne T, Cruz F, Garciamina J, Yvin J, Etienne P (2014) Copper-deficiency in Brassica napus induces copper remobilization, molybdenum accumulation and modification of the expression of chloroplastic proteins. PLOS ONE 9(10):e109889
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91(2):179–194
Bo L, Xu D, Ann HB, Hassan NEH (2017) Advances in methods for identification and characterization of plant transporter function. J Exp Bot 68(15):4045–4056
Briat JF (2005) Iron from soil to plant products. Bull Lacad Nat Méd 189(8):1609–1619
Brunetti P, Zanella L, De Paolis A, Litta DD, Cecchetti V, Falasca G, Barbieri M, Altamura MM, Costantino P, Cardarelli M (2015) Cadmium-inducible expression of the ABC-type transporter AtABCC3 increases phytochelatin-mediated cadmium tolerance in Arabidopsis. J Exp Bot 66(13):3815–3829
Bu Y, Takano T, Liu S (2019) The role of ammonium transporter (AMT) against salt stress in plants. Plant Signal Behav 14(8):1625696–1625696
Bughio N, Yamaguchi H, Nishizawa NK, Nakanishi H, Mori S (2002) Cloning an iron-regulated metal transporter from rice. J Exp Bot 53(374):1677–1682
Burkhead JL, Reynolds KAG, Abdelghany SE, Cohu CM, Pilon M (2009) Copper homeostasis: Tansley review. New Phytol 182(4):799–816
Cai H, Huang S, Che J, Yamaji N, Ma JF (2019) The tonoplast-localized transporter OsHMA3 plays an important role in maintaining Zn homeostasis in rice. J Exp Bot 70(10):2717–2725
Cao Y, Sun D, Chen J, Mei H, Ai H, Xu G, Chen Y, Ma LQ (2018) Phosphate transporter PvPht1;2 enhances phosphorus accumulation and plant growth without impacting arsenic uptake in plants. Environ Sci Technol 52(7):3975–3981
Chai H, Guo J, Zhong Y, Hsu CC, Zou C, Wang P, Zhu JK, Shi H (2020) The plasma-membrane polyamine transporter PUT3 is regulated by the Na+/H+ antiporter SOS1 and protein kinase SOS2. New Phytol 226(3):785–797
Chen G, Hu Q, Luo L, Yang T (2015) Rice potassium transporter OsHAK1 is essential for maintaining potassium-mediated growth and functions in salt tolerance over low and high potassium concentration ranges. Plant Cell Environ 38(12):2747–2765
Chen G, Liu C, Gao Z, Yu Z, Jiang H, Li Z, Ren D, Ling Y, Xu G, Qian Q (2017a) OsHAK1, a high-affinity potassium transporter, positively regulates responses to drought stress in rice. Frontiers Plant Sci 8:1885
Chen P, Yi L, Zhao L, Hou Z, Yan M, Hu B, Liu Y, Muhammad AS, Zhang Z, Ur RZ (2017b) Genome-Wide Identification and Expression Profiling of ATP-Binding Cassette (ABC) Transporter Gene Family in Pineapple (Ananas comosus (L.) Merr.) Reveal the Role of AcABCG38 in Pollen Development. Frontiers in Plant Science 8:2150
Chen H, Zhang C, Guo H, Hu Y, He Y, Jiang D (2018) Overexpression of a Miscanthus sacchariflorus yellow stripe-like transporter MsYSL1 enhances resistance of Arabidopsis to cadmium by mediating metal ion reallocation. Plant Growth Regul 85:101–111
Chen L, Qin L, Zhou L, Li X, Chen Z, Sun L, Wang W, Lin Z, Zhao J, Yamaji N (2019) A nodule-localized phosphate transporter Gm PT 7 plays an important role in enhancing symbiotic N2 fixation and yield in soybean. New Phytol 221(4):2013–2025
Choi H, Jin J, Choi S, Hwang J, Kim Y, Suh MC, Lee Y (2011) An ABCG/WBC-type ABC transporter is essential for transport of sporopollenin precursors for exine formation in developing pollen. Plant J 65(2):181–193
Clarkson DT (1996) Marschner H. 1995. Mineral nutrition of higher plants. second edition. 889pp. London: Academic Press, £29.95 (paperback). Ann Bot 78:527–528
Connorton JM, Jones ER, Rodriguezramiro I, Fairweathertait SJ, Uauy C, Balk J (2017) Wheat vacuolar iron transporter TaVIT2 transports Fe and Mn and Is effective for biofortification. Plant Physiol 174(4):2434–2444
Conte SS, Walker EL (2011) Transporters contributing to iron trafficking in plants. Mol Plant 4(3):464–476
Cui Y-N, Li X-T, Yuan J-Z, Wang F-Z, Wang S-M, Ma Q (2019) Nitrate transporter NPF7 3/NRT1 5 plays an essential role in regulating phosphate deficiency responses in Arabidopsis. Biochem Biophys Res Commun 508(1):314–319
Do THT, Martinoia E, Lee Y (2018) Functions of ABC transporters in plant growth and development. Curr Opin Plant Biol 41:32–38
Dong Y, Fan G, Zhao Z, Deng MJF (2014) Compatible solute, transporter protein, transcription factor, and hormone-related gene expression provides an indicator of drought stress in Paulownia fortunei. Funct Integr Genomics 14(3):479–491
Dong J, Pineros MA, Li X, Yang H, Liu Y, Murphy AS, Kochian LV, Liu D (2017) An Arabidopsis ABC transporter mediates phosphate deficiency-induced remodeling of root architecture by modulating iron homeostasis in roots. Mol Plant 10(2):244–259
Eisenhut M, Hoecker N, Schmidt SB, Basgaran RM, Flachbart S, Jahns P, Eser T, Geimer S, Husted S, Weber APM (2018) The plastid envelope chloroplast manganese transporter1 is essential for manganese homeostasis in Arabidopsis. Mol Plant 11(7):955–969
Evens NP, Buchner P, Williams LE, Hawkesford MJ (2017) The role of ZIP transporters and group F bZIP transcription factors in the Zn-deficiency response of wheat (Triticum aestivum). Plant J 92(2):291–304
Fan W, Liu C, Cao B, Qin M, Long D, Xiang Z, Zhao A (2018) Genome-wide identification and characterization of four gene families putatively involved in cadmium uptake, translocation and sequestration in Mulberry. Frontiers Plant Sci 9:879–879
Fang Y, Xiong L (2014) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci Cmls 72(4):673–689
Feng H, Li B, Zhi Y, Chen J, Li R, Xia X, Xu G, Fan X (2017a) Overexpression of the nitrate transporter, OsNRT23b, improves rice phosphorus uptake and translocation. Plant Cell Rep 36(8):1287–1296
Feng S, Tan J, Zhang Y, Liang S, Xiang S, Wang H, Chai T (2017b) Isolation and characterization of a novel cadmium-regulated Yellow stripe-like transporter (SnYSL3) in Solanum nigrum. Plant Cell Rep 36(2):281–296
Fu S, Lu Y, Zhang X, Yang G, Chao D, Wang Z, Shi M, Chen J, Chao D-Y, Li R (2019) The ABC transporter ABCG36 is required for cadmium tolerance in rice. J Exp Bot 70(20):5909–5918
Gallardo K, Courty P, Signor CL, Wipf D, Vernoud V (2014) Sulfate transporters in the plant’s response to drought and salinity: regulation and possible functions. Frontiers Plant Sci 5:580–580
Gao H, Xie W, Yang C, Xu J, Li J (2017) NRAMP2, a trans-Golgi network-localized manganese transporter, is required for Arabidopsis root growth under manganese deficiency. New Phytol 217:179–193
Garciaoliveira AL, Benito C, Guedespinto H, Martinslopes P (2018) Molecular cloning of TaMATE2 homoeologues potentially related to aluminium tolerance in bread wheat (Triticum aestivum L.). Plant Biol 20(5):817–824
Gebert M, Meschenmoser K, Svidova S, Weghuber J, Schweyen RJ, Eifler K, Lenz H, Weyand K, Knoop VPC (2009) A root-expressed magnesium transporter of the MRS2/MGT gene family in Arabidopsis thaliana allows for growth in low-Mg2+ environments. Plant Cell 21(12):4018–4030
Gong X, Liu M, Zhang L, Ruan Y, Ding R, Ji Y, Zhang N, Zhang S, Farmer J, Wang C (2015) Arabidopsis AtSUC2 and AtSUC4, encoding sucrose transporters, are required for abiotic stress tolerance in an ABA-dependent pathway. Physiol Plant 153(1):119–136
Guan MY, Zhu YX, Liu XX, Jin C (2019) Induction of S-nitrosoglutathione reductase reduces root cadmium uptake by inhibiting Iron-regulated transporter 1. Plant Soil 438(1):251–262
Guo F-Q, Young J, Crawford NM (2003) The nitrate transporter AtNRT1.1 (CHL1) functions in stomatal opening and contributes to drought susceptibility in Arabidopsis. Plant Cell 15(1):107–117
Hirayama T, Shinozaki K (2010) Research on plant abiotic stress responses in the post-genome era: past, present and future. Plant J 61(6):1041–1052
Hu AY, Zheng MM, Sun LM, Zhao XQ, Shen RF (2019) Ammonium alleviates manganese toxicity and accumulation in rice by down-regulating the transporter gene through rhizosphere acidification. Frontiers Plant Sci 10:1194
Huang C, Yamaji N, Mitani N, Yano M, Nagamura Y, Ma JF (2009) A bacterial-type ABC transporter is involved in aluminum tolerance in rice. Plant Cell 21(2):655–667
Huang W, Hu B, Liu J, Zhou Y, Liu S (2020) Identification and characterization of tonoplast sugar transporter (TST) gene family in cucumber. Horticult Plant J 6(3):145–157
Ibraheem O, Dealtry G, Roux S, Bradley G (2011) The effect of drought and salinity on the expressional levels of sucrose transporters in rice (‘Oryza sativa’ Nipponbare) cultivar plants. Plant Omics 4(2):68–74
Jarzyniak KM, Michał J (2014) Membrane transporters and drought resistance—a complex issue. Frontiers Plant Sci 5:687
Jiang S, Sun J, Tian Z, Hu H (2017) Root extension and nitrate transporter up-regulation induced by nitrogen deficiency improves nitrogen status and plant growth at the seedling stage of winter wheat (Triticum aestivum L.). Environ Exp Bot 141:28–40
Junli Zhou, Xiangfeng Wang, Yuling Jiao, Yonghua Qin, Xigang Liu (2007) Global genome expression analysis of rice in response to drought and high-salinity stresses in shoot, flag leaf, and panicle. Plant Mol Biol 63:591–608
Kang J, Hwang JU, Lee M, Kim YY, Assmann SM, Martinoia E, Lee Y (2010) PDR-type ABC transporter mediates cellular uptake of the phytohormone abscisic acid. Proc Natl Acad Sci USA 107:2355–2360
Kariya K, Sameeullah M, Sasaki T, Yamamoto Y (2017) Overexpression of the sucrose transporter gene NtSUT1 alleviates aluminum-induced inhibition of root elongation in tobacco (Nicotiana tabacum L.). Soil Science and Plant Nutrition 63(1):45–54
Kavitha PG, Kuruvilla S, Mathew MK (2015) Functional characterization of a transition metal ion transporter, OsZIP6 from rice (Oryza sativa L.). Plant Physiol Biochem 97:165–174
Khatun MA, Hossain MM, Bari MA, Abdullahil KM, Parvez MS, Alam MF, Kabir AH (2018) Zinc deficiency tolerance in maize is associated with the up-regulation of Zn transporter genes and antioxidant activities. Plant Biol 20(4):765–770
Kim DY, Jin JY, Alejandro S, Martinoia E, Lee Y (2010) Overexpression of AtABCG36 improves drought and salt stress resistance in Arabidopsis. Physiol Plant 139(2):170–180
Klein M, Perfus-Barbeoch L, Frelet A, Gaedeke N, Forestier C (2003) The plant multidrug resistance ABC transporter AtMRP5 is involved in guard cell hormonal signalling and water use. Plant J 36(1):119–129
Klein M, Geisler M, Suh SJ, Kolukisaoglu HÜ, Azevedo L, Plaza S, Curtis MD, Richter A, Weder B, Schulz B, Martinoia E (2004) Disruption of AtMRP4, a guard cell plasma membrane ABCC-type ABC transporter, leads to deregulation of stomatal opening and increased drought susceptibility. Plant J 39(2):219–236
Kuromori T, Sugimoto E, Shinozaki K (2011) Arabidopsis mutants of AtABCG22, an ABC transporter gene, increase water transpiration and drought susceptibility. Plant Journal 67(5):885–894
Kuromori T, Fujita M, Urano K, Tanabata T, Sugimoto E, Shinozaki K (2016) Overexpression of AtABCG25 enhances the abscisic acid signal in guard cells and improves plant water use efficiency. Plant Sci 251:75–81
Lee M, Choi Y, Burla B, Kim YY, Jeon B, Maeshima M, Yoo JY, Martinoia E, Lee Y (2008) The ABC transporter AtABCB14 is a malate importer and modulates stomatal response to CO2. Nat Cell Biol 10:1217–1223
Lee Eu, Kim KY, Zhang J, Yamaoka Y, Lee Y (2020) Arabidopsis seedling establishment under waterlogging requires ABCG5—mediated formation of a dense cuticle layer. New Phytol. https://doi.org/10.1111/nph.16816
Leigh RA, Jones RGW (1984) A hypothesis relating critical potassium concentrations for growth to the distribution and functions of this ion in the plant cell. New Phytol 97(1):1–13
Li J, Fu Y, Pike S, Bao J, Tian W, Zhang Y, Chen C, Zhang Y, Li H, Huang J (2010) The Arabidopsis nitrate transporter NRT18 functions in nitrate removal from the xylem sap and mediates cadmium tolerance. Plant Cell 22(5):1633–1646
Li F, Lei H, Zhao X, Shen X, Liu A (2012) Isolation and characterization of two sorbitol transporter gene promoters in micropropagated apple plants (Malus × domestica) regulated by drought stress. Plant Growth Regul 68:475–482
Li J, Liu J, Dong D, Jia X, Mccouch SR, Kochian LV (2014) Natural variation underlies alterations in Nramp aluminum transporter (NRAT1) expression and function that play a key role in rice aluminum tolerance. Proc Natl Acad Sci U S A 111(17):6503–6508
Li H, Wang N, Ding J, Liu C, Du H, Huang K, Cao M, Lu Y, Gao S, Zhang S (2017a) The maize CorA/MRS2/MGT-type Mg transporter, ZmMGT10, responses to magnesium deficiency and confers low magnesium tolerance in transgenic Arabidopsis. Plant Mol Biol 95(3):269–278
Li N, Meng H, Xing H, Liang L, Zhao X, Luo K (2017b) Genome-wide analysis of MATE transporters and molecular characterization of aluminum resistance in Populus. J Exp Bot 68(20):5669–5683
Li Q, Zheng J, Li S, Huang G, Skilling SJ, Wang L, Li L, Li M, Yuan L, Liu P (2017c) Transporter-mediated nuclear entry of jasmonoyl-isoleucine is essential for jasmonate signaling. Mol Plant 10(5):695–708
Li H, Liu C, Zhou L, Zhao Z, Li Y, Qu M, Huang K, Zhang L, Lu Y, Cao M (2018a) Molecular and functional characterization of the magnesium transporter gene ZmMGT12 in maize. Gene 665:167–173
Li Y, Li J, Yan Y, Liu W, Zhang W, Gao L, Tian Y (2018b) Knock-down of CsNRT2.1, a cucumber nitrate transporter, reduces nitrate uptake, root length, and lateral root number at low external nitrate concentration. Frontiers Plant Sci 9:722–722
Li J, Wang Y, Zheng L, Li Y, Zhou X, Li J, Gu D, Xu E, Lu Y, Chen X (2019) The intracellular transporter AtNRAMP6 is involved in Fe homeostasis in Arabidopsis. Frontiers Plant Sci 10:1124
Li W, Du J, Feng H, Wu Q, Xu G, Shabala S, Yu L (2020) Function of NHX-type transporters in improving rice tolerance to aluminum stress and soil acidity. Planta 251(3):1–13
Lin Y-F, Zeshan H, Sangita T, Henk S (2016) Expression of the ZNT1 zinc transporter from the metal hyperaccumulator Noccaea caerulescens confers enhanced zinc and cadmium tolerance and accumulation to Arabidopsis thaliana. PLoS ONE 11(3):e0149750
Lin W, Lin Y, Chiang S, Syu C, Hsieh L, Chiou T (2018) Evolution of microRNA827 targeting in the plant kingdom. New Phytol 217(4):1712–1725
Liu J, Yang L, Luan M, Wang Y, Zhang C, Zhang B, Shi J, Zhao F, Lan W, Luan S (2015) A vacuolar phosphate transporter essential for phosphate homeostasis in Arabidopsis. Proc Natl Acad Sci U S A 112(47):6571–6578
Liu J, Li Y, Wang W, Gai J, Li Y (2016a) Genome-wide analysis of MATE transporters and expression patterns of a subgroup of MATE genes in response to aluminum toxicity in soybean. BMC Genom 17(1):1–15
Liu TY, Huang TK, Yang SY, Hong YT, Huang SM, Wang FN, Chiang SF, Tsai SY, Lu WC, Chiou TJ (2016b) Identification of plant vacuolar transporters mediating phosphate storage. Nat Commun 7:11095
Liu J, Zhou M, Delhaize E, Ryan PR (2017a) Altered expression of a malate-permeable anion channel, OsALMT4, disrupts mineral nutrition. Plant Physiol 175(4):1745–1759
Liu R, Li B, Qin G, Zhang Z, Tian S (2017b) Identification and functional characterization of a tonoplast dicarboxylate transporter in tomato (Solanum lycopersicum). Frontiers Plant Sci 8:186–186
Liu F, Xu Y, Han G, Wang W, Li X, Cheng B (2018a) Identification and functional characterization of a maize phosphate transporter induced by mycorrhiza formation. Plant Cell Physiol 59(8):1683–1694
Liu MY, Lou HQ, Chen WW, Pineros MA, Xu JM, Fan W, Kochian LV, Zheng SJ, Yang JL (2018b) Two citrate transporters coordinately regulate citrate secretion from rice bean root tip under aluminum stress. Plant Cell Environ 41(4):809–822
Liu Q, Dang H, Chen Z, Wu J, Chen Y, Chen S, Luo L (2018c) Genome-wide identification, expression, and functional analysis of the sugar transporter gene family in cassava (Manihot esculenta). Int J Mol Sci 19(4):987
Lu K, Wu B, Wang J, Zhu W, Nie H, Qian J, Huang W, Fang Z (2018) Blocking amino acid transporter OsAAP3 improves grain yield by promoting outgrowth buds and increasing tiller number in rice. Plant Biotechnol J 16(10):1710–1722
Ma Q, Sun M, Lu J, Kang H, You C, Hao Y (2019a) An apple sucrose transporter MdSUT2.2 is a phosphorylation target for protein kinase MdCIPK22 in response to drought. Plant Biotechnol J 17(3):625–637
Ma QJ, Sun MH, Kang H, Lu J, You CX, Hao YJ (2019b) A CIPK protein kinase targets sucrose transporter MdSUT2.2 at Ser(254) for phosphorylation to enhance salt tolerance. Plant Cell Environ 42(3):918–930
Maclean AM, Bravo A, Harrison M (2017) Plant signaling and metabolic pathways enabling arbuscular mycorrhizal symbiosis. Plant Cell 29(10):2319–2335
Magalhaes JV, Liu J, Guimaraes CT, Lana UGP, Alves VMC, Wang Y, Schaffert RE, Hoekenga OA, Pineros MA, Shaff J (2007) A gene in the multidrug and toxic compound extrusion (MATE) family confers aluminum tolerance in sorghum. Nat Genet 39(9):1156–1161
Mao D, Chen J, Tian L, Liu Z, Yang L, Tang R, Li J, Lu C, Yang Y, Shi J (2014) Arabidopsis transporter MGT6 mediates magnesium uptake and is required for growth under magnesium limitation. Plant Cell 26(5):2234–2248
Matsuda S, Takano S, Sato M, Furukawa K, Nagasawa H, Yoshikawa S, Kasuga J (2016) Rice stomatal closure requires guard cell plasma membrane ATP-binding cassette transporter RCN1/OsABCG5. Mol Plant 009(003):417–427
Meng JG, Zhang XD, Tan SK, Zhao KX, Yang ZM (2017) Genome-wide identification of Cd-responsive NRAMP transporter genes and analyzing expression of NRAMP 1 mediated by miR167 in Brassica napus. Biometals 30(6):917–931
Mohammed AS, Kapri A, Goel R (2011) Heavy metal pollution: source, impact, and remedies. Biomanag Met Contam Soil. https://doi.org/10.1007/978-94-007-1914-9_1
Monfared HH, Chew JK, Azizi P, Xue G, Ee S, Kadkhodaei S, Hedayati P, Ismail I, Zainal Z (2020) Overexpression of a rice monosaccharide transporter gene (OsMST6) confers enhanced tolerance to drought and salinity stress in Arabidopsis thaliana. Plant Mol Biol Rep 38(1):151–164
Murcia G, Pontin M, Piccoli P (2018) Role of ABA and Gibberellin A3 on gene expression pattern of sugar transporters and invertases in Vitis vinifera cv. Malbec during berry ripening. Plant Growth Regul 84(2):275–283
Nadal M, Sawers RJH, Naseem S, Bassin B, Kulicke C, Sharman A, An G, An K, Ahern KR, Romag A (2017) An N-acetylglucosamine transporter required for arbuscular mycorrhizal symbioses in rice and maize. Nat Plants 3(6):17073
Nath M, Tuteja N (2016) NPKS uptake, sensing, and signaling and miRNAs in plant nutrient stress. Protoplasma 253(3):767–786
Nguyen VNT, Moon S, Jung KH (2014) Genome-wide expression analysis of rice ABC transporter family across spatio-temporal samples and in response to abiotic stresses. J Plant Physiol 171(14):1276–1288
Nio MC, Nogoy FM, Kang KK, Cho YG (2018) Low-affinity cation transporter 1 improves salt stress tolerance in Japonica rice. Plant Breed Biotechnol 6:82–93
Noelia J, Benito P, Begona G, Alejandro A, Asmini A, Caitlin SB, Raquel O, Maria JA, Matthew G, Vicente M (2017) The sodium transporter encoded by the HKT1;2 gene modulates sodium/potassium homeostasis in tomato shoots under salinity. Plant Cell Environ 40(5):658–671
Nussaume L, Kanno S, Javot H, Marin E, Pochon N, Ayadi A, Nakanishi TM, Thibaud M (2011) Phosphate import in plants: focus on the PHT1 transporters. Frontiers Plant Sci 2:83–83
Oh D, Leidi EO, Zhang Q, Hwang S, Li Y, Quintero FJ, Jiang X, Durzo MP, Lee SY, Zhao Y (2009) Loss of halophytism by interference with SOS1 expression. Plant Physiol 151(1):210–222
Park W, Kim H, Park T, Lee Y, Ahn S (2017) Functional characterization of plasma membrane-localized organic acid transporter (CsALMT1) involved in aluminum tolerance in Camelina sativa L. Plant Biotechnol Rep 11(3):181–192
Peng F, Wang Chao, Zhu J, Zeng J (2018) Expression of TpNRAMP5, a metal transporter from Polish wheat (Triticum polonicum L), enhances the accumulation of Cd, Co and Mn in transgenic Arabidopsis plants. Planta 247(6):1395–1406
Peret B, Desnos T, Jost R, Kanno S, Berkowitz O, Nussaume L (2014) Root architecture responses, search of phosphate. Plant Physiol 166(4):1713–1723
Phukan UJ, Jeena GS, Tripathi V, Shukla RK (2018) MaRAP2-4, a waterlogging-responsive ERF from Mentha, regulates bidirectional sugar transporter AtSWEET10 to modulate stress response in Arabidopsis. Plant Biotechnol J 16(1):221–233
Planchet E, Kaiser WM (2006) Nitric oxide production in plants: facts and fictions. Plant Signal Behav 1(2):46–51
Puig S, Andrés-Colás N, García-Molina A, Pearrubia L (2010) Copper and iron homeostasis in Arabidopsis: responses to metal deficiencies, interactions and biotechnological applications. Plant Cell Environ 30(3):271–290
Qin Y, Wu W, Wang Y (2019) ZmHAK5 and ZmHAK1 function in K + uptake and distribution in maize under low K + conditions. J Integr Plant Biol 61(6):691–705
Rai H, Yokoyama S, Satohnagasawa N, Furukawa J, Nomi T, Ito Y, Fujimura S, Takahashi H, Suzuki R, Yousra E (2017) Cesium uptake by rice roots largely depends upon a single gene, HAK1, which encodes a potassium transporter. Plant Cell Physiol 58(9):1486–1493
Ranjan A, Pandey N, Lakhwani D, Dubey NK, Pathre UV, Sawant SV (2012) Comparative transcriptomic analysis of roots of contrasting Gossypium herbaceum genotypes revealing adaptation to drought. BMC Genom 13(1):680
Remy E, Cabrito TR, Baster P, Batista RA, Teixeira MC, Friml J, Sa-Correia I, Duque P (2013) A major facilitator superfamily transporter plays a dual role in polar auxin transport and drought stress tolerance in Arabidopsis. Plant Cell 25(3):901–926
Ren Y, Guo S, Zhang J, He H, Sun H, Tian S, Gong G, Zhang H, Levi A, Tadmor Y (2018) A tonoplast sugar transporter underlies a sugar accumulation QTL in Watermelon. Plant Physiol 176(1):836–850
Rentsch D, Hirner B, Schmelzer E, Frommer WB (1996) Salt stress-induced proline transporters and salt stress-repressed broad specificity amino acid permeases identified by suppression of a yeast amino acid permease-targeting mutant. Plant Cell 8:1437–1446
Reynallorens I, Corrales I, Poschenrieder C, Barcelo J, Cruzortega R (2015) Both aluminum and ABA induce the expression of an ABC-like transporter gene (FeALS3) in the Al-tolerant species Fagopyrum esculentum. Environ Exp Bot 111:74–82
Ribeiro AP, De Souza WR, Martins PK, Vinecky F, Duarte KE, Basso MF, Cunha BADBD, Campanha RB, De Oliveira PA, Centeno DDC (2017) Overexpression of BdMATE gene improves aluminum tolerance in Setaria viridis. Frontiers Plant Sci 8:865–865
Seyed AH, Anne M, Mohammad RH, Ali N (2017) Induction of barley silicon transporter HvLsi1 and HvLsi2, increased silicon concentration in the shoot and regulated starch and ABA homeostasis under osmotic stress and concomitant potassium deficiency. Frontiers Plant Sci 8:1359–1359
Shahzad R, Harlina PW, Ayaad M, Ewas M, Nishawy E, Fahad S, Subthain H, Amar MH (2018) Dynamic roles of microRNAs in nutrient acquisition and plant adaptation under nutrient stress: a review. Plant Omics 11(01):58–79
Shen C, Bai Y, Wang S, Zhang S, Wu Y, Chen M, Jiang D, Qi Y (2010) Expression profile of PIN, AUX/LAX and PGP auxin transporter gene families in Sorghum bicolor under phytohormone and abiotic stress. FEBS J 277(14):2954–2969
Song ZZ, Yang SY, Zuo J, Su YH (2014) Over-expression of ApKUP3 enhances potassium nutrition and drought tolerance in transgenic rice. Biol Plant 58(4):649–658
Song Z, Shao H, Huang H, Shen Y, Wang L, Wu F, Han D, Song J, Jia H (2017) Overexpression of the phosphate transporter gene OsPT8 improves the Pi and selenium contents in Nicotiana tabacum. Environ Exp Bot 137(137):158–165
Steffens B, Kovalev A, Gorb SN, Sauter M (2012) Emerging roots alter epidermal cell fate through mechanical and reactive oxygen species signaling. Plant Cell 24(8):3296–3306
Suzuki M, Sato Y, Wu S, Kang B, Mccarty DR (2015) Conserved functions of the MATE transporter BIG EMBRYO1 in regulation of lateral organ size and initiation rate. Plant Cell 27(8):2288–2300
Takemoto Y, Tsunemitsu Y, Fujiikashino M, Mitaniueno N, Yamaji N, Ma JF, Kato S, Iwasaki K, Ueno D (2017) The tonoplast-localized transporter MTP82 contributes to manganese detoxification in the shoots and roots of Oryza sativa L. Plant Cell Physiol 58(9):1573–1582
Thomine S, Lelievre F, Debarbieux E, Schroeder JI, Barbierbrygoo H (2003) AtNRAMP3, a multispecific vacuolar metal transporter involved in plant responses to iron deficiency. Plant J 34(5):685–695
Upadhyay N, Kar D, Mahajan BD, Nanda S, Rahiman R, Panchakshari N, Bhagavatula L, Datta S (2019) The multitasking abilities of MATE transporters in plants. J Exp Bot 70(18):4643–4656
Vahdati K, Leslie C (2013) Abiotic stress - plant responses and applications in agriculture. Press Univ France. https://doi.org/10.5772/45842
Vance CP, Uhdestone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157(3):423–447
Veraestrella R, Gomezmendez MF, Amezcuaromero JC, Barkla BJ, Rosassantiago P, Pantoja O (2017) Cadmium and zinc activate adaptive mechanisms in Nicotiana tabacum similar to those observed in metal tolerant plants. Planta 246(3):433–451
Wang Y, Cai Y, Cao Y, Liu J (2018a) Aluminum-activated root malate and citrate exudation is independent of NIP1;2-facilitated root-cell-wall aluminum removal in Arabidopsis. Plant Signal Behav 13(1):e1422469
Wang Y, Juan XU, Zhang M, Tian X, Zhaohu LI (2018b) GhKT2: a novel K+ transporter gene in cotton (Gossypium hirsutum). Frontiers Agric Sci Eng 5(2):226–235
Wang X, Yang G, Shi M, Hao D, Wei Q, Wang Z, Fu S, Su Y, Xia J (2019a) Disruption of an amino acid transporter LHT1 leads to growth inhibition and low yields in rice. BMC Plant Biol 19(1):268
Wang Y, Wang Y, Li B, Xiong C, Eneji AE, Zhang M, Li F, Tian X, Li Z (2019b) The cotton high-affinity K+ transporter, GhHAK5a, is essential for shoot regulation of K+ uptake in root under potassium deficiency. Plant Cell Physiol 60(4):888–899
Wen S, Bao T, Zeng X, Bie Z, Cheng J (2020) CsHT11 encodes a pollen-specific hexose transporter and is induced under high level sucrose in pollen tubes of cucumber (Cucumis sativus). Plant Growth Regul 90:237–248
Wormit A, Trentmann O, Feifer I, Lohr C, Tjaden J, Meyer S, Schmidt UG, Martinoia E, Neuhaus HE (2006) Molecular identification and physiological characterization of a novel monosaccharide transporter from arabidopsis involved in vacuolar sugar transport. Plant Cell 18(12):3476–3490
Xu G, Li C, Yao Y (2009) Proteomics analysis of drought stress-responsive proteins in Hippophae rhamnoides L. Plant Mol Biol Rep 27(2):153–161
Xu Q, Pan W, Zhang R, Lu Q, Xue W, Wu C, Song B, Du S (2018) Inoculation with Bacillus subtilis and Azospirillum brasilense produces abscisic acid that reduces Irt1-mediated cadmium uptake of roots. J Agric Food Chem 66(20):5229–5236
Yamada K, Osakabe Y, Mizoi J, Nakashima K, Fujita Y, Shinozaki K, Yamaguchishinozaki K (2010) Functional analysis of an Arabidopsis thaliana abiotic stress-inducible facilitated diffusion transporter for monosaccharides. J Biol Chem 285(2):1138–1146
Yan L, Peng L, Xie C, Shi X, Dong C, Shen Q, Xu Y (2018) Genome-wide identification, characterization, and expression analyses of the HAK / KUP / KT potassium transporter gene family reveals their involvement in K + deficient and abiotic stress responses in pear rootstock seedlings. Plant Growth Regul 85:187–198
Yang Y, Guo Y (2018) Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol 217(2):523–533
Yang T, Zhang S, Hu Y, Wu F, Hu Q, Chen G, Cai J, Wu T, Moran N, Yu L (2014) The role of a potassium transporter OsHAK5 in potassium acquisition and transport from roots to shoots in rice at low potassium supply levels. Plant Physiol 166(2):945–959
Yang Q, Ma X, Luo S, Gao J, Yang X, Feng Y (2018a) SaZIP4, an uptake transporter of Zn/Cd hyperaccumulator Sedum alfredii Hance. Environ Exp Bot 155:107–117
Yang WT, Baek D, Yun D, Lee KS, Hong SY, Bae KD, Chung YS, Kwon YS, Kim DH, Jung K (2018b) Rice OsMYB5P improves plant phosphate acquisition by regulation of phosphate transporter. PLoS ONE 13(3):e0194628
Ye J, Wang X, Hu T, Zhang F, Wang B, Li C, Yang T, Li H, Lu Y, Giovannoni JJ (2017) An InDel in the promoter of Al-ACTIVATED MALATE TRANSPORTER9 selected during tomato domestication determines fruit malate contents and aluminum tolerance. Plant Cell 29(9):2249–2268
Yokosho K, Yamaji N, Fujiikashino M, Ma JF (2016) Retrotransposon-mediated aluminum tolerance through enhanced expression of the citrate transporter OsFRDL4. Plant Physiol 172(4):2327–2336
Yong H, Shuya Y, Lu H, Xuelong W, Jianbin Z (2018) A sodium transporter HvHKT1;1 confers salt tolerance in barley via regulating tissue and cell ion homeostasis. Plant Cell Physiol 59(10):1976–1989
Yuan M, Li X, Xiao J, Wang S (2011) Molecular and functional analyses of COPT/Ctr-type copper transporter-like gene family in rice. BMC Plant Biol 11(1):69
Zhai Z, Gayomba SR, Jung H, Vimalakumari NK, Pineros MA, Craft E, Rutzke M, Danku J, Lahner B, Punshon T (2014) OPT3 is a phloem-specific iron transporter that is essential for systemic iron signaling and redistribution of iron and cadmium in Arabidopsis. Plant Cell 26(5):2249–2264
Zhang J-T, Mu C-S (2010) Effects of saline and alkaline stresses on the germination, growth, photosynthesis, ionic balance and anti-oxidant system in an alkali-tolerant leguminous forage Lathyrus quinquenervius. Soil Sci Plant Nutr 55(5):685–697
Zhang J, Jia W, Yang J, Ismail AM (2006) Role of ABA in integrating plant responses to drought and salt stresses. Field Crops Res 97(1):111–119
Zhang H, Zhu H, Pan Y, Yu Y, Luan S, Li L (2014) A DTX/MATE-type transporter facilitates abscisic acid efflux and modulates ABA sensitivity and drought tolerance in Arabidopsis. Mol Plant 7(10):1522–1532
Zhang Z, Ruan Y, Zhou N, Wang F, Guan X, Fang L, Shang X, Guo W, Zhu S, Zhang T (2017) Suppressing a putative sterol carrier gene reduces plasmodesmal permeability and activates sucrose transporter genes during cotton fiber elongation. Plant Cell 29(8):2027–2046
Zhang C, Lu W, Yang Y, Shen Z, Ma JF, Zheng LJP, Physiology C (2018a) OsYSL16 is required for preferential Cu distribution to floral organs in rice. Plant Cell Physiol 59(10):2039–2051
Zhang L, Wu X, Wang J, Qi C, Wang X, Wang G, Li M, Li X, Guo Y (2018b) BoALMT1, an Al-induced malate transporter in cabbage, enhances aluminum tolerance in Arabidopsis thaliana. Frontiers Plant Sci 8:2156
Zhang M, Lu X, Li C, Zhang B, Zhang C, Zhang XS, Ding Z (2018c) Auxin efflux carrier ZmPGP1 mediates root growth inhibition under aluminum stress. Plant Physiol 177(2):819–832
Zhang XD, Zhao KX, Yang ZM (2018d) Identification of genomic ATP binding cassette (ABC) transporter genes and Cd-responsive ABCs in Brassica napus. Gene 664:139–151
Zhang L, Peng Y, Li J, Tian X, Chen Z (2019a) OsMGT1 confers resistance to magnesium deficiency by enhancing the import of Mg in rice. Int J Mol Sci 20(1):207
Zhang Q, Chen H, Xu C, Zhu H, Zhu Q (2019b) Heavy metal uptake in rice is regulated by pH-dependent iron plaque formation and the expression of the metal transporter genes. Environ Exp Bot 162:392–398
Zhi CC, Yamaji N, Horie T, Jing C, Jian L (2017) A magnesium transporter OsMGT1 plays a critical role in salt tolerance in rice. Plant Physiol 174(3):1837–1849
Zhou A, Ma H, Feng S, Gong S, Wang J (2018a) DsSWEET17, a tonoplast-localized sugar transporter from Dianthus spiculifolius, affects sugar metabolism and confers multiple stress tolerance in Arabidopsis. Int J Mol Sci 19(6):1564
Zhou A, Ma H, Shuang F, Gong S, Wang J (2018b) A novel sugar transporter from Dianthus spiculifolius, DsSWEET12, affects sugar metabolism and confers osmotic and oxidative stress tolerance in Arabidopsis. Int J Mol Sci 19(2):497
Acknowledgements
The National Natural Science Foundation of China (31971410 for LL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Dawei Xue.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, P., Luo, T., Pu, X. et al. Plant transporters: roles in stress responses and effects on growth and development. Plant Growth Regul 93, 253–266 (2021). https://doi.org/10.1007/s10725-020-00684-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00684-3