Abstract
In the early years of my GI fellowship, a healthy 40-year-old man came to my clinic and announced that he was going to die of pancreatic cancer. His brothers, father and uncles had all died of the disease; he felt his fate was inescapable. I asked whether his family members had seen doctors or had any tests. His answer was yes to both. Even so, doctors could not diagnose the pancreatic cancer at early stages. CT scans were always negative. I thought to myself, in order to help this patient—CT scans may not be reliable for early detection. Perhaps other methods of imaging the pancreas might be of more benefit. This patient opened a door that led to a 30-year journey of trying to detect pancreatic cancer at earlier stages when it is curable.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The initial patient was a member of Family X; a large kindred that inherited pancreatic cancer in an autosomal dominant fashion with a prodrome of diabetes [1]. Other families followed. Over the next several years, our team studied the role of pancreatic cancer surveillance in 14 high-risk individuals from 3 families that inherited pancreatic cancer. Every test for the pancreas was evaluated including CA19.9, CEA, endoscopic ultrasound (EUS), endoscopic retrograde cholangiopancreatography (ERCP), magnetic resonance imaging (MRI) and computer tomography (CT) scan. Of these tests, the EUS, ERCP and MRI were most useful in detecting the early stages of pancreatic cancer including carcinoma in-situ, and the other tests were of no value [2]. This was the first report of pancreatic cancer surveillance. Physicians and scientists studying pancreatic cancer were intrigued that early detection was possible in such a uniformly deadly disease. Similar programs for pancreatic cancer surveillance were soon set up in centers of excellence, including the CAPS program at Johns Hopkins, programs at Nebraska, University of Pittsburgh and the Mayo Clinic, the PRECEDE consortium and many international centers across the globe [3, 4]. Interestingly, 30 years later, EUS and MRI remain the foundational tests for pancreatic cancer surveillance. ERCP has become unnecessary, as the accuracy of MRI markedly improved over time.
Pathology and nomenclature
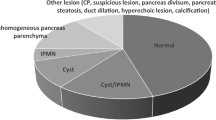
To identify a thing, one must name it. As my friend and superb pathologist, the late Roger Haggitt, would tell me: The eye sees what the mind knows. Familial pancreatic cancer was initially described in 1973, however the nomenclature for the precursors of pancreatic cancer had been in a state of disarray for decades prior to the 1970s and even after that [5]. Despite the work of renowned pathologists, such as Klimstra, Longnecker, Cubilla, Takeuchi, and Hruban, who had studied pancreatic neoplasia, a central problem remained regarding the wide variety of names used to describe neoplasia [6,7,8,9]. The precursor names varied from dysplasia, to papillary hyperplasia with atypia, to intraductal proliferation and so on. As mouse models were developed emulating the disease process in humans, a uniform approach to the language of pancreatic tumorigenesis was clearly needed. In 2004, a workshop at University of Pennsylvania assembled a remarkable group of 100 physicians and scientists, who formed working groups to develop a consensus report on a wide range of topics from genetically engineered mouse models, to biology, to therapy and early detection [10]. One of the most important products of the conference was the consensus report on the pathology of pancreatic cancer and it’s precursor lesions. The extraordinary value of this consensus report cannot be overestimated. It became the basis for our understanding of pancreatic cancer biology and for clinical diagnosis of pre-cancerous disease. A revised classification system was developed in 2015, such that is 2-tiered, low-grade pancreatic intraepithelial neoplasia (PanIN) and high-grade PanIN, is now used [11]. Lesion size could help distinguish PanIN from intraductal papillary mucinous neoplasm (IPMN).
Cost-effectiveness
While surveillance programs became more widely available and patient numbers escalated, it was initially difficult to get financial coverage of pancreatic cancer surveillance for asymptomatic high-risk individuals. Most surveillance exams were not covered by insurance in these early years. To create a proof of principle that insurance should cover the cost of surveillance, we needed to demonstrate that it was cost effective. Cost effectiveness studies could also help illuminate which patients were best suited for surveillance and when to start and stop. Our initial study, in 2002, showed that surveillance was cost-effective if a patient had a life-time risk of pancreatic cancer more than 15% and surveillance lost cost effectiveness as the patient aged above age 70 [12]. Subsequent studies were better performed, but showed fairly similar results [13, 14]. An economic analysis, performed in 2019 by Corral et al., revealed that surveillance with EUS was the dominant strategy in highest risk individuals (greater than 20-fold increased risk of pancreatic cancer); the highest risk individuals would be those with FAMMM, Peutz Jeghers, hereditary pancreatitis, and those families with 2 or 3 affected first degree relatives (FDR)---where lifetime risks can range from 15 to 36%. Surveillance with MRI was the dominant strategy if pancreatic cancer risk was only 5-fold greater than the population [13]. Hereditary Breast and Ovarian Cancer Syndrome (HBOCS) individuals without other risk factors, in general, did not reach the 5-fold risk cut off. Once patients reach age 76, surveillance was no longer cost effective. This data is helpful in triaging patients who would be best served in a setting of limited resources.
Risk stratification
There is a unanimous agreement that surveillance should be done at centers of expertise. However, endoscopic ultrasound, one of the foundational imaging modalities for discovering early cancers, is a subjective test and the number of physicians trained to evaluate high-risk individuals is limited. Recent AGA guidelines suggest that pancreatic cancer surveillance should be available for even lower-risk individuals, such as Hereditary Breast and Ovarian Cancer Syndrome (HBOCS) patients [15]. The prevalence of BRCA1 or 2 is estimated to be one in 400–500 people. In the US, the number of BRCA1/2 carriers between the age of 50 and 70 would approximate 192,000. About 14% of these individuals will know that they carry a BRCA1 or 2 mutation [16]; this provides 27,000 mutation carriers who would need pancreatic surveillance every year if AGA guidelines are followed. The demand exceeds our capacity. Cost effectiveness studies, above, and risk stratification can help triage this group of individuals. For example, some consensus guidelines suggest performing surveillance on lower-risk HBOCS individuals who also have a family member with pancreatic cancer [17]. Other non-genetic risk factors could be included in risk assessment, such as smoking and diabetes, for lower at-risk patients [18, 19]. Our current management for BRCA1/2 carriers, who do not have a family history of pancreatic cancer or other non-genetic factors, is to check annually for diabetes via hemoglobin A1c. If new onset diabetes develops (HgbA1c ≥ 6.0), the patient now approaches a 10% lifetime risk of pancreatic cancer and starts surveillance. Only prospective studies of this approach will determine if it is practical and cost effective. Further research can help discover other risk factors.
Biomarkers
Who doesn’t love a good biomarker? Together with my outstanding collaborators, I have spent nearly 30 years pursuing ideal biomarkers for pancreatic cancer. Issues that remain difficult to overcome include the need for an extremely high specificity; false positives would require expensive work-ups and substantial worry for the biomarker-positive patient. Other issues also slow the ability of a biomarker reaching from the bench to the bedside. If a biomarker cannot be patented, it will be difficult to move into the commercial realm. Biomarkers need to be tested on many cohorts of patients, in keeping with the best design for development [20]. Even when a biomarker panel reaches initial benchmarks, such as recently done through Immunovia and the IMMray PanCan-d test, if the biomarker test is not reimbursable by insurance, then the utility in the clinic will be limited due to costs [21]. Machine-learning algorithms to improve pre-test probability, population scalability, and lowered cost for engineered testing may help resolve some these issues.
Future endeavors
This is an exciting time to work on the earlier detection of pancreatic cancer. Risk assessment and outcomes will be better understood through prospective epidemiologic studies of larger cohorts of patients. Consortiums such as the PRECEDE and CAPS studies will make this possible [3, 4]. Through these consortiums, it is possible to perform further gene discovery to determine inherited causes of FPC. Imaging assessment may undergo an evolution with the use of artificial intelligence. This is a particularly optimistic area of research because clinical application is not as burdensome as the hurdles that are faced by biologic biomarkers. Studies in the early detection of pancreatic cancer in high risk patients will hopefully lead to better assessment in the sporadic form of the disease. Initial data suggests that earlier detection can provide improved 5-year survivor rates [22, 23]. This is an objective that is needed for a disease that has doubled in the general population in the past 20 years, as predicted by the late Gloria Petersen [10]. More alarming is the recent increased incidence in young patients, particularly women [24]. I believe that the challenges before us are surmountable. The remarkable efforts of early pioneers in the field have laid a foundation for the next generation of scientists and doctors. Hopefully, these individuals working together, will bring clinical knowledge forward to place where early detection is no longer an anomaly, but rather a common occurrence.
Data availability
No datasets were generated or analysed during the current study.
References
Evans JP, Burke W, Chen R et al (1995) Familial pancreatic Adenocarcinoma: Association with diabetes and exocrine insufficiency and early molecular diagnosis. J Med Genet 32:330–335
Brentnall TA, Bronner MP, Byrd DR, Haggitt RC, Kimmey MB (1999) Early diagnosis and treatment of pancreatic dysplasia in patients with a family history of pancreatic cancer. Ann Intern Med 131:247–255
Canto MI, Harinck F, Hruban RH et al (2013) International Cancer of the pancreas Screening (CAPS) Consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 62:339–347
Gonda TA, Everett JN, Wallace M et al (2021) Recommendations for a more Organized and Effective Approach to the early detection of pancreatic Cancer from the PRECEDE (pancreatic Cancer Early Detection) Consortium. Gastro 161:1751–1757
MacDermott RP, Kramer P (1973) Adenocarcinoma of the pancreas in four siblings. Gastro 65:137–139
Longnecker DS (1994) Preneoplaastic lesions of the exocrine pancreas. Int J Pancreatol 16:201–204
Pour PM, Sayed S, Sayed G (1982) Hyperplastic, preneoplastic and neoplastic lesions found in 83 human pancreases. Am J Clin Pathol 77:137–152
Cubilla AL, Fitzgerald PJ (1976) Morphological lesions associated with human primary invasive nonendocrine pancreas cancer. Cancer Res 36:2690–2698
Brat DJ, Lillemoe KD, Yeo CJ, Warfield PB, Hruban RH (1998) Progression of pancreatic intraductal neoplasia to infiltrating adenocarcinoma of the pancreas. Am J Surg Pathol 22:163–169
Hruban RH, Rustgi AK, Brentnall TA, Tempero MA, Wright CV, Tuveson DA (2006) Pancreatic cancer in mice and man: the Penn Workshop 2004. Cancer Res 66:14–17
Basturk O, Hong SM, Wood LD et al (2015) A revised classification system and recommendations from the Baltimore Consensus Meeting for neoplastic precursor lesions in the pancreas. Am J Surg Pathol 39:1730–1741
Rulyak SJ, Kimmey MB, Veenstra D, Brentnall TA (2003) Cost-effectiveness of pancreatic Cancer Screening in Familial Pancreatic Cancer kindreds. Gastrointest Endosc 57:23–29
Corral JE, Das A, Bruno MJ, Wallace MB (2019) Cost-effectiveness of pancreatic cancer surveillance in high-risk individuals. Pancreas 48:526–536
Ibrahim IS, Vasen HFA, Wasser MNJM et al (2023) Cost effectiveness of pancreas surveillance: the CDKN2A-p16-Leiden cohort. United Eur Gastroenterol J 11:163–170
Aslanian HR, Lee JH, Canto MI (2020) AGA clinical practice update on pancreas cancer screening in high-risk individual: Expert review. Gastro 159:358–362
Drohan B, Roche CA, Cusack JC, Hughes KS (2012) Hereditary breast and ovarian Cancer and other Hereditary syndromes: using Technology to identify carrier. Ann Surg Onc 19:132–137
Goggins M, Overbeek KA, Brand R et al (2020) Management of patient with increased risk for familial pancreatic cancer: updated recommendation from the International Cancer of the pancreas Screening (CAPS) Consortium. Gut 69:7–17
Rulyak SJ, Lowenfels AB, Maisonneuve P, Brentnall TA (2003) Risk factors for the development of pancreatic cancer in familial pancreatic cancer kindreds. Gastro 124:1291–1299
Matubayashi H, Maeda A, Kanemoto H et al (2011) Risk factors of familial pancreatic cancer in Japan: current smoking and recent onset diabetes. Pancreas 40:974–978
Pepe MS, Feng Z, Janes H, Bossuyt PM, Potter JD (2008) Pivotal evaluation of the Accuracy of a Biomarker used for classification or prediction: standards for Study Design. J Natl Cancer Inst 100:1432–1438
Brand RE, Persson J, Bratlie SO et al (2022) Detection of early-stage pancreatic ductal adenocarcinoma from blood samples: results of a Multiplex Biomarker signature validation study. Clin Transl Gastroenterol 13:e00468
Vasen H, Ibrahim I, Ponce CG et al (2016) Benefit of surveillance for pancreatic cancer in high-risk individuals: outcome of long-term prospective follow-up studies from three European expert centers. J Clin Oncol 34:2010–2019
Canto MI, Kerdsirichairat T, Yeo CJ et al (2020) Surgical outcomes after pancreatic resection of screening-detected lesions in individuals at high risk for developing pancreatic cancer. J Gastrointest Surg 24:1101–1110
Abboud Y, Samaan JS, Oh J et al (2023) Increasing pancreatic Cancer incidence in Young women in the United States: a Population-Based Time-Trend Analysis, 2001–2018. Gastro 164:978–989
Author information
Authors and Affiliations
Contributions
TAB wrote the text and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Brentnall, T.A. Familial pancreatic cancer: a long fruitful journey. Familial Cancer 23, 217–220 (2024). https://doi.org/10.1007/s10689-024-00364-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-024-00364-5