Abstract
The present study was conducted in one of the most densely cultivated area of Al-Qassim region in Kingdom of Saudi Arabia to (i) monitor trace metal (Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, and Zn) contents in surface and subsurface soils, (ii) assess the pollution and potential ecological risk levels of trace metals, and (iii) identify trace metal sources using enrichment factor (EF), correlation matrix, and principal component analysis (PCA). The pollution levels of the analyzed trace metals calculated by the geoaccumulation index (I geo) and contamination factor (C f) suggested that the soils were highly contaminated with Cd and moderately contaminated with Pb. Based on the average values of EF, soil samples were found to present extremely high enrichment for Cd, significant enrichment for Pb, moderate enrichment for Zn, and deficient to minimal enrichment for other trace metals. Among the analyzed trace metals, a very high ecological risk was observed only in the case of Cd at some sampling sites. Meanwhile, other investigated trace metals had a low ecological risk. The results of PCA combined with correlation matrix suggested that Fe, Mn, Zn, Cu, Cr Ni, Cu, and Co represent natural abundance in soil, but Cd, Pb, and Cu are of anthropogenic inputs, mainly due to agrochemical and fertilizer applications. It could be generally concluded that the obtained results can be useful for assessing and conducting a future program for trace metal monitoring in agricultural areas of Saudi Arabia.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trace metals are ubiquitous in the environment and are considered to be one of the main inorganic contaminants, mainly due to their negative impacts on the ecosystem (Bortey-Sam et al. 2015; Xiao et al. 2015; Bai et al. 2015). Natural processes and anthropogenic inputs are two main sources responsible for entering trace metals into ecosystem compartments. Trace metals are of a great threat to the living organisms because of their persistence, nonbiodegradability, toxicity, and abundance in water, air, and soil. Soil acts as a sink of trace metals by receiving them from the air (particulate deposition) and water (industrial wastewater). However, soil contaminated with trace metals is recognized to be a pollution source for groundwater and crops, thereby negatively affecting human health through water consumption and the food chain (Bortey-Sam et al. Xiao et al. 2015; Mico et al. 2006a). Agricultural soils have recently gained attention with respect to trace metal contamination because of agricultural practices, resulting in deteriorating environmental conditions (Kelepertzis 2014; Wu et al. 2016; Toth et al. 2016). In this context, trace metals can enter the agricultural soils through domestic sewage, sewage irrigation, synthetic fertilizers, pesticides, agricultural waste inputs, and manure application (Bai et al. 2011a; Khan et al. 2013). Bai et al. (2011a) reported higher content of trace metals (Cd, Cr, Cu, Ni, and Zn) in the soil samples from the old reclaimed wetland compared to those from a young one. They suggested that the long-term tillage remarkably increased the levels of trace metals in the soils due to domestic sewage and discharge of agrochemicals. In their study, both Fe and Mn were found to be controlled by the natural processes, while other trace metals of Cd, Cr, Cu, Ni, Pb, and Zn mainly originated from anthropogenic inputs. Both the essential trace nutrients (Cu, Zn, Mn, Fe, Ni) and nonessential trace metals (As, Cd, Pb) could be toxic to human health, if present at elevated concentrations (Naveedullah et al. 2013). It is, therefore, of importance to regularly monitor and assess trace metal levels in agricultural soils, to prevent the contamination of food crops and groundwater. More recently, Toth et al. (2016) monitored As, Cd, Cr, Cu, Hg, Pb, Zn, Sb, Co, and Ni in agricultural soils of the European Union and estimated that 6.24% of the agricultural land needs metal remedial action.
Arid and semiarid soils are more susceptible to agricultural practices due to frequent drought conditions and water deficiency. Moreover, the soil of arid regions generally lacks nutrients for plant growth. Therefore, external sources of nutrients in the form of synthetic fertilizers or compost are extensively utilized to increase soil fertility in these relatively infertile soils (Reeve et al. 2012). Agrochemicals are reported to contain trace metals such as As, Cd, Pb, Hg, Cu, Mn, and Zn, as active ingredients or impurities (Zoffoli et al. 2013). Continuous application of the agrochemicals may lead to the accumulation of trace metals in soils, thus resulting in increasing contamination levels of trace metals in groundwater and food crops. Kelepertzis (2014) reported the accumulation of Cu, Zn, Cd, Pb, and As in the Mediterranean agricultural soils receiving large amounts of fertilizers, herbicides, and fungicides. In a study conducted by Atafar et al. (2010) investigating trace metal levels in agricultural soils, Cd, Pb, and As were found to be accumulated in the cultivated soils due to chemical fertilizer application. Among the trace metals, Cd is considered to be a common impurity in phosphate fertilizers and their incorporation into the soil could increase levels and availability of soil Cd, resulting in increasing uptake by plants (Mar et al. 2012). Hence, there is a need to evaluate the cultivated areas of arid regions for trace metal levels, particularly where agrochemicals and irrigation are extensively used to increase crop productivity.
In the Kingdom of Saudi Arabia (KSA), the soils are of poor quality, lacking sufficient amounts of nutrients, organic matter, and moisture. Despite that, rapid and successful agricultural development has occurred in KSA since the 1980s. Therefore, huge amounts of limited water resources and fertilizers have been introduced for agricultural purposes, resulting in the accumulation of organic and inorganic contaminants in agricultural soils (Al-Wabel et al. 2011; Balkhair and Ashraf 2016). Among different regions of the KSA, Al-Qassim is considered to be one of the most productive agricultural areas. Soils of this region are intensively used for agricultural farming to produce more food for the growing population. Recently, Al-Wabel et al. (2016) found high levels of pesticides in groundwater in a densely agricultural area of Al-Qassim region, which were associated with agricultural activities. Nazzal et al. (2015) reported also that the increases in toxic metals in the soils of the central Saudi Arabia were attributed to agricultural activities including the use of chemical and organic fertilizers and pesticides. Therefore, there is a need for the assessment of trace metal levels in such agricultural area susceptible to the intensive use of agrochemicals and fertilizers. In literature, the pollution levels of trace metals and their enrichment in soil have been assessed using different indices such as the geoaccumulation index, contamination factor, and enrichment factor (Bai et al. 2010, 2011b, 2015; Lu et al. 2016). Ecological risk caused by trace metals has been also evaluated based on the potential ecological risk index to assess the quality of the soil environment (Bortey-Sam et al. 2015). Additionally, multivariate analysis (i.e., principal component analysis) and correlation matrix are considered to be an effective tool for the identification of the source of trace metals in the soil (Bai et al. 2011a, b; Lu et al. 2016). However, limited information is available on the pollution levels, enrichment, and sources of trace elements in agricultural soils of KSA. Therefore, studies on the assessment of trace metal levels, ecological risk, and sources in agricultural areas of KSA using pollution and ecological indices as well as principal component analysis, which are recognized to be useful in assessment and monitoring studies, are still needed. Our hypothesis, in this context, is that extensive agricultural activities in this region may lead to trace metal accumulation, and there could be substantial pollution levels and ecological risk for trace metals in these soils. The present study is, therefore, conducted in a densely cultivated agricultural area of Al-Qassim region in KSA to (i) monitor trace metal contents in surface and subsurface soils, (ii) assess the pollution and potential ecological risk levels of trace metals, and (iii) identify trace metal sources (natural or anthropogenic) using enrichment factor, lithogenic and anthropogenic contributions, and principal component analysis.
Materials and methods
Study area and sample collection
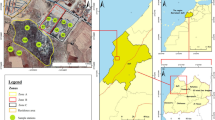
The study area of Al-Qassim is located in central Saudi Arabia, approximately 400 km northwest of the capital of the country (Riyadh). The Al-Qassim region occupied an area of 65,000 km−1 and its land height is about 600–750 m above sea level, with gliding from west to east in general. The weather in Al-Qassim is typical desert climate characterized by cold rainy winters, hot summers, and low humidity (around 20%). The temperature of the Al-Qassim region ranges from 36 to 41 °C in summer and drops to an average of 11 °C in winter. Average rainfall in the area is 100 mm per annum. Soil samples of the study area were collected from the surface (0–30 cm) and subsurface (30–60 cm) of 33 soil profiles (Fig. 1).
Soil analyses
The collected soil samples were air-dried and sieved <2 mm for the following physicochemical analyses. The particle size distribution of soil samples was determined by the hydrometer method (Gee and Bauder 1994). Calcium carbonate content was determined using a calcimeter. The soil organic matter content was determined following the oxidation by K2Cr2O7 (Nelson and Sommers 1996). The pH and EC of soil samples were measured electrometrically by a digital pH meter (HI 2210, HANNA) and EC meter (Test kit Model 1500_20 Cole and Parmer) in 1:1 soil-water suspensions. Soluble cations (Ca2+, Na+, K+, and Mg2+) and anions (Cl−, HCO3 −, SO4 2−, and CO3 2−) were measured according to standard procedures (Sparks 1996). The soluble Ca2+ and Mg2+ were determined by versenate titration method (EDTA), and the soluble Na+ and K+ were determined by using a flame photometer (Corning 400). The soluble CO3 2− and HCO3 − were determined by titration against sulfuric acid (H2SO4), whereas soluble Cl− was determined by titration against silver nitrate (AgNO3) according to standard methods (Sparks 1996). The soluble sulfate (SO4 2−) concentrations were determined by using the turbidity method (Tabatabai 1996). The total contents of trace metals (Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, and Zn) in soil samples were determined following the USEPA method 3051 using a microwave-assisted digestion unit (MARS) (USEPA 1994). Briefly, 0.5 g of each soil sample was digested using 10 mL of concentrated nitric acid for 10 min in the microwave digestion unit. The digestate was filtered, diluted, and analyzed using inductively coupled plasma optical emission spectrometer (ICP-OES, Perkin Elmer Optima 4300 DV, USA) for trace metal concentrations.
Assessment of pollution levels and enrichment in soil
To determine the heavy metal pollution levels and enrichment in the cultivated study area of Al-Qassim region, the following different pollution indices for Cd, Co, Cr, Cu, Ni, Mn, Pb, and Zn were calculated: the geoaccumulation index (I geo), contaminant factor (C f), degree of contamination (DC), and enrichment factor (EF).
Geoaccumulation index
The I geo has been widely used to assess the soil contamination with heavy metals. It was calculated as follows:
where C n is the metal content in the investigated surface and subsurface soil samples, B n is the geochemical background value of trace metal in average shale (Turekian and Wedepohl 1961), and 1.5 is the factor to minimize the effect of possible lithogenic variations in the soil. The following criteria based on the values of I geo were applied to assess the metal contamination level in the soil:
- I geo ≤ 0:
-
uncontaminated soil
- 0 < I geo < 1:
-
uncontaminated to moderately contaminated soil
- 1 < I geo < 2:
-
moderately contaminated soil
- 2 < I geo < 3:
-
moderately to strongly contaminated soil
- 3 < I geo < 4:
-
strongly contaminated soil
- 4 < I geo < 5:
-
strongly to extremely contaminated soil
- I geo ≥ 5:
-
extremely contaminated soil
Contamination factor and degree of contamination
The C f is used often to assess the contamination level of heavy metals in soil. It was calculated as the ratio between metal concentration in the investigated soil (C n) and background or normal concentration of that metal (B n) (Muller 1979):
In this study, worldwide average concentrations of trace metals in noncontaminated soils are applied to normal trace metal concentration (B n) (Kabata-Pendias 2011). The following criteria were used to evaluate the contamination levels in soils based on the C f:
- C f < 1:
-
low degree of metal contamination
- 1 ≤ C f ≥ 3:
-
moderate degree of metal contamination
- 3 ≤ C f ≥ 6:
-
considerable degree of metal contamination
- C f > 6:
-
very high degree of metal contamination
In addition, DC was calculated as the sum of the C f for the identified trace metals as follows (Hakanson 1980):
The following criteria were used to evaluate the degree of contamination:
- DC < 8:
-
low degree of contamination
- 8 ≤ DC < 16:
-
moderate degree of contamination
- 16 ≤ DC < 32:
-
considerable degree of contamination
- DC > 32:
-
very high degree of contamination
Enrichment factor
The EF provides information about the normalized metal concentration in soil above the uncontaminated level. For normalization, a reference metal concentration is used. The most widely used reference element for this purpose is Fe, because of its natural abundance and less interaction with other heavy metals. The EF was calculated by the following equation (Sutherland 2000):
where C n and C Fe are the respective metal and Fe concentrations in the investigated soil samples, and Cnref and Feref are the respective metal and Fe contents in the earth’s crust. The following criteria were employed to determine the metal enrichment in soil based on the EF values:
- EF < 2:
-
deficiently to minimal enrichment in soil
- 2 ≤ EF < 5:
-
moderate enrichment in soil
- 5 ≤ EF < 20:
-
significant enrichment in soil
- 20 ≤ EF < 40:
-
very high enrichment in soil
- EF > 40:
-
extremely high enrichment in soil
Lithogenic and anthropogenic contributions
The lithogenic and anthropogenic contributions for each investigated heavy metal were also calculated in this study, as suggested by Hernandez et al. (2003). To calculate the metal content of lithogenic source, the following equation was used:
where M L: metal content from a lithogenic source; Fes: the total Fe content in the soil samples; and [M ref/Feref]: the ratio of metal concentration and Fe in the earth’s crust. Meanwhile, the metal content derived from anthropogenic source was calculated using the following equation:
where M A: the anthropogenic content of trace metal; M T: the total metal content in the soil samples; and M L: trace metal content from the lithogenic source.
Ecological risk assessment
The potential ecological risk index is used to assess the quality of the soil environment. The potential ecological risk index was calculated by the following equations (Hakanson 1980; Gan et al. 2000):
where Ei is a single ecological index, RI is considered to be the potential ecological risk index, C f is the contamination factor, C s i is the concentration of heavy metal in the investigated soil samples, and B n is the respective trace metal in the worldwide average soil. Ti is the toxic response factor (Cd = 5; Pb, Cu, and Ni = 5; Cr = 2; Zn = 1). The single ecological index is classified into five categories (Gan et al. 2000; Effendi et al. 2016): Ei of 30 (low risk), Ei of 30–50 (moderate risk), Ei of 50–100 (considerable risk), Ei of 100–150 (high risk), and Ei of >150 (very high risk). The integrated potential ecological risk index (RI) is classified into five categories, namely RI of <100 (low risk), RI of 100–150 (moderate risk), RI of 150–200 (considerable risk), RI of 200–300 (very high risk), and RI of >300 (disastrous risk).
Quality assurance and quality control
The collected soil samples for determining trace metal content were digested and analyzed with four replicates. For quality control, the reference soil samples (Till-1, Till-2, Till-3, and Till-4) were used for analyzing trace metals. To ensure the analytical performance of the ICP-OES, a standard of known concentration was run after each set of 10 samples. The imprecision of the method was computed as relative standard deviation (RSD) targeted at ≤5%. The detection limits of the ICP-OES were <0.1 μg L−1 for Cd and Fe and 1 μg L−1 for Co, Cu, Cr, Pb, Mn, Ni, and Zn. A multielement quality control standard 21 (PerkinElmer, USA) was used for quality control check of the equipment.
Statistical analysis and data mapping
The mean, minimum, maximum, and standard deviation (±SD) of the analytical data were computed using Microsoft Excel. Pearson correlation coefficients between the analyzed trace metals and principal component analysis (PCA) were achieved using statistical software Statistica for Windows (StatSoft 1992). The obtained data from different indices, namely C f, DC, Ei, and RI for the analyzed potentially toxic trace metals in surface and subsurface soil samples of the study area at Al-Qassim region were mapped using a MATLAB Software, which was used to interpolate scattered data. A built-in function (grid data) fits a surface of the form v = f (x, y) to the scattered data in the vectors (x, y, v). The grid data function interpolates the surface at the query points specified by (x1, y2) and returns the interpolated values, v1. The surface always passes through the data points defined by x and y.
Results and discussion
Soil physicochemical properties of the study area
Table 1 presents descriptive statistical analysis results of soil texture, CaCO3 content, organic matter content, EC, pH, and soluble ionic concentrations in the surface (0–30 cm) and subsurface (30–60 cm) soil samples at Al-Qassim region. Most of the soil samples had higher sand content than silt and clay contents with an average value of 71.4 and 69.8 for sand, 15.6 and 15.9 for silt, and 13.0 and 14.4 for clay in surface and subsurface soils, respectively. Soil EC was between 1.0 and 104.7 dS m−1 with an average value of 10.1 dS m−1 in the surface soil samples and between 0.50 and 43.4 dS m−1 with an average of 6.40 dS m−1 in subsurface soil. All the surface soil samples indicated alkaline conditions with pH values ranging from 7.5 to 8.1 in surface soil and from 7.6 to 8.5, except the surface soil samples collected from one profile having a pH value of 6.7. The high pH of the soil samples is mainly because of neutralization of the soil acidity by a high content of calcium carbonate. Soil CaCO3 content was between 11.2 and 454 g kg−1 with an average value of 91.3 g kg−1 in the surface soil samples and between 7.84 and 340 g kg−1 with an average of 103 g kg−1 in subsurface soil. Soil organic matter content ranged from 0.57 to 13.7 g kg−1 with an average value of 3.61 g kg−1 in the surface soil samples and from 0.28 to 6.40 g kg−1 with an average of 1.96 g kg−1 in subsurface soil. Among soluble cations, Na+ was dominant with mean values of 105.7 and 50.3 mmol L−1 in subsurface soil followed by Ca2+ with mean values of 7.5 and 7.3 mmol L−1 and Mg2+ with mean values of 5.65 and 4.7 mmol L−1 in surface and subsurface soil samples, respectively. The corresponding dominant anions were Cl− with mean of 103.1 and 54.5 mmol L−1 and SO4 2− with mean of 11.5 and 8.45 mmol L−1 in the surface and subsurface soil samples, respectively The mean values of Na+ (50.3 mmol L−1), K+ (1.3 mmol L−1), and Cl− (54.5 mmol L−1) were almost two times less than those observed in the surface soils. This indicated that these ions may have accumulated on the top soil as a consequence of brackish water used for irrigation purpose in the region (Al-Salamah and Nassar 2009). These high soluble cations and anions contributed to high EC values in soils of Al-Qassim region. The average EC values in the surface (10.1 dS m−1) and subsurface soils (6.4 dS m−1) indicated that the soils of the studied region were moderate to strongly saline (Wentz 2001). The soil ionic properties and pH and EC values indicated that soils of Al-Qassim are typical of an Arab region representing arid, saline, calcareous, and alkaline soils.
Trace metal contents of surface and subsurface soil samples
The statistical summary of the total trace metal (Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, and Zn) contents in surface and subsurface soil samples of the study area at Al-Qassim region is given in Table 2. The average total contents of Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, and Zn in the surface soil samples were 1.65, 1.34, 17.4, 5.84, 5529, 125, 2.61, 26.9, and 21.1 mg kg−1, respectively. Likewise, the average total contents of Cd, Co, Cr, Cu, Fe, Mn, Ni, Pb, and Zn in the subsurface soil samples were 1.32, 1.86, 18.1, 7.36, 5945, 151, 2.84, 20.7, and 14.3 mg kg−1, respectively. These results indicated that the surface soil contained high average contents of Cd, Pb, and Zn, whereas Co, Cr, Cu, Fe, Mn, and Ni contents were slightly higher in the subsurface soils. It was observed that the contents of Zn followed by Ni showed higher coefficient of variation and positive skewness than the other analyzed trace metals in surface soils, suggesting that there is a strong skewness factor for these heavy metals. However, among all identified metals in surface and subsurface soil samples, Cu content in subsurface soil samples showed the highest coefficient of variation and positive skewness.
It should be mentioned that there are no quality guidelines for soil trace metals in Saudi Arabia. Therefore, the trace metal contents were compared with other guidelines including the common range in the earth crust, average shale values, worldwide average soil, and the Dutch optimum and Act target values, as shown in Table 2. The results indicated that all analyzed heavy metals (except for Cd) in surface and subsurface soil samples at Al-Qassim region were within the corresponding values of common range in the earth crust reported by Lindsay (1979). The maximum and average values of Cd contents accounted for 5.40 and 1.65 mg kg−1 in surface soil and for 2.70 and 1.32 mg kg−1, respectively, which were higher than the values of the common range, average shale value, and worldwide average soil. The results also showed that the maximum contents of Pb (in surface and subsurface soils) and Zn (in the surface soil) were higher than those of average shale value and worldwide average soil. Similarly, the maximum content of these three metals (Cd, Pb, and Zn) was higher than the Dutch optimum value but lower than the Dutch Act target value.
The results indicated also that the contents of Cd and Pb in surface soil samples from the study area were higher when compared with other studies conducted in different regions of the world. For instance, the contents of Cd and Pb in the present study were higher than the reported average values of 0.38 and 19.6 mg kg−1, respectively, for agricultural soils of Mediterranean semiarid area of Spain (Mico et al. 2006a). Bao et al. (2014) reported 0.15 mg kg−1 (Cd) and 26.49 mg kg−1 (Pb) in agricultural soils of Beijing, China, irrigated with sewage water, which were lower than the corresponding contents obtained in the current study. In a study conducted by Abbaslou et al. (2014) investigating trace element contents and background values in the arid soils of Hormozgan Province of southern Iran, the Cd (0.28 mg kg−1) and Pb (22.4 mg kg−1) contents were also lower than those of the current study.
Identification of trace metal pollution and ecological risk levels
To identify trace metal pollution levels, various methods have been proposed. In this study, various indices that include the I geo, C f, and DC were applied to identify the levels of metal pollution. In addition, the potential ecological risk for trace metals was calculated to identify the ecological risk levels of the analyzed trace metals. The minimum, maximum, and average values of I geo for different trace metals in the surface and subsurface soils at Al-Qassim region are shown in Fig. 2. Based on the I geo, the contamination of the investigated surface and subsurface soil samples by Cd is characterized by the following criteria: uncontaminated to moderately contaminated (I geo < 1), strongly contaminated (3 < I geo < 4), and moderately to strongly contaminated (2 < I geo < 3) based on minimum, maximum, and average values, respectively. The maximum values of I geo indicated that the surface soil of profile 11 is strongly contaminated with Pb (I geo = 3.48). However, its average I geo values were 0.72 and 0.55 in surface and subsurface soils, respectively, suggesting uncontaminated to moderately contaminated soil. The values of I geo for most of the other trace metals of the most investigated soil samples are <0 indicating that the soils are not contaminated with Co, Cr, Cu, Mn, Ni, and Zn. However, only in the surface soil of profile 2 and subsurface soil of profile 3, respectively, that Zn and Cu showed I geo values of 1.37 and 1.83, suggesting moderately contaminated soil. The average values of C f of an individual metal in the surface and subsurface soil samples ranged from 0.09 to 4.04 and from 0.10 to 3.23, respectively (Table 3). Overall, the highest C f values were pronounced for Cd. The maximum and average C f values for Cd accounted for 13.2 and 4.04 in surface samples and for 6.59 and 3.23 in subsurface soil samples, respectively. This result indicates that the soils of the study area have a very high degree of Cd contamination based on maximum C f values or a considerable degree of metal contamination based on average C f values, especially in the surface soil samples. The other trace metal of concern from a contamination point of view was Pb with an average value of 1.90 and 1.39 for surface and subsurface soil samples, respectively, indicating that the soil samples of the study area are moderately contaminated with Pb. However, the average C f values of all other trace metals (Co, Cr, Cu, Mn, Ni, and Zn) are less than 1, suggesting that the soils are not contaminated with these trace metals. Among these six metals, however, Zn and Cu showed C f value of 2.34 and 2.75 only in the surface soil of profile 2 and subsurface soil of profile 3, respectively, indicating moderately contaminated with these two metals. It is clear that condensed agricultural soils reveal some degree of trace metal contamination. For determining DC, it was calculated as the sum of the contamination factor for the eight analyzed trace elements (Cd, Pb, Zn, Cu, Mn, Ni, Co, and Cr). Table 3 shows the DC in surface and subsurface soils of the study area. The range of DC values was 1.4–16.6 and 1.74–13.1 in the surface and subsurface soil samples, respectively, indicating a low to moderate degree of contamination in a study area at Al-Qassim region. Among all metals, the C f values for Cd showed the highest contribution to the degree of contamination with an average contribution of 59.5% and 57.2% in surface and subsurface soil samples, respectively (Fig. 3). While the average contributions of other trace metals to the degree of contamination were low or very low, accounting for 16.2 and 15.7% (Pb), 5.10 and 4.25% (Zn), 1.85 and 2.15% (Cu), 2.50 and 2.78% (Ni), 5.54 and 6.90% (Mn), 3.19 and 4.08% (Co), and 6.05 and 6.94% (Cr) in surface and subsurface soil samples, respectively.
The ecological risk potential was also assessed in this study to identify its level of the analyzed trace metals of Cd, Pb, Cu, Cr, Ni, and Zn. Table 4 shows the values of single ecological risk for each analyzed trace metal as well as the values of the integrated potential ecological risk of the study area. Based on the obtained average values of single ecological risk (Ei), the order of trace metals in surface and subsurface soils, respectively, were as follows: Cd (121 and 96.9) > Pb (4.98 and 3.85) > Cu (0.75 and 0.95) > Cr (0.58 and 0.61) > Ni (0.45 and 0.49) > Zn (0.30 and 0.20). In our study, the values of single ecological risk index revealed that about 39% (at 0–30 cm depth) and 30% (at 30–60 cm depth) of soil samples for Cd can be categorized to very high ecological risk with maximum Ei value of 395 in surface soil and with maximum Ei value of 198 in subsurface soil. However, other investigated trace metals had low-risk criteria. On the other hand, the average RI in surface and subsurface soils was categorized to moderate risk criteria. Based on the maximum value of RI, however, the disastrous risk (407) and very high risk (206) criteria existed in the surface and subsurface soils, respectively. Generally, the average contribution of Cd to the integrated potential ecological risk was very high and amounted to 90.6 and 90.5% in surface and subsurface soil samples, respectively (Fig. 3), while the average contributions of other trace metals to the integrated ecological risk were very low and accounted for 5.37 and 5.43% (Pb), 0.43 and 0.36% (Zn), 0.49 and 0.61% (Cu), 2.00 and 1.96% (Ni), and 1.07 and 1.16% (Cr) in surface and subsurface soil samples, respectively.
Figures 4 and 5 show the contour maps of C f and Ei for Cd and Pb as well as the DC and the RI in the agricultural area of Al-Qassim region. The results of contour maps showed that the investigated agricultural sites were highly impacted by Cd contamination, resulting in higher contamination degree and ecological risk in the study area of Al-Qassim region. Based on the C f for Cd, the contour maps indicated that the investigated surface and subsurface soil samples are characterized by low, moderate, considerable, or very high degree of Cd contamination. However, the investigated surface soil samples are characterized by a low, moderate, or considerable degree of Pb contamination. The contour maps of single ecological risk (Ei) for Cd indicated that the soil samples are classified into five categories, namely low, moderate, considerable, high, or very high risk. Meanwhile, all the soil samples of the study area have a low ecological risk for Pb. Generally, the contour maps suggested that higher levels of contamination and ecological risk for Cd and Pb are mainly distributed in the southern parts of Al-Qassim region. It was observed that there are some hot spots with high contamination and ecological risk levels for Cd at the southern parts of Al-Qassim region, suggesting a considerable degree of contamination or an ecological disastrous risk. Therefore, the agricultural soils located at a southern part of Al-Qassim region should be monitored on a regular basis for potentially toxic metals in the vegetation and agricultural crops. However, the lowest degree of contamination and ecological risk levels were observed in the northern parts of Al-Qassim region, mainly because the potentially toxic metal (especially for Cd) concentrations were not detected or low in these sites indicating lower anthropogenic activities. The variations in the degree of contamination with potentially toxic metals among the investigated sites of agricultural soils could be explained by variations in agricultural activities used by farmers that are responsible for introducing the toxic metals into the soils such as the type of agrochemicals (fertilizers, pesticides, and fungicides) applied, application rates, and application frequency. To our knowledge, the intensive agriculture and farming practices including long-term use of agrochemicals were present at the farms of the southern parts of the study area, which are older than those located at the northern parts. This can confirm our finding that the highest degree of contamination and ecological risk levels were found in the southern parts of Al-Qassim region.
Trace metal enrichment and sources
The EF has been proposed as a useful method for measuring heavy metal sources (Sutherland 2000; Hu et al. 2013; Bai et al. 2015). Table 5 presents a descriptive statistical analysis of trace metal EF. It was observed that the average EF values of an individual trace metal in the surface and subsurface soil samples, respectively, were decreased in the following order: 84 and 72.2 (Cd) > 16.5 and 13.2 (Pb) > 2.43 and 1.48 (Zn) > 1.29 and 1.59 (Mn) > 0.81 and 0.82 (Cr) > 0.79 and 0.71 (Cu) > 0.28 and 0.32 (Co) > 0.21 and 0.20 (Ni). Based on the average and maximum values of EF, soil samples were found to present extremely high enrichment for Cd in surface and subsurface soil samples. The maximum values of EF have also shown extremely high enrichment for Pb in surface soil and very high enrichment in subsurface soil. Meanwhile, its average EF values showed significant Pb enrichment in soil. On the contrary, the average EF values for other trace metals (Zn, Co, Cr, Cu, Ni, Mn, and Fe) were <2, indicating deficient to minimal enrichment in soil. It has been previously reported that the soil samples with EF values less than 2 indicate that heavy metals are mainly governed by the soil parent materials and natural weathering process (Ye et al. 2011), while the soil samples with EF values >2 indicate that heavy metal could be impacted by anthropogenic activity. In this study, the percentage of soil samples with EF> and <2 were computed, as shown in Fig. 6. The results indicated that around 97.0 and 100% of soil samples had EF >2 for Cd and Pb, respectively. Thus, the trace metals of Cd and Pb with much higher EF in surface and subsurface soil samples may be a consequence of human activities and anthropogenic inputs. Meanwhile, the other analyzed trace metals with much lower EF values (EF < 2 in most collected soil samples) may be a result of lithogenic origin. The contribution percentage of lithogenic and anthropogenic sources for each trace metal was also calculated and presented in Fig. 7. The average values of anthropogenic contribution of the analyzed trace metals in surface and subsurface soil samples from the study area amounted to 94.9 and 94.5% for Cd, 1.45 and 3.10% for Cr, 29.3 and 27.6% for Cu, 19.2 and 22.4% for Mn, 90.8 and 89.3% for Pb, and 34.6 and 27.2% for Zn, respectively. In addition, the anthropogenic and lithogenic contribution of Co and Ni in all soil samples of the study area amounted to 0.0 and 100%, respectively. The obtained data from the calculated lithogenic and anthropogenic contribution were in line with those of the enrichment factor, indicating that the soil samples have a much higher significant anthropogenic contribution for Cd and Pb than the other trace metals. The soils of the study area at Al-Qassim are intensively used for agricultural farming, and the use of inorganic fertilizers and organic farming are commonly practiced (El-Mergawi and Al-Redhaiman 2010). Therefore, agrochemicals including inorganic fertilizers and pesticides as well as manure application may be an anthropogenic source for Cd and Pb inputs. Additionally, Cd incorporated into agricultural soils and edible parts of plants could be through long-term application of sewage sludge and irrigation with wastewater (Gupta et al. 2008; Mapanda et al. 2005; Balkhair and Ashraf 2016). Similar to our findings, Nazzal et al. (2015) found that the contents of Cd, Hg, and Pb in the soils of central Saudi Arabia were higher than the average values for normal soils and they attributed the increases in the levels of trace metals in soils to agricultural activities including the use of chemical and organic fertilizers and pesticides. They also reported that other anthropogenic inputs such as municipal waste, quarries, local industries, and runoff might contribute to the increase in the levels of trace metals in the soil. Several other researchers reported that the main source of Cd accumulation in agriculture soils is manures and phosphate fertilizers (Xu et al. 2015; Ju et al. 2007; Atafar et al. 2010). Phosphate fertilizers have been reported to contain Cd and Pb as trace metal impurities (Roberts 2014), thus being responsible for substantial pollution levels for Cd and Pb in agriculture soils. In a study conducted by Atafar et al. (2010) investigating trace metal levels in agricultural soils, Cd, Pb, and As were accumulated in the soils due to chemical fertilizer applications. Oyedele et al. (2006) found also that potentially toxic metals (Cd, Pb, and Hg) in soils significantly increased following phosphate fertilizer application. Previous results have shown that total soil Cd, hydrochloric acid (HCl) extractable Cd, water extractable Cd, and Cd content of plants increased due to the application of superphosphate (Mar et al. 2012). In a study conducted by He and Singh (1994a, b) investigating Cd uptake by crops from phosphate fertilizers, applying Cd-containing NPK fertilizers caused significant increases in soil Cd availability, resulting in increasing Cd contents in crops. In our recent work, pesticide residues were detected in groundwater samples in most investigated locations at the study area of Al-Qassim region and their concentrations were higher than the maximum permissible levels (Al-Wabel et al. 2016). This finding suggested that extreme levels of pesticides were associated with agricultural activities and intensive use of agrochemicals in the study area of Al-Qassim region. In this context, long-term application of agrochemicals (fertilizers, pesticides, and fungicides) could be a reasonable reason for increasing trace metal levels in agricultural soils (Kelepertzis 2014; Atafar et al. 2010; Bai et al. 2011a).
Both the correlation matrix and PCA are considered to be an effective tool for the identification of trace metal sources in the soil (Almasoud et al. 2015; Bai et al. 2011a, b; Lu et al. 2016). Therefore, in the current study, the correlation matrix and PCA were applied to the total contents of the analyzed trace metals to predict and distinguish trace metal sources. As shown in Table 6, there were significant positive correlations among the analyzed trace metals in surface and subsurface soils. Fe showed a positive significant correlation with Co (r = 0.67), Cr (r = 0.76), Mn (r = 0.72), and Ni (r = 0.62) in surface soil and with Co (r = 0.95), Cr (r = 0.71), Cu (r = 0.76), Mn (r = 0.39), Ni (r = 0.59), and Zn (r = 0.89) in subsurface soil. Mn is also significantly correlated with Co (r = 0.63), Cr (r = 0.67), Ni (r = 0.49), and Zn (r = 0.40) in surface soil and with Co (r = 0.45) and Zn (r = 0.40) in subsurface soil. Significant correlations were also observed among Cd, Pb, and Cu in surface and subsurface soils. Moreover, the data showed that a positive correlation exists between Cd and Zn only in the surface soil. It has been previously reported that the significant positive correlation among trace metals indicates a similar contamination source (Nazzal et al. 2015; Lu et al. 2016; Bai et al. 2015). Therefore, in the current study, the obtained significant positive correlations between the trace metals suggest that the analyzed trace metals could be derived from different sources. However, a single correlation matrix should be applied in combination with other analysis tools such as multivariate analysis (e.g., PCA) to be an effective assessment tool for predicting and identifying trace metal sources (Lu et al. 2016). Therefore, in the current study, PCA was performed on the analyzed trace metals.
The results of PCA showed that five principal components were extracted, which amounted to 91.4 and 97.3% of the total variance in surface and subsurface soil samples, respectively (Table 7). Factor 1 was heavily and positively loaded with Fe, Mn, Co, and Cr in surface soil and with Fe, Co, Cu, and Zn in subsurface soil, which accounted for 39.97 and 50.97% of the total variance, respectively. Factor 2 was dominated by Pb in surface soil with 29.92% of the total variance and by Cd and Pb in subsurface soil with 27.47% of the total variance. The other remaining factors, in surface soil, were dominated by Zn (factor 3 with 10.16% of the total variance), by Cd and Cu (factor 4 with 6.44% of the total variance), and by Ni (factor 5 with 4.39% of the total variance). Meanwhile, in subsurface soil, factors 3, 4, and 5 were dominated by Mn, Cr, and Ni with 10.13, 5.63, and 3.10% of the total variance, respectively. The 3D plot of the PCA suggested that the analyzed trace metals originate from different sources (Fig. 8). The results of the 3D plot of the PCA showed high loading of most analyzed trace metals including Fe, Mn, Ni, Co, and Cr at 0–30 cm depth and Fe, Zn, Ni, Cu, Co, and Cr at 30–60 cm depth. The presence of these trace metals in the same component and their significant positive correlations with Fe and Mn suggest that these trace metals represent natural abundance in soil, as indicated by EF values less than 2 in most soil samples. It was also observed that there was a loading between Cd, Pb, and Cu in surface soil and between Cd and Pb in subsurface soil. As shown in Table 6, significant correlations among Cd, Pb, and Cu further indicate that these trace metals had a common origin, which is consistent with the result of the principal component analysis. Among these three metals, Cd and Pb have shown to have EF >2, indicating that Cd and Pb might have a similar source and a consequence of anthropogenic inputs (e.g., intensive agrochemical and fertilizer application). Though the EF values of Cu in most soil samples were less than 2 suggesting its possible lithogenic origin, it was associated with Cd and Pb in the same component of PCA that is of anthropogenic source. Therefore, this finding speculates that Cu in soil may possibly represent a mixed source (natural and anthropogenic). The results of the 3D plot of the PCA showed that Zn stands alone with significant positive correlation with Cd in subsurface soil, suggesting another different source. Cd and Pb could be incorporated into the soil through P fertilizers. Additionally, the anthropogenic portion of Cu may be incorporated into the soil with Cu-based agrochemicals (e.g., fungicidal sprays and copper sulfate) related to specific agronomic practices (Bai et al. 2011a). Similar to our findings, Mico et al. (2006b) found that Co, Cr, Fe, Mn, Ni, and Zn were grouped into the first component and associated with parent rocks (lithogenic component), whereas Cd, Cu, and Pb were related with anthropogenic components. They also reported that Cd contents were affected by anthropogenic activities such as phosphate fertilizer application to agricultural soils. Kelepertzis (2014) reported that the long-term application of fertilizers, pesticides, and fungicides resulted in accumulation of Cu, Zn, Cd, Pb, and As in agricultural soils, while Ni, Cr, Co, and Fe could be controlled by lithogenic effects. In a study conducted by Lu et al. (2016) to identify the sources of trace metals in marsh soils of a Tamarix chinensis wetland, As, Cd, Pb, and Zn were found to be grouped into the first principal component of PCA, suggesting that these trace metals might originate from agrochemicals, fertilizers, and oil. They also observed that Pb and Zn were significantly correlated with soil P and N, implying that these two metals are from similar sources (e.g., agricultural runoff and intensive fertilizer application). Bai et al. (2011a) found also that long-term tillage remarkably increased the levels of trace metals such as Cd, Cr, Cu, Ni, Pb, and Zn, which were associated with anthropogenic inputs of domestic sewage and discharge of agrochemicals. In addition to the soil contamination caused by intensive application of agrochemicals and fertilizers, atmospheric deposition of heavy traffic is considered to be the main pollution source for Pb in agricultural soils near the roadside (Azeez et al. 2014). Due to rapid urbanization and increased development in Saudi Arabia, deposition of contaminants emitted from traffic and industries may be considered to be the main sources of soil pollution. It could be concluded that the principal component analysis together with correlation matrix and EF may be applied as useful tools for identifying trace metal sources in agricultural soils. Our findings suggest generally that the potentially toxic trace metals of Cd and Pb in soil samples of the study area mainly originated from anthropogenic activity, mainly due to the continuous application of agrochemicals including chemical fertilizers, pesticides, and organic manure applications resulting in the high accumulation of potentially toxic metals in the condensed agricultural soils of Al-Qassim region. Thus, this may lead to toxic effects of Cd and Pb on vegetation and food crops of the study area. Therefore, monitoring of trace metals in vegetation and food crops of Al-Qassim region should be taken into account.
Conclusions
The cultivated area of Al-Qassim region in the Kingdom of Saudi Arabia was investigated to assess the pollution and potential ecological risk levels of trace metals using pollution and ecological risk indices and to identify trace metal sources (natural or anthropogenic) using enrichment factor and principal component analysis. The results indicated that the soils were highly contaminated with Cd and moderately contaminated with Pb. Moreover, the ecological risk index revealed that Cd was categorized to very high ecological risk and its contribution to the integrated potential ecological risk was very high accounting for 90.6 and 90.5% in surface and subsurface soil samples, respectively, while the contribution of other trace metals to the integrated ecological risk was very low (0.35–5.27%). Based on the principal component analysis and correlation matrix, Cd, Pb, and Cu represent the anthropogenic inputs and other trace metals represent natural abundance in soil. Therefore, substantial information of the current study was extracted from trace metal pollution levels, sources, and ecological risk levels in a cultivated area of KSA by available tools of pollution and ecological risk indices as well as multivariate analysis. So future studies should focus on monitoring and assessing potentially toxic metal levels in all agricultural areas of KSA.
References
Abbaslou, H., Martin, F., Abtahi, A., & Moore, F. (2014). Trace element concentrations and background values in the arid soils of Hormozgan Province of southern Iran. Archives of Agronomy and Soil Science, 60, 1125–1143.
Almasoud, F. I., Usman, A. R., & Al-Farraj, A. S. (2015). Heavy metals in the soils of the Arabian Gulf coast affected by industrial activities: analysis and assessment using enrichment factor and multivariate analysis. Arabian Journal of Geosciences, 8, 1691–1703.
Al-Salamah, I. S., & Nassar, I. N. (2009). Trends in drinking water quality for some wells in Qassim, Saudi Arabia, 1997–2009. Journal of Applied Sciences, 9, 3555–3561.
Al-Wabel, M. I., El-Saeid, M. H., Al-Rurki, A. M., & Abdel-Nasser, G. (2011). Monitoring of pesticide residues in Saudi Arabia agricultural soils. Research Journal of Environmental Sciences, 5, 269–278.
Al-Wabel, M., El-Saeid, M. H., El-Naggar, A. H., Al-Romian, A., Osman, K., Elnazi, K., & Sallam, A. S. (2016). Spatial distribution of pesticide residues in the groundwater of a condensed agricultural area. Arabian Journal of Geosciences. doi:10.1007/s12517-015-2122-y.
Atafar, Z., Mesdaghinia, A., Nouri, J., Homaee, M., Yunesian, M., Ahmadimoghaddam, M., & Mahvi, A. H. (2010). Effect of fertilizer application on soil heavy metal concentration. Environmental Monitoring and Assessment, 160, 83–89.
Azeez, J. O., Mesele, S. A., Sarumi, B. O., Ogundele, J. A., Uponi, A. O., & Hassan, A. O. (2014). Soil metal pollution as a function of traffic density and distance from road in emerging cities: a case study of Abeokuta, southwestern Nigeria. Archives of Agronomy and Soil Science, 60, 275–295.
Bai, J., Xiao, R., Cui, B., Zhang, K., Wang, Q., Liu, X., Gao, H., & Huang, L. (2011a). Assessment of heavy metal pollution in wetland soils from the young and old reclaimed regions in the Pearl River estuary, South China. Environmental Pollution, 159, 817–824.
Bai, J., Xiao, R., Gong, A., Gao, H., & Huang, L. (2011b). Assessment of heavy metal contamination of surface soils from typical paddy terrace wetlands on the Yunnan Plateau of China. Physics and Chemistry of the Earth, 36, 447–450.
Bai, J., Yang, Z., Cui, B., Gao, H., & Ding, Q. (2010). Some heavy metals distribution in wetland soils under different land use types along a typical plateau lake, China. Soil & Tillage Research, 106, 344–348.
Bai, J., Zhao, Q., Lu, Q., Wanga, J., & Reddy, K. R. (2015). Effects of freshwater input on trace element pollution in salt marsh soils of a typical coastal estuary, China. Journal of Hydrology, 520, 186–192.
Balkhair, K. S., & Ashraf, M. A. (2016). Field accumulation risks of heavy metals in soil and vegetable crop irrigated with sewage water in western region of Saudi Arabia. Saudi Journal of Biological Sciences, 23, S32–S44.
Bao, Z., Wu, W., Liu, H., Chen, H., & Yin, S. (2014). Impact of long-term irrigation with sewage on heavy metals in soils, crops, and groundwater—a case study in Beijing. Polish Journal of Environmental Studies, 23, 309–318.
Bortey-Sam, N., Nakayama, S. M. M., Akoto, O., Ikenaka, Y., Baidoo, E., Mizukawa, H., & Ishizuka, M. (2015). Ecological risk of heavy metals and a metalloid in agricultural soils in Tarkwa, Ghana. International Journal of Environmental Research and Public Health, 12, 11448–11465.
Effendi, H., Kawaroe, M., Mursalin, & Lestari, D. F. (2016). Ecological risk assessment of heavy metal pollution in surface sediment of Mahakam Delta, East Kalimantan. Procedia Environmental Sciences, 33, 574–582.
El-Mergawi, R. A., & Al-Redhaiman, K. (2010). Effect of organic and conventional production practices on antioxidant activity, antioxidant constituents and nutritional value of tomatoes and carrots in Saudi Arabia markets. Journal of Food, Agriculture and Environment, 8, 253–258.
Gan, J. L., Jia, X. P., Lin, Q., Li, C. H., Wang, Z. H., Zhou, G. J., Wang, X. P., Cai, W. G., & Lu, X. Y. (2000). A primary study on ecological risk caused by the heavy metals in coastal sediments. Journal of Fisheries of China, 24, 533–538.
Gee, G.W., & Bauder, J.W. (1994). Particle-size analysis. In A. Klute (Ed.), Methods of soil analysis. Part 1, 3rd edition. Physical and mineralogical methods. Madison: SSSA and ASA.
Gupta, N., Khan, D. K., & Santra, S. C. (2008). An assessment of heavy metal contamination in vegetables grown in wastewater-irrigated areas of Titagarh, West Bengal, India. Bulletin of Environmental Contamination and Toxicology, 80, 115–118.
Hakanson, L. (1980). An ecological risk index for aquatic pollution control. A sedimentological approach. Water Research, 14, 975–1001.
He, Q. B., & Singh, B. R. (1994a). Crop uptake of cadmium from phosphorus fertilizers: II. Relationship with extractable soil cadmium. Water, Air, and Soil Pollution, 74, 267–280.
He, Q. P., & Singh, B. R. (1994b). Crop uptake of cadmium from phosphorus fertilizers: I. Yield and cadmium content. Water, Air, and Soil Pollution, 74, 251–265.
Hernandez, L., Probst, A., Probst, J. L., & Ulrich, E. (2003). Heavy metal distribution in some French forest soils: evidence for atmosphere contamination. Science of the Total Environment, 312, 195–210.
Hu, Y., Liu, X., Bai, J., Shih, K., Zeng, E. Y., & Cheng, H. (2013). Assessing heavy metal pollution in the surface soils of a region that had undergone three decades of intense industrialization and urbanization. Environmental Science and Pollution Research, 20, 6150–6159.
Ju, X. T., Kou, C. L., Christie, P., Dou, Z. X., & Zhang, F. S. (2007). Changes in the soil environment from excessive application of fertilizers and manures to two contrasting intensive cropping systems on the North China Plain. Environmental Pollution, 145, 497–506.
Kabata-Pendias, A. (2011). Trace elements in soils and plants (Fourth ed.). London: Taylor &Francis.
Kelepertzis, E. (2014). Accumulation of heavy metals in agricultural soils of Mediterranean: insights from Argolida basin, Peloponnese, Greece. Geoderma, 221-222, 82–90.
Khan, K., Lu, Y., Khan, H., Ishtiaq, M., Khan, S., Waqas, M., Wei, L., & Wang, T. (2013). Heavy metals in agricultural soils and crops and their health risks in Swat District, northern Pakistan. Food and Chemical Toxicology, 58, 449–458.
Lindsay, W. L. (1979). Chemical equilibrium in soils. NewYork: Wiley Interscience.
Lu, Q., Bai, J., Gao, Z., Zhao, Q., & Wang, J. (2016). Spatial and seasonal distribution and risk assessments for metals in a Tamarix chinensis wetland, China. Wetlands, 36, S125–S136.
Mapanda, F., Mangwayana, E. N., Nyamangara, J., & Giller, K. E. (2005). The effect of long-term irrigation using wastewater on heavy metal contents of soils under vegetables in Harare, Zimbabwe. Agriculture, Ecosystems & Environment, 107, 151–165.
Mar, S. S., Okazaki, M., & Motobayashi, T. (2012). The influence of phosphate fertilizer application levels and cultivars on cadmium uptake by Komatsuna (Brassica rapa L. var. perviridis). Soil Science and Plant Nutrition, 58, 492–502.
Mico, C., Peris, M., Sanchez, J., & Recatala, L. (2006a). Heavy metal content of agricultural soils in a Mediterranean semiarid area: the Segura River Valley (Alicante, Spain). Spanish Journal of Agricultural Research, 4, 363–372.
Mico, C., Recatala, L., Peris, M., & Sanchez, J. (2006b). Assessing heavy metal sources in agricultural soils of an European Mediterranean area by multivariate analysis. Chemosphere, 65, 863–872.
Muller, G. (1979). Schwermetalle in den sediments des Rheins-Veran-derungen seitt. Umschan, 79, 778–783.
Naveedullah., Hashmi, M. Z., Yu, C., Shen, H., Duan, D., Shen, C., Lou, L., & Chen, Y. (2013). Risk assessment of heavy metals pollution in agricultural soils of Siling Reservoir Watershed in Zhejiang Province, China. BioMed Research International, 2013, Article ID 290306, 10 pages.
Nazzal, Y., Howari, F. M., Jafri, M. K., Naeem, M., & Ghrefat, H. (2015). Risk ssessment through evaluation of potentially toxic metals in the surface soils of the Qassim area, Central Saudi Arabia. Italian Journal of Geosciences, 135, 210–216.
Nelson D. W., & Sommers L. E. (1996). Total carbon, organic carbon, and organic matter. In Methods of soil analysis, part 3. Chemical methods (pp. 961–1009). Madison: SSSA. Book ser. N°5.
Oyedele, D. J., Asonugho, C., & Awotoye, O. O. (2006). Heavy metals in soil and accumulation by edible vegetables after phosphate fertilizer application. Electronic Journal of Environmental, Agricultural and Food Chemistry, 5, 1446–1453.
Reeve, J. R., Endelman, J. B., Miller, B. E., & Hole, D. J. (2012). Residual effects of compost on soil quality and dryland wheat yield sixteen years after compost application. Soil Science Society of America Journal, 76, 278–285.
Roberts, T. L. (2014). Cadmium and phosphorous fertilizers: the issues and the science. Procedia Engineering, 83, 52–59.
Sparks, D. L. (1996). Methods of soil analysis part 3—chemical methods. Madison: SSSA, American Society of Agronomy.
StatSoft Inc. (1992). Statistica for Windows (computer program manual). Tulsa: StatSoft, Inc.
Sutherland, R. A. (2000). Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environmental Geology, 39, 611–627.
Tabatabai, M. A. (1996). Sulfur. In D. L. Sparks, A. L. Page, P. A. Helmke, & R. H. Loeppert (Eds.), Methods of soil analysis part 3—chemical methods. Madison: SSSA, American Society of Agronomy.
Toth, G., Hermann, T., Da Silva, M. R., & Montanarella, L. (2016). Heavy metals in agricultural soils of the European Union with implications for food safety. Environment International, 88, 299–309.
Turekian, K. K., & Wedepohl, K. H. (1961). Distribution of the elements in some major units of the Earth’s crust. Geological Society of America Bulletin Journal, 72, 175–192.
USEPA (1994). Method 3051, microwave assisted acid digestion of sediments, sludges, soils and oils. In Test methods for evaluating solid waste, physical/chemical methods, 3rd ed. U.S.
VROM. (2001). The new Dutch list. Soil quality standards, Netherlands Ministry of Housing, Spatial Planning and Environment. Department of Soil Protection, The Hague.
Wentz, D. (2001). Salt tolerance of plants. Agri-facts—practical information for Alberta’s agriculture industry. Agdex 518–17.
Wu, J., Song, J., Li, W., & Zheng, M. (2016). The accumulation of heavy metals in agricultural land and the associated potential ecological risks in Shenzhen, China. Environmental Science and Pollution Research, 23, 1428–1440.
Xiao, R., Bai, J., Lu, Q., Zhao, Q., Gao, Z., Wen, X., & Liu, X. (2015). Fractionation, transfer, and ecological risks of heavy metals in riparian and ditch wetlands across a 100-year chronosequence of reclamation in an estuary of China. Science of the Total Environment, 517, 66–75.
Xu, Y. G., Yu, W. T., Ma, Q., & Zhou, H. (2015). Potential risk of cadmium in a soil-plant system as a result of long-term (10 years) pig manure application. Plant, Soil and Environment, 61, 352–357.
Ye, C., Li, S., Zhang, Y., & Zhang, Q. (2011). Assessing soil heavy metal pollution in the water-level-fluctuation zone of the Three Gorges Reservoir, China. Journal of Hazard Materials, 191, 366–372.
Zoffoli, H. J. O., Amaral-Sobrinho, N. M., Zonta, E., Luisi, M. V., Marcon, G., & Tolon-Becerra, A. (2013). Inputs of heavy metals due to agrochemical use in tobacco fields in Brazil’s Southern Region. Environmental Monitoring and Assessment, 185, 2423–2437.
Acknowledgements
The investigators of research project no. ALC012 thank the Promising Research Center in Biological Control and Agricultural Information, Al-Qassim University, Buraydah, KSA, for providing funding and unlimited support and help. The authors are also grateful to the Department of Soil Science, College of Food, and Agricultural Sciences, King Saud University, for providing research facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Wabel, M.I., Sallam, A.EA.S., Usman, A. et al. Trace metal levels, sources, and ecological risk assessment in a densely agricultural area from Saudi Arabia. Environ Monit Assess 189, 252 (2017). https://doi.org/10.1007/s10661-017-5919-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-017-5919-1