Abstract
Symptoms of leaf yellowing, reddening, witches’ broom and declining were observed in peach, apricot, and plum orchards from two states and a union territory [Himachal Pradesh (H.P.), Uttarakhand and Jammu & Kashmir (J&K)] of India during 2019–2021. Association of three groups (16SrI, 16SrII,16SrV) of phytoplasma were confirmed in symptomatic peach, plum and apricot samples by amplifying DNA using 16S rRNA and multiple non-ribosomal primers (secA, secY, tuf). Pair wise sequence comparison, phylogenetic analysis and virtual RFLP analysis using multiple non-ribosomal gene sequences confirmed the presence of ‘Candidatus Phytoplasma asteris’ (16SrI-B), ‘Ca. Phytoplasma australasia' (16SrII-D), elm yellows (new subgroup variant of 16SrV) and ‘Ca. Phytoplasma ziziphi' (16SrV-B) related strains in peach, plum and apricot trees. Besides, other suspected symptomatic plant hosts and weeds in and around stone fruit orchards were also identified positive with phytoplasma strains belonging to 16SrI-B and 16Sr II-D subgroups by amplifying 16SrRNA and secA genes and sequence analysis. Association of similar strains of phytoplasma was identified in leafhopper species, Hishimonus phycitis from J&K and Empoasca sp. from Uttarakhand by utilizing the same set of primer pairs and 16S rRNA and secA gene sequence comparison. The presence of ‘Ca. P. asteris’ (16SrI-B) and ‘Ca. P. australasia’ (16SrII-D) related strains detected in peach are new host records from India and the association of elm yellows (a variant of 16SrV subgroup) related strain in plum is the first report in the world.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The temperate stone fruits such as apricot, peach, nectarine, plum and cherry are the most important fruit crops cultivated throughout the temperate regions of the world with an area of 7.6 million ha with 49.9 million tons of production (FAOSTAT, 2019). Temperate stone fruits are mainly grown in the north-western Himalayan states/union territory [Himachal Pradesh (H.P.), Uttarakhand and Jammu & Kashmir (J&K)] of India and a few locations in the north-eastern regions of India. About 77,742 ha of temperate stone fruits are grown in India with an annual production of about 2, 55,000 MT (FAOSTAT, 2019). Stone fruit trees are infected by several pathogens among which ‘Candidatus Phytoplasmas’ members are a serious emerging pathogen causing severe yield losses all over the world (Fiore et al., 2018; Žežlina et al., 2016).
Phytoplasmas are small sized (0.3–1.2 μm), wall-less, non-helical and polymorphic obligate plant pathogenic bacteria, belonging to the class Mollicutes with a genome size of 530 bp to 1350 kb and having low G + C content in their DNA (23.0–29.5 mol%) (Bertaccini & Lee, 2018; Marcone et al., 2014). Phytoplasma diseases are of serious concern in many countries of the Americas, Europe and the Middle East (Fiore et al., 2018; Hemmati et al., 2021). Temperate stone fruit diseases associated with phytoplasma infection includes X-disease in sweet cherry, leptonecrosis of the plum, apricot chlorotic leaf roll (ACLR), leaf yellowing and reddening in peach, declining of plum, peach and almond. The major phytoplasma strains association was identified as European stone fruit yellows (ESFY) (Fiore et al., 2018; Lorenz et al., 1994; Zirak et al., 2021). Plants species of the Prunus genus were reported to be associated with more than eight phytoplasma groups worldwide (Cieślińska & Morgaś, 2011; Allahverdi et al., 2014; Bertaccini et al., 2014; Fiore et al., 2018; Hemmati et al., 2021; Marcone et al., 2014). Only few reports are available on the characterization of phytoplasma strains associated with stone fruit crops in India (Rao et al., 2020).
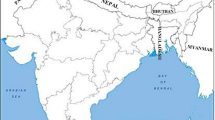
In this study, we investigated the phytoplasmas in three stone fruit crops viz., peach, plum, and apricot in orchards situated at Rajouri from Jammu & Kashmir; Solan, Mashobra and Rajgarh from Himachal Pradesh; Pantnagar and Mukteshwar from Uttarakhand (Fig. 1) utilizing multiple non-ribosomal gene primers and their sequence comparison analysis. Weeds and other plant hosts showing suspected phytoplasma symptoms in and around stone fruit orchards, and leafhoppers feeding on symptomatic plants and near vegetations were also investigated for the presence of phytoplasmas.
Materials and methods
Plant sample survey and symptomatology
Leaf samples were collected from three symptomatic and two asymptomatic plants each of peach, plum, and apricot from 2019 to 2021 (Table 1). Weeds and other plant hosts showing suspected phytoplasma symptoms in and around stone fruit orchards were also collected from the same locations and analyzed to verify the presence of phytoplasmas. The disease incidence was recorded in the orchards by counting the number of trees displaying symptoms over total number of trees in 0.5 ha. All the collected leaf and stalk samples were stored at 4 °C till further analysis.
Insect samples
The leafhoppers (LH) were collected from the fruit orchards during from June to October, 2019–21 using yellow sticky traps and sweeping nets. The LH species were identified at the Division of Entomology, ICAR-Indian Agricultural Research Institute, New Delhi, India.
DNA extraction and PCR assays
DNA was extracted from 100 mg of young growing shoot tissue and leaf midrib of symptomatic and asymptomatic stone fruits, weed species and from LH by the CTAB method (Ahrens & Seemüller, 1992; Marzachi et al. 1998). The DNA was resuspended in 80 μl of nuclease free water and kept at −20 °C for further PCR assays. The concentration of DNA was quantified with a Nanodrop spectrophotometer (ND-1000 UV/ VIS, USA).
PCR assays were carried out in a reaction volume of 25 μl containing nuclease-free water (Sisco Research Laboratories Pvt. Ltd., India), One PCR™ 2X PCR Master Mix (GeneDireX, Taiwan), for/rev primers 10 pmol/μl (final concentration 0.2 μM), and DNA template (=100 ng). Universal phytoplasma primer pairs P1/P7 (Deng & Hiruki, 1991; Schneider et al., 1995) followed by nested primer pair R16F2n/R2 (Gundersen & Lee, 1996) were used to amplify the phytoplasma 16S rRNA gene in PCR assays. The specific primers for secA, secY and tuf genes were also used to confirm and validate the presence of phytoplasma in the symptomatic fruit trees and other plant host samples (Table 2). The amplified products of the first round of PCR were diluted 1:20 with nuclease free water and used as template in nested PCR assays. DNA extracted from the asymptomatic plants was used as negative control.
The cycling conditions for the secA gene were 94 °C for 5 min, 94 °C for 30 s, 53 °C for 60 s, 72 °C for 60 s for 30 cycles and a final extension step of 72 °C for 10 min. The same reaction conditions were followed in the first and second round of PCR assays. A total of 38 cycles were carried out for the secY gene under the cycling conditions: 94 °C for 10 min, 94 °C for 60 s, 55 °C for 120 s, 72 °C for 3 min and a final extension step of 72 °C for 7 min. The cycling conditions for the tuf gene in the first round are: 94 °C for 90s, 94 °C for 30 s, 45 °C for 30 s, 72 °C for 60 sec for 35 cycles and a final extension step of 72 °C for 10 min and for the nested round: 94 °C for 4 min, 94 °C for 30 s, 53 °C for 30 s, 72 °C for 60 sec for 35 cycles and a final extension step of 72 °C for 5 min. PCR reactions were carried out in a thermal cycler (Mastercycler, Eppendorf, Germany). DNA extracted from the Catharanthus roseus infected with sesame phyllody phytoplasma maintained in a glasshouse (16SrI group, GenBank Acc. No. KC920747) was used as positive control. The PCR products were subjected to electrophoresis in 1.0% (w/v) agarose gel, stained with GoodView™ Nucleic Acid stain and the amplifications were observed in the Gel Doc system (Azure Biosystems, USA). The amplified products were purified, ligated into pGEM®T vector (Promega, Madison, USA) and cloned in Escherichia coli (DH5-α). The cloned products were sequenced using the M13Fwd/M13Rev universal primer pair through AgriGenome, Kerala, India.
Sequence analysis
For sequence data analysis, Qiagen CLC Main work bench was used (https: //digitalinsights.qiagen.com). The sequences were assembled using DNA Base V.4 (http://www.dnabaser.com), aligned with phytoplasma ribosomal group/subgroup representatives available in GenBank using Clustal W software, and the consensus sequences were submitted to the GenBank. The 16S rRNA gene sequences generated in the present study were aligned with those of ‘Ca. Phytoplasma’ strains, retrieved from NCBI GenBank, and the sequence identity values were determined. A phylogenetic tree was constructed by the neighbour-joining method using MEGA 7.0 (Kumar et al., 2016) using 1000 bootstrap replications and Acholeplasma laidlawii (GenBank Acc. No. AB680603) for the 16S rRNA gene, Acholeplasma oculi for the secY and Bacillus subtillis (GenBank Acc. No. D10279) for the secA and the tuf genes, respectively to root the trees.
Virtual RFLP analysis
About ~1.25 kb of phytoplasma DNA sequences corresponding to the R16F2n/R2 fragments of stone fruits (peach, plum and apricot), other hosts (Toona sinensis, Cannabis sativus, Celosia argentia, Datura stramonium, C. roseus, Phyllanthus niruri and Ageratum conyzoides) and leafhoppers (H. phycitis and Empoasca sp.) phytoplasma strains were subjected to in silico RFLP comparison analysis using the iPhyClassifier online tool by comparing the computer generated restriction patters and the similarity coefficient value was calculated (Zhao et al., 2009).
Results
Survey, disease incidence and symptomatology
Leaf yellowing & reddening along with decline was observed in peach cvs Peach Pant 1, Red June and Para Deluxe from Pantnagar, Uttarakhand, and little leaf, yellowing and decline of plum cv Fla12 was recorded from Mukteshwar, Uttarakhand (Table 1; Figs. 1a, b, c and 2b, c, d). Leaf rolling, little leaf, leaf yellowing & reddening and decline were observed in peach and apricot trees from Rajouri, J&K (Figs. 1d and 2e). Little leaf, yellowing and reddening in peach cv July Elberta from Mashobra, H.P.; leaf yellowing & reddening and decline symptoms on peach cv Sun Haven from Solan, H.P. and yellowing, reddening and decline in peach cv Red Haven, Florida sun from Rajgarh, H.P. (Figs. 1e–g and 2a) were recorded.
Phytoplasma disease symptoms in peach orchards: a peach leaf yellowing & reddening cultivar cv. Red June, Pantnagar, Uttarakhand; b peach leaf yellowing & reddening with less vigour cv. Peach Pant-1, Pantnagar; c peach leaf reddening and decling cv. Para Deluxe, Pantnagar; d peach little leaf and yellowing and reddening and decline, Rajouri, Jammu and Kashmir e peach little leaf, yellowing & reddening cv. July Elberta, Mashobra H.P., f peach leaf yellowing and reddening and declining cultivar Sun Heaven from Solan, g peach leaf yellowing and reddening and decline in Red Heaven, Rajgarh, H.P
Phytoplasma disease symptoms in stone fruit orchards: a peach leaf yellowing & reddening and declining in cultivar Florida sun, Rajgarh, H.P.; b plum little leaf, yellowing and decling in cultivar Fla12, Mukteshwar, Uttarakhand c, d section of plum with yellowing and deformed leaves, Mukteshwar, Uttarakhand; e little leaf, yellowing and reddening in apricot, Rajouri, J&K
Symptoms of severe witches’ broom in Cannabis sativa and Toona sinensis, flat stem in Celosia argentea, little leaf and witches’ broom in D. stramonium and little leaf and yellowing in C. roseus was recorded from Rajouri, J & K nearby fruit orchards (Fig. 3a–e). Witches’ broom in Toona sinensis from Solan and Rajgarh, H.P., leaf rolling and chlorosis in P. niruri from Rajgarh, H.P (Fig. 3f, g) was also recorded. Further, severe leaf yellowing in Ageratum conyzoides was also recorded from Pantnagar, Uttarakhand in peach orchards (Fig. 3h).
Phytoplasma disease symptoms in weeds/alternate host plants: a Toona sinensis witches’ broom, b Cannabis sativa little leaf and witches’ broom, c Catharanthus roseus leaf yellowing, d Celosia argentea flat stem, e Datura stramonium little leaf and witches’ broom from Rajouri, Jammu & Kashmir; f, Toona sinensis witches’ broom, g Phyllanthus niruri leaf rolling and chlorosis, Rajgarh, H. P.; h Ageratum conyzoides leaf yellowing, Pantnagar, Uttarakhand
Identification of the leafhoppers
Ten different leafhoppers species, Bothrogonia alternata (Melichar), Olidinia indica (Walker), Atkinsoniella opponens (Walker), Idioscopus spp., Kolla paulula (Walker), Phlogotettix subhimalayanus (Meshram & Ramamurthy), Idioscopus nitidulus (Walker), Hishimonus phycitis (Distant) (H.P) and Empoasca sp. captured from stone fruit trees and nearby plant species were identified on the basis of morphology. H. phycitis (H.P) and Empoasca sp. were identified as the main leafhopper species at surveyed locations by sweeping net method and counts on yellow sticky traps.
Phytoplasma detection by PCR assays
The DNA fragments of ~1.25 kb were consistently amplified in nested PCR assays with the R16F2n/R16R2 primer pair from the samples of symptomatic peach and apricot trees from Rajouri, peach (cvs Peach Pant 1, Red June and Para Deluxe) and plum (cv Fla12) samples from Uttarakhand and peach (cvs July Elbert, Sun Haven, Red Haven and Florida sun) from H.P. along with the positive control (data not shown). Amplification was also obtained in the symptomatic weeds and tree (C. sativa, T. sinensis, D. stramonium, C. roseus, P. niruri and A. conyzoides) from different locations and in insect samples of H. phycitis from Rajouri, J&K and Empoasca sp. from Pantnagar, Uttarakhand (Table 1). However, no phytoplasma DNA amplifications were achieved in any of the non-symptomatic samples from any stone fruits and weeds/tree from all the surveyed locations (data not shown). The sequences achieved from the amplified products of three stone fruits (17 samples), seven alternate plant hosts and two leafhoppers (H. phycitis and Empoasca sp.) were analyzed and deposited in GenBank (Table 1).
A BLASTn identity search of GenBank’s database revealed that the stone fruit phytoplasma isolates from the two states were associated with three phytoplasma groups. The peach leaf rolling and reddening (PPRR -1& -2), apricot little leaf and yellowing (PARD -1& -2) from Jammu; peach cv July Elberta leaf yellowing Mashobra strains (PLDM-1 &-2), peach cv Sun Haven reddening & declining Solan strains (PPLS -1& -2), peach cv Red Haven reddening and declining strains from Rajgarh (PPLR-1 &-2) revealed 99.34–99.80% 16S rRNA gene sequence identity with ‘Candidatus Phytoplasma asteris’ group (16SrI) related strains. The peach leaf yellowing & reddening and declining Pantnagar strains (cvs Peach Pant-PYRU-1, Red June- PYRU-2 and Para Deluxe-PYRU-3) had 99.30–100% sequence identity with ‘Candidatus Phytoplasma australasia’-related strains (16SrII). However, the peach reddening & declining Rajgarh strains (cv Florida sun PPYR-1 & -2) and plum little leaf and yellowing Mukteshwar strains (cv Fla12-PLYD-1 & -2) had shown 99.92 and 100% sequence identities with ‘Candidatus Phytoplasma ziziphi’ and elm yellows related strain (very similar to 16SrV-C), respectively. Besides, C. sativa witches’ broom (CSWB-1 & -2), T. sinensis witches’ broom (TSWB-1 & -2), D. stramonium little leaf (DALY-1), C. argentea flat stem (CAPF-1) and C. roseus little leaf and yellowing (CLLY-1) from Jammu; T. sinensis witches’ broom Rajgarh and Solan (TWBR-1 & -2), P. niruri leaf rolling & yellowing Rajgarh strains (PLLR-1 & -2) from H.P. revealed 99.34–99.80% sequence identity with ‘Candidatus Phytoplasma asteris’-related strains. A. conyzoides leaf yellowing Pantnagar strain (ALY-1 & -2) had 100% sequence identity with ‘Candidatus Phytoplasma australasia’-related strains. Besides, H. phycitis Jammu strains (HPP-1 & -2) and Empoasca sp. strains (GLH-1 & -2) from Pantnagar revealed 99.34% and 100% sequence identity with ‘Candidatus Phytoplasma asteris’ and ‘Candidatus Phytoplasma australasia’- related strains, respectively.
Pair wise sequence comparison of partial secA gene sequences revealed that peach and apricot phytoplasma strains from J & K and peach from H.P. as well as weeds phytoplasma strains from both Jammu and H.P. shared 99.34% to 99.80% sequence identity with other previously identified phytoplasma strains related to ‘Candidatus Phytoplasma asteris’ group, while peach and weed phytoplasma strains from Uttarakhand showed 100% sequence similarity with other previously identified phytoplasma related strains to ‘Candidatus Phytoplasma australasia’ group (Table 1). Similarly, the pairwise comparison of ~1400 bp sequences of secY and ~ 940 bp of tuf gene sequences of peach and weeds phytoplasma strains of H.P. (Table 1) had shown 99.62–99.77% and 99.89% sequence identities with phytoplasma strains belonging to ‘Candidatus Phytoplasma asteris’-related strains, respectively.
A phylogenetic tree was constructed using partial 16S rRNA gene sequences showing clustering of all the peach and weeds phytoplasma strains from H.P. and peach, apricot, H. phycitis and weeds phytoplasma strains from J&K together with Ca. P. asteris. While phytoplasma strains of peach, Empoasca sp. and weeds from Pantnagar were clustered together with ‘Candidatus Phytoplasma australasia’ related strains. However, the phytoplasma strains associated with plum from Mukteshwar, Uttarakhand and peach (cv Florida sun) from Rajgarh, H.P. were clustered together with the elm yellows group (16SrV) (Fig. 4). Similarly, secA gene sequences showed clustering of peach and weeds phytoplasma strains from H.P. and peach, apricot, H. phycitis and weeds phytoplasma strains from J&K together with ‘Candidatus Phytoplasma asteris’, while phytoplasma strains of peach, Empoasca sp. and weeds from Pantnagar were clustered together with ‘Candidatus Phytoplasma australasia’-related strains (Fig. 5). Besides, secY and tuf gene sequences of all the peach and weeds phytoplasma strains from H.P. also showed clustering together with ‘Candidatus Phytoplasma asteris’ related strains (Figs. 6 and 7).
Phylogenetic tree constructed by neighbor-joining method based on 16S rDNA gene sequences of stone fruits, weeds and insects with other selected phytoplasma strains form GenBank. Accession numbers are specified in the tree. Acholeplasma laidlawii was used as an out group. Numbers on branches are bootstrap values obtained for 1000 replicates. The bar represents a phylogenetic distance of 1
Phylogenetic tree of secA gene sequences constructed by neighbor-joining method and Kimura’s three- parameter model, showing the relationships among stone fruit phytoplasma strains, weeds and alternate host as well as leaf hopper phytoplasma strains and reference phytoplasma strains. Bacillus subtilis was used as an out-group. Numbers on branches are bootstrap values obtained for 1000 bootstrap replicates. The bar represents a phylogenetic distance of 0.1
Phylogenetic tree of tuf gene sequences constructed by neighbor-joining method and Kimura’s three- parameter model, showing the relationships among stone fruit phytoplasma strains, weeds and alternate host phytoplasma strains and reference phytoplasma strains. Bacillus subtilis was used as an out-group. Numbers on branches are bootstrap values obtained for 1000 bootstrap replicates. The bar represents a phylogenetic distance of 0.5
Phylogenetic tree of secY gene sequences constructed by neighbor-joining method and Kimura’s three- parameter model, showing the relationships among stone fruit phytoplasma strains, weeds and alternate host phytoplasma strains and reference phytoplasma strains. Acholeplasma oculi was used as an out-group. Numbers on branches are bootstrap values obtained for 1000 bootstrap replicates. The bar represents a phylogenetic distance of 0.5
In silico RFLP analysis
Virtual RFLP analysis results of the 16S rDNA gene fragments of peach, apricot, weeds and H. phycitis phytoplasma strains from J & K and peach (except cv Florida Sun, PPYR-1) and weeds phytoplasma strains from H.P. showed identical restriction profiles with Oenothera phytoplasma reference strains belonging to 16SrI-B with a similarity coefficient of 1.0 [shown only one representative strain Peach (PPLS-1) Acc. No. OL527667 with reference strain Acc. No. M30790; Fig. 8a, b]. The restriction profiles of the 16Sr RNA sequence of peach, weeds and Empoasca sp. phytoplasma strains from Pantnagar produced identical patterns with the papaya yellow crinkle disease reference strain [Acc. No. OL454916 with reference strain 16SrII-D, Acc. No. Y10097; Fig. 8c, d]. However, peach phytoplasma strains from Rajgarh (PPYR-1, Acc. No. OL469604) produced identical RFLP profiles with the jujube witches’ broom phytoplasma reference strain belonged to the 16SrV-B subgroup (Acc. No. AB052876; Fig. 8e, f). The predicted digestion fragments of the plum phytoplasma strains (PLYD-1, Acc. No.OL455729) matched the RFLP patterns very closely with the alder yellows phytoplasma reference strain which belonged to 16SrV-C sub group, except the restriction profile of DraI and MseI endonucleases (Acc. No. AY197642; Fig. 8g, h). Hence, the results of the present study confirmed affiliation of four subgroups of phytoplasmas (16SrI-B, 16SrII-D, 16SrV-B & variant of V-C) in three stone fruit trees in the western Himalaya region.
Virtual RFLP patterns from in silico digestion of 16SrDNA R16F2n/R2 fragments of phytoplasma strains infecting stone fruits in India and the phytoplasma reference strains with 17 restriction enzymes (AluI, BamHI, BfaI, BstUI, DraI, EcoRI, HaeIII, HhaI, HinfI, HpaI, HpaII, KpnI, MboI, MseI, RsaI, SspI and TaqI) using iPhyClassifier program. The patterns are compared for 16SrI-B reference strain (GenBank Acc. No. M30790 (a) with peach decline (PPLS-1) Acc.no. OL527667 (b); for16SrII-D reference strain (Acc. No. Y10097 (c) with Peach reddening (PYRU-2) Acc. No. OL454916 (d); for16SrV-B reference strain (Acc. No. AB052876 (e) with peach yellowing & reddening (PPYR-1) Acc. no. OL469604 (f); for 16SrV-C reference strain (GenBank Acc. No. AY197642 (g) with plum yellowing and reddening (PLYD-1) Acc. No. OL455729 (h)
Discussion
In the present study, four phytoplasma subgroup strains, 16SrI-B, 16Sr II-D, 16SrV-B, and 16SrV-C, associated with peach, plum, apricot,alternate hosts and two leafhoppers were identified and characterized. The secA, tuf, and secY genes were reported to show more genetic variation than the 16S rRNA gene and are hence useful for the genetic diversity determination among the phytoplasma strains (Bertaccini & Lee, 2018; Martini et al., 2019). Keeping this in mind, in the present study, three non-ribosomal gene markers (secA, secY, tuf) were validated to confirm the presence of phytoplasmas (16SrI-B and 16SrII-D) association in stone fruits, alternate hosts and the leafhoppers. There were found valid and authentic in phytoplasma characterization of all the stone fruit and weed/tree samples.
In the present investigation, ‘Ca. P. asteris’ (16SrI-B) from Jammu & Kashmir and Himachal Pradesh and ‘Ca. Phytoplasma australasia (16SrII-D) from Uttarakhand have been confirmed associated with peach leaf yellowing, reddening and decline symptoms. Peach isa new host for these subgroups of phytoplasmas. Peach cv Florida sun from Rajgarh, Himachal Pradesh was shown to be associated with ‘Ca. Phytoplasma ziziphi' (16SrV-B subgroup) which has already been reported in peach from India (Khan et al., 2013). Peach is susceptible to several phytoplasma strains worldwide including, 16SrI-A, 16SrII, 16SrIII-B, 16SrVI-B,16SrIX-B&C, 16SrX-B&C and 16SrXV-B groups and subgroups in USA, Canada, Argentina, Europe, China, Iran and India (Balakishiyeva et al., 2010; Singh et al., 2014; Blomquist & Kirkpatrick, 2002; Fiore et al., 2018; Marcone et al., 2010; Rao, 2021).
Apricot is reported to be an important host of phytoplasma strains belonging to the aster yellows group (16SrI-B &-F) and X-disease (16SrIII-A) in India, Italy, Spain, USA, Singapore and Iran (Fiore et al., 2018). Our study also confirmed and established the association of ‘Ca. P. asteris’ (16Sr I-B) related strain with leaf rolling and decline symptoms of apricot trees in Rajouri, Jammu & Kashmir, India. A similar strain was also reported earlier in apricot from J&K, India (Rao et al., 2020).
Plum is reported to be infected with aster yellows, peanut witches’ broom and stolbur groups in Iran and Poland (Zirak et al., 2009; Zwolińska et al., 2019). Recently, Zirak et al. (2021) reported associations of ‘Ca. P. asteris’ and ‘Ca. P. trifolii’ in plum orchards from East Azerbaijan (Iran). Our present investigation established the association of the elm yellows related strain (variant of 16Sr V-C subgroup) with little leaf and yellowing symptoms in plum trees in Mukteshwar, Uttarakhand, India. However, the plum phytoplasma strain (Acc. No. OL455729) had a 1.0 similarity coefficient with the Alder yellows phytoplasma reference strain (16SrV-C, Acc. No. AY197642), but the gel pattern showed difference in restriction profiles with DraI and MseI endonucleases which suggest that the associated phytoplasma strain is a variant of the elm yellows (16SrV) group which needs further investigation. To our knowledge, this is the first report of a new variant of 16SrV group to be associated with plum in the world.
Out of four subgroups of phytoplasma strains identified with three stone fruit crops (peach, apricot and plum), the 16SrI-B and 16SrII-D phytoplasma subgroups are identified as widespread strains infecting vegetables, fruits, ornamentals, legumes and spice crops in India (Rao, 2021). Besides stone fruits, the present study also suggested the role of weeds and other plant hosts growing in and around the fruit orchards as natural host reservoirs for the transmission of the 16SrI-B and 16SrII-D subgroup phytoplasmas possibly with the help of leafhopper vectors. In the present study, H. phycitis was found positive for the presence of the 16SrI-B phytoplasma strain. Different leaf hoppers, plant hoppers and psyllids have been reported as vectors of stone fruit related phytoplasma strains (Abou-Jawdah et al., 2011,; Dakhil et al., 2011; Mozaffarian & Wilson, 2016). In Lebanon, Ca. P. phoenicim was detected in the leafhopper, A. decedens, and in the cixiids, Cixius sp., Tachycixius spp. and Eumecurus spp. Transmission studies also demonstrated that A. decedens, T. viperinus and T. cypricus are able to transmit ‘Candidatus Phytoplasma phoenicium’ (Abou-Jawdah et al., 2014). The X- disease in peach is also reported to be transmitted by the psyllid Cacopsylla pyricola (Blomquist & Kirkpatrick, 2002; Purcell, 1982).
In India, different species of leafhoppers and plant hoppers are already identified as putative or natural vectors of phytoplasma strains belonging to the 16SrI-B and II-D subgroups of phytoplasmas (Rao, 2021). H. phycitis and Empoasca sp., identified as putative vector in the present study, for the two different groups of phytoplasma strains of stone fruits may play an important role in transmission of associated phytoplasma strains. Results of this study will facilitate further work on phytoplasma screening and indexing during production of quality planting material. It will also help in the studies related to epidemiology, diversity and management of phytoplasmas infecting stone fruit trees.
Results of the present study further confirmed that all the multiple non-ribosomal gene specific primers (secA, secY and tuf genes) are equally sensitive and effective in phytoplasma detection in stone fruit crops. These results also suggests the validity of these molecular markers for identification and characterization of phytoplasma strains belonging to 16SrI, 16SrII and 16SrV groups in other plant species (Martini et al., 2019).
Since the stone fruits are vegetatively propagated crops, transmission can take place via cuttings used as planting material.. Hence, it would be important to index established nurseries of stone fruit for phytoplasma disease free mother stocks. The timely and accurate detection of phytoplasmas in stone fruits utilizing multiple non-ribosomal gene primers would further enable the designing of appropriate diagnostic tools for sensitive and reliable detection of phytoplasmas in stone crops.
References
Abou-Jawdah, Y., Dakhil, H., Lova, M. M., Sobh, H., Nehme, M., Fakhr-Hammad, E. A., & Bianco, P. A. (2011). Preliminary survey of potential vectors of Candidatus Phytoplasma phoenicium in Lebanon and probability of occurrence of apricot chlorotic leaf roll (ACLR) phytoplasma. Bulletin of Insectology, 64(Supplement), S123–S124.
Abou-Jawdah, Y., Abdel Sater, A., Jawhari, M., Sobh, H., Abdul-Nour, H., Bianco, P. A., & Alma, A. (2014). Asymmetrasca decedens (Cicadellidae, Typhlocybinae), a natural vector of ‘Candidatus Phytoplasma phoenicium. Annals of Applied Biology, 165(3), 395–403.
Ahrens, U., & Seemüller, E. (1992). Detection of DNA of plant pathogenic mycoplasma like organisms by a polymerase chain reaction that amplifies a sequence of the 16S rRNA gene. Phytopathology, 82, 828–832.
Allahverdi, T., Rahimian, H., & Babaeizad, V. (2014). Association of ‘Candidatus Phytoplasma prunorum’ associated with plum yellow leaf stunt in Iran. Iranian Journal Plant Pathology, 50(2), 99.
Balakishiyeva, G., Danet, J. L., Qurbanov, M., Mamedov, A., Kheyr-Pour, A., & Foissac, X. (2010). First report of phytoplasma infections in several temperate fruit trees and vegetable crops in Azerbaijan. Journal of Plant Pathology, 92, S4.115.
Bertaccini, A., & Lee, I.-M. (2018). Phytoplasma: An update. In G. P. Rao, A. Bertaccini, N. Fiore, & L. W. Liefting (Eds.), Phytoplasmas: Plant pathogenic bacteria-I, characterization and epidemiology of phytoplasma-associated diseases (pp. 1–29). Springer.
Bertaccini, A., Duduk, B., Paltrinieri, S., & Contaldo, N. (2014). Phytoplasmas and phytoplasma diseases: A severe threat to agriculture. American Journal of Plant Sciences, 5, 1763–1788.
Blomquist, C. L., & Kirkpatrick, B. C. (2002). Identification of phytoplasma taxa and insect vectors of peach yellow leaf roll disease in California. Plant Disease, 86(7), 759–763.
Cieślińska, M., & Morgaś, H. (2011). Detection and identification of ‘Candidatus Phytoplasma prunorum’, Candidatus Phytoplasma mali’and ‘Candidatus Phytoplasma pyri’ in stone fruit trees in Poland. Journal of Phytopathology, 159(4), 217–222.
Dakhil, H. A., Hammad, E. A. F., El-Mohtar, C., & Abou-Jawdah, Y. (2011). Survey of leafhopper species in almond orchards infected with almond witches’-broom phytoplasma in Lebanon. Journal of Insect Science, 11, 60.
Deng, S., & Hiruki, C. (1991). Amplification of 16S rRNA genes from culturable and nonculturable mollicutes. Journal of Microbiological Methods, 14, 53–61.
FAOSTAT (2019). Agriculture data, agricultural statistics databases. http://faostat.fao.org, Organization of the United Nations, Rome, Italy.
Fiore, N., Bertaccini, A., Bianco, P. A., Cieślińska, M., Ferretti, L., Hoat, T. X., & Quaglino, F. (2018). Fruit crop phytoplasmas. In G. P. Rao, A. Bertaccini, N. Fiore, & L. W. Liefting (Eds.), In Phytoplasmas: Plant pathogenic bacteria-I (pp. 153–190). Springer.
Gundersen, D. E., & Lee, I.-M. (1996). Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathologia Mediterranea, 35, 144–151.
Hemmati, C., Nikooei, M., Al-Subhi, A. M., & Al-Sadi, A. M. (2021). History and current status of phytoplasma diseases in the Middle East. Biology, 10(3), 226.
Hodgetts, J., Boonham, N., Mumford, R., Harrison, N., & Dickinson, M. (2008). Phytoplasma phylogenetics based on analysis of secA and 23S rRNA gene sequences for improved resolution of candidate species of Candidatus Phytoplasma. International Journal of Systematic and Evolutionary Microbiology, 58, 1826–1837.
Khan, J. A., Kumar, J., Thakur, P. D., Handa, A., & Jarial, K. (2013). First report of a 'Candidatus Phytoplasma Ziziphi'-related strain associated with peach decline disease in India. Journal of Plant Pathology, 95, S4–S76.
Kumar, S., Stecher, G., & Tamura, K. (2016). MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Molecular Biology and Evolution, 33(7), 1870–1874.
Lee, I.-M., Zhao, Y., & Bottner, K. D. (2006). SecY gene sequence analysis for finer differentiation of diverse strains in the aster yellows phytoplasma group. Molecular and Cellular Probes, 20(2), 87–91.
Lorenz, K. H., Dosba, F., Poggi-Pollini, C., Llacer, G., & Seemüller, E. (1994). Phytoplasma diseases of Prunus species in Europe are caused by genetically similar organisms. Journal of Plant Diseases and Protection, 101, 567–575.
Marcone, C., Jarausch, B., & Jarausch, W. (2010). ‘Candidatus Phytoplasma prunorum’, the causal agent of European stone fruit yellows: An overview. Journal of Plant Pathology, 92, 19–34.
Marcone, C., Guerra, L. J., & Uyemoto, J. K. (2014). Phytoplasmal diseases of peach and associated phytoplasma taxa. Journal of Plant Pathology, 96(1), 15–28.
Martini, M., Quaglino, F., & Bertaccini, A. (2019). Multilocus genetic characterization of phytoplasma. In A. Bertaccini, K. Oshima, M. Kube, & G. P. Rao (Eds.), Phytoplasmas: Plant pathogenic bacteria-III, genomics, host pathogen interactions and diagnosis (pp. 161–200). Springer.
Marzachi, C., Verati, F., & Bosco, D. (1998). Direct PCR detection of phytoplasmas in experimentally infected insects. Annals of Applied Biology, 153, 45-54.
Mozaffarian, F., & Wilson, M. R. (2016). A checklist of the leafhoppers of Iran (Hemiptera: Auchenorrhyncha: Cicadellidae). Zootaxa, 4062(1), 1–63.
Purcell, A. H. (1982). Insect vector relationships with prokaryotic plant pathogens. Annual Review of Phytopathology, 20, 397–417.
Rao, G. P. (2021). Our understanding about phytoplasma research scenario in India. Indian Phytopathology, 74(5), 371–401.
Rao, G. P., Rao, A., Kumar, M., Ranebennur, H., Mitra, S., & Singh, A. K. (2020). Identification of phytoplasma in six fruit crops in India. European Journal of Plant Pathology, 156(4), 1197–1206.
Salehi, E., & Quaglino, F. (2020). Peach witches’-broom, an emerging disease associated with ‘Candidatus Phytoplasma phoenicium’ and ‘Candidatus Phytoplasma aurantifolia’ in Iran. Crop Protection, 127, 104946.
Schneider, B., Seemüller, E., Smart, C. D., & Kirkpatrick, B. C. (1995). Phylogenetic classification of plant pathogenic mycoplasma-like organisms or phytoplasma. Molecular and Diagnostic Procedures in Mycoplasmology, 1, 369–380.
Schneider, B., Gibb, K. S., & Seemüller, E. (1997). Sequence and RFLP analysis of the elongation factor Tu gene used in differentiation and classification of phytoplasmas. Microbiology, 143(10), 3381–3389.
Singh, J., Rani, A., Kumar, P., Baranwal, V. K., Saroj, P. L., Sirohi, A., Pandey, A. N., & Schenk, P. M. (2014). New host record of a ‘Candidatus Phytoplasma asteris’-related strain infecting peach in India. Australasian Plant Disease Notes, 9(1), 125.
Žežlina, I., Rot, M., Kač, M., & Trdan, S. (2016). Causal agents of stone fruit diseases in Slovenia and the potential for diminishing their economic impact–a review. Plant Protection Science, 52(3), 149–157.
Zhao, Y., Wei, W., Lee, I. M., Shao, J., Suo, X., & Davis, R. E. (2009). Construction of an interactive online phytoplasma classification tool, iPhyClassifier and its application in analysis of the peach X-disease phytoplasma group (16SrIII). International Journal Systematic and Evolutionary Microbiology, 59, 2582–2593.
Zirak, L., Bahar, M., & Ahoonmanesh, A. (2009). Molecular characterization of phytoplasmas related to peanut witches’ broom and “stolbur” groups infecting plum in Iran. Journal of Plant Pathology, 91, 713–716.
Zirak, L., Khakvar, R., Zarrini, G., & Hasanpour, K. (2021). Detection and molecular characterization of phytoplasmas associated with stone fruit trees in northwest of Iran. Crop Protection, 142, 105526.
Zwolińska, A., Borodynko-Filas, N., Nowaczyk, D., & Hasiów-Jaroszewska, B. (2019). First report of Prunus domestica as the host of a phytoplasma belonging to group 16SrI, subgroup B/L. Plant Disease, 103(1), 145–146.
Acknowledgements
The authors are thankful to the Director, ICAR-Indian Agricultural Research Institute, New Delhi, India, for providing financial assistance. Also, wish to express sincere thanks to the Head, Division of Plant Pathology, Indian Agricultural Research Institute, for providing laboratory facilities.
Funding
Consumables funding provided by Indian Agricultural Research Institute, New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This paper does not contain studies on human or animal participants.
Informed consent
The paper has not been submitted elsewhere for publication, in whole or in part. Y.S.S. collected the samples and recorded field data and analyses the samples for PCR assays. PVDK analysed the data. AKS, SW and KPS helped in survey of stone fruit crops in different states, GPR drafted the first manuscript and made corrections. All authors contributed to improve the manuscript, reviewed and approved the manuscript.
Conflict of interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Shreenath, Y.S., Singh, A.K., Kumar, P.V.D. et al. Characterization and distribution of phytoplasma strains associated with temperate stone fruits and their possible natural reservoirs in the north-western Himalayan states of India. Eur J Plant Pathol 164, 93–108 (2022). https://doi.org/10.1007/s10658-022-02541-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-022-02541-7