Abstract
Arsenic (As) and mercury (Hg) are ubiquitous elements known to disrupt thyroid function in vertebrates. To explore the underlying mechanisms of Hg and As on the fish thyroid system, we investigated the associations between muscle concentrations of Hg and As with thyroid-related gene transcription in flathead (Platycephalus bassensis) from a contaminated estuary. We sampled fish at several sites to determine the hepatic expression of genes including deiodinases (D1 and D2), transthyretin (TTR), thyroid hormone receptors (TRα and TRβ) and related them to Hg and As levels in the same individuals. Negative correlations were observed between Hg levels and D2, TTR, TRα and TRβ, whereas positive associations were found between As concentrations and TTR and TRβ. These results suggest that Hg and As exposures from environmental pollution affect the regulation of genes important for normal thyroid function in fish. These thyroid-related genes could be used as biomarkers for monitoring environmental thyroid-hormone disrupting chemicals.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arsenic (As) and mercury (Hg) are environmental contaminants commonly associated with industrial pollution, which can be bioaccumulated and biomagnified and pose adverse effects on humans and wildlife (Abernathy et al. 2003; Eisler 2004; Mason et al. 1996; Ward et al. 2010). In recent years, there has been substantial evidence suggesting that both Hg and As can disrupt the thyroid system of vertebrates (Ciarrocca et al. 2012; Iavicoli et al. 2009; Tan et al. 2009). The thyroid system is highly conserved throughout vertebrates and plays a crucial role in many bioactivities such as normal metabolism, reproduction and development (Power et al. 2001; Zoeller et al. 2007). Therefore, the impairment of the thyroid system by an exposure to Hg and As may exert profound effects on the health of vertebrates. Previous studies reported Hg concentrations were positively associated with the circulating free thyroxine (T4)/free triiodothyronine (T3) ratio in humans (Barregard et al. 1994; Ellingsen et al. 2000). In mammals, low level As exposure altered thyroid hormone receptor-mediated gene expression in pituitary cells (Davey et al. 2008). Likewise, associations between Hg exposure and thyroid hormone concentrations have been observed in birds (Wada et al. 2009) and exposure to As affected the thyroid hormone receptor dependent developmental processes of amphibians (Davey et al. 2008). In fish, chronic As exposure decreased the plasma T4 concentration in coho salmon (Oncorhynchus kisutch) (see Nichols et al. 1984), while in juvenile rainbow trout (Oncorhynchus mykiss), both plasma T4 and T3 concentrations were increased when exposed to Hg (Bleau et al. 1996). Although Hg and As have been proven to be associated with thyroid parameters in different vertebrate species, the relationships between Hg, As, and thyroid related genes are poorly understood. In particular, the synergistic effects of Hg and As on these genes are still unknown (Iavicoli et al. 2009).
Fish can be used to provide valuable information on potential thyroid disrupting effects of environmental factors in fish or other animals that prey upon fish (Brown et al. 2004; Leatherland 2000). Thyroid hormone endocrine disruptors affect thyroid hormone synthesis, thyroid hormone metabolism, thyroid hormone transport and thyroid hormone receptors (Patrick 2009). In teleost fish, two types of thyroid hormone (T4 and T3) are synthesized in the thyroid follicles and then released into the circulation (Brown et al. 2004). Transthyretin (TTR) is one of the major transporter proteins that carry thyroid hormone into different target tissues in fish (Power et al. 2009). Thus, TTR has been suggested as one of the major targets for endocrine disrupting chemicals (Lans et al. 1994). In the target tissues, the majority of T4 is converted into T3, a more biologically active form of hormone by deiodinases (D1 and D2) and this is a crucial step in regulating the peripheral thyroid status (Orozco and Valverde-R 2005). In target cells, T3 binds to thyroid hormone receptors (TRs) as a transcription factor to regulate gene expression (Yamano 2005). Therefore, the transthyretin (TTR), deiodinases (D1 and D2) and thyroid hormone receptors (TRs) are important for thyroid hormone disruption.
Southern sand flathead (Platycephalus bassensis) have been used as an indicator species to assess the health of contaminant-exposed fish populations (Ayling et al. 1975). P. bassensis are demersal, non-migratory and they accumulate heavy metals from their diet consisting of benthic organisms (Parry and Scientific 1995). The Derwent estuary in Tasmania has been recognized as a river polluted with both Hg and As (Bloom and Ayling 1977; Green and Coughanowr 2003) and flathead in the Derwent River have been monitored for heavy metal residues since the 1970s (http://www.derwentestuary.org.au/). Metallurgical liquid effluent has been discharged into the estuary for decades (Bloom and Ayling 1977; Langlois et al. 1987). The bioaccumulations of heavy metals in fish and shellfish from the Derwent estuary are much higher than elsewhere in Tasmanian waters (Eustace 1974; Ratkowsky et al. 1975; Verdouw et al. 2011).
The Derwent estuary is located in the southern part of Tasmania and is close to the capital city Hobart, which has a high level of industrial and urban activities, suggesting the accumulation of not only heavy metals but organic pollutants in flathead is high. Therefore, cytochrome P4501A (CYP1A) gene, a commonly used molecular marker for monitoring organic pollutants such as polyaromatic hydrocarbons (PAHs), polychlorinated biphenyls (PCBs), furans and dioxins (Rees et al. 2003), was employed as an additional marker. The metallothionein (MT) gene, the most commonly used molecular marker of metal pollution in fish, was also assessed in this study. However, previous studies have suggested that the use of the MT gene as a biomarker in fish may be inappropriate as it can be influenced by a variety of abiotic and biotic factors such as salinity, temperature, pH, fish species and tissues (Chen et al. 2004; Cho et al. 2008). Nonetheless, the use of the MT gene as a pollution biomarker has been widely adopted in a range of fish species even though most of the studies assessing the validity of MT as a biomarker were conducted under laboratory conditions (Knapen et al. 2007). The suitability of the MT gene as a molecular marker for metals exposure under field conditions in sand flathead is unknown.
In the present study, P. bassensis from the historically polluted Derwent estuary were used as to explore the relationships between As, Hg and thyroid-related gene expression levels. Thyroid related genes (D1, D2, TTR, TRα and TRβ), CYP1A and MT were cloned and examined from the flathead liver. The hepatic mRNA levels of these genes were compared in flathead from different sites to investigate the effects of heavy metal exposure on thyroid related genes.
Materials and methods
Site descriptions
Three sites were chosen in Derwent River based on historical data showing different levels of As and Hg residues in flathead. The sites are as follows: Cornelian Bay (CB), Kingston Beach North (KBN) and Ralphs Bay (RB) (Fig. 1). Cornelian Bay is located in the upstream of Derwent River and is a graduating area of salt water to fresh water. This site is close to the main industrial inputs, but the previous data have indicated that As and Hg in sand flathead are lower than in flathead of Kingston Beach North and Ralphs Bay respectively (Unpublished data from Zinifex monitoring program, 2011). Kingston Beach North and Ralphs Bay are located on the western shore and eastern shore respectively. These two sites are primarily sea water and are approximately 20 km from the major pollution sources such as Hobart city and Nyrstar. However, the previous studies have shown that sand flathead from Kingston Beach North have the highest As level and the fish from Ralphs Bay have the highest Hg level among a number of sampling sites in Derwent River (Unpublished data from Zinifex monitoring program, 2011).
Sample collection
Juvenile P. bassensis were caught from Kingston Beach North (n = 8), Ralphs Bay (n = 8) and Cornelian Bay (n = 9) between October and November, 2012 (Table 1). Only juvenile fish were used in gene expression analysis. The gender was not identified due to these fish being sexually immature. All the fish used in this study were opportunistically sampled as part of a heavy metal monitoring project conducted by Nyrstar N. V Hobart (a mining and metals industry) under a permit issued under section 14 of the Tasmanian Governments Living Marine Resources Management Act 1995 (2011–2012 Annual Environment Review of Nyrstar N.V). Fish were captured from a small boat using a fishing rod with baited hook. Body weight (W) and standard length (L) were measured for each individual to calculate the condition factor (K) using the following formula: K = 100 × W (kg) × L (cm)−3. For As and Hg analysis, approximately 50 g of muscle was collected from individual fish and then put in a labeled plastic bag placed on ice for transport to the laboratory. All the samples were stored at −20 °C until Hg and As analyses. A sample of liver was collected from a small ventral incision in each fish and immediately stored in a RNA preservation reagent (25 mM sodium citrate, 10 mM EDTA, 4 M ammonium sulphate, pH 5.2). The samples were held on ice until transport to the laboratory and stored at −80 °C until further processing.
Metals determination
All samples were analysed by Analytical Services Tasmania that is accredited by the National Association of Testing Authorities (NATA). The determination of total Hg in each sample was used as a proxy of methylmercury (MeHg) due to the assumption that MeHg is the primary form of Hg in fish muscle (Harris et al. 2003). Samples were digested and analysed using cold vapour atomic fluorescence spectrometry conducted on an atomic fluorescence analyser as described by (Verdouw et al. 2011). For the analysis of other metals including, As, Cd, Co, Cr, Cu, Fe, Zn, Se, Pb, Ni and Mn, approximately 12 g of skinless and boneless muscle samples from individuals were dried and ground into powder. 1 g of each sample was digested and analyzed using an inductively coupled plasma atomic emission spectrophotometer. All the data of residues were given as milligrams per wet kilogram of sample with a minimum detection level of 0.02 mg/kg.
RT-qPCR analysis
Total RNA was isolated from the liver of P. bassensis using TRI Reagent (Molecular Research Centre, OH, USA) and treated with DNAse (Turbo DNase, Ambion, TX, USA) to remove any contaminating genomic DNA. RNA quality was assessed by electrophoresis in a 1.0% agarose gel. The RNA concentrations were determined using an Invitrogen Qubit fluorometer and Quant-iT RNA assay kit (Invitrogen, VIC, Australia). Total RNA (1 µg) was used for the first-strand cDNA synthesis using BioScript reverse transcriptase (Bioline, NSW, Australia) with Oligo (dT) 18 priming as per manufacturers instructions.
Degenerate oligonucleotide primers (Macrogen, South Korea) were designed from the conserved regions of other teleost sequences retrieved from Genbank (Table 2) and used to obtain nine target partial genes by PCR amplification of P. bassensi hepatic cDNA; deiodinase type I (D1), deiodinase type II (D2), transthyretin (TTR) thyroid hormone receptor α (TRα), hormone receptor β (TRβ), cytochrome P4501A (CYP1A), metallothionein (MT), β-actin and elongation factor 1-alpha (EF1α). PCR amplification of each gene was conducted in a volume of 20 μl containing 2× PCR mixtures (Bioline, NSW, Australia), 0.5–0.75 μM of each primer and approximate 50 ng template cDNA. Amplification was carried out on an Eppendorf thermal cycler using the following PCR profile: 2 min at 94 C, then 32 cycles of 94 °C for 30 s, 50–52 °C for 45 s and 72 °C for 1 min, followed by a final extension at 72 °C for 10 min. PCR products were separated via 1.5% agarose gel and purified from gel by using a gel extraction kit (Qiagen, NSW, Australia). Purified PCR products were Sanger sequenced directly in the forward direction (Macrogen). Homologs of these genes were confirmed with published sequences by BLASTx (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and the gene sequences deposited in Genbank (Table 2). The sequence identity of thyroid related genes was analysed by comparing with other known teleost homologues (Table 3).
The deiodinases (D1 and D2), transthyretin (TTR), thyroid hormone receptors (TRα and TRβ), metallothionein (MT), and cytochrome P4501A (CYP1A) mRNA expression were determined by quantitative real-time RT-PCR using gene-specific primers (Table 1). Real-time PCR was performed on an iQ5 Real-time PCR Detection System (Bio-Rad, NSW, Australia) in a volume of 10 μl containing 0.1 μM of each primer, 5 μl 2× SensiMixPlus SYBR & Fluorescein PCR master mix (Bioline, NSW, Australia), and 1 μl of cDNA. The PCR cycling consisted of an initial denaturation at 95 °C for 10 min, followed by 40 cycles of 95 °C for 10 s, 60 °C for 30 s and 72 °C for 20 s with signal collection in each cycle. To assess the specificity of the PCR amplification, a melting curve was obtained at the end of the cycling and a single peak for each target gene was observed. The data analysis of the qPCR results was performed using qBasePlus Biogazelle software (Hellemans et al. 2007). The target genes expression in different samples was normalized to β-actin and EF1α quantities. The geometric mean was used to normalize relative quantity (MNRQ) and was calculated for each gene of grouped replicates from the site. The heavy metal determination data have indicated that fish from CB have the relatively lower As level than those from KBN and have the lower Hg level than fish from RB. Therefore, the expression level of all the genes was relative to that of expression in fish from CB.
Statistical analysis
The Bartlett’s test was used to assess homogeneity of variance for heavy metal data prior to statistical analysis. Spatial differences in Hg and As concentrations and hepatic gene expression were performed using one-way analysis of variance (ANOVA) followed by the Tukey HSD test to determine differences among the sites. To investigate the relationship between gene expression and heavy metal levels, correlation analyses were performed on As, Hg and hepatic genes expression from all fish. The Pearson’s correlation coefficient was used to assess the relationships between parameters (heavy metal levels and relative genes expression after all samples pooled together). All statistical analyses were carried out using GraphPad Prism version 6.0 (GraphPad software Inc) with statistical significance at P < 0.05.
Results
Metal concentrations
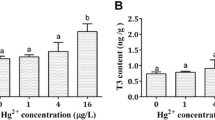
As and Hg residues in flathead muscle from Cornelian Bay, Kingston Beach North and Ralphs Bay showed significant spatial differences (Fig. 2). As residues in flathead from Kingston Beach North were significantly higher than those of Cornelian Bay and Ralphs Bay (P < 0.0001, Fig. 2a).The Hg concentrations in flathead from Cornelian Bay and Kingston Beach North were significantly lower than in Ralphs Bay (P < 0.0001, Fig. 2b). No significant differences were found for the concentrations of Zn and Fe. Other metals were below the minimum detection level (0.02 mg/kg).
Hepatic genes expression
The combined reference genes (EF1α, β-actin) were considered stable by the qBase plus software and therefore suitable for normalisation with a stability value (M-value) of < 0.7 and a CV of 0.25. Hepatic expression levels of D2 mRNA were significantly down-regulated in flathead caught at Ralphs Bay compared to those from Cornelian Bay (CB) (P < 0.05, Fig. 3b), but not significantly different from fish at Kingston Beach North. There was no significant difference among these three sites for the expression levels of D1 (Fig. 3a). For the transcripts of TTR, the highest expression level was observed in fish from Kingston Beach North (3.62-fold that of Cornelian Bay, P < 0.0001, Fig. 3c). Similarly, TRβ mRNA levels were significantly higher in flathead from Kingston Beach North than those from Cornelian Bay and Ralphs Bay (P < 0.0001, Fig. 3e). In contrast, TRα hepatic mRNA levels were not significantly different in flathead sampled at different sites (Fig. 3d). There were no significant differences in hepatic mRNA levels of CYP1A in flathead among all research sites (Fig. 3f). MT mRNA expression was significantly higher in flathead from the Kingston Beach North (KBN) than in the fish from Cornelian Bay (CB) and Ralphs Bay (RB) (P < 0.05) (Fig. 3g).
Hepatic mRNA levels of D1, D2, TTR, TRα, TRβ, CYP1A and MT, relative to two reference genes (β-actin and EF1α) were analysed by qPCR in flathead captured at three sites of Derwent River estuary. The values are shown as geometric means of normalized relative quantity (MNRQ) ± 95% confidence interval. Different letters indicated significant differences between sites
Relationships between gene transcripts, morphometric measurements, Hg and As levels
There were statistically significant positive correlations between As levels and expression of TTR (r = 0.6584, P = 0.0001, Fig. 4a), expression of TRβ (r = 0.6455, P = 0.0002, Fig. 4b) and expression of MT (r = 0.4834, P = 0.0062, Fig. 4g). Significant negative correlations were observed between the levels of Hg and the mRNA expression of D2 (r = −0.4021, P = 0.0232, Fig. 4c), TTR (r = −0.4953, P = 0.0050, Fig. 4d), TRα (r = −0.3663, P = 0.0329, Fig. 4e), TRβ (r = −0.6241, P = 0.0003, Fig. 4f) and MT (r = −0.4101, P = 0.0187, Fig. 4h). No correlation was observed between the morphometric measurements (fork length, weight and condition factor) and gene transcripts (Fig. 5).
Correlations between gene expression and heavy metal concentration in sampled fish. The gene expression of TTR a, TRβ b and MT g were positively correlated with As concentration. The significantly negative correlations were found between D2 c, TTR d, TRα e, TRβ f and MT h gene expressional levels and Hg concentrations
Discussion
D2 mRNA in flathead livers was significantly lower in Ralphs Bay, where flathead had the highest Hg concentrations. In addition, a significantly negative correlation was observed between D2 transcripts and Hg levels for fish from all sites. It has been proposed that the synthesis and activity of 5′-deiodinases are generally inhibited by Hg exposure (Barregard et al. 1994; Mori et al. 2006). In previous studies, Hg exposure was associated with the decreased T4 concentrations of snakehead fish (Channa punctatus) (see Bhattacharya et al. 1989) and decreased both T3 and T4 in plasma of catfish (Clarias batrachus) (see Kirubagaran and Joy 1994). In birds, a recent study of the tree swallow (Tachycineta bicolor) inhabiting an area near a Hg polluted river found that both plasma T3 and T4 were suppressed compared to those in reference sites, suggesting Hg may exert inhibitory effects on 5′-deiodinases (Wada et al. 2009). Deiodinases are selenoproteins, and selenium (Se) is the essential element required by deiodinases (Khan and Wang 2009; Orozco and Valverde-R 2005). Methylmercury (MeHg), the predominant form of Hg in the fish body, could impact on the availability of Se (Raymond and Ralston 2004). In zebrafish (Danio rerio), selenium protein genes could be down-regulated by elevated MeHg level, suggesting MeHg regulates the selenium protein transcripts may be affected through low Se availability (Penglase et al. 2014). More recently, it has been confirmed that low levels of Se in sediments were associated with elevated MeHg bioaccumulation in flathead from the Derwent estuary (Jones et al. 2014). Hence, it is reasonable to hypothesise that the interaction between MeHg and Se may result in decreasing the bioavailability of Se thus contributing to down-regulation of the deiodinase gene. The D1 gene showed a similar spatial expression pattern to D2 but there was no significant effect of site and it was not significantly correlated with Hg levels when all the samples were pooled together. It has been suggested that the teleost D1 is resistant to 5′-deiodinase inhibitors (Orozco and Valverde-R 2005). A field study on walleye (Sander vitreus) has shown that the D1 gene was not as sensitive as the D2 gene as a molecular marker for environmental pollutants (Picard-Aitken et al. 2007). The data in our study suggest D2 gene was sensitive to Hg exposure.
In this study, TTR gene expression levels were negatively correlated with Hg residues in flathead. In teleost fish, the TTR gene has been cloned from few species (Richardson 2009) and therefore the transcriptional regulation of TTR by thyroid endocrine disrupting chemicals is largely unknown. In the rat, it has been reported that hepatic mRNA levels of TTR were significantly decreased due to selenium deficiency (Kendall and Christensen 1997). As stated above, the accumulation of MeHg could cause Se deficiency which results in down-regulation of transcripts of deiodinases. Thus, one possible explanation for the negative correlation between TTR mRNA levels and Hg residues is that Se deficiency in the fish liver may result in a decrease of TTR transcripts. However, further investigations are needed to confirm this and explain the detailed mechanisms.
The expression levels of two TRs genes, TRα and TRβ, were both negatively correlated with Hg concentrations in the flathead. To the best of our knowledge, no studies on the effects of Hg on TRs genes regulations in fish have been reported. Previously, it has been demonstrated that the TRs genes could be regulated by thyroid hormones levels. For instance, a study in frog (Xenopus laevis) reported that expression levels of TRα and TRβ were up-regulated by T3 at 2-fold and 20-fold, respectively (Yaoita and Brown 1990). Similar results were observed in zebrafish (Danio rerio) and striped parrotfish (Scarus iseri), TRα and TRβ transcripts were increased by T3 (Johnson and Lema 2011; Liu et al. 2000). In this context, it can be hypothesized that the down-regulations of these two TRs might be due to the decrease of T3 in the fish livers. Consistent with this, in flathead, the expression levels of D2, the predominant 5′-deiodinase in the fish liver that is responsible for the synthesis of T3, was also negatively correlated with Hg concentrations. However, an additional endpoint, such as measurement of T3 levels in fish liver is required to support this hypothesis.
The mRNA levels of TTR in fish liver were higher in Kingston Beach North and positively correlated with As concentrations in fish body. The precise action of As as a toxin is not known, but it is believed to be a result of As binding to cellular proteins (Yan et al. 2009). Proteins such as tubulin, PARP-1, thioredoxin reductase and estrogen receptor-alpha could be the As binding targets (Kitchin and Wallace 2008). In fish, it has been reported that sea bream (Sparus auratus) TTR has high affinity with a number of common pollutants including loxinyl and several polybrominated diphenyl ethers (PBDEs) (Morgado et al. 2007). In other vertebrates, several environmental pollutants, such as bisphenol A (BPA), nonylphenol and polychlorinated biphenyls (PCBs), could bind to TTR and disrupt the thyroid hormones homeostasis (Simon et al. 2011; Yamauchi et al. 2003). Based on this observation, it could be assumed that As may bind to TTR and up-regulation of TTR mRNA levels could be a requirement of keeping hormone homeostasis in the fish liver.
Similarly, the thyroid hormone receptor gene TRβ was up-regulated in fish from Kingston Beach North as compared to flathead from Cornelian Bay and its transcripts were positively correlated with As concentrations. It has been reported that As has strong disruptive effects on thyroid hormones by altering gene regulation through the thyroid hormone receptors (Davey et al. 2008; Freitas et al. 2011), but the precise mechanism of As interfering with thyroid hormone receptors is still unclear. In the present study, the high expression of TRβ could contribute to maintaining the normal biological processes when TRs were inhibited by As.
No significant differences were found for TRα mRNA expression among sampling sites and there was no correlation with As concentrations. Previous study reported that mRNA of TRα was not changed in the Chinese rare minnow (Gobiocypris rarus) liver but decreased in fish brain when exposed to amitrole (Li et al. 2009). A similar phenomenon was observed in the frog (Xenopus laevis) when TRα was down-regulated in the brain but there was no effect on the transcription of this gene in the tail after MMI treatment (Zhang et al. 2006). These results indicated that TRα expression was organ or tissue-specific (Li et al. 2009). The observation in the present study indicates TRα in flathead liver is not sensitive to As exposure. Therefore, these results may imply that thyroid hormone transporter proteins and their receptors could be the target sites for fish exposed to As and that both TTR and TRβ could be regulated by high levels of As in fish.
The highest expression level of MT in flathead was at Kingston Beach North and its transcript was positively correlated with the concentrations of As in flathead muscles. The toxicity of As has been associated with the generation of reactive oxygen species (ROS) (Kondoh et al. 2001; Thornalley and Vasak 1985). Therefore, high levels of ROS could result in lipid peroxidation and cellular damage. In previous studies, it has been proposed that MTs acting as scavengers of ROS could be induced by cumulative ROS. However, a study on the effects of As compounds on hepatic MT expression in catfish liver, showed the induction of hepatic MT by As was not through an oxidative stress mechanism (Schlenk et al. 1997). Additionally, a recent study in mammals suggested that the metal-activated transcription factor 1 (MTF1) was involved in As induction of metallothioneins (Shen et al. 2013). It has been demonstrated that As covalently bound to the C-terminal cysteine cluster of MTF1, which results in inducing the binding of MTF1 to the metal response elements of metallothioneins (He and Ma 2009). In the present study, the up-regulation of MT was present in flathead from Kingston Beach North where the fish had the highest concentrations of As, suggesting MT in flathead could be induced by As exposure. The positive correlation between As and MT transcripts strongly suggests that MT plays a critical role in protecting the liver cells from arsenic-mediated toxicity. These lines of evidence indicate MT could be used as a molecular marker to monitor the As exposure in the Derwent River.
The failure of MT mRNA expression to reflect Hg bioaccumulation in flathead is consistent with previous studies. For example, none of the investigated organs displayed significant correlations between total Hg and MT levels for two fish species, Dicentrarchus labrax and Liza aurata (Mieiro et al. 2011). Although several investigations have associated an increased MT expression with Hg exposure (Schlenk et al. 1995), the use of fish MTs gene as biomarkers of exposure to Hg is problematic and has been questioned (Mieiro et al. 2011). It is well documented that metallic Hg (II) could induce MT in the injected mouse, while MeHg and Hg (0) are inert in inducing MT(Yasutake and Nakamura 2011). MeHg is considered to be the primary form of Hg in fish muscle (Harris et al. 2003). In our study, MT expression was not regulated at Ralphs Bay where the fish had the highest levels of Hg accumulation, suggesting the mRNA expression of MT is not sensitive for Hg exposure in flathead. Interestingly, a significant negative correlation was observed between the MT mRNA expression levels and the Hg concentrations in flathead. These results suggest that MT mRNA expression is not suitable for monitoring Hg levels in flathead.
In the present study, there were no significant differences among the relative expressions of CYP1A at different sites. The hepatic mRNA expression of the CYP1A gene in fish has been used to reflect the degree of organic pollution, such as PCBs, PAHs and PBDEs (An et al. 2011; Ibhazehiebo et al. 2011; Shirey et al. 2006). The expression profile of CYP1A in the present study supports the previous report of the Derwent Estuary Program (DEP) that organic pollutants were at low levels in the sediments of the Derwent estuary (http://www.derwentestuary.org.au/).
Conclusion
Flathead from sites contaminated with As and Hg showed changes in hepatic thyroid hormone-related gene expression. The transcription of TTR and TRβ genes were positively correlated with As concentration, suggesting they were sensitive to As exposure, while D2, TTR, TRα and TRβ gene expression were suppressed with increased Hg levels. Whether the relationships between these pollutants and thyroid-related genes transcripts are direct contaminant effects or compensatory reactions remain to be confirmed in a controlled laboratory experiment. Apart from this, further studies of the thyroid hormone levels in fish blood, and histological assessment of fish thyroid follicles are required to adequately address the biological impact of the environmental Hg and As exposure.
References
Abernathy CO, Thomas DJ, Calderon RL (2003) Health effects and risk assessment of arsenic. J Nutr 133:1536S–1538S
An LH, Hu JY, Yang M, Zheng BH, Wei A, Shang JJ, Zhao XR (2011) CYP1A mRNA expression in redeye mullets (Liza haematocheila) from Bohai Bay, China. Mar Pollut Bull 62:718–725. doi:10.1016/j.marpolbul.2011.01.019
Ayling G, Wilson K, Ratkowsky D (1975) Sand flathead (Platycephalus bassensis), an indicator species for mercury pollution in Tasmanian waters. Mar Pollut Bull 6:142–144
Barregard L, Lindstedt G, Schutz A, Sallsten G (1994) Endocrine function in mercury exposed chloralkali workers. Occup Environ Med 51:536–540
Bhattacharya T, Bhattacharya S, Ray AK, Dey S (1989) Influence of industrial pollutants on thyroid-function in Channa punctatus (Bloch). Indian J Exp Biol 27:65–68
Bleau H, Daniel C, Chevalier G, vanTra H, Hontela A (1996) Effects of acute exposure to mercury chloride and methylmercury on plasma cortisol, T3, T4, glucose and liver glycogen in rainbow trout (Oncorhynchus mykiss). Aquat Toxicol 34:221–235. doi:10.1016/0166-445x(95)00040-B
Bloom H, Ayling G (1977) Heavy metals in the Derwent estuary environmental. Geology 2:3–22
Brown SB, Adams BA, Cyr DG, Eales JG (2004) Contaminant effects on the teleost fish thyroid. Environ Toxicol Chem 23:1680–1701
Chen WY, John JAC, Lin CH, Lin HF, Wu SC, Lin CH, Chang CY (2004) Expression of metallothionein gene during embryonic and early larval development in zebrafish. Aquat Toxicol 69:215–227. doi:10.1016/j.aquatox.2004.05.004
Cho YS, Lee SY, Kim KY, Bang IC, Kim DS, Nam YK (2008) Gene structure and expression of metallothionein during metal exposures in Hemibarbus mylodon. Ecotoxicol Environ Safe 71:125–137. doi:10.1016/j.ecoenv.2007.08.005
Ciarrocca M et al. (2012) Exposure to arsenic in urban and rural areas and effects on thyroid hormones. Inhal Toxicol 24:589–598. doi:10.3109/08958378.2012.703251
Davey JC et al. (2008) Arsenic as an endocrine disruptor: arsenic disrupts retinoic acid receptor- and thyroid hormone receptor-mediated gene regulation and thyroid hormone-mediated amphibian tail metamorphosis. Environ Health Perspect 116:165–172. doi:10.1289/ehp.10131
Eisler R (2004) Arsenic hazards to humans, plants, and animals from gold mining. In: Ware G (ed.) Reviews of environmental contamination and toxicology. Springer, New York, p 133–165
Ellingsen DG, Efskind J, Haug E, Thomassen Y, Martinsen I, Gaarder PI (2000) Effects of low mercury vapour exposure on the thyroid function in chloralkali workers. J Appl Toxicol 20:483–489
Eustace I (1974) Zinc, cadmium, copper, and manganese in species of finfish and shellfish caught in the Derwent Estuary, Tasmania. Mar Freshw Res 25:209–220
Freitas J, Cano P, Craig-Veit C, Goodson ML, Furlow JD, Murk AJ (2011) Detection of thyroid hormone receptor disruptors by a novel stable in vitro reporter gene assay. Toxicol in Vitro 25:257–266. doi:10.1016/j.tiv.2010.08.013
Green GJ, Coughanowr C (2003) State of the Derwent Estuary: a review of pollution sources, loads and environmental quality data from 1997 to 2003. Department of Primary Industries, Water and Environment.
Harris HH, Pickering IJ, George GN (2003) The chemical form of mercury in fish. Science 301:1203–1203. doi:10.1126/science.1085941
He XQ, Ma Q (2009) Induction of Metallothionein I by arsenic via metal-activated transcription factor 1 critical role of C-terminal cysteine residues in arsenic sensing. J Biol Chem 284:12609–12621. doi:10.1074/jbc.M901204200
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19. doi:10.1186/gb-2007-8-2-r19
Iavicoli I, Fontana L, Bergamaschi A (2009) The effects of metals as endocrine disruptors. J Toxicol Environ Health B 12:206–223. doi:10.1080/10937400902902062
Ibhazehiebo K, Iwasaki T, Kimura-Kuroda J, Miyazaki W, Shimokawa N, Koibuchi N (2011) Disruption of thyroid hormone receptor-mediated transcription and thyroid hormone-induced purkinje cell dendrite arborization by polybrominated diphenyl ethers. Environ Health Perspect 119:168–175. doi:10.1289/ehp.1002065
Johnson KM, Lema SC (2011) Tissue-specific thyroid hormone regulation of gene transcripts encoding iodothyronine deiodinases and thyroid hormone receptors in striped parrotfish (Scarus iseri). Gen Comp Endocr 172:505–517. doi:10.1016/j.ygcen.2011.04.022
Jones H, Swadling K, Butler E, Macleod C (2014) Complex patterns in fish–sediment mercury concentrations in a contaminated estuary: the influence of selenium co-contamination? Estuar Coast Shelf Sci 137:14–22
Kendall SD, Christensen MJ (1997) Selenium deficiency decreases expression of the genes for transthyretin and citrate transport protein in rat liver. Nutr Res 17:351–362. doi:10.1016/S0271-5317(96)00264-3
Khan MAK, Wang FY (2009) Mercury-selenium compounds and their toxicological significance: toward a molecular understanding of the mercury-selenium antagonism. Environ Toxicol Chem 28:1567–1577
Kirubagaran R, Joy KP (1994) Effects of short-term exposure to methylmercury chloride and its withdrawal on serum levels of thyroid-hormones in the catfish Clarias batrachus. Environ Contam Toxicol 53:166–170
Kitchin KT, Wallace K (2008) The role of protein binding of trivalent arsenicals in arsenic carcinogenesis and toxicity. J Inorg Biochem 102:532–539. doi:10.1016/j.jinorgbio.2007.10.021
Knapen D, Reynders H, Bervoets L, Verheyen E, Blust R (2007) Metallothionein gene and protein expression as a biomarker for metal pollution in natural gudgeon populations. Aquat Toxicol 82:163–172. doi:10.1016/j.aquatox.2007.02.008
Kondoh M, Inoue Y, Atagi S, Futakawa N, Higashimoto M, Sato M (2001) Specific induction of metallothionein synthesis by mitochondrial oxidative stress. Life Sci 69:2137–2146. doi:10.1016/S0024-3205(01)01294-2
Langlois D, Cooper RJ, Clark NH, Ratkowsky DA (1987) The Effect of a mercury containment program at a zinc smelting plant on the mercury content of sand flathead in the Derwent estuary. Mar Pollut Bull 18:67–70. doi:10.1016/0025-326x(87)90569-8
Lans M, Klasson-Wehler E, Brouwer A (1994) Thyroid hormone binding proteins as targets for hydroxylated PCB, PCDD and PCDF metabolites; an overview. Organohalogen Compd 20:481–485
Leatherland JF (2000) Contaminant-altered thyroid function in wildlife. Taylor & Francis, New York, NY
Li W, Zha JM, Spear PA, Li ZL, Yang LH, Wang ZJ (2009) Changes of thyroid hormone levels and related gene expression in Chinese rare minnow (Gobiocypris rarus) during 3-amino-1,2,4-triazole exposure and recovery. Aquat Toxicol 92:50–57. doi:10.1016/j.aquatox.2009.01.006
Liu YW, Lo LJ, Chan WK (2000) Temporal expression and T3 induction of thyroid hormone receptors alpha 1 and beta 1 during early embryonic and larval development in zebrafish, Danio rerio. Mol Cell Endocrinol 159:187–195. doi:10.1016/S0303-7207(99)00193-8
Mason RP, Reinfelder JR, Morel FM (1996) Uptake, toxicity, and trophic transfer of mercury in a coastal diatom. Environ Sci Technol 30:1835–1845
Mieiro CL, Bervoets L, Joosen S, Blust R, Duarte AC, Pereira ME, Pacheco M (2011) Metallothioneins failed to reflect mercury external levels of exposure and bioaccumulation in marine fish—considerations on tissue and species specific responses. Chemosphere 85:114–121. doi:10.1016/j.chemosphere.2011.05.034
Morgado I, Hamers T, Van der Ven L, Power DM (2007) Disruption of thyroid hormone binding to sea bream recombinant transthyretin by ioxinyl and polybrominated diphenyl ethers. Chemosphere 69:155–163. doi:10.1016/j.chemosphere.2007.04.010
Mori K, Yoshida K, Tani J, Hoshikawa S, Ito A, Watanabe C (2006) Methylmercury inhibits type II 5 ‘-deiodinase activity in NB41A3 neuroblastoma cells. Toxicol Lett 161:96–101. doi:10.1016/j.toxlet.2005.08.001
Nichols JW, Wedemeyer GA, Mayer FL, Dickhoff WW, Gregory SV, Yasutake WT, Smith SD (1984) Effects of freshwater exposure to arsenic trioxide on the parr‐smolt transformation of coho salmon (Oncorhynchus kisutch). Environ Toxicol Chem 3:143–149
Orozco A, Valverde-R C (2005) Thyroid hormone deiodination in fish. Thyroid 15:799–813. doi:10.1089/thy.2005.15.799
Parry GD, Scientific C (1995) The distribution, abundance and diets of demersal fish in Port Phillip Bay. CSIRO INRE Port Phillip Bay Environment Study.
Patrick L (2009) Thyroid disruption: mechanisms and clinical implications in human health. Altern Med Rev 14:326–346
Penglase S, Hamre K, Ellingsen S (2014) Selenium prevents downregulation of antioxidant selenoprotein genes by methylmercury. Free Radic Biol Med 75:95–104. doi:10.1016/j.freeradbiomed.2014.07.019
Picard-Aitken M, Fournier H, Pariseau R, Marcogliese DJ, Cyr DG (2007) Thyroid disruption in walleye (Sander vitreus) exposed to environmental contaminants: cloning and use of iodothyronine deiodinases as molecular biomarkers. Aquat Toxicol 83:200–211. doi:10.1016/j.aquatox.2007.04.004
Power DM et al. (2001) Thyroid hormones in growth and development of fish. Comp Biochem Phys C 130:447–459. doi:10.1016/S1532-0456(01)00271-X
Power DM, Morgado I, Cardoso JC (2009) Evolutionary insights from fish transthyretin. In: Richardson SJ, Cody V (eds) Recent advances in transthyretin evolution, structure and biological functions, Springer, Berlin, p 59–75
Ratkowsky D, Dix T, Wilson K (1975) Mercury in fish in the Derwent Estuary, Tasmania, and its relation to the position of the fish in the food chain. Mar Freshw Res 26:223–231
Raymond LJ, Ralston NV (2004) Mercury: selenium interactions and health implications. Seychelles Med Dent J 7:72–77
Rees CB, McCormick SD, Vanden Heuvel JP, Li WM (2003) Quantitative PCR analysis of CYP1A induction in Atlantic salmon (Salmo salar). Aquat Toxicol 62:67–78
Richardson SJ (2009) Evolutionary changes to transthyretin: evolution of transthyretin biosynthesis. FEBS journal 276:5342–5356
Schlenk D, Wolford L, Chelius M, Steevens J, Chan KM (1997) Effect of arsenite, arsenate, and the herbicide monosodium methyl arsonate (MSMA) on hepatic metallothionein expression and lipid peroxidation in channel catfish. Comparat Biochem Physiol C 118:177–183. doi:10.1016/S0742-8413(97)00083-2
Schlenk D, Zhang YS, Nix J (1995) Expression of hepatic metallothionein messenger-rna in feral and caged fish species correlates with muscle mercury levels. Ecotoxicol Environ Safe 31:282–286. doi:10.1006/eesa.1995.1075
Shen S, Li X-F, Cullen WR, Weinfeld M, Le XC (2013) Arsenic binding to proteins. Chem Rev 113:7769–7792
Shirey EAL, Langerveld AJ, Mihalko D, Ide CF (2006) Polychlorinated biphenyl exposure delays metamorphosis and alters thyroid hormone system gene expression in developing Xenopus laevis. Environ Res 102:205–214. doi:10.1016/j.envres.2006.04.001
Simon E et al. (2011) Blood plasma sample preparation method for the assessment of thyroid hormone-disrupting potency in effect-directed analysis. Environ Sci Technol 45:7936–7944. doi:10.1021/es2016389
Tan SW, Meiller JC, Mahaffey KR (2009) The endocrine effects of mercury in humans and wildlife. Crit Rev Toxicol 39:228–269. doi:10.1080/10408440802233259
Thornalley PJ, Vasak M (1985) Possible role for metallothionein in protection against radiation-induced oxidative stress—kinetics and mechanism of its reaction with superoxide and hydroxyl radicals. Biochim Biophys Acta 827:36–44. doi:10.1016/0167-4838(85)90098-6
Verdouw JJ, Macleod CK, Nowak BF, Lyle JM (2011) Implications of age, size and region on mercury contamination in estuarine fish species. Water Air Soil Pollut 214:297–306. doi:10.1007/s11270-010-0424-y
Wada H, Cristol DA, McNabb FM, Hopkins WA (2009) Suppressed adrenocortical responses and thyroid hormone levels in birds near a mercury-contaminated river. Environ Sci Technol 43:6031–6038
Ward DM, Nislow KH, Folt CL (2010) Bioaccumulation syndrome: identifying factors that make some stream food webs prone to elevated mercury bioaccumulation. Ann N Y Acad Sci 1195:62–83
Yamano K (2005) The role of thyroid hormone in fish development with reference to aquaculture. JARQ 39:161–168
Yamauchi K, Ishihara A, Fukazawa H, Terao Y (2003) Competitive interactions of chlorinated phenol compounds with 3,3 ‘,5-triiodothyronine binding to transthyretin: detection of possible thyroid-disrupting chemicals in environmental waste water. Toxicol Appl Pharm 187:110–117. doi:10.1016/S0041-008x(02)00045-5
Yan HM, Wang N, Weinfeld M, Cullen WR, Le XC (2009) Identification of arsenic-binding proteins in human cells by affinity chromatography and mass spectrometry. Anal Chem 81:4144–4152. doi:10.1021/ac900352k
Yaoita Y, Brown DD (1990) A correlation of thyroid-hormone receptor gene-expression with amphibian metamorphosis. Gene Dev 4:1917–1924. doi:10.1101/gad.4.11.1917
Yasutake A, Nakamura M (2011) Induction by mercury compounds of metallothioneins in mouse tissues: inorganic mercury accumulation is not a dominant factor for metallothionein induction in the liver. J Toxicol Sci 36:365–372
Zhang F, Degitz SJ, Holcombe GW, Kosian PA, Tietge J, Veldhoen N, Helbing CC (2006) Evaluation of gene expression endpoints in the context of a Xenopus laevis metamorphosis-based bioassay to detect thyroid hormone disruptors. Aquat Toxicol 76:24–36. doi:10.1016/j.aquatox.2005.09.003
Zoeller RT, Tan SW, Tyl RW (2007) General background on the hypothalamic-pituitary-thyroid (HPT) axis. Crit Rev Toxicol 37:11–53. doi:10.1080/10408440601123446
Acknowledgements
We thank the Nyrstar N. V, Hobart, for allowing us to sample on their heavy metal survey program and for providing access to the heavy metals dataset. Mark Stalker, Catarina Norte dos Santos, Stuart Dick and Neil Linton Warnock are thanked for their help in the field. This work was funded by John Bicknell Scholarship and IPRS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Fu, D., Leef, M., Nowak, B. et al. Thyroid hormone related gene transcription in southern sand flathead (Platycephalus bassensis) is associated with environmental mercury and arsenic exposure. Ecotoxicology 26, 600–612 (2017). https://doi.org/10.1007/s10646-017-1793-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-017-1793-4