Abstract
Fish species distribution is commonly influenced by aquatic macrophytes. Despite of the usage of these plants as habitats for refuge, feeding and reproduction by fish, too dense macrophyte stands make microhabitats unsuitable for certain fish species. Moreover, the distance from the open water within macrophyte stands may also affect fish species distribution because of increasingly harsh conditions. In order to test differences in species distribution of small-sized fish within macrophyte stands we sampled stands of Eichhornia spp presenting low and high levels of macrophyte density and at several distances from the open water (0, 3, 6, 9, 12 and 15 m). We measured depth, temperature, dissolved oxygen, conductivity and pH of the water column and attributes of the fish assemblages. We captured 1,167 individuals of fish belonging to 24 species. Oxygen was significantly higher in lower levels of macrophyte density and similar patterns were found for fish abundance and species richness. These results indicate that, in general, small sized-fish prefer less dense macrophyte stands. In addition, both depth and oxygen were significantly higher at the closest distance from open water, where the composition of fish species was also distinct from those found in other distances. In accordance with changes in species composition, different fish species showed divergent distribution along distances from the open water. Together these results demonstrate that oxygen content influences fish species composition, and indicate that fish species are able to use less suitable microhabitats most likely because of morphological, physiological and behavioral adaptations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Small-sized fish species are diverse and abundant in neotropical aquatic ecosystems, where they commonly inhabit macrophyte stands. These fish usually seek macrophytes for refuge (Persson and Eklov 1995; Sánchez-Botero et al. 2007; Meerhoff et al. 2007b) because the physical structure provided by these plants reduce the chance of being visualized by active predators (Kovalenko et al. 2012), and consequently, they become less likely to be predated (Rozas and Odum 1988). In addition, small-sized fish may also be attracted toward macrophytes because of food availability (Werner et al. 1983a; Werner et al. 1983b). The structure provided by these plants commonly harbor a great density and diversity of periphytic algae (Rodrigues et al. 2003; Schneck et al. 2011) and associated invertebrates (Tanigushi et al. 2003; Warfe and Barmuta 2006; Thomaz et al. 2008), which are, in turn, the most common feeding items consumed by small-sized fish species (Agostinho et al. 2003; Sánchez-Botero et al. 2007). Considering such conditions of safety and food availability, these fish species may grow and reproduce successfully in macrophyte stands (Conrow et al. 1990; Sánchez-Botero and Araújo-Lima 2001; Sánchez-Botero et al. 2007), explaining the common use of these plants as habitats.
Despite of commonly using structured habitats in contrast to open areas, small-sized fish may select more dense or sparse vegetation depending on how they interact with the habitat. For example, small-sized fish may avoid too dense vegetation stands because of conditions of hypoxia or because macrophyte steams, leaves and roots work as a physical barrier, hampering the movement and obstructing the vision (Harrel and Dibble 2001). Alternatively, some fish species may particularly use habitats provided by dense vegetation due to specific biological adaptations (e.g., tolerance to low levels of oxygen) and particular strategies of life (e.g., ambushers).
In addition to being affected by differences in vegetation density, the spatial distribution of small-sized fish may differ within macrophyte stands (e.g., Agostinho et al. 2007). The patch formed by these plants commonly provide a mosaic of physical and chemical conditions (Miranda et al. 2000), which is most likely driven by differences in the morphology of lakes and decreasing influence of limnetic zone as the distance from the open water increases (e.g., Agostinho et al. 2007). Consequently, the distribution of small-sized fish is a result of tradeoffs between fish avoidance and selectivity regarding environmental aspects.
Studies describing the distribution of small-sized fish in macrophyte stands commonly focused in testing differences between vegetated and non-vegetated environments (Agostinho et al. 2003; Pelicice et al. 2005; Gomes et al. 2012), between stands of macrophytes composed by different macrophyte species (Fernandez et al. 1998; Meschiatti et al. 2000; Suarez et al. 2001; Vono and Barbosa 2001; Sánchez-Botero et al. 2003; Meerhoff et al. 2003, 2007a; Prado et al. 2010; Dibble and Pelicice 2010), between macrophytes providing different habitat complexities (Dibble and Pelicice 2010) and between stands of macrophytes of the same species that vary across spatial scales (Delariva et al. 1994; Pacheco and Da-Silva 2009; Teixeira de Mello et al. 2009; Milani et al. 2010). Despite of this, as far as we know, no investigations concerned to describe concomitantly the distribution of small-sized fish species along a refined gradient of macrophyte patches and vegetation density. This may provide basis for a better understanding of the distribution of small-sized fish within macrophyte stands.
Regarding these aspects, we hypothesized that abundance, species richness, evenness and composition of small-sized fish using habitats of different levels of vegetation density and position along macrophyte stands differ. In addition, we hypothesized that the distribution of different species along the horizontal gradient differ because of particular biological aspects and adaptations of fish species. The rationale behind these hypotheses relies on the differences concerning habitat use. In addition, in this study we aimed to describe the variation in the spatial use of habitat by small-sized fish in different vegetation density and in different location of horizontal gradients within macrophyte stands. In order to achieve this objective we sampled large stands of Echhornia azurea (~300 m2), a rooted floating leaved macrophyte, which are broadly distributed in neotropical aquatic ecosystems and provide habitat for small-sized fish.
Materials and methods
Study area
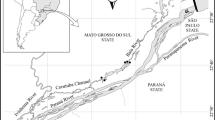
We surveyed three different lakes (Maria Luiza, Careca and Onça) connected to Baia River, a tributary of the upper Paraná River floodplain (Fig. 1). The lakes are shallow (3 m on average) and have extensive stands of aquatic macrophytes along their shores, colonizing the littoral zone for more than 15 m towards open water. Most common macrophyte species dominating the waterscape is Eichhornia azurea (Sw.) Kunth (rooted floating leaves), but some Eichhornia crassipes (Mart.) Solms, a free floating species which is quite similar regarding morphology, also occur. Taking into account the typical configuration of the stands of aquatic macrophytes, we considered for sampling those stands which were at least 300 m2 (20–40 m along the margins and 15–25 m towards open water), ensuring large availability of habitat for fish and reduced effect of borders.
Sampling
In December 2011, two macrophyte stands were haphazardly selected in each lake, with at least 100 m of distance between each other. As a rule-of-thumb, one of the stands presented a visually expressive higher macrophyte density (termed hereafter vegetation density) than the other. This characterizes two levels of vegetation density as habitat for fish, for which statistical differences were formalized after the fact (see methods and results). In each stand, we determined six different distances from the threshold between the pelagic zone and macrophyte patch toward the shore (0, 3, 6, 9, 12 and 15 m), characterizing habitat for fish regarding six different levels of separation from open water. Distances were considered from the threshold between the pelagic zone toward the shore due to three main reasons. First, this area commonly harbor great density of fish; second, some stands may extend further than 15 m towards open water; and third, the morphology of floodplain lakes produce tenuous shorelines, usually presenting harsh conditions for fish.
In each combination of vegetation density and distance from the open water we characterized physical and chemical aspects of water column, measured plant biomass and sampled fish. Physical and chemical characteristics of the water column were described by depth (cm), temperature (°C), dissolved oxygen (mg.L-1), conductivity (μS.cm-1) and pH. Measurements were taken twice, early in the morning and in the evening, and mean values were used for analyses. The plant biomass were measured by removing all the submerged structures of macrophytes (including roots and stems) inside a 0.5 × 0.5 m square and drying (60 °C) to constant weight. Values were expressed as gDW.m-3.
Fish were sampled using plexigas-type minnow traps (described elsewhere in Dibble and Pelicice 2010 and Cunha et al. 2011) (Fig. 6 in Appendix), which were set early in the morning and checked for fish each 12 h to a total sampling effort of 24 h. This type of gear captures small-sized fish (size range: 0.6–4.7 cm; mean size: 2.4 cm) (Dibble and Pelicice 2010), which are the most common inhabiting macrophyte stands, and provide an representative overview of the assemblage by capturing fish in an effective way and requiring no changes in habitat structure to set up the device (Ribeiro and Zuanon 2006). Fish captured along the 24 h were counted and identified to species according to Graça and Pavanelli (2007) and Mirande (2010), and expressed in terms of abundance index (individuals per trap per 24 h), species richness and evenness.
Data analysis
We used two-way ANOVA to test differences in depth, temperature, dissolved oxygen, conductivity, pH, plant biomass, fish abundance, species richness and evenness between vegetation densities and distances from the open water. Tukey tests were used as a posteriori comparisons in order to assess differences between treatments. These analyses were run using the Statistica™ software (Statsoft 2007).
To summarize the structure of the fish assemblage we used a non-metric multidimensional scaling (NMDS) on the Bray-Curtis resemblance matrix obtained from square root transformation of relative abundance of species (Legendre and Gallagher 2001). Differences between vegetation density, distances from the open water and their interaction for species composition were assessed using a permutational analysis of variance (PERMANOVA main test) applied to the Bray-Curtis resemblance matrix (Anderson et al. 2008). When the PERMANOVA was significant, we conducted a pair-wise test to assess differences between treatments. Monte Carlo permutations (n = 9999) were used to test for statistical significance (p < 0.05). These analyses were run using the PRIMER 6 with the add on PermanovaTM software (Clarke and Gorley 2006; Anderson et al. 2008).
The relationships of the environmental variables, other than vegetation density and distance from the open water, with the fish assemblage were evaluated using BioEnv (Clarke and Ainsworth 1993). The purpose of BioEnv is to find the best match between the multivariate among-sample patterns of an assemblage and the environmental variables. BioEnv creates models using all possible combinations of environmental variables. The extent to which the two patterns expressed as dissimilarity matrices match reflects the degree to which the chosen abiotic data are related with the biotic patterns (Clarke and Gorley 2006). For this analysis the abundance matrix was square-root-transformed to obtain Bray-Curtis dissimilarity matrix and environmental variables were normalized before obtaining the environmental matrices based on Euclidean distance. The Spearman correlation coefficient (ρ) was used to measure the association between dissimilarity matrices. This analysis were run using the PRIMER 6 software with the add on PermanovaTM (Clarke and Gorley 2006; Anderson et al. 2008).
Additionally, to investigate the preference of most abundant species in regard to habitat use along horizontal gradients we tested differences in optimum abundance of fish along transects between fish species and vegetation densities. The optimum abundance indicates at which point of a gradient a given species presents the highest abundance. To estimate the optimum abundance we used the weighted averaging approach, which consists in average the distances where the species occur weighted by its abundance (Šmilauer and Lepš 2014). For calculation, we summed 1 to the values of distance from the open water in order to avoid weighting for 0 and before statistical analysis and graphical representation this constant were subtracted. We used two-way ANOVA to test differences in optimum abundance along transects between vegetation densities, most abundant fish species, and their interactions. Tukey tests were used as a posteriori comparisons in order to assess differences between treatments. These analyses were run using the Statistica™ software (Statsoft 2007).
Results
As the distance within the macrophyte stands increased from the pelagic towards the littoral zone, the depth decreased for both levels of vegetation density (stands with high and low densities) (Fig. 2a). Depth varied significantly with distances from the open water (two-way ANOVA; F = 3.76; p = 0.01), but not with vegetation density or interaction (two-way ANOVA; F < 0.13; p > 0.50). The depth at the distance of 0 m differed from the depth at distances of 12 m (Tukey test; p = 0.04) and 15 m (p = 0.03) (Fig. 2a). Water temperature did not differ according to any factor (two-way ANOVA; F < 0.06; p > 0.98) (Fig. 2b).
Mean and standard error of depth (cm) (a), temperature (°C) (b), dissolved oxygen (mg.L-1) (c), conductivity (μS.cm-1) (d), pH (e) and plant biomass (gDW m-3) (f) in macrophyte stands with low (grey) and high (black) vegetation densities (M) and at distances (D) of 0, 3, 6, 9, 12 and 15 meters from open water toward shoreline. Values in bold correspond to p < 0.05 in a two-way ANOVA
The concentration of oxygen was, in general, greater in the stands with less vegetation density. In general, the highest concentration of oxygen was registered near the pelagic zone (at distance 0 m), and it decreased with increasing distance from the open water (Fig. 2c). This variable differed significantly between levels of vegetation density (two-way ANOVA; F = 8.28; p < 0.01) and distance from the open water (two-way ANOVA; F = 15.60; p < 0.01), but the interaction was not significant (two-way ANOVA; F = 2.22; p = 0.08). Considering pair-wise comparisons, only the concentration of oxygen registered at the distance of 0 m from open water differed from the oxygen concentrations registered in other distances (Tukey test; p < 0.01) (Fig. 2c).
Regarding mean conductivity, this variable generally presented higher values in stands with higher vegetation density, resulting in statistical significant differences (two-way ANOVA; F = 9.66; p < 0.01). Despite of decreasing values of this variable in response to distance from the open water in the stands with low density, no statistical differences were detected for this factor or for the interaction term (two-way ANOVA; F < 2; p > 0.10) (Fig. 2d). Finally, pH did not differ according to any factor (two-way ANOVA; F < 0.80; p > 0.58; check Table 2 in Appendix for more detailed information about variables) (Fig. 2e).
Plant biomass was significantly different between the stands with low and high levels of vegetation density (two-way ANOVA, F = 9.14, p = 0.006), thus confirming the previous visual determination of vegetation density treatments (Fig. 2f). Despite of the difference between low and high vegetation densities, no differences were detected for distance from the open water or for the interaction term (F < 0.35; p > 0.50) (Fig. 2f).
Regarding fish assemblage, a total of 1,167 individuals were collected in the stands. They belonged to two orders (Characiformes and Perciformes), seven families, 18 genera and 24 species of fish. The most abundant species were in general characids, represented by Hyphessobrycon eques (Steindachner, 1882) (n = 249), Moenkhausia bonita (Benine, Castro & Sabino, 2004) (n = 248), Serrapinnus heterodon (Eigenmann, 1915) (n = 179), Aphyocharax anisitsi (Eigenmann & Kennedy, 1903) (n = 147), Moenkhausia aff. sanctaefilomenae (Steindachner, 1907) (n = 90) and Psellogrammus kennedyi (Eigenmann, 1903) (n = 77) (Table 3 in Appendix).
Considering the abundance of fish, this variable significantly differed between vegetation densities (two-way ANOVA; F = 10.2; p < 0.01); this response variable was higher in stands with low vegetation density (Fig. 3a). With regards to species richness, this variable differed significantly only between levels of vegetation density (two-way ANOVA; F = 7.06; p = 0.01). Despite the low richness near to the pelagic zone (0 m from the open water), when compared to richness at other distances from the open water, neither the distance from the open water nor the interaction term was significant (two-way ANOVA; F < 2.1; p > 0.10) (Fig. 3b). Evenness was also higher in stands with less density, but the factors and their interaction had no significant effects (two-way ANOVA; F < 2.2; p > 0.15) (Fig. 3c).
Mean and standard error of the fish assemblage attributes: abundance (a), species richness (b) and evenness (c) in macrophyte stands with low (grey) and high (black) levels of vegetation density (M) at distances from the open water (D) varying from 0 to 15 meters. Values in bold correspond to p < 0.05 of a two-way ANOVA
The two-dimensional configuration of the NMDS presented a good representation of the data (stress = 0.17 after 9999 interactions). In general, composition of fish species overlapped in different levels of vegetation density, indicating that fish assemblages did not differ (PERMANOVA; Pseudo-F = 2.6; p = 0.30; Fig. 4a). Despite of the high variability found among distances from the open water, this variable significantly affected fish species composition (Fig. 4b; PERMANOVA; Pseudo-F = 2.2; p < 0.01). This difference was evidenced by the fish assemblage sampled at the distance of 0 m, which was the only level of distance from the open water differing from other levels (Pair-wise comparisons; p < 0.05 for comparisons between 0 m and all other distances). Considering the interaction between vegetation density and distance from the open water, no significant difference was detected (PERMANOVA; Pseudo-F = 1.1; p = 0.37).
The BioEnv analysis showed all possible combinations of physical and chemical variables related to abundance of fish species. The ten models with the highest Spearman correlations (ρ) were selected to demonstrate the most important variables (Table 1). Dissolved oxygen was present in all of the best models (10), followed by pH (7) and depth (6). Therefore, among the considered variables, dissolved oxygen was the most important in explaining assemblage structure (ρ = 0.44, p < 0.01 (Table 1)).
The optimum abundance of small-sized fish along horizontal gradients differed between fish species (two-way ANOVA; F = 4.18; p < 0.05), but no differences were found between levels of vegetation density (two-way ANOVA; F = 3.5; p = 0.07) or interaction term (two-way ANOVA; F = 1.28; p = 0.29; Fig. 5). In general, M. bonita presented its optimum abundance in regions close the open water, and differed from H. eques, S. heterodon and P. kennedyi in terms of habitat use (Tukey test; p < 0.05). Both H. eques and S. heterodon presented greatest abundances from the middle of the transect to the farthest distances from the open water, differing significantly from S. notomelas. Psellogrammus kennedyi had its distributions restricted to the regions farthest from the open water, but was only significant different from S. notomelas (Tukey test; p < 0.05). The species H. eques, M. aff. sanctaefilomena and S. heterodon was found in all of the distances from the open water and vegetation density of stands (Table 3 in Appendix).
Optimum abundance of small-sized fish along distances from the open water in macrophyte stands with low (grey) and high (black) levels of vegetation density (M) and different species of fish (S). Horizontal markers and rectangles respectively indicate mean and standard error of the distances from the open water where individuals of fish species most commonly occurred
Discussion
The mean number of fish species and abundance were lower in macrophyte stands with high vegetation density than stands with low vegetation density, indicating that in general small sized-fish prefer less dense macrophyte stands. These patterns may be a result of interaction with different aspects of the environment. For example, species richness decreased with gradually distances from the open water toward shoreline only in stands with low vegetation density. These trends in habitat use by different species were probably related to the reduction of dissolved oxygen, given that oxygen content followed the same patterns. The variability in oxygen concentration is commonly associated with the shallow waters of the littoral region and the horizontal variation in vegetation density (through the width of the stand) and can be explained by different but not excluding mechanisms.
The morphology of floodplain lakes is generally marked by greater depths in the center than near the margins due to depositional processes. This morphology was evident in this study among distances closer to the open water that did not significantly differ in depth. Thus, a greater distance from the limnetic region (i.e., further inside the stand and closer to the shoreline) indicates a shallower depth and a greater influence of the heterotrophic process in the sediment, which in turn lead to low pH because of oxygen consumption caused by decomposition (Miranda and Hodges 2000; Esteves and Gonçalves 2011). The difference between the vegetation density of the stands and the horizontal variation in plant biomass (distances from the open water) can be caused by differences in the densities of roots and leaves, the degree of maturity of the macrophytes, the stage of colonization of epiphyton and the species composition (Miranda et al. 2000). In addition, the stands of macrophytes in this study were composed predominantly of the E. azurea, a species with an emergent photosynthetic portion and root structures disposed in the water column. The emergent structures resist the wind action on the water surface, which hampers the diffusion of atmospheric oxygen (Sand-Jensen 1989). These kinds of macrophytes also release most part of the oxygen produced by photosynthesis directly to the atmosphere (Pokorný and Rejmánková 1984; Goodwin et al. 2008). Moreover, macrophyte roots retains organic matter (Poi de Neiff et al. 1994), thereby raising oxygen consumption. All of these factors contribute to the low oxygen concentrations in regions close to the shoreline (distances higher than 15 m from the open water), where vegetation tends to be denser, presenting harsh conditions to fish species.
Plant biomass may also lead to abiotic constraints that affect the distribution of small-sized fish. When the entire stand was considered, the ones with higher vegetation density had fewer fishes probably due to the physical restrictions. Regarding the position within the stands, the difference in the fish assemblages of the limnetic region (0 m from open water) compared with those inside the macrophytes stands was previously expected, given that it has been demonstrated in other studies conducted in the same area (Agostinho et al. 2007; Gomes et al. 2012). Though the oxygen content was adequate in the limnetic region, the fish species richness was higher in stands with lower density and at the distance of 3 m from open water toward shoreline for both levels of vegetation density, which is in accordance with the results obtained by Agostinho et al. (2007). They find the highest fish richness near the edge of the macrophyte stands. In the limnetic region (at distance 0 m), we also found lower values for species richness and evenness due to the dominance of M. bonita. The preference of small-bodied fishes for the edges of the stands is related to specific levels of vegetation density offered by these environments, which can reduce predation (Sánchez-Botero et al. 2003; Pelicice et al. 2005), and to the widespread availability of food resources due to the adequate concentrations of oxygen.
Despite the changes with increasing distance from the open water, we did not find significant differences in the assemblage structure in relation to this environmental variable. One reason for this was that the range of plant biomass was not a continuum, i.e., vegetation densities varying proportionally with the distance from the edge, and so, detectable changes in assemblage structure are restricted to closest distance from the open water. Other explanation consider the fact that in some stands the vegetation density decreased at distances of 12 and 15 m, forming oxygenated patches. This may occasionally favor more species of small-sized fish increasing the variation in assemblage structure.
Notwithstanding, we found that the most abundant species seems to select a preferred habitat within macrophyte stands. Moenkhausia bonita, for instance, most commonly used the borders of the macrophyte stand (lower distances within the stands) and this might probably mirrors adaptive strategies of exploiting high quality resources, while successfully protecting themselves through schooling behavior. This species are commonly found foraging in groups from 10 to 30 individuals (Benine et al. 2004). In contrast, a congeneric species, M. aff. sanctaefilomenae, does not share the microhabitat with M. bonita, which may indicate displacement caused by lower competitive capabilities. Despite of these, the adaptive plasticity of M. aff. sanctaefilomenae may allow these species to coexist in broader scale (stand scale), which is also suggested by its feeding plasticity (Crippa et al. 2009). Similar to M. aff. sanctaefilomenae, A. anisitsi and S. notomelas coexist because of the food segregation (Hahn and Crippa 2006).
Otherwise, H. eques and P. kennedyi were quietly restricted to farthest distances from the open water. The distribution of these species remarkably mirrors adaptations to low oxygen conditions, which is also a reasonable explanation to the distribution of other species which were found in many levels of vegetation density and distances from the open water, even at oxygen concentrations below 1.5 mg.L-1. Previous studies show that species of the genera Hyphessobrycon, Aphyocharax, Astyanax and Acestrorhynchus may develop dermal lip protuberance, an adaptation for breathing in the water-air layer, in conditions of hypoxia (Scarabotti et al. 2011). Species sampled in our study such as H. eques, A. anisitsi, Astyanax altiparanae (Garutti & Britski, 2000) and Acestrorhynchus lacustris (Lütken, 1875) should also have the same adaptation, considering phylogenetic conservatism (Winemiller 1989; Scarabotti et al. 2011). Other abundant species found in macrophyte stands such as M. aff. sanctaefilomenae, S. heterodon and P. kennedyi are considered opportunistic species, which are commonly adapted to survive in extreme conditions (Winemiller 1992; Lourenço et al. 2008; Gonçalves et al. 2011). Other examples of tolerance to low oxygen conditions include less common species, for example, Hoplerythrinus unitaeniatus (Agassiz, 1829) and Erythrinus erythrinus (Bloch & Schneider, 1801). These species are facultative air breathers (Oyakawa 2003), and have the ability to diffuse atmospheric oxygen into their bloodstream through the swim bladder, capturing gas through frequent ascents to the water surface (Lima-Filho et al. 2012).
In summary, our findings suggest that fish may use different levels of vegetation density and position within stand of E. azurea, which is the most widespread macrophyte structuring habitat for fish in neotropical ecosystems. Despite of some restrictive conditions, the horizontal gradient may mediate segregation between different species in terms of distribution. Thus, these results indicate that environmental factors may be determinant to habitat specialization within large stands of macrophytes. This is probably favored by morphological respiratory (air breathing and superficial air breathing), physiological and behavioral adaptations.
References
Agostinho AA, Gomes LC, Julio HF Jr (2003) Relações entre macrófitas e fauna de peixes. In: Thomaz SM, Bini LM (eds) Ecologia e manejo de macrófitas aquáticas. Eduem, Maringá, pp 261–279
Agostinho AA, Thomaz SM, Gomes LC, Baltar SLMA (2007) Influence of the macrophyte Eichhornia azurea on fish assemblage of the upper Paraná river floodplain (Brazil). Aquat Ecol 41:611–619
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA for PRIMER: Guide to software and statistical methods. PRIMER-E, Plymouth
Benine RC, Castro RMC, Sabino J (2004) Moenkhausia bonita: A New Small Characin Fish from the Rio Paraguay Basin, Southwestern Brazil (Characiformes: Characidae). Copeia 1:68--73
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–219
Clarke KR, Gorley RN (2006) PRIMER v6: User manual/tutorial. PRIMER-E, Plymouth
Conrow R, Zale AV, Gregory RW (1990) Distributions and abundances of early life stages of fishes in a Florida lake dominated by aquatic macrophytes. Trans Am Fish Soc 119:521–528
Crippa VEL, Hahn NS, Fugi R (2009) Food resource used by small-sized fish in macrophyte patches in ponds of the upper Paraná river floodplain. Acta Sci Biol Sci 31:119--125
Cunha ER, Thomaz SM, Evangelista HBA, Carniato J, Souza CF, Fugi R (2011) Small-sized fish assemblages do not differ between a native and a recently established non-indigenous macrophyte in a neotropical ecosystem. Nat Cons 9:61–66
Delariva RL, Agostinho AA, Nakatani K, Baumgartner G (1994) Ichthyofauna associated to aquatic macrophytes in the upper Paraná river floodplain. Rev Unimar 16(3):41–60
Dibble ED, Pelicice FM (2010) Influence of aquatic plant-specific habitat on an assemblage of small neotropical floodplain fishes. Ecol Freshw Fish 9:381–389
Esteves FA, Gonçalves JF Jr (2011) Etapas do metabolismo aquático. In: Esteves FA (ed) Fundamentos de limnologia. Interciência, Rio de Janeiro, pp 119–124
Fernandez OA, Murphy KJ, Lopes-Cazorla A, Sabbatini MR, Lazzari MA, Domaniewski JCJ, Irigoyen JH (1998) Interrelationship of fish and channel environmental conditions with aquatic macrophytes in an Argentine irrigation system. Hydrobiologia 380:15–25
Gomes LC, Bulla CKC, Agostinho AA, Vasconcelos LP, Miranda LE (2012) Fish assemblage dynamics in a Neotropical floodplain relative to aquatic macrophytes and the homogenizing effects of a flood pulse. Hydrobiologia 685:97–107
Gonçalves CS, Souza UP, Braga FMS (2011) Population structure, feeding and reproductive aspects of serrapinnus heterodon (characidae, cheirodontinae) in a Mogi guaçu reservoir (SP), upper Paraná river basin. Biol Limnnol 23(1):13–22
Goodwin K, Caraco N, Cole J (2008) Temporal dynamics of dissolved oxygen in a floating-leaved macrophyte bed. Freshw Biol 53:1632–1641
Graça WJ, Pavanelli CS (2007) Peixes da planície de inundação do Alto rio Paraná e áreas adjacentes. Eduem, Maringá
Hahn NS, Crippa VEL (2006) Estudo comparativo da dieta, hábitos alimentares e morfologia trófica de duas espécies simpátricas, de peixes de pequeno porte, associados à macrófitas aquáticas. Acta Sci Biol Sci 28:359--364
Harrel SL, Dibble ED (2001) Foraging efficiency of juvenile bluegill, Lepomis macrochirus, among different vegetated habitats. Env Biol Fish 62:441–453
Kovalenko KE, Thomaz SM, Warfe DM (2012) Habitat complexity: approaches and future directions. Hidrobiologia 685:1--17
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Ecology 129:271–290
Lima-Filho JA, Martins J, Arruda R, Carvalho LN (2012) Air-breathing behaviour of the jeju fish Hoplerythrinus unitaeniatus in Amazonian Streams. Biotropica 44:512–520
Lourenço LS, Mateus LA, Machado NG (2008) Sincronia na reprodução de Moenkhausia sanctaefilomenae (steindachner) (characiformes: characidae) na planície de inundação do rio cuiabá, pantanal mato-grossense, Brasil. Revi Bras Zool 25(1):20–27
Meerhoff M, Mazzeo N, Moss B, Rodriguez-Gallego L (2003) The structuring role of free-floating versus submerged plants in a subtropical shallow lake. Aquat Ecol 37:377–391
Meerhoff M, Clemente JM, Teixeira de Mello F, Iglesias C, Pedersen AR, Jeppesen E (2007a) Can warm climate-related structure of littoral predator assemblies weaken clear water state in shallow lakes? Global Change Biol 13:1888–1897
Meerhoff M, Iglesias C, Teixeira de Mello F, Clemente JM, Jensen E, Lauridsen TL, Jeppesen E (2007b) Effects of habitat complexity on community structure and predator avoidance behavior of littoral zooplankton in temperate versus subtropical shallow lakes. Freshw Biol 52:1009–1021
Meschiatti AJ, Arcifa MS, Fenerich-Verani N (2000) Fish communities associated with macrophytes in Brazilian floodplain lakes. Environ Biol Fish 58:133–143
Milani V, Machado FA, Silva VCF (2010) Assembleias de peixes associados às macrófitas aquáticas em ambientes alagáveis do Pantanal de Poconé, MT, Brasil. Rev Biol Neotrop 10:261–270
Miranda LE, Hodges KB (2000) Role of aquatic vegetation coverage on hypoxia and sunfish abundanc in bays of eutrophic reservoir. Hydrobiologia 427:51–57
Miranda LE, Driscoll MP, Allen MS (2000) Transient physicochemical microhabitats facilitate fish survival in inhospitable aquatic plant stands. Freshw Biol 44:617–628
Mirande JM (2010) Phylogeny of the family Characidae (Teleostei: Characiformes): from characters to taxonomy. Neotrop Ichthyol 8(3):385–568
Oyakawa OT (2003) Family erythrinidae. In: Reis RE, Kullander SO, Ferraris CJ Jr (eds) Check list of the freshwater fishes of south and Central America. Edipucrs, Porto Alegre, pp 238–240
Pacheco EB, Da-Silva CJ (2009) Fish associated with macrophytes in the Chacororé-Sinhá Mariana lake system and Mutum river, Pantanal of Mato Grosso Brazil. Braz J Biol 69:101–108
Pelicice FM, Agostinho AA, Thomaz SM (2005) Fish assemblages associated with Egeria in a tropical reservoir: investigating the effects of plant biomass and diel period. Acta Oecol Int J Ecol 27:9–16
Persson L, Eklov P (1995) Prey refuges affecting interactions between piscivorous perch and juvenile perch and roach. Ecology 76:70–81
Poi de Neiff A, Neiff JJ, Orfeo O, Carignan R (1994) Quantitative importance of particulate matter retention by the roots of Eichhornia crassipes in the Paraná floodplain. Aquat Bot 47:213–223
Pokorný J, Rejmánková E (1984) Oxygen regime in a fishpond with duckweeds (Lemnaceae) and Ceratophyllum. Aquat Bot 17:125–137
Prado KLL, Freitas CEC, Soares MGM (2010) Assembleias de peixes associadas às macrófitas aquáticas em lagos de várzea do baixo rio Solimões. Biotemas 23:131–142
Ribeiro OM, Zuanon J (2006) Comparação da eficiência de dois métodos de coleta de peixes em igarapés de terra firme da Amazônia Central. Acta Amazon 36(3):389–394
Rodrigues L, Bicudo DC, Moschinicarlos V (2003) O papel do perifíton em áreas alagáveis e nos diagnósticos ambientais. In: Thomaz SM, Bini LM (eds) Ecologia e Manejo de Macrófitas Aquáticas. Eduem, Maringá, pp 211–230
Rozas LP, Odum WE (1988) Occupation of submerged aquatic vegetation by fishes: testing the roles of food and refuge. Oecologia 7:101–106
Sánchez-Botero JI, Araújo-Lima CARM (2001) As macrófitas aquáticas como berçário para a ictiofauna da várzea do rio Amazonas. Acta Amazon 3:437–447
Sánchez-Botero JI, Farias ML, Piedade MT, Garcez DS (2003) Ictiofauna associada às macrófitas aquáticas Eichhornia azurea (SW.) Kunth. e Eichhornia crassipes (Mart.) Solms. no lago Camaleão, Amazônia Central, Brasil. Acta Sci Biol Sci 25:369–375
Sánchez-Botero JI, Leitão RP, Caramaschi EP, Garcez DS (2007) The aquatic macrophytes as refuge, nursery and feeding habitats for freshwater fish from Cabiúnas lagoon, restinga de Jurubatiba National Park, Rio de Janeiro, Brazil. Acta Limnol Brasil 19:143–153
Sand-Jensen K (1989) Environmental variables and their effect on photosynthesis of aquatic plant communities. Aquat Bot 34:5–25
Scarabotti PA, López JA, Ghirardi R, Parma MJ (2011) Morphological plasticity associated with environmental hypoxia in characiform fishes from neotropical floodplain lakes. Env Biol Fish 92:391–402
Schneck F, Schwarzbold A, Melo AS (2011) Substrate roughness affects stream benthic algal diversity, assemblage composition, and nestedness. J N Am Benthol Soc 30:1049–1056
Šmilauer P, Lepš J (2014) Multivariate Analysis of Ecological Data using CANOCO 5 (2nd Ed.). Cambridge University Press, New York, 373 p
Statsoft (2007) http://www.statsoft.com/textbook/. Accessed 20 June 2012
Suarez YR, Petrere M Jr, Catella AC (2001) Factors determining the structure of fish communities in pantanal lagoons (MS, Brazil). Fisheries Manag Ecol 8:173–186
Tanigushi H, Shigeru N, Tokeshi M (2003) Influences of habitat complexity on the diversity and abundance of epiphytic invertebrates on plants. Freshw Biol 48:718–728
Teixeira de Mello F, Meerhooff M, Pekcan-Hekim Z, Jeppensen E (2009) Substantial diferences in litoral fish community structure and dynamics in subtropical and temperate shallow lakes. Freshw Biol 54:1202--1215
Thomaz S, Dibble E, Evangelista L, Higuti J, Bini L (2008) Influence of aquatic macrophyte habitat complexity on invertebrate abundance and richness in tropical lagoons. Freshw Biol 53:358–367
Vono V, Barbosa FAR (2001) Habitats and littoral zone fish community structure of two natural lakes in southeast Brazil. Environ Biol Fish 61:371–379
Warfe DM, Barmuta LA (2006) Habitat structural complexity mediates food web dynamics in a freshwater macrophyte community. Oecologia 150:141–154
Werner EE, Gilliam JF, Hall DJ, Mittelbach GG (1983a) An Experimental Test of the Effects of Predation Risk on Habitat Use in Fish. Ecology 64(6):1540--1548
Werner EE, Mittelbach GG, Hall DJ, Gilliam, JF (1983b) Experimental tests of optimum habitat use in fish: the role of relative habitat profitability. Ecology 64(6):1525--1539
Winemiller KO (1989) Developmental of dermal lip protuberances for aquatic surface respiration in South American characid fishes. Copeia 2:382–390
Winemiller KO (1992) Life-history strategies and the effectiveness of sexual selection. Oikos 63:2
Acknowledgments
The authors acknowledge the ‘Conselho Nacional de Desenvolvimento Científico e Tecnológico’ (CNPq) for providing scholarship (T.M.L.; J.C.B.S.; R.D.L.B.) and Research Productivity grants (L.C.G.). The ‘Coordenação de Aperfeiçoamento de Pessoal de Nível Superior’ (CAPES-PROEX) for providing scholarship (E.R.C.) and funding this research. The Núcleo de Pesquisas em Limnologia, Ictiologia e Aquicultura (NUPELIA) for logistical support. The ‘Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis’ (IBAMA) for providing Animal Care license (Process n°224421 - Project PELD n° 558118/ 2009-7). We also thank F. A. Teixeira, J. R. Gonçalves, S. Rodrigues, V. R. Casaré, V. A. Capatti and F. G. Oliveira for field assistance; S. M. Thomaz and H. F. Júlio-Júnior for comments on this paper.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Schematic illustration of the minnow trap used to collect fish in the stands of macrophytes (available in Cunha et al. 2011 - Supplementary Material). The letter a represents a whole overview of the trap, letter b indicates the dimensions of the trap and letter c demonstrates the parts which constitutes the trap
Rights and permissions
About this article
Cite this article
Lopes, T.M., Cunha, E.R., Silva, J.C.B. et al. Dense macrophytes influence the horizontal distribution of fish in floodplain lakes. Environ Biol Fish 98, 1741–1755 (2015). https://doi.org/10.1007/s10641-015-0394-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-015-0394-4