Abstract
Habitat selection is a shared process among animals where individuals choose areas that differ in biotic and abiotic characteristics to maximize individual fitness. We used manipulative laboratory mesocosm choice experiments to examine hierarchical and interactive relationships influencing this habitat selection process of estuarine fishes. We assessed selection among substrate, dissolved oxygen (DO) concentration, food availability, and predation risk using two common juvenile estuarine fish species, pinfish (Lagodon rhomboides) and Atlantic croaker (Micropogonias undulatus). For both species oxygen concentration greatly influenced selection patterns; fishes strongly avoided low DO conditions, while in higher levels of DO factors such as substrate or food influenced selection patterns. However, both species strongly avoided predators even when alternative habitat was severely oxygen limited. These results suggest that predation risk may be the greatest determinant of habitat selection of the factors considered. Expansion of low DO areas in the world’s oceans is a major anthropogenic disturbance and is rapidly increasing. Assessing impacts of hypoxia on habitat usage of mobile organisms is critical as changes in environmental metrics including predator distribution and DO levels may alter habitat selection patterns disrupting critical ecosystem processes and trophic interactions. Our results indicate that juvenile fishes may forgo emigration from hypoxia due to predation risk. If similar patterns occur for juvenile fishes in estuaries they may potentially suffer from reduced growth, reproductive output, and survivorship.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat selection is a nearly universal process among animals where individuals must choose among habitats that differ in biotic and abiotic characteristics (Johnson 1980; Huey 1991). The hierarchy of habitat selection for an individual should reflect factors potentially limiting an individual’s fitness. For example, ideal habitats would provide high net energetic return rate (to maximize growth and reproduction) and low mortality (Gilliam and Fraser 1987). These conditions rarely exist in nature, and individuals must choose between sub-optimal environments to balance physiological performance with predation risk.
Habitat selection patterns of fishes have been well-described and offer a good model to test the relative roles of abiotic versus biotic habitat characteristics. For example, estuaries are recognized as high quality habitat types as they are food-rich, structurally complex, and provide refuge from predation (Beck et al. 2001). However, human activities have dramatically altered both abiotic and biotic properties of coastal ecosystems at an alarming rate (Altieri 2008; Halpern et al. 2008). For example, in many coastal estuaries, predator densities have been dramatically reduced (Lotze et al. 2006), important habitats such as seagrass meadows and oyster reefs have declined (Levin and Stunz 2005), and dissolved oxygen (DO) concentrations are falling (i.e., hypoxia) worldwide (Rabalais et al. 2007; Diaz and Rosenburg 2008; Montagna and Froeschke 2009). These habitat alterations may influence habitat selection of ecologically important species and could precipitate large-scale community changes in marine ecosystems (Utne-Palm et al. 2010).
A critical abiotic attribute often compromised in marine ecosystems is dissolved oxygen (DO). In aquatic systems, low oxygen levels are a widespread problem (Diaz and Rosenburg 2008), not limited to particular areas, and interact with biotic habitat characteristics in very complex ways (Breitburg 2002; Rabalais et al. 2002; Altieri 2008), ultimately altering the distribution of individuals (Lenihan et al. 2001; Bell and Eggleston 2005; Utne-Palm et al. 2010). Successful avoidance of hypoxia (DO concentration <2 mg O2 l−1) is dependent upon movement responses and physiological tolerances which are typically species specific (Pihl et al. 1991). In aquatic ecosystems, low levels of oxygen have been associated with reduced abundance, biomass, diversity, growth, and have also been attributed to population declines of some estuarine fishes (Eby et al. 2005; Long and Seitz 2008; Vaquer-Sunyer and Duarte 2008; Montagna and Froeschke 2009). Mitigation of human impacts to ecosystems requires an improved understanding of attributes animals use for habitat selection, particularly as it relates to importance of abiotic and biotic factors on habitat choice.
Hypoxia is becoming a more common phenomenon in estuaries and is thought to be increasing in occurrence worldwide including the Gulf of Mexico (Diaz and Rosenburg 2008). The Gulf of Mexico supports a variety of economically and ecologically important species that may be impacted by declining DO levels. The seasonal presence of a large dead zone in the Gulf of Mexico at the mouth of the Mississippi river is well documented and also occurs within estuarine waters along the Texas coast (Montagna and Ritter 2006). The presence of a hypoxic zone has been documented annually in Corpus Christi Bay, Texas since 1988 and there is a long-term trend of decreasing dissolved oxygen levels in these waters (Applebaum et al. 2005). This hypoxic zone also had reduced diversity of both benthic and mobile organisms (including fishes) and lower abundance and biomass of epifauna (Montagna and Froeschke 2009).
Pinfish (Lagodon rhomboides) and Atlantic croaker (Micropogonias undulatus) are two of the most abundant estuarine fishes in the Gulf of Mexico and shallow coastal estuaries comprise important nursery habitat for these species (Reese et al. 2008). Both can be found in a variety of habitats including seagrass meadows and non-vegetated substrate and are sensitive to hypoxia (Wannamaker and Rice 2000). Thus, these species provide excellent models for testing hypotheses about factors influencing habitat selection patterns of estuarine fishes.
The goal of this study was to assess the relative importance of biotic and abiotic factors on habitat selection. Specifically, we compared the relative importance of predator density, substrate, and food availability with varying levels of DO concentration to assess ecological impacts of declining oxygen concentrations on habitat selection patterns. We used a series of replicated laboratory mesocosm choice experiments with two ecologically important estuarine fishes as models.
Methods
Collection of study organisms
Juvenile Atlantic croaker (“croaker”) and pinfish were collected from shallow estuarine habitats in Aransas and Corpus Christi Bays, Texas USA using bag seines. Mean standard length (SL) of croaker and pinfish was: (mean ± standard error) 38 ± 5 mm SL and 35 ± 3 mm SL respectively. Animals were held at wet laboratory facilities in aerated and filtered 38-l aquaria. Fish were maintained in holding tanks at 23–25°C and 30–35 psu; DO concentrations were maintained between 6.0 and 6.5 mg O2 l−1. Fish were acclimated to aquaria for at least 3 d prior to experimental procedures and fed frozen mysid shrimp (mysid sp.) daily to satiation and kept on a 12-h light/dark photoperiod.
Experimental design
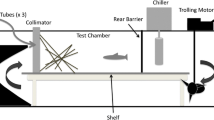
We used a sequential series of replicated experimental mesocosm trials to test for habitat selection patterns for both species at varying levels of predator density, substrate type, DO concentration, and food. Replicated two-way choice trials were completed in a 225-cm × 60-cm × 75-cm mesocosm filled to 25 cm with filtered seawater (Fig. 1). Washed sand was used to simulate non-vegetated habitat (Stunz et al. 2001), and shoal grass (Halodule wrightii) was simulated using an artificial seagrass unit (ASU). Artificial seagrass units are replicate seagrass habitat and are created from polyethylene ribbon attached to a mesh base placed under a sand substrate. ASU’s were used as a proxy for seagrass because preliminary trials on selection patterns of juvenile pinfish and croaker showed no significant selection patterns between shoal grass and the ASU’s (one sample t-test, pinfish t = 1.88, df = 7, p = 0.10, croaker t = 0.95, df = 9, p = 0.36).
a Diagram of experimental mesocosm set up prior to fish introduction where the seagrass treatment is placed in the low DO treatment, and b after the fish are introduced. c Experimental set-up prior to fish introduction where the sand treatment is placed in the low DO treatment, and d set-up after the fish are introduced
A DO gradient was established using a Plexiglass divider, and releasing nitrogen and oxygen gas into each chamber of the mesocosm. During experimental trials, the divider was raised 60 mm to permit fish movement (Wannamaker and Rice 2000). Dissolved oxygen concentrations were measured at the center and at each end of the mesocosm prior to introduction of experimental fish and at the conclusion of each experiment using a Thermo Scientific Orion 3-Star DO meter (Thermo Electron Corporation Beverly, MA USA). Trials were not run if DO levels deviated more than ± 0.40 mg O −12 from target levels. Wannamaker and Rice (2000) demonstrated that pinfish and croaker could detect and avoid the hypoxia in a laboratory mesocosm and our preliminary experiments showed similar patterns.
Experimental procedure
To determine the relative importance of substrate and DO concentrations on habitat selection patterns, we established a range of substrate-DO combinations. First, we conducted a substrate control experiment to determine selection patterns based on the habitat type (seagrass vs. non-vegetated) without a DO treatment (both chambers 6.0 mg O −l2 ). We then conducted a series of experiments, each testing fish habitat selection patterns between two substrate-DO combinations (Fig. 1). Experiments were conducted at DO levels from 1 to 6 mg O −l2 to identify potential graded, threshold or interactive responses. Trials were first set up with the low DO treatment within the seagrass substrate and high DO treatment within the non-vegetated substrate. Reciprocal experiments were completed by establishing low oxygen treatment in non-vegetated substrate and high oxygen concentration in seagrass.
For all experiments, three fish (approximating natural densities; Stunz et al. 2002) were randomly selected from holding aquaria for each trial. Fish were released in the center of the mesocosm as close as possible to the divider near the bottom and acclimated for 10 min prior to each experiment. The acclimation period occurred after the divider was raised to permit movement and exploration of all treatments. For every trial the locations of all three fish were averaged into a single response as individual fish within a trial were not independent. The low and high oxygen chambers of the mesocosm were re-assigned for each replicate to eliminate bias toward any particular area. Observations were made by a single observer >2 m away from the tank with the observer peering over an opaque barrier to prevent observer interference to fish behavior. During preliminary trials, this method of observation did not elicit a startle or other behavioral responses from the fish. Since these experiments were relatively short-term, we were able to make live direct observations during the entirety of the experiment without disturbing the fish.
To assess the relative importance of food availability relative to hypoxia for both species, the experimental mesocosm was modified by placing a food enriched treatment (mysid shrimp) in one chamber, while food was absent in the other. Fish were fasted for 24 h and three fish were placed in the mesocosm with DO concentration at 4.0 mg O −l2 . One chamber was enriched with 6.07 ± 0.07 g (mean ± standard error) of frozen mysid shrimp per trial and habitat choice was monitored every 20 s for 10 min after acclimation (n = 6). Excess food was removed after every trial. A second experiment (n = 6) was repeated, but mysid shrimp were placed in a low oxygen treatment (1.0 mg O2 l−l), while the other chamber remained at 4.0 mg O2 l−l but without the mysid shrimp food treatment.
The relative influence of predator-presence and DO concentration on habitat selection patterns was also examined by introducing predatory fish. Three sub-adult red drum (Sciaenops ocellatus) (mean ± standard error) 190 ± 8 mm SL were used for the predation trials. Predators were acclimated for 48 h. The Plexiglass partition separating sides of the mesocosm was modified by cutting eight 4-cm round holes near the bottom of the divider to permit movement of juvenile fishes throughout the entire mesocosm but restricted the movement of the red drum predators to one chamber. We performed preliminary trials that showed juvenile fish would readily pass through holes in the partition. For the initial experiment (n = 6) both sections of the mesocosm were set to 4.0 mg O2 l−1. A second experiment (n = 6) was conducted in which the predatory red drum were placed in the 4.0 mg O2 l−1 DO treatment and the side without predators set to 1.0 mg O2 l−1.
Statistical analyses
The response variable for all habitat selection trials was the mean proportion of time three fish spent in each chamber in the mesocosm. Residuals were not normally distributed, therefore, data were arc-sin square root transformed and tested against the null hypothesis of 50% of the time spent in each side of the mesocosm using a two-tailed, one-sample, student’s t-test (α = 0.05). All data management and analyses were conducted using SAS 9.1.3 software (SAS Institute, Inc., 2000).
Results
Habitat selection experiments
In normoxic conditions (both chambers 6 mg O2 l−1), both pinfish and croaker displayed significant habitat preferences. Pinfish selected seagrass treatment (t = 3.5, df = 5, p = 0.016) (Fig. 2a), while croaker selected non-vegetated bottom (t = 4.5, df = 5, p = 0.007) (Fig. 2c). Based on significant habitat selection preference patterns in previous trials, we designed experiments to test the importance of habitat type (e.g., seagrass and non-vegetated bottom) and oxygen concentration on selection patterns. For pinfish, both habitat type and DO levels influenced selection patterns. Dissolved oxygen exerted greater influence on selection patterns during hypoxic conditions (i.e., DO ≤ 2.0 mg O2 l−1), while habitat type was more important during moderate hypoxia or normoxic conditions (i.e., DO ≥ 2.0 mg O2 l−1; Fig. 2a).
Mean ± SE percentage occurrence of pinfish and croaker in each habitat*DO treatment combination. Each solid-open bar pair represents six replicate 30 min mesocosm trials with the following treatments: a high DO-non-vegetated, low DO-seagrass; b low DO- non-vegetated, high DO-seagrass; c high DO- non-vegetated, low DO-seagrass; d low DO-sand, high DO-seagrass. Significant results from one-sample Student’s t-tests are indicated by * = p < 0.05 and *** = p <0.001
Despite the preference for vegetated habitat in the control experiment, pinfish avoided the low oxygen-seagrass treatment when DO levels were decreased to 1.0 mg O2 l−1 and selected the alternative 4.0 mg O2 l−1 sand bottom treatment (t = 3.8, df = 5, p = 0.011) (Table 1). A similar pattern was observed during 1.0 mg O2 l−1 seagrass vs. 2.0 mg O2 l−1 sand bottom experiment as pinfish displayed significant selection for increased oxygen treatment despite a relatively small difference in DO concentrations between treatments. However, this pattern was not observed when oxygen levels were increased, simulating moderate levels of hypoxia. During the 2.0 mg O2 l−1 seagrass vs. 4.0 mg O2 l−1 non-vegetated substrate experiment, pinfish selection patterns were more variable, although fish spent more time in the lower oxygen seagrass chamber. Overall, no significant selection patterns were detected for this experiment (t = 1.1, df = 5, p = 0.33). In the 4.0 mg O2 l−1 seagrass vs. 6.0 mg O2 l−1 non-vegetated substrate experiment, a similar pattern was observed as fish selected the lower oxygen seagrass chamber, but the response was variable and a significant selection pattern was not detected (t = 1.9, df = 5, p = 0.121). Habitat selection patterns of pinfish were also determined when DO concentrations were reduced in the sand bottom treatment relative to their preferred seagrass habitat. In this situation, pinfish displayed significant selection patterns for the increased DO-seagrass treatment, as this treatment contained both the favored abiotic and biotic conditions within one chamber of the mesocosm (Fig. 2b).
Habitat selection patterns were also determined for croaker. In contrast to pinfish, croaker is a habitat generalist (Petrik et al. 1999). However, the influence of habitat type and DO concentration was similar between both species. Dissolved oxygen exerted greater influence on selection patterns during hypoxic conditions (e.g. DO ≤ 2.0 mg O2 l−1), while habitat type was more important during moderate hypoxia or normoxic conditions. Habitat selection experiments with croaker were investigated by placing the favored substrate (non-vegetated bottom as determined from preliminary trials) and high oxygen treatment in separate chambers. Similar to pinfish, croaker avoided their preferred habitat type when DO levels were 1.0 mg O2 l−1. This was observed for both the 4.0 mg O2 l−1 seagrass vs. 1.0 mg O2 l−1 non-vegetated bottom (t = 6.0, df = 5, p = 0.002) and 2.0 mg O2 l−1 seagrass vs. 1.0 mg O2 l−1 non-vegetated bottom experiments (t = 30.5, df = 5, p < 0.001) (Table 1, Fig. 2c). When minimum DO levels were increased in the sand bottom chamber to 2.0 and 4.0 mg O2 l−1, respectively, selection patterns were more variable. Overall, croaker selection patterns were similar to pinfish, as the mean proportion of time spent in elevated DO-seagrass chamber was higher; although, significant patterns were not detected for either the 4.0 mg O2 l−1 seagrass vs. 2.0 mg O2 l−1 sand bottom (t = 2.5, df = 5, p = 0.056) or 6.0 mg O2 l−1 seagrass vs. 4.0 mg O2 l−1 sand bottom (t = 1.7, df = 5, p = 0.151) experiments.
Habitat selection patterns of croaker were also determined during a reciprocal set of experiments where oxygen levels were reduced in the seagrass substrate relative to non-vegetated substrate. Croaker demonstrated significant avoidance of seagrass when DO levels were reduced to 1.0 mg O2 l−1 within this treatment. This pattern was observed during both the 1.0 mg O2 l−1 seagrass vs. 2.0 mg O2 l−1 non-vegetated (t = 2.9, df = 5, p = 0.034) and the 1.0 mg O2 l−1 seagrass vs. 4.0 mg O2 l−1 non-vegetated (t = 4.3, df = 5, p = 0.008) experiments (Fig 2d). However, when oxygen levels were increased to 2.0 mg O2 l−1 in the seagrass and to 4.0 mg O2 l−1 in the sand bottom chambers, selection patterns deviated from prior experiments. In both the 2.0 mg O2 l−1 seagrass vs. 4.0 mg O2 l−1 sand bottom and the 4.0 mg O2 l−1 seagrass vs. 6.0 mg O2 l−1 no significant selection patterns were detected (t = 1.7, df = 5, p = 0.140; t = 1.4, df = 5, p = 0.209, respectively).
Food vs. DO selection experiments
The addition of food resources influenced selection patterns of pinfish during moderate hypoxia (4.0 mg O2 l−1) but did not influence selection patterns at 1.0 mg O2 l−1. Both chambers of the mesocosm were initially set to 4.0 mg O2 l−1 and a mysid shrimp food supplement was added to one side. Pinfish showed a marginally significant preference for food (t = 2.5, df = 5, p = 0.052) (Fig. 3a). During a second experiment food treatment was placed in a reduced oxygen treatment (1.0 mg O2 l−1), while the other chamber remained at 4.0 mg O2 l−1 without a food supplement. Pinfish avoided the food enriched chamber and exhibited a significant selection for the 4.0 mg O2 l−1 treatment despite the lack of food (t = 3.4, df = 5, p = 0.020). Both experiments were repeated for croaker. During moderate hypoxia (4.0 mg O2 l−1) food enrichment did not influence selection patterns (t = 2.1, df = 5, p = 0.089), and croaker avoided the food supplemented treatment when placed in the low oxygen treatment (t = 3.9, df = 5, p = 0.011) (Fig. 3b).
Mean ± SE percentage of time (a) pinfish and (b) croaker spent in each food enhanced*DO treatment combination. Each solid-open bar pair represents six replicate 10 min. mesocosm trials with the following treatments: 1) 4.0 mg O2 l−1 and mysid shrimp food supplement vs. 4.0 mg O2 l−1 without food supplement treatment; 2) 1.0 mg O2 l−1 and mysid shrimp food supplement vs. 4.0 mg O2 l−1 without food supplement treatment. Mean percentage of time (c) pinfish and (d) croaker spent in each chamber of the mesocosm in the presence of a three red drum predators with two different DO*predator combinations. Significant results from one-sample Student’s t-tests are indicated by * = p < 0.05
Predator presence vs. DO selection experiments
The presence of predators exerted a strong influence on selection patterns for both species at all levels of DO concentration tested. An initial experiment was conducted to determine the effect of predator presence on the habitat selection patterns in absence of a DO difference (both chambers set to 4 mg O2 l−1). Both species strongly avoided predators in the control experiment (no DO concentration difference, pinfish, t = 6.4, df = 5, p = 0.001; croaker, t = 6.6, df = 5, p = 0.001) (Fig. 3c-d). In a second experiment fish had a choice between predators with 4.0 mg O2 l−1 and no predators with 1.0 mg O2 l−1. Both species chose chambers without predators, despite the low oxygen concentration (pinfish, t = 6.6, df = 5, p = 0.001; croaker, t = 4.5, df = 5, p = 0.007).
The response of habitat selection patterns to DO, substrate, and food was strongly interactive (Fig. 4). During hypoxic conditions, DO concentration was an important determinant of habitat selection patterns. However, as oxygen levels increased, the relative importance on fish habitat selection decreased and substrate preference became more important. As with substrate, food availability influenced selection patterns of pinfish at 4.0 mg O2 l−1, however, food availability was unimportant at 1.0 mg O2 l−1. The presence of predators exerted the greatest influence on habitat selection. Both species strongly avoided predators even when the alternative habitat was severely hypoxic (1.0 mg O2 l−1).
A general mechanistic hypothesis of habitat selection of juvenile estuarine fishes based on our experimental observations. Dissolved oxygen concentration was an important determinant of habitat selection patterns when concentrations were below 2 mg O2 l−l. The presence of preferred habitat type (substrate) and food availability became increasingly important with increasing oxygen concentration. During low oxygen conditions food availability or substrate type did not influence habitat selection however the relative importance increased with DO concentration. Patterns of habitat selection in response to predation risk and the DO concentrations were similar between species. Predation risk (dashed line), at least in the case of high predator density in this study, exerted the greatest influence on habitat selection patterns of juvenile estuarine fishes (of the factors examined in the current study) across all levels of DO concentration considered
Discussion
Habitat selection influences distribution, abundance, and population dynamics of mobile organisms (Johnson 1980; Levin et al. 1997; Stunz et al. 2001; Morris 2003). Preferential selection for high quality habitats may increase growth rates or survivorship, ultimately contributing disproportionately to adult populations (Beck et al. 2001). However, habitat quality may be influenced by a myriad of abiotic and biotic factors and improved management of marine resources requires a detailed understanding of the mechanism used by fishes to select the highest quality habitat available (Morris 2003).
Abundance of estuarine organisms is typically higher in structurally complex habitats such as seagrass meadows (Jordan et al. 1997; Levin et al. 1997). Seagrass meadows are particularly important habitats for newly recruited juvenile fishes (Burfeind and Stunz 2006). In experiments without DO treatments, pinfish showed significant selection preference for seagrass habitat although croaker, a habitat ‘generalist’ preferred non-vegetated habitat. This is consistent with previous laboratory experiments (Petrik et al. 1999) and field observations (Jordan et al. 1997) for these species. Despite significant substrate preferences, both species avoided their “preferred” substrate when placed in low DO concentrations while at higher levels, adequate DO levels allowed other factors such as “preferred” substrate or prey availability to influence habitat usage patterns. In a Galveston TX estuary, both recruitment and growth rates of pinfish were higher in seagrass as compared to non-vegetated habitats (Levin et al. 1997). Juvenile red drum also grew significantly faster in vegetated as compared to sand substrates in experimental field enclosures (Stunz et al. 2002). Similar to the substrate treatment, the addition of food only influenced selection patterns of pinfish in absence of hypoxic conditions. Pinfish avoided the food enriched treatment when placed in the low oxygen treatment, suggesting that food availability is not a strong driver of habitat selection in estuarine ecosystems where food is typically abundant (Heck et al. 2003).
The ability of estuarine organisms to detect and avoid hypoxia in laboratory mesocosms was previously reported (Wannamaker and Rice 2000; Stierhoff et al. 2009). As with the current study, avoidance patterns were greatest when DO treatments were below 2 mg O2 l−1. This coincides with the level at which fish emigrate from hypoxic areas and is associated with significant reductions in abundance (Breitburg 2002), and diversity (Vaquer-Sunyer and Duarte 2008; Montagna and Froeschke 2009). Habitat selection patterns of flatfishes in the Gulf of Mexico were altered by low DO levels with reduced habitat suitability in regions with hypoxia and increased suitability in nearby refuges (Switzer et al. 2009). This study suggests potential wide-scale alteration of habitat selection patterns due to hypoxia and suggests that this factor alone may induce emigration or avoidance of otherwise suitable habitats (Utne-Palm et al. 2010). In estuaries, hypoxia may reduce quality of nursery habitat even if preferred habitat types and food resources are abundant.
Hypoxia impacts may be most severe for juveniles as the dispersal potential may be limited due to their small size and increased risk of predation during movement away from hypoxic zones. In a study of intermittent hypoxia in Chesapeake Bay, juvenile fishes were less able to escape than adults and mortality rates of juveniles was extremely high (Breitburg 1992). Increased mortality rates of small fish due to hypoxia may be associated with increased oxygen demands of juveniles, reduced swimming speeds (Breitburg 1992), or increased predation risk associated with emigration. Results from the current study provide further evidence for predator-mediated habitat selection (Jordan et al. 1997), and the critical role that predators play in ecosystem regulation (Heck and Valentine 2007). Long and Seitz (2008) reported increased susceptibility of benthic prey to predators from hypoxia in Chesapeake Bay. However, Altieri (2008) suggests that responses to hypoxia may reduce predation and hypoxia tolerant species such as quahog clam (Mercenaria mercenaria) may benefit from non-lethal hypoxia events. However reductions in abundance and diversity of species sensitive to hypoxia have been observed (Altieri 2008; Montagna and Froeschke 2009). If juvenile fishes forgo emigration from hypoxia due to predation risk, they are subject to the physiological effects of hypoxia and long-term impacts on fish populations may be observed. Landry et al (2007) reported reduced reproductive output and Eby et al. (2005) predicted long-term population declines of croaker resulting from exposure to hypoxic conditions. However, species specific responses to hypoxia is typical and has been reported in both laboratory and field studies (Wannamaker and Rice 2000; Montagna and Froeschke 2009; Switzer et al. 2009). These results suggest that environmental stressors such as hypoxia can be important determinants on community structure (Menge and Sutherland 1987; Lenihan et al. 2001), where some species may benefit but net declines in diversity and resilience may be expected from ecosystem stressors.
Hypoxia may exert direct or indirect effects on population dynamics of juvenile fishes. Populations may be affected directly from hypoxia either through increased mortality or decreased recruitment due to avoidance of hypoxic areas. Indirect effects including reduced growth rate, increased density dependent competition in normoxic refuges and greater predation risk have been hypothesized previously. While indirect effects are more difficult to empirically demonstrate (Heck and Valentine 2007), they may exert greater long-term effects on the population dynamics and community structure of estuarine systems. For example, the rate of juvenile survival is often cited as the best predictor of subsequent adult population size (Caley et al. 1996; Levin and Stunz 2005) and hypoxia induced exposure has been shown to reduce growth rates of some fishes (Chabot and Dutil 1999; Eby et al. 2005; Stierhoff et al. 2006) and increase their duration in critical life stages where predation risk is high (Levin et al. 1997; Levin and Stunz 2005). Moreover, reduced growth rate may delay sexual maturity and reduce total reproductive output leading to long-term reductions in population size. Growth rates of croaker were reduced significantly inside a hypoxic estuary and subsequent reductions of demographic rates were predicted to result in long-term population declines in the estuary (Eby et al. 2005). Stierhoff et al. (2006) reported reduced feeding and growth rates of Paralichthys dentatus and Pseudopleuronectes americanus due to moderate hypoxia while similar results were also reported for Gadus morhua (Chabot and Dutil 1999). Reduced growth rates may ultimately lead to substantial reductions in fisheries productivity, predator densities, and ultimately a disruption of vital ecosystem links and trophic interactions to the detriment of ecosystem based management goals.
Expansion of low oxygen areas is currently considered among the most damaging environmental problems (Diaz and Rosenburg 2008). This problem will intensify as low oxygen zones increase both temporally and spatially throughout coastal and estuarine regions from enhanced nutrient deposition and warming seas (Diaz and Rosenburg 2008; Vaquer-Sunyer and Duarte 2008). Assessing impacts of these changes on habitat usage of mobile organisms is critical as changes in environmental metrics including predator distribution and DO levels may alter habitat selection patterns and reduce fitness levels of individuals and potentially disrupting vital ecosystem links and trophic interactions to the detriment of ecosystem-based management goals.
References
Altieri AH (2008) Dead zones enhance key fisheries species by providing predation refuge. Ecology 89:2808–2818
Applebaum S, Montagna PA, Ritter C (2005) Status and trends of dissolved oxygen in Corpus Christi Bay, Texas, U.S.A. Environ Monitor Assess 107:297–311
Beck MW, Heck KL, Able KW, Childers DL, Eggleston DB, Gillanders BM, Halpern B, Hays CG, Hoshino K, Minello TJ, Orth RJ, Sheridan PF, Weinstein MR (2001) The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates. Bioscience 51:633–641
Bell GW, Eggleston DB (2005) Species-specific avoidance responses by blue crabs and fish to chronic and episodic hypoxia. Mar Biol 146:761–770
Breitburg D (2002) Effects of hypoxia, and the balance between hypoxia and enrichment, on coastal fishes and fisheries. Estuaries 25:767–781
Breitburg DL (1992) Episodic hypoxia in Chesapeake Bay: Interacting effects of recruitment, behavior, and physical disturbance. Ecol Monogr 62:525–546
Burfeind DD, Stunz GW (2006) The effects of boat propeller scarring intensity on nekton abundance in subtropical seagrass meadows. Mar Biol 148:953–962
Caley MJ, Carr MH, Hixon MA, Hughes TP, Jones GP, Menge BA (1996) Recruitment and the local dynamics of open marine populations. Annu Rev Ecol Syst 27:477
Chabot D, Dutil JD (1999) Reduced growth of Atlantic cod in non-lethal hypoxic conditions. J Fish Biol 55:472–491
Diaz JR, Rosenburg R (2008) Spreading dead zones and consequences for marine ecosystems. Science 321:926–929
Eby LA, Crowder LB, McClellan CM, Peterson CH, Powers MJ (2005) Habitat degradation from intermittent hypoxia: Impacts on demersal fishes. Mar Ecol Prog Ser 291:249–261
Gilliam JF, Fraser DF (1987) Habitat selection under predation hazard: Test of a model with foraging minnows. Ecology 68:1856–1862
Halpern BS, Walbridge S, Selkoe KA, Kappel CV, Micheli F, D’Agrosa C, Bruno JF, Casey KS, Ebert C, Fox HE, Fujita R, Heinemann D, Lenihan HS, Madin EMP, Perry MT, Selig ER, Spalding M, Steneck R, Watson R (2008) A global map of human impact on marine ecosystems. Science 319:948–952
Heck KL, Hays G, Orth RJ (2003) Critical evaluation of the nursery role hypothesis for seagrass meadows. Mar Ecol Prog Ser 253:123–136
Heck KL, Valentine JF (2007) The primacy of top-down effects in shallow benthic ecosystems. Estuaries Coasts 30:371–381
Huey RB (1991) Physiological consequences of habitat selection. Am Nat 137:S91
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65–71
Jordan F, Bartolini M, Nelson C, Patterson PE, Soulen HL (1997) Risk of predation affects habitat selection by the pinfish Lagodon rhomboides (Linnaeus). J Exp Mar Biol Ecol 208:45–56
Landry et al (2007) Long Term Hypoxia suppresses reproductive capacity in the estuarine fish. Comp Biochem Physiol A 143:317–323
Lenihan HS, Peterson CH, Byers JE, Grabowski JH, Thayer GW, Colby DR (2001) Cascading of habitat degradation: Oyster reefs invaded by refugee fishes escaping stress. Ecol App 11:764–782
Levin P, Petrik R, Malone J (1997) Interactive effects of habitat selection, food supply and predation on recruitment of an estuarine fish. Oecologia 112:55–63
Levin PS, Stunz GW (2005) Habitat triage for exploited fishes: Can we identify essential fish habitat?. Est Coast Mar Sci. 70–78
Long WC, Seitz RD (2008) Trophic interactions under stress: hypoxia enhances foraging in an estuarine food web. Mar Eco Prog Ser 362:59–68
Lotze HK, Lenihan HS, Bourque BJ, Bradbury RH, Cooke RG, Kay MC, Kidwell SM, Kirby MX, Peterson CH, Jackson JBC (2006) Depletion, degradation, and recovery potential of estuaries and coastal seas. Science 312:1806–1809
Menge BA, Sutherland JP (1987) Community regulation: variation in disturbance, competition, and predation in relation to environmental stress and recruitment. Am Nat 130:730
Montagna PA, Ritter C (2006) Direct and indirect effects of hypoxia on benthos in Corpus Christi Bay, Texas, U.S.A. J Exp Mar Biol Ecol 330:119–1313
Montagna PA, Froeschke J (2009) Long-term biological effects of coastal hypoxia in Corpus Christi Bay, Texas, USA. J Exp Mar Biol Ecol 381:S21–S30
Morris DW (2003) Toward an ecological synthesis: a case for habitat selection. Oecologia 136:1–13
Petrik R, Levin PS, Stunz GW, Malone J (1999) Recruitment of Atlantic croaker, Micropogonias undulatus: do postsettlement processes disrupt or reinforce initial patterns of settlement? Fish Bull 97:954–961
Pihl L, Baden SP, Diaz RJ (1991) Effects of periodic hypoxia on distribution of demersal fish and crustaceans. Mar Biol 108:349–360
Rabalais NN, Turner RE, Scavia D (2002) Beyond science into policy: Gulf of Mexico hypoxia and the Mississippi River. BioScience 52:129–142
Rabalais NN, Turner RE, Gupta BKS, Platon E, Parsons ML (2007) Sediments tell the history of eutrophication and hypoxia in the northern Gulf of Mexico. Ecol App 17:S129–S143
Reese MM, Stunz GW, Bushon AM (2008) Recruitment of estuarine-dependentnekton through a new tidal inlet: the opening of Packery Channel in Corpus Christi, TX, USA. Estuaries Coasts 31:1143–1157
Stierhoff KL, Targett TE, Miller K (2006) Ecophysiological responses of juvenile summer and winter flounder to hypoxia: experimental and modeling analyses of effects on estuarine nursery quality. Mar Ecol Prog Ser 325:255–266
Stierhoff KL, Tyler RM, Targett TE (2009) Hypoxia tolerance of juvenile weakfish (Cynoscion regalis): Laboratory assessment of growth and behavior avoidance responses. J Exp Mar Biol Ecol 381:S173–S179
Stunz GW, Levin PS, Minello TJ (2001) Selection of estuarine nursery habitats by wild-caught and hatchery-reared juvenile red drum in laboratory mesocosms. Environ Biol Fish 61:305–313
Stunz GW, Minello TJ, Levin PS (2002) A comparison of early juvenile red drum densities among various habitat types in Galveston Bay, Texas. Estuaries 25:76–85
Switzer TS, Chesney EJ, Baltz DM (2009) Habitat selection by flatfishes in the northern Gulf of Mexico: Implications for susceptibility to hypoixa. J Exp Mar Biol Ecol 381:S51–S64
Utne-Palm AC, Salvanes AGV, Currie B, Kaartvedt S, Nilsson GE, Braithwaite VA, Stecyk JAW, Hundt M, van der Bank M, Flynn B, Sandvik GK, Klevjer TA, Sweetman AK, Brüchert V, Pittman K, Peard KR, Lunde IG, Strandabø RAU, Gibbons MJ (2010) Trophic structure and community stability of an overfished ecosystem. Science 329:333–336
Vaquer-Sunyer R, Duarte CM (2008) Thresholds of hypoxia for marine biodiversity. PNAS 105:15452–15457
Wannamaker CM, Rice JA (2000) Effects of hypoxia on movements and behavior of selected estuarine organisms from the southeastern United States. J Exp Mar Biol Ecol 249:145–163
Acknowledgments
Funding for this study was provided by the Texas Research Development Fund. We thank the members of the Fisheries Ecology Laboratory at Texas A&M University-Corpus Christi for their help with this project. Special thanks to M. Reese and L. Brown for their field and laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Froeschke, J.T., Stunz, G.W. Hierarchical and interactive habitat selection in response to abiotic and biotic factors: The effect of hypoxia on habitat selection of juvenile estuarine fishes. Environ Biol Fish 93, 31–41 (2012). https://doi.org/10.1007/s10641-011-9887-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-011-9887-y