Abstract
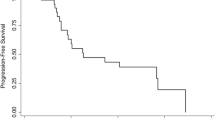
Ninety per cent of pancreatic adenocarcinomas (PC) contain mutations of the K-Ras proto-oncogene resulting in constitutively activated Ras protein. A critical step in Ras activation is farnesylation of Ras protein. Farnesyl transferase inhibitors are compounds that inhibit farnesylation. We report the results of a phase II trial of R115777, an oral farnesyl transferase inhibitor, in patients with surgically incurable locally advanced or metastatic PC. Between 6/1/2000 and 11/20/2001, 58 cases were accrued, 53 of whom were eligible and analyzable. Patients were required to have a performance status (PS) 0 to 1, be able to take oral medications, and to have adequate renal, hepatic, and hematologic functions. Fifty-five percent were male. Median age was 64.7 years (38.9 to 80.6), and patients had no previous systemic therapy for advanced PC. Treatment consisted of R115777 300 mg po bid given for 3 out of every 4 weeks. Toxicities were as follows: Grade 3 in 19/53 (36%), grade 4 in 53 (173%), and grade 5 in 53 (8%). Most frequent toxicities were: anemia 35/53 (66%), fatigue and malaise 33/53 (62%), nausea 31/53 (58%). Grade 5 toxicities included: thromboembolism 1, infection 2, other 1. Median survival was 2.6 months (mo) (95% CI 2.1–3.6), 6-mo survival is 19% (95% CI, 8–29%), median time to treatment failure was 1.4 mo (95% GI 1.1–1.6). R115777 is ineffective as monotherapy in advanced pancreatic cancer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jemal A, Thomas A, Murray T, Thun M: Cancer statistics, 2002. CA Cancer J Clin 52: 23–47, 2002.
Schnall, SF, Macdonald JS: Chemotherapy of adenocarcinoma of the pancreas. Semin Oncol 23: 220–228, 1996.
Yeo CJ, Cameron JL, Sohn TA: Six hundred and fifty consecutive pancreaticoduodenectomies in the 1990s: Pathology, complications, and outcomes. Ann Surg 226: 248–257, 1997.
Burris HA, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P, Nelson F, Dorr FA, Stephens CD, Von Hoff DD: Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreatic cancer: A randomized trial. J Clin Oncol 15: 2403–2413, 1997.
End DW, Smets G, Todd AV, Applegate TL, Fuery CJ, Angibaud P, Venet M, Sanz G, Poignet H, Skrzat S, Devine A, Wouters W, Bowden C: Characterization of the antitumor effects of the selective farnesyl protein transferase inhibitor R115777 in vivo and in vitro. Cancer Res 61: 131–137, 2001.
Grünewald K, Lyons J, Fröhlich A: High frequency of Ki-ras codon 12 mutations in pancreatic adenocarcinomas. Int J Cancer 43: 1037–1041, 1989.
Cohen SJ, Linus H, Sulabha R, Abbruzzese JL, Alpaugh RK, Beard M, Lewis NL, McLaughlin S, Rogatko A, Perez-Ruixo JJ, Thistle AM, Verhaeghe T, Wang H, Weiner LM, Wright JJ, Hudes GR, Meropol NJ: Phase II and pharmacodynamic study of the farnesyltransferase inhibitor R115777 as initial therapy in patients with metastatic pancreatic adenocarcinoma. J Clin Oncol 21(7): 1301–1306, 2003.
Van Custem E, Karasek P, Oettle H, Vervenne W, Szawlowski A, Schoffski P, Post S, Neumann H, Safran H, Humblet Y, van de Velde H, Ma Y, Von Hoffl: Phase III trial comparing gemcitabine +R11577 (Zarnestra) versus gemcitabine +placebo in advanced pancreatic cancer (PC). Proc Am Soc Clin Oncol 21: 130a, 2002 (abstr 21).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Macdonald, J.S., McCoy, S., Whitehead, R.P. et al. A phase II study of farnesyl transferase inhibitor R115777 in pancreatic cancer: A Southwest oncology group (SWOG 9924) study. Invest New Drugs 23, 485–487 (2005). https://doi.org/10.1007/s10637-005-2908-y
Issue Date:
DOI: https://doi.org/10.1007/s10637-005-2908-y