Abstract
Background
Barrett’s esophagus (BE) is a complication of gastroesophageal reflux disease (GERD) that is a precursor to esophageal adenocarcinoma. There is limited information regarding whether medications can reduce the risk of developing BE.
Aim
We analyzed medical records at a large veterans hospital to determine the effects of statins, aspirin, non-aspirin NSAIDs, calcium, or multivitamins on the risk of developing BE.
Methods
In this retrospective case–control study, 250 patients with biopsy-confirmed Barrett’s esophagus were compared with 250 controls with acid-peptic symptoms but no endoscopic BE. Medication histories were reviewed for the use of the above substances prior to endoscopic evaluation. Logistic and linear regression was used to determine predictors of the outcomes.
Results
Mean age at diagnosis was significantly older in the Barrett’s population compared with controls (61.2 vs. 56.7 years, P < 0.001), with no difference in mean BMI (29.1 vs. 29.0, respectively). On multivariate analysis, independently significant factors for risk of BE were found with multivitamins (OR 0.41, P = 0.001), statins (OR 0.53, P = 0.003), age (OR 1.033/year, P = 0.001), and Hispanic ethnicity (OR 0.38, P = 0.007). Furthermore, statin use was associated with less long-segment (3 cm or longer) BE and was inversely correlated with continuous BE length.
Conclusions
GERD patients with BE are less likely to use multivitamins and statins, as well as less likely to be of Hispanic ethnicity. Additionally, statins were inversely associated with BE length. Prospective studies of this topic are indicated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Barrett’s esophagus (BE) is a metaplastic change in the esophageal epithelium induced by gastroesophageal reflux disease (GERD) [1]. It is a common condition, with a prevalence of 6.8 % in an American study [2] and 1.6 % in a Swedish study [3]. It is the largest known risk factor for esophageal adenocarcinoma (EAC), increasing the risk by at least 30-fold compared with the general population [4]. In addition, the incidence of EAC has been increasing over recent years [5]. Current practice is to perform endoscopic surveillance of BE [6], with the intended benefit of decreased mortality from EAC. However, more than 90 % of all EAC patients were not known to have BE before diagnosis [7]. Chemoprevention presents a readily assessable method of analyzing the effects of certain medications on the risks of BE and EAC.
Nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, have been studied both in the setting of preventing BE [8] and prevention of EAC in patients with established BE [9, 10]. These studies have suggested a benefit in both settings. This is presumably due to their inhibition of the cyclooxygenase-2 (COX-2) enzyme, which has increased expression in BE and EAC [11]. A large trial is ongoing in the UK, which is evaluating the efficacy and safety of aspirin for cancer prevention in BE (the AspECT trial).
Another class of medications that has shown promise in chemoprevention is the 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statins). Statins have shown some preventive effect on development of EAC in BE patients in several studies [12–15], although not all studies have demonstrated this protective effect [16]. Recently, statins have been studied in the setting of primary prevention of BE, with a protective effect found [17]. Calcium has been studied for chemoprevention in colorectal cancer to mixed results [18, 19], but has not been evaluated in BE. Similarly, multivitamin use has been shown to have possible benefit in reducing total cancer [20], but other results have been inconsistent, and these data have not been correlated with the risk of developing BE.
In this case–control study, we analyzed pharmacy records at a large veterans hospital to determine whether there were protective effects of statins, aspirin, NSAIDs, calcium, and multivitamins on the development of BE.
Methods
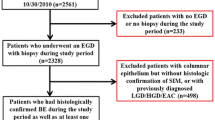
To conduct this case–control study, patient data were retrospectively obtained from the Phoenix Veterans Affairs (VA) Hospital electronic medical records, as well as from a separate secure database of endoscopic procedural data [Clinical Outcomes Research Initiative (CORI), Portland, OR]. CORI records were searched for patients who underwent esophagogastroduodenoscopy (EGD) for the indication of BE during the years 2005 through 2009. A random subset of patients underwent chart review until a case population of 250 patients was established. Patients were included if they had biopsy-proven BE of at least 2 cm length, as assessed via pathology reports in VA records. There were no limitations placed on age or comorbidities. However, patients were excluded if pathology data were not available. The control population was identified in a similar manner, with CORI records searched for EGD indication of reflux symptoms or dyspepsia, with BE either not visible on exam, or proven biopsy negative for intestinal metaplasia. Also, the clinical range was expanded to 2012 to identify an adequate cohort. No matching was performed. The number in each group was not determined based on a power calculation, but on availability of records and logistical needs.
Once the cases and controls were identified, the following data were recorded: gender, age at diagnosis, body mass index (BMI) closest to the time of diagnosis, ethnicity (Hispanic or not), H. pylori status, prior or current proton pump inhibitor (PPI) use, prior or current H2-receptor antagonist use, and tobacco and alcohol use. Only ethnicity data were obtained directly from CORI, with the remainder taken from VA medical records. Patient pharmacy medication records were reviewed to see whether there was previously a filled prescription for calcium, statins, multivitamins, aspirin, and other NSAIDs. Prescription fills were only counted if they were dated at least 1 month prior to either the initial diagnosis of BE, or the date of endoscopy for the control group.
Statistical analysis was performed using IBM SPSS version 21.0. Study outcomes were assessed using Fisher’s exact test for categorical variables and the Mann–Whitney U test for continuous variables. Logistic and linear regression was used for multivariate analysis. A two-tailed P < 0.05 was considered significant. There were few missing data, and there were no imputations performed. This study was approved by the Phoenix VA Hospital Institutional Review Board.
Results
Study Population
There were 250 patients in each the case and control groups. Baseline characteristics are summarized in Table 1. The majority in both populations were men, with only eight women (3.2 %) in the case group and 36 women (14.4 %) in the control group. Due to the small number of women in both groups, it was felt that their inclusion would diminish the homogeneity of the sample. As such, the female patients were not included in the following data analysis of 242 male BE patients and 214 male controls. Additionally, pharmacy data were not available for 15 patients in the case group and eight patients in the control group, leaving 227 BE patients and 206 control patients for the medication analysis. For the univariate analysis of Hispanic ethnicity, non-White patients (2 in BE, 13 in controls) were excluded given the small number. This resulted in available data for 239 BE patients and 199 controls.
The mean age of the Barrett’s population was 61.2 (SD 9.1), which was significantly older than the mean age in the control population of 56.7 years (SD 13.7, P < 0.001). There was no difference in mean body mass index (BMI) in BE patients (29.1, SD 5.6) versus controls (29.0, SD 5.7, P = 0.849). However, the BE population was significantly more likely to be of non-Hispanic ethnicity (94.2 vs. 87.3 %, P = 0.013). While proton pump inhibitor use was marginally more common in controls than BE patients (80.6 vs. 72.7 %, P = 0.055), there was no significant difference in H2-receptor antagonist use (53.4 vs. 56.4 %, 0.562), H. pylori positivity (15.8 vs. 16.7 %, P = 0.929), or current/former alcohol use (37.7 vs. 43.2 %, P = 0.572). Tobacco use data were also collected, but as there was a lack of a standardized method for recording these data, it was felt to be inaccurate and was not included.
Demographics and Risk of BE
In univariate analysis (Table 2), Hispanic ethnicity was significantly associated with BE risk, with a protective effect found (OR 0.40, P = 0.007). Increasing age also correlated with BE risk (OR 1.04 per year, P < 0.001). On multivariate analysis (Table 3), all univariate predictors with a P < 0.10 were considered. Forward selection using LR was used. Hispanic ethnicity retained a protective effect (OR 0.38, P = 0.007), while age remained correlated with BE risk (OR 1.033, P = 0.001).
Medications and Risk of BE
Statins, calcium, and multivitamins proved to be significant factors for risk of BE in univariate analysis (Table 2). With usage of multivitamins, the odds ratio for BE was 0.40 (P < 0.001). A protective effect was seen for calcium (OR 0.45, P = 0.016) and statins (OR 0.65, P = 0.034). Aspirin (OR 0.70, P = 0.086) and NSAIDs (OR 0.71, P = 0.081) both suggested some degree of benefit, although not reaching statistical significance. In the multivariate model, calcium lost statistical significance, yet multivitamins (OR 0.41, P = 0.001) and statins (OR 0.53, P = 0.003) retained significant associations (Table 3).
Barrett’s Length
Length of BE was analyzed both as a continuous variable, as well as in terms of long segment (3 cm or greater) versus short segment (under 3 cm). In the continuous assessment, mean length of BE was 4.0 cm (SD 2.9). In multivariate analysis, age was associated with an increased BE length of 0.63 cm for every 10 years older (95 % CI 0.23–1.04, P = 0.002), and statin use was associated with a 0.89 cm decrease in BE length (95 % CI 0.13–1.65, P = 0.023). Long-segment BE was also predicted by age with an OR of 1.37 for every 10 years older (95 % CI 1.02–1.86, P = 0.036), and a protective effect was seen with statin use (OR 0.47, 95 % CI 0.27–0.81, P = 0.007).
Discussion
BE remains the single largest risk factor for EAC [4], but little is known regarding ways to potentially prevent its onset. By assessing medication exposures in patients both with BE and with GERD symptoms alone, we aimed to better characterize the effects of these medications on the risk of developing BE.
In this case–control study, a multiple regression model found significant associations between the use of multivitamins and statins and BE. Not surprisingly, increasing age correlated with increased risk of BE. Furthermore, Hispanic ethnicity was found to demonstrate a protective effect on presence of BE. While our finding of a high percentage of males is expected in a veteran study, significantly more men were found in the BE group versus the control group. This is also consistent with prior data, which suggests a male to female ratio of approximately 2:1 [21]. However, as previously noted, the female patients were not included in any further data analysis to improve homogeneity.
Statins demonstrated an additional association with decreased overall BE length and decreased long-segment BE. Prior studies have demonstrated a positive effect of statin medications on risk of EAC in BE patients [12–15], and a recent VA-based study has also demonstrated a protective effect on the de novo development of BE [17]. Statins have been shown to be anti-proliferative and lead to apoptosis in BE cells in vitro [22]. This anti-proliferative effect could potentially prevent metaplastic change, thereby providing a mechanism for the observed decrease in risk of BE in our study.
The data for multivitamins were also suggestive of a beneficial effect, a novel finding. This benefit might lie within the antioxidant effects of constituent vitamins A, C, and E. A large, randomized, placebo-controlled trial of a daily multivitamin has shown a decrease in total cancer incidence compared with placebo (HR 0.92, P = 0.04), although cancer-specific and all-cause mortality were unchanged [23]. Despite this, a recently updated literature review by the United States Preventive Services Task Force (USPSTF) continues to state that the evidence for routine use of multivitamins for cancer prevention remains insufficient [24].
Calcium has been hypothesized to reduce the proliferative effect of bile acids and fatty acids on the colonic epithelium by binding these substances [25, 26]. In this study, calcium showed a positive effect in the univariate analysis. However, after adjusting for other variables, this lost statistical significance.
Aspirin has previously been analyzed in a case–control study of 434 patients with BE [8], in which the odds ratio for BE was 0.56 (95 % CI 0.39–0.80). In contrast, this study showed that aspirin and other NSAIDs trended toward a protective effect, yet did not attain statistical significance. This discrepancy may be explained by inadequate power in the present study. The putative mechanism for the effect of aspirin and NSAIDs is through COX-2 inhibition. This has been demonstrated to inhibit cellular proliferation and angiogenesis, as well as restore apoptosis [27].
There do remain limitations to our study. As the study was retrospective, causality cannot be proven, and there may be uncontrolled confounding variables present. For example, data on tobacco use were not included, since it was not consistently clear whether a non-smoker had been a prior or never smoker. As tobacco use is considered a risk factor for BE, lack of such data could have influenced the above results. However, multivariate analysis was used in an attempt to minimize the influence of confounding. Also, we ensured that biopsy-proven intestinal metaplasia was used in defining the BE population. Furthermore, the control group had undergone endoscopy for GERD-related symptoms, which would tend to provide similar populations for the cases and controls. In addition, patient medication use was taken from chart pharmacy records, but duration of medication use was not assessed. Also, undocumented over-the-counter medication use could have been unaccounted for. Furthermore, the male veteran population used in this study limits the generalizability of the data. However, despite the above limitations, our study’s findings are statistically sound, consistent with prior research, and can be related to putative mechanisms of action.
In conclusion, we have demonstrated that GERD patients with BE are less likely to use multivitamins or statins, as well as less likely to be of Hispanic ethnicity. Furthermore, statin use was associated with shorter BE length and a lower proportion of patients with long-segment BE. These data are promising in the search for ways to reduce the risk of progressing along the BE–EAC continuum. Although widespread adoption of these medications for preventing BE would be premature, there is support for the necessity of future prospective studies on prevention of BE and EAC.
References
Shaheen NJ, Richter JE. Barrett’s oesophagus. Lancet. 2009;373:850.
Rex DK, Cummings OW, Shaw M, et al. Screening for Barrett’s esophagus in colonoscopy patients with and without heartburn. Gastroenterology. 2003;125:1670.
Ronkainen J, Aro P, Storskrubb T, et al. Prevalence of Barrett’s esophagus in the general population: an endoscopic study. Gastroenterology. 2005;129:1825.
Van der Veen AH, Dees J, Blankensteijn JD, Van Blankenstein M. Adenocarcinoma in Barrett’s oesophagus: an overrated risk. Gut. 1989;30:14.
American Cancer Society. ACS: cancer facts and figures 2012. Atlanta: American Cancer Society; 2012.
American Gastroenterological Association. American Gastroenterological Association medical position statement on the management of Barrett’s esophagus. Gastroenterology. 2011;140:1084.
Verbeek RE, Leenders M, ten Kate FJW, et al. Surveillance of Barrett’s esophagus and mortality from esophageal adenocarcinoma: a population-based cohort study. Am J Gastroenterol. 2014;109:1215.
Omer ZB, Ananthakrishnan AN, Nattinger KJ, et al. Aspirin protects against Barrett’s esophagus in a multivariate logistic regression analysis. Clin Gastroenterol Hepatol. 2012;10:722.
Corley DA, Kerlikowske K, Verma R, Buffler P. Protective association of aspirin/NSAIDs and esophageal cancer: a systematic review and meta-analysis. Gastroenterology. 2003;124:47.
Abnet CC, Freedman ND, Kamangar F, Leitzmann MF, Hollenbeck AR, Schatzkin A. Non-steroidal anti-inflammatory drugs and risk of gastric and oesophageal adenocarcinomas: results from a cohort study and a meta-analysis. Br J Cancer. 2009;100:551.
Wilson KT, Fu S, Ramanujam KS, Meltzer SJ. Increased expression of inducible nitric oxide synthase and cyclooxygenase-2 in Barrett’s esophagus and associated adenocarcinomas. Cancer Res. 1998;58:2929.
Beales IL, Vardi I, Dearman L. Regular statin and aspirin use in patients with Barrett’s oesophagus is associated with a reduced incidence of oesophageal adenocarcinoma. Eur J Gastroenterol Hepatol. 2012;24:917.
Kantor ED, Onstad L, Blount PL, Reid BJ, Vaughan TL. Use of statin medications and risk of esophageal adenocarcinoma in persons with Barrett’s esophagus. Cancer Epidemiol Biomarkers Prev. 2012;21:456.
Kastelein F, Spaander MC, Biermann K, et al. Nonsteroidal anti-inflammatory drugs and statins have chemopreventative effects in patients with Barrett’s esophagus. Gastroenterology. 2011;141:2000.
Nguyen DM, Richardson P, El-Serag HB. Medications (NSAIDs, statins, proton pump inhibitors) and the risk of esophageal adenocarcinoma in patients with Barrett’s esophagus. Gastroenterology. 2010;138:2260.
Nguyen DM, El-Serag HB, Henderson L, Stein D, Bhattacharyya A, Sampliner RE. Medication usage and the risk of neoplasia in patients with Barrett’s esophagus. Clin Gastroenterol Hepatol. 2009;7:1299.
Nguyen T, Khalaf N, Ramsey D, El-Serag HB. Statin use is associated with a decreased risk of Barrett’s esophagus. Gastroenterology. 2014;147:314.
Wu K, Willett WC, Fuchs CS, Colditz GA, Giovannucci EL. Calcium intake and risk of colon cancer in women and men. J Natl Cancer Inst. 2002;94:437.
Wactawski-Wende J, Kotchen JM, Anderson GL, et al. Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med. 2006;354:684.
Sesso HD, Christen WG, Bubes V, et al. Multivitamins in the prevention of cardiovascular disease in men: the physicians’ health study II randomized controlled trial. JAMA. 2012;308:1751.
Cook MB, Wild CP, Forman D. A systematic review and meta-analysis of the sex ratio for Barrett’s esophagus, erosive reflux disease, and nonerosive reflux disease. Am J Epidemiol. 2005;162:1050.
Ogunwobi OO, Beales IL. Statins inhibit proliferation and induce apoptosis in Barrett’s esophageal adenocarcinoma cells. Am J Gastroenterol. 2008;103:825.
Gaziano J, Sesso HD, Christen WG, et al. Multivitamins in the prevention of cancer in men: the physicians’ health study II randomized controlled trial. JAMA. 2012;308:1871.
Moyer VA, on behalf of the U.S. Preventive Services Task Force. Vitamin, mineral, and multivitamin supplements for the primary prevention of cardiovascular disease and cancer: U.S. preventive services task force recommendation statement. Ann Intern Med. 2014;160:558.
Wargovich MJ, Eng VW, Newmark HL. Calcium inhibits the damaging and compensatory proliferative effects of fatty acids on mouse colon epithelium. Cancer Lett. 1984;23:253.
Van der Meer R, Kleibeuker JH, Lapre JA. Calcium phosphate, bile acids and colorectal cancer. Eur J Cancer Prev. 1991;1:55.
Anderson LA, Johnston BT, Watson RG, et al. Nonsteroidal anti-inflammatory drugs and the esophageal inflammation–metaplasia–adenocarcinoma sequence. Cancer Res. 2006;66:4975.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goldberg, A., Gerkin, R.D. & Young, M. Medical Prevention of Barrett’s Esophagus: Effects of Statins, Aspirin, Non-aspirin NSAIDs, Calcium, and Multivitamins. Dig Dis Sci 60, 2058–2062 (2015). https://doi.org/10.1007/s10620-015-3569-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3569-0