Abstract
Intra-abdominal hypertension (IAH) and abdominal compartment syndrome (ACS) may result from several clinic situations and carries high morbidity and mortality risk, particularly in intensive care unit patients. The clinical spectrum changes from splanchnic hypoperfusion and intestinal ischemia to multiple organ failure. Previous studies demonstrated that serum D-lactate levels may be an early indicator in intestinal ischemia. This study aimed to investigate the relationship between intestinal ischemia and serum D-lactate levels during experimental IAH. Thirty-two male Wistar Albino rats weighing 250±50 g were divided into four groups. Three different intra-abdominal pressure (IAP) levels supplied by placement of an intraperitoneal Peritofix catheter and iso-osmotic polyethylene glycol infusion. Each of the IAP levels (15, 20, and 25 mm Hg groups) was checked with the monitor system and fixed for an hour. Control-group animals were not subjected to increased IAP. One hour later, 5-ml blood samples were taken for measurement of serum D-lactate levels and 2-cm intestinal tissue samples were taken 5 cm proximal to the ileocecal valve for histopathologic examination. Elevated serum D-lactate levels were recorded in animals with higher IAP levels.There was a positive correlation between serum D-lactate levels and IAP levels. Histological examinations of the intestinal tissue samples showed no significant pathologic changes in concordance with intestinal ischemia. Serum D-lactate levels may be an early indicator for increased IAP pressure before intestinal ischemic changes occur.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The abdominal cavity has a limited capacity for enlargement. Many clinical situations may cause acute elevations in intra-abdominal pressure (IAP), termed intra-abdominal hypertenion (IAH). Sustained and uncontrolled IAH adversely affects organ functions. Organ dysfunction due to IAH has been defined as abdominal compartment syndrome (ACS). ACS is a late and serious complication of sustained IAH [1, 2].
IAH and ACS are frequently encountered entities in intensive care patients, more than expected [1–3]. Untreated IAH converts to ACS and results in multiorgan failure with high risk of morbidity and mortality [1, 2, 4]. Thus early diagnosis of IAH, before ACS occurs, is extremely important. Experimental and clinical reports indicate that splanchnic hypoperfusion and reduced intestinal mucosal blood flow are early physiopathologic alterations due to IAH [3–8]. It has also been shown that serum D-lactate levels may be an early indicator for detection of intestinal ischemia [9–12].
The purpose of this study was to investigate the relationship between serum D-lactate levels and increased IAP levels.
Materials and methods
Experimental design
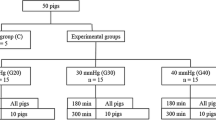
The local ethics committee of Ankara Numune Teaching and Research Hospital approved the study protocol. Thirty-two male Wistar albino rats weighing 250±50 g were randomly divided into four groups. The rats were allowed free access to normal food and water before experimentation. The animals were anesthetized with an intramuscular injection of ketamine hydrochloride (50 mg/kg; Ketalar) and xylazine hydrochloride (5 mg/kg; Rompun). Abdominal skin was shaved. It was cleaned with povidone iodine solution.
A 1-cm midline incision was performed and a Peritofix catheter (B/Braun, Melsungen AG, Germany) was placed intraperitoneally for infusion of a polyethylene glycol (PEG) solution in all animals except those in the control group. Iso-osmotic PEG solution was infused into the abdominal cavity via a three-way stopcock. The intra-abdominal pressure (IAP) was measured continuously with this catheter connected to a pressure transducer from a monitor system (Petaş, KMA 275, Turkey).
Group 1 served as the controls; animals were not subjected to increased IAP. The IAP was recorded continuously until it reached a level of 15 mm Hg in Group II (n = 8), 20 mm Hg in Group III (n = 8), and 25 mm Hg in Group IV (n = 8) animals. The IAP was maintained for 60 min using monitoring, and additional PEG infusion was applied when needed. One hour later, relaparotomy was done in all groups and 2-cm intestinal tissue specimens were taken 5 cm proximal to the ileocecal valve for histological examinations. Then 5-ml blood samples were taken in heparinized syringes for serum D-lactate levels.
Measurement of serum D-Lactate levels
Sera were separated from 5 ml of heparinized blood (20 IU/ml) by centrifugation at 3200 rpm for 10 min and stored at –70°C before measurements. We used a UV-spectrophotometric method for measurement of D-lactate levels. All reagents were purchased from Roche (R-Biopharm Catalog No. 11 112 821 035; Mannheim, Germany). We used a Shimadzu 1201 UV-visible spectrophotometer (Shimadzu, Kyoto, Japan). Results are presented as millimolar concentration [13].
Histological evaluation
Small bowel segments were placed in 10% formaldehyde and stained with hematoxylin-eosin. They were evaluated according to Park-Chiu classification [14].
Statistical analyses
All data were recorded using SPSS 11.00 for Windows for statistical analyses. Statistical comparisons were performed using the Tukey post hoc test. P values <0.05 were considered significant.
Results
Mean serum D-lactate levels and ranges in all groups are detailed in Table 1 (see also Fig. 1).Footnote 1 Serum D-lactate levels were significantly elevated in groups with 25 mm Hg IAP compared with the other groups. Mean serum D-lactate levels were also relatively high in groups with 15 mm Hg IAP and in groups with 20 mm Hg IAP (0.43 and 0.46 mM, respectively) compared with the control group. These elevations were not statistically significant. The degrees of statistical significance of the elevated serum D-lactate levels according to group are shown in Table 2.
Histopathologic evaluation of the intestinal tissue samples revealed no ischemic changes in elevated-IAP groups. Decreased intestinal wall thickness was a unique histopathologic alteration, noted only in groups with 25 mm Hg IAP.
Discussion
Elevated IAP or IAH is a frequently encountered problem in critically ill patients and carries a high risk of morbidity and mortality [15]. There has been an exponential increase in investigations focused on increased IAP and subsequent adverse effects such as ACS [16]. There is still controversy about the exact level of IAP at which ACS occurs [1, 17]. Increased IAP and ACS are not synonymous. Although ACS is a late and highly lethal complication of increased IAP but not an inevitable result of IAH [12, 18]. Thus a clinician should be aware of the physiopathological implications of IAH and ACS in patients at risk for increased IAP. Acute ACS is increasingly recognized as a cause for multiorgan dysfunction. Early detection and timely prevention of IAH are extremely important and require suspicion. Of course the aphorism “Prevention is better than treatment“ is always valid for this challenging problem [4, 19].
Physical abdominal examination is not reliable for detecting increased IAP. However, routine IAP monitoring is not acceptable for practice. A variety of ACS models and IAP measurement methods has been suggested in animal models and clinical investigations [6, 17, 18, 20–25]. The main goal of all these studies is to clarify the pathophysiologic alterations related to IAH and ACS.
PEG, Ringer's lactate, saline infusions, and carbon dioxide insufflation have been applied to increase IAP in different experimental abdominal compartment syndrome models [6, 21–23, 25]. In this study, PEG was used to increase the IAP, as it is an economical and easily applicable agent.
Various pressure levels and durations have been used in various experimental models. Bloomfield et al. maintained an IAP level of 25 mm Hg for 0, 30, 60, 90, and 120 min in their study [22]. Rezende-Neto et al. examined the physiopathologic changes related to IAH at an IAP level of 20 mm Hg for 60 and 90 min [16]. Doty et al. observed the effects of a 30 mm Hg IAP level for 60 min [26]. All experimental studies showed that IAP levels of ≥25 mm Hg resulted in decreased urine output and mesenteric blood flow, elevated interleukin-1b cytokine levels, and derangement of cardiopulmonary functions in a 1-hr period. We examined the adverse effects of IAP levels of 15, 20, and 25 mm Hg for a 1-hr period in rats. Elevated serum D-lactate levels recorded were significantly higher than those of the control group only at the 25 mm Hg IAP level in the current study.
There is no consensus about the exact IAP level that is associated with clinically significant impairment of organ function [2, 19, 27]. Although an IAP >20 mm Hg is clinically significant in most patients, even at the relatively low pressure of 10–15 mm Hg significant impairments of organ function may occur. Hunter et al. demonstrated a significant decrease in mesenteric arterial and mucosal blood flow at an IAP level of 20 mm Hg despite minimal changes in cardiac output [27]. It has also been shown that hepatic arterial blood flow and portal venous blood flow significantly decrease when the IAP is as low as 10 mm Hg. Barnes et al. showed that there was a 61% reduction in superior mesenteric blood flow at an IAP level of 40 mm Hg [6]. Diebel et al. demonstrated that significant reductions in intestinal mucosal blood flow and mesenteric, hepatic, and portal blood flows occurred even when cardiac output and systemic blood pressure were maintained at normal levels in a porcine model of increased IAP [6].
These studies also showed that splanchnic hypoperfusion and reduced intestinal mucosal blood flow without histopathological findings of intestinal ischemia are early adverse effects of raised IAP. Intestinal and hepatic ischemia possibly lead to multiorgan dysfunction syndrome secondary to bacterial translocation and cytokine release. Thus early determination of intestinal ischemia is very important for prevention of multiorgan dysfunction syndrome [6, 26, 28, 29]. Poeze et al. reported that D-lactate is a better marker of splanchnic hypoperfusion than its isomer L-lactate [12]. Murray et al. showed that serum D-lactate level measurements may be a useful marker of acute intestinal ischemia [12, 13]. In our study, significantly increased serum D-lactate levels were attributed to decreased intestinal mucosal blood flowFootnote 2. However, we found no distinct histopathologic changes except decreased intestinal wall thickness in animals with an IAP of 25 mm Hg. We believe that serum D-lactate levels may be an early marker of intestinal hypoperfusion in the presence of IAH.
Conclusion
In conclusion, there is a positive correlation between serum D-lactate levels and increased IAP levels. An IAP of 25 mm Hg has significant adverse effects on splanchnic and intestinal mucosal blood flow in our experimental IAH model. We believe that significantly increased serum D-lactate levels are likely to be a useful determinant of intestinal hypoxia due to increased IAP.
Notes
Citation of Fig. 1 here OK? If not, pls. cite in correct location. Thank you.
This sentence as meant? If not, pls. correct. Thank you.
References
Sanchez N, Tenofsky, et al. (2001) What is the abdominal pressure? Am Surg 67:243–248
Hong JJ, Cohn JM, Perez, Dolich MO, Brown M, McKenney (2002) Prospective study of the incidence and outcome of intra-abdominal hypertension and the abdominal compartment syndrome. Br J Surg 89:591–596
McNellis J, Soffer S, Marini C, Ritter G (2002) Abdominal compartment syndrome in the surgical intensive care unit. Am Surg 68:18–23
Sieh KM, Kent, Chu M (2001) Intra-abdominal hypertension and abdominal compartment syndrome. Arch Surg 386:53–61
Ivatury R, Diebel L, Porter J, Simon R (1997) Intra-abdominal hypertension and the abdominal compartment syndrome. Surg Clin North Am 77:783–800
Diebel L, Dulchavsky S, et al. (1997) Splanchnic ischemia and bacterial translocation in the abdominal compartment syndrome. J Trauma 43:852–856
Kologlu M, Sayek I, Kologlu B, Demirali O (1999) Effect of persistently elevated intra-abdominal pressure on healing of colonic anastomoses. Am J Surg 178:293–297
Meldrum D, Moore F, Moore E, Francois R (1997) Prospective characterization and selective management of the abdominal compartment syndrome. Am J Surg 174:667–672
Szalay L, Umar F, Kham A, et al. (2003) Increased plasma D-lactate is associated with the severity of hemorrhagic/traumatic shock in rats. Shock 3:245–251
Murray M, Barbose J, Cobb C (1993) Serum D-lactate levels as a predictor of acute intestinal ischemia in a rat model. J Surg Res 54:507–509
Murray M, Gonze M, Nowak L, Cobb C (1994) Serum D-lactate levels as an aid to diagnosing acute intestinal ischemia. Am J Surg 167:575–578
Poeze M, Solberg B, Greve JW (2003) Gastric PgO2 and Pg-aCO2 gaps are related to D-lactate and not to L-lactate levels in patients with septic shock. Intensive Care Med 29:2081–2085
Marti R, Vanela E, et al. (1997) Determination of D-lactate by enzymatic methods in biological fluids; study of interferences. Clin Chem 43:1010–1015
Chiu CT, McArdle AH, Brown R, Scott H, Gurd F (1970) Intestinal mucosal lesion in low flow states. Arch Surg 101:478–483
Malbrain M, Davide C, Pelosi P, Luciano G (2005) Incidence and prognosis of intra-abdominal hypertension in a mixed population of critically ill patients: a multiple-center epidemiological study. Crit Care Med 33:315–322
Rezende-Neto J, Moore E, Vinicius M, et al. (2002) Systemic inflammatory responses secondary to abdominal compartment syndrome; stage for muliple organ failure. J Trauma 53:1121–1128
Malbrain MLNG (1999) Abdominal pressure in the critically ill:measurement and clinical relevance. Intens Care 25:1453–1458
Engum SA, Kogon B, Jensen E (2002) Gastric tonometry and direct intra-abdominal pressure monitoring in abdominal compartment syndrome. J Pediatr Surg 37:214–218
Moore AFK, Hargest R, Martin M, Delicata RJ (2004) Intraabdominal hypertension and the abdominal compartment syndrome. Br J Surg 91:1102–1110
Kirkpatrick A, Brenneman F, McLean R, Boulanger B (2000) Is clinical examination an accruate indicator of raised intra-abdominal pressure in critically injured patients? Can J Surg 43:207–211
Robotham J, Wise R, Bromberger B (1985) Effects of changes in abdominal pressure on left ventricular performance and regional blood flow. Crit Care Med 13:803–809
Bloomfield G, Saggi B, Blocher C, Sugerman H (1999) Physiologic effects of externally applied continuous negative abdominal pressure for intra-abdominal hypertension. J Trauma 46:1009–1013
Oda J, Ivatury R, Blocher R, Malhotra A (2002) Amplified cytokine response and lung injury by sequential abdominal hemorrhagic shock and compartment syndrome in laboratory model of ischemia-reperfusion. J Trauma 52:625–632
Gudmundson FF, Gislason A, et al. (2001) Effects of prolonged increased intra-abdominal pressure on gastrointestinal blood flows in pigs. Surg Endosc 15:854–860
Schachtrupp A, Graf J, Tons C, Hoer J (2003) Intravasculer volume depletion in a hour porcine model of intra abdominal hypertension. J Trauma 55:734–740
Doty J, Oda J, Ivatury R (2002) The effects of hemodynamic shock and increased intra-abdominal pressure on bacterial translocation. J Trauma 52:13–17
Hunter JD, Damani Z (2004) Intra-abdominal hypertension and the abdominal compartment syndrome. Anaesthesia 59:899–907
Hsu YP, Chen RJ, Feng J (2004) Increases suscepility to oxidant injury in hepatocytes from rats with intra-abdominal hypertension. J Trauma 57:569–575
Gracias V, Braslow B, Johnson J (2002) Abdominal compartment syndrome in the open abdomen. Arch Surg 137:1300–1306
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duzgun, A.P., Gulgez, B., Özmutlu, A. et al. The Relationship Between Intestinal Hypoperfusion and Serum D-Lactate Levels During Experimental Intra-Abdominal Hypertension. Dig Dis Sci 51, 2400–2403 (2006). https://doi.org/10.1007/s10620-006-9334-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9334-7